Pterosaurs from the Late Cretaceous of Angola
Abstract
:1. Introduction
2. Materials and Methods
3. Nomenclatural Acts
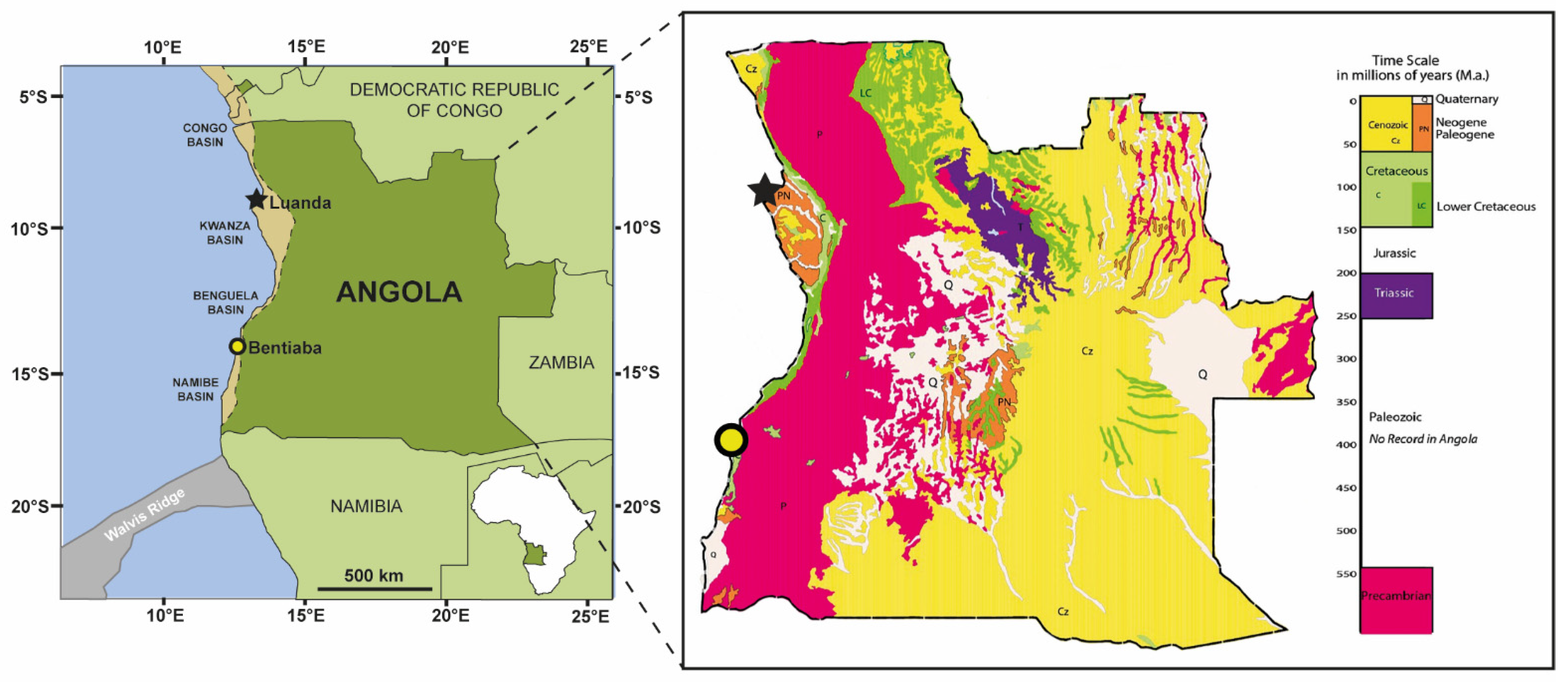
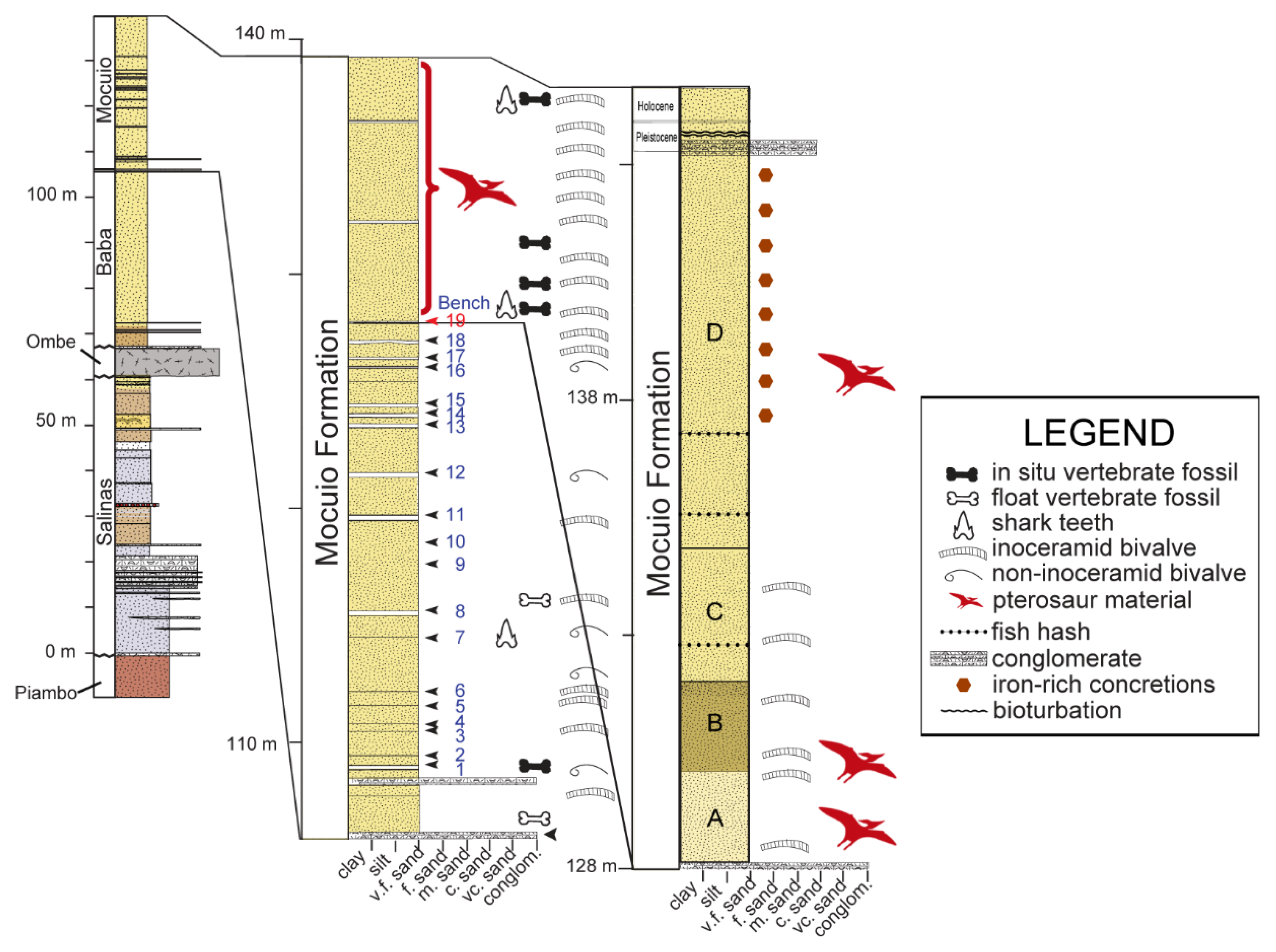
4. Geographical and Geological Setting
5. Description
5.1. Epapatelo otyikokolo, Gen. et sp. Nov
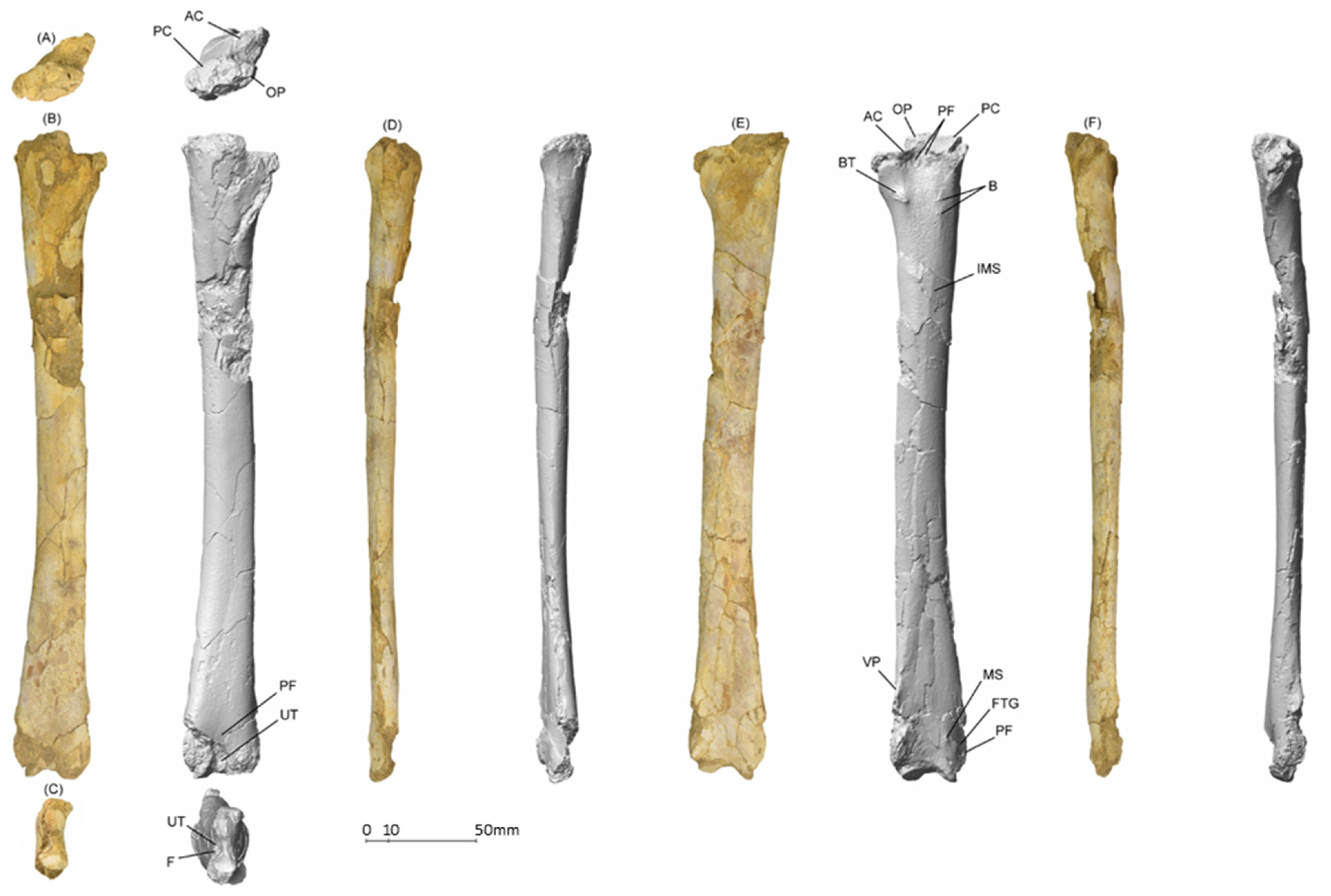
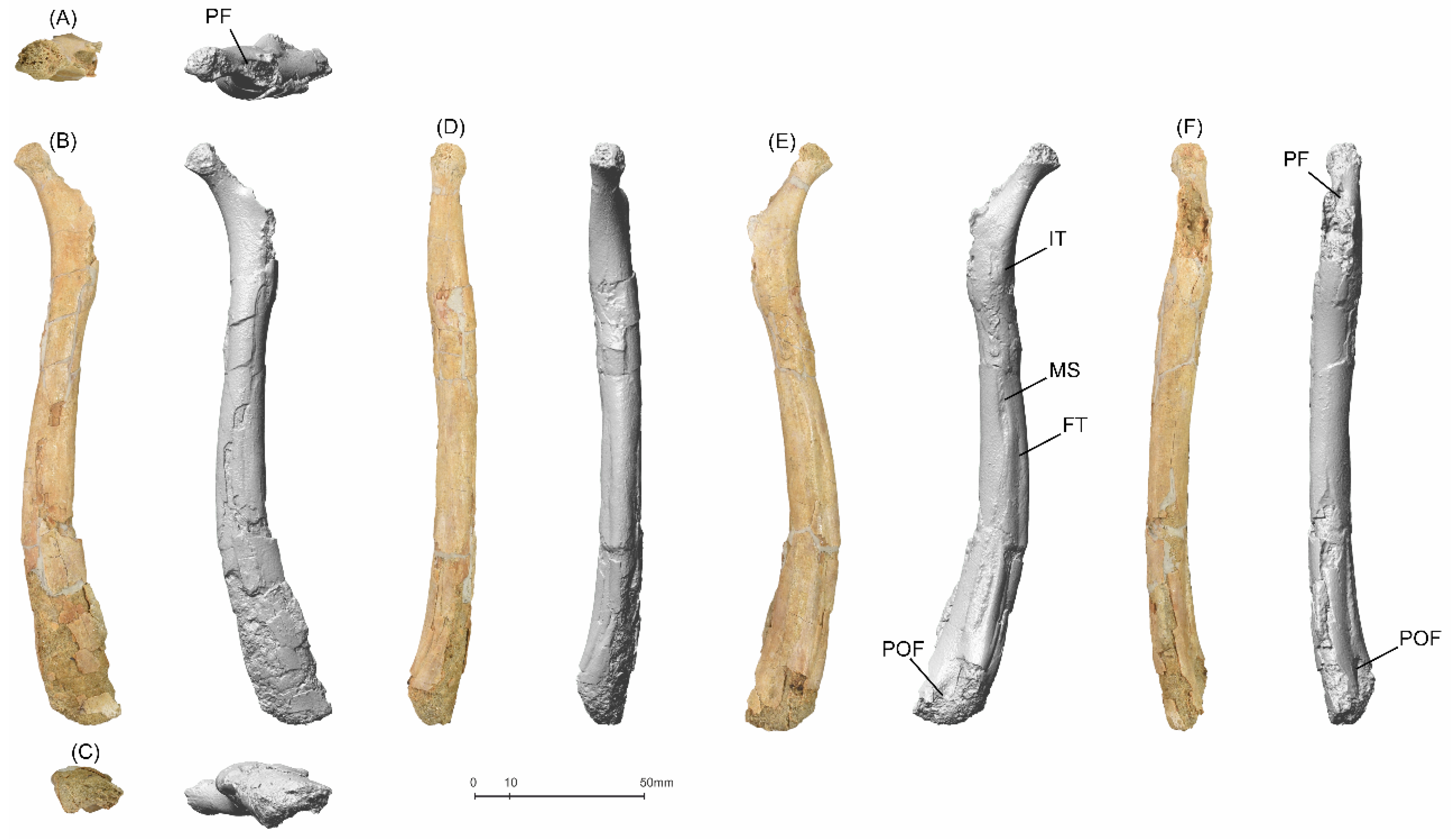
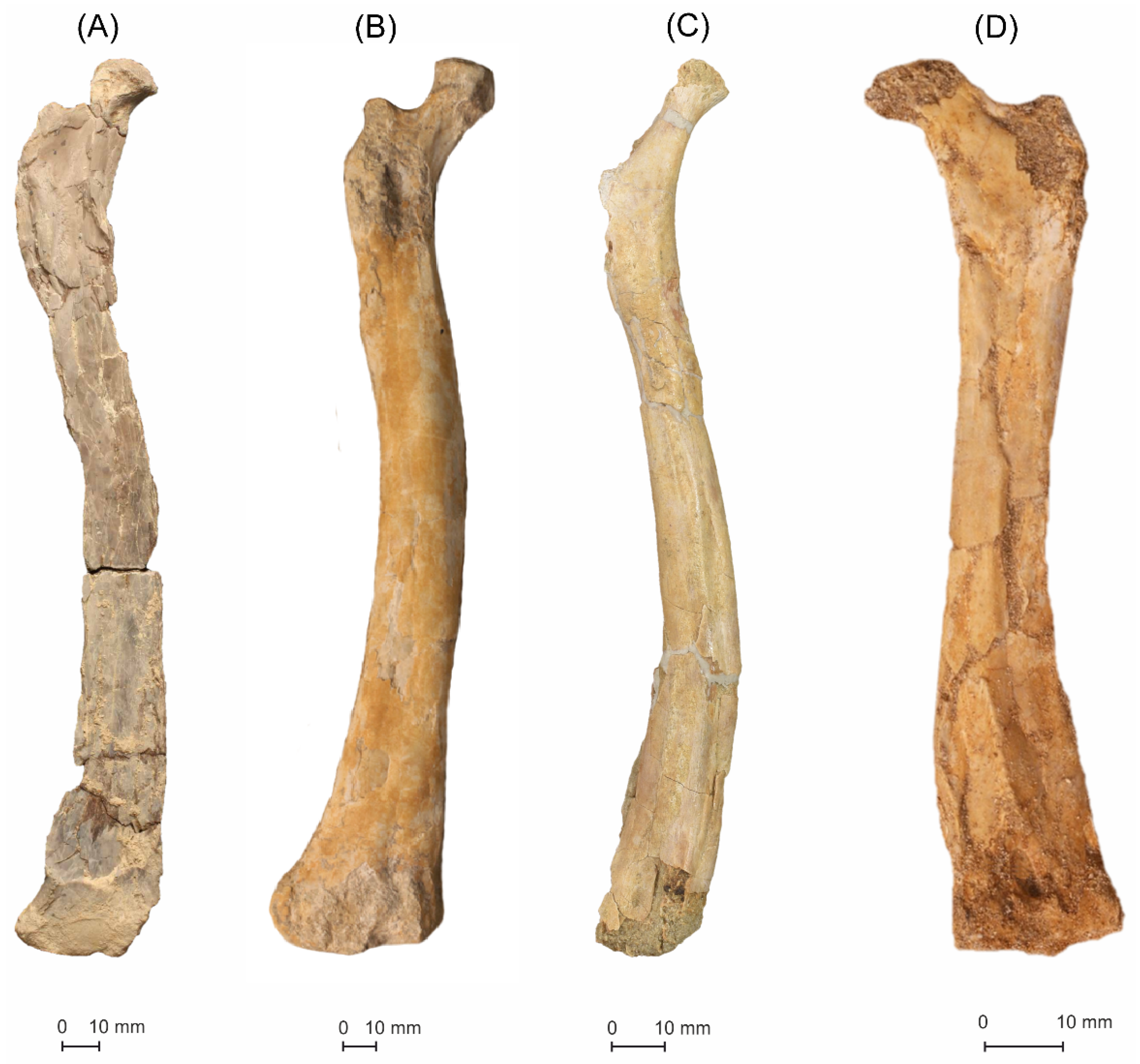
5.1.1. MGUAN-PA650 Humerus
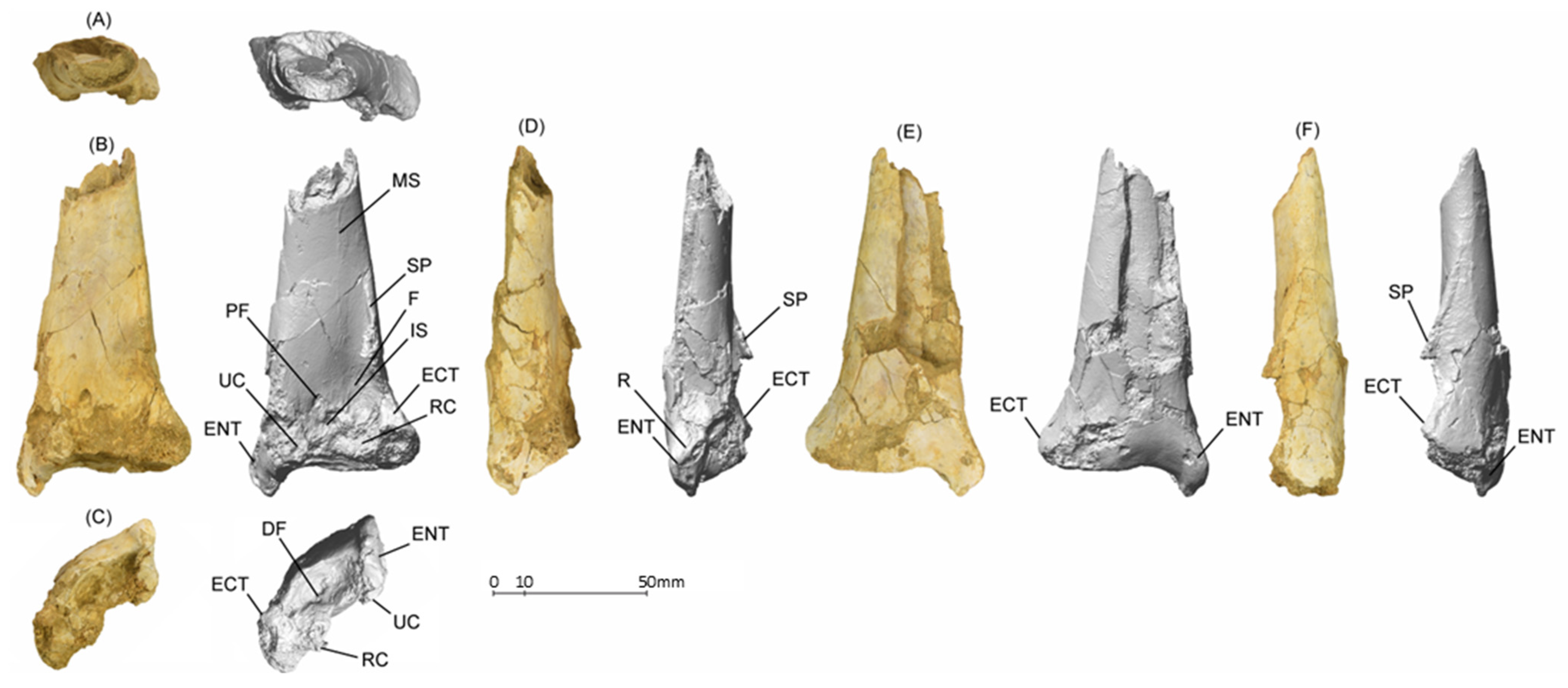
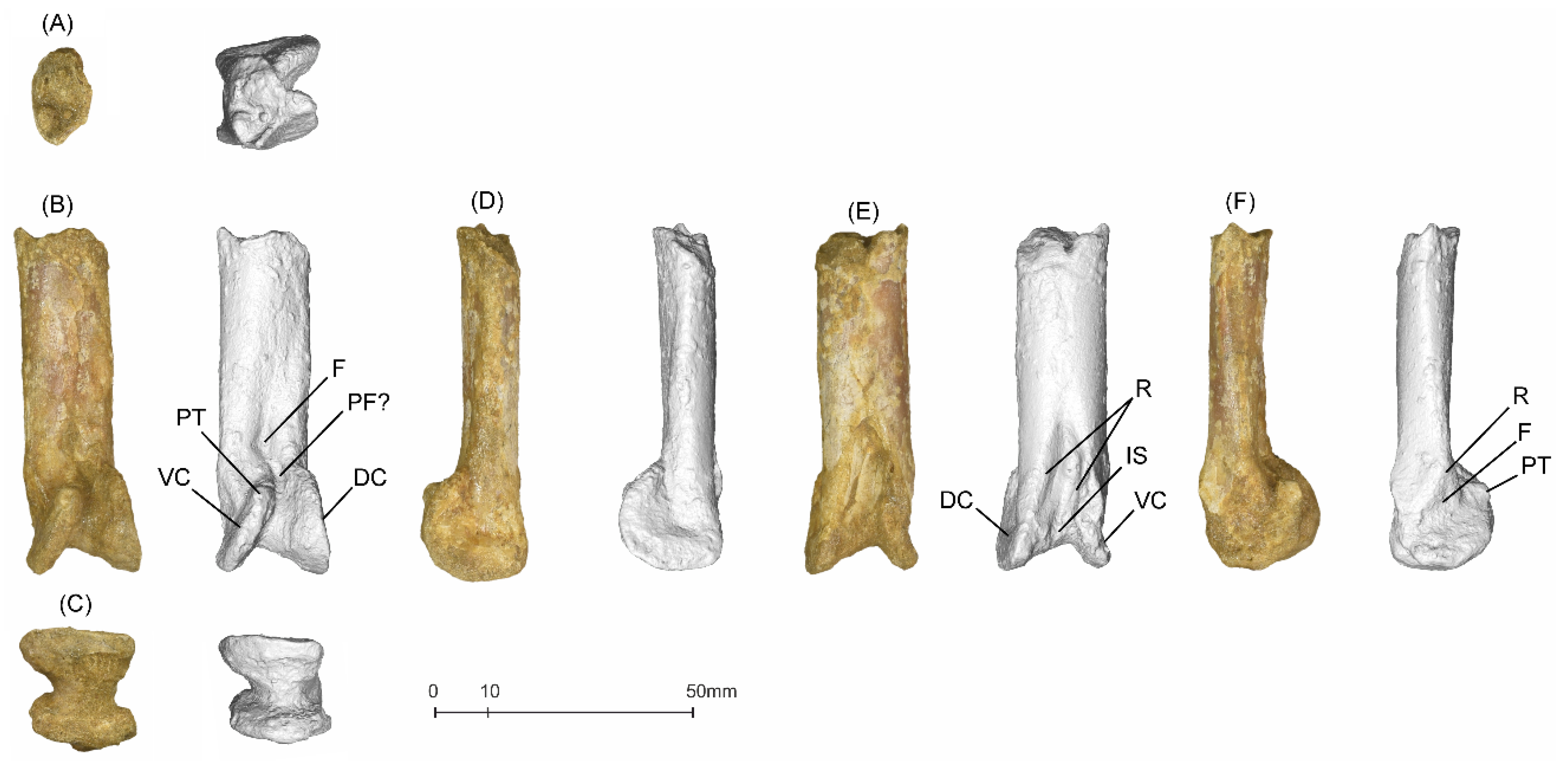
5.1.2. MGUAN-PA650 Ulna
5.1.3. MGUAN-PA661 Ulna and Radius
5.2. Pteranodontia Indet
MGUAN-PA163 Femur
5.3. Pteranodontoidea Indet
5.3.1. MGUAN-PA654 Metacarpal IV
5.3.2. MGUAN-PA654 Manual Digit IV Phalanx 1
6. Additional Material
7. Results
7.1. Systematic Paleontology
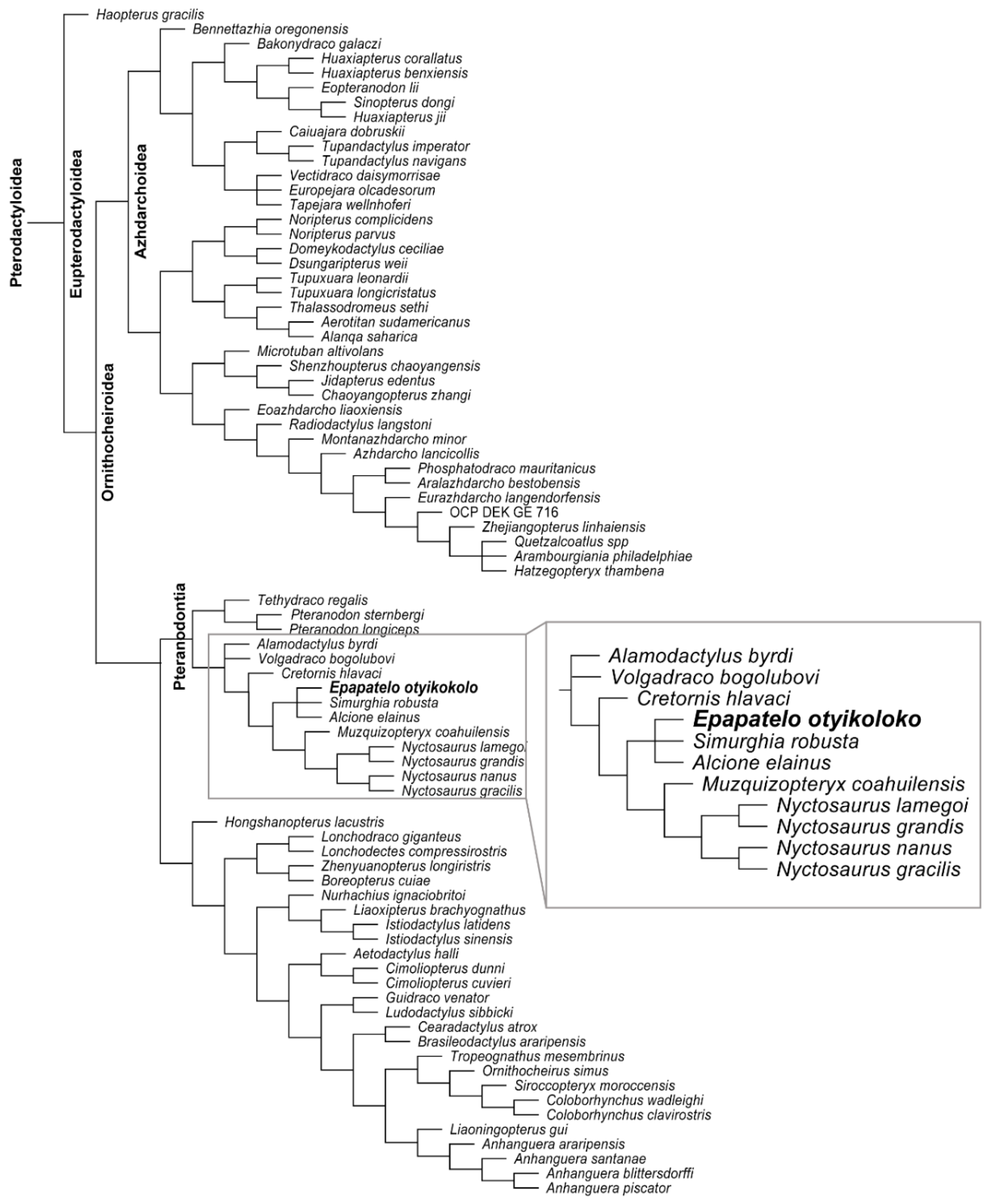
7.2. Phylogenetic Analysis
7.3. Wingspan Calculation
7.4. Bone Histology
7.5. Comparison and Discussion
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barrett, P.M.; Butler, R.J.; Edwards, N.P.; Milner, A.R. Pterosaur distribution in time and space: An atlas. Zitteliana 2008, B28, 61–107. [Google Scholar]
- Kellner, A.W.A.; Mello, A.M.S.; Ford, T. A survey of pterosaurs from Africa with the description of a new specimen from Morocco. In Paleontologia: Cenários da Vida; Carvalho, I.S., Cassab, R.C.T., Schwanke, C., Carvalho, M.A., Fernandes, A.C.S., Eds.; Interciência: Rio de Janeiro, Brazil, 2007; pp. 257–267. [Google Scholar]
- Mateus, O.; Callapez, P.M.; Polcyn, M.J.; Schulp, A.S.; Gonçalves, A.O.; Jacobs, L.L. The fossil record of biodiversity in Angola through time: A paleontological perspective. In Biodiversity of Angola; Huntley, B.J., Russo, V., Lages, F., Ferrand, N., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 53–76. [Google Scholar]
- Araújo, R.; Polcyn, M.J.; Schulp, A.S.; Mateus, O.; Jacobs, L.L.; Gonçalves, A.O.; Morais, M.-L. A new elasmosaurid from the Early Maastrichtian of Angola and the implications of girdle morphology on swimming style in plesiosaurs. Neth. J. Geosci. 2015, 94, 109–120. [Google Scholar] [CrossRef]
- Araújo, R.; Polcyn, M.J.; Lindgren, J.; Jacobs, L.L.; Schulp, A.S.; Mateus, O.; Gonçalves, A.O.; Morais, M.-L. New aristonectine elasmosaurid plesiosaur specimens from the Early Maastrichtian of Angola and comments on paedomorphism in plesiosaurs. Neth. J. Geosci. 2015, 94, 93–108. [Google Scholar] [CrossRef]
- Jacobs, L.L.; Mateus, O.; Polcyn, M.J.; Schulp, A.S.; Antunes, M.T.; Morais, M.L.; Da Silva Tavares, T. The occurrence and geological setting of Cretaceous dinosaurs, mosasaurs, plesiosaurs, and turtles from Angola. J. Paleontol. Soc. Korea 2006, 22, 91–110. [Google Scholar]
- Jacobs, L.L.; Mateus, O.; Polcyn, M.J.; Schulp, A.S.; Scotese, C.R.; Goswami, A.; Ferguson, K.M.; Robbins, J.; Vineyard, D.P.; Neto, A.B.; et al. Cretaceous paleogeography, paleoclimatology, and amniote biogeography of the low and mid-latitude South Atlantic Ocean. Bull. Société Géologique Fr. 2009, 180, 333–341. [Google Scholar] [CrossRef]
- Marx, M.P.; Mateus, O.; Polcyn, M.J.; Schulp, A.S.; Gonçalves, A.O. The cranial anatomy and relationships of Cardiocorax mukulu (Plesiosauria: Elasmosauridae) from Bentiaba, Angola. PLoS ONE 2021, 16, 1–51. [Google Scholar]
- Mateus, O.; Jacobs, L.; Polcyn, M.; Schulp, A.S.; Vineyard, D.; Buta Neto, A.; Antunes, M.T. The oldest African eucryptodiran turtle from the Cretaceous of Angola. Acta Palaeontol. Pol. 2009, 54, 581–588. [Google Scholar] [CrossRef]
- Mateus, O.; Polcyn, M.J.; Jacobs, L.L.; Araújo, R.; Schulp, A.S.; Marinheiro, J.; Pereira, B.; Vineyard, D. Cretaceous amniotes from Angola: Dinosaurs, pterosaurs, mosasaurs, plesiosaurs, and turtles. In Actas de V Jornadas Internacionales sobre Paleontología de Dinosaurios y su Entorno; Hurtado, P.H., Fernández-Baldor, F.T., Canudo Sanagustín, J.I., Eds.; Colectivo Arqueológico y Paleontológico de Salas, C.A.S.: Burgos, Spain, 2012; pp. 71–105. [Google Scholar]
- Polcyn, M.J.; Jacobs, L.L.; Schulp, A.S.; Mateus, O. The North African mosasaur Globidens phosphaticus from the Maastrichtian of Angola. Hist. Biol. 2010, 22, 175–185. [Google Scholar] [CrossRef]
- Schulp, A.S.; Polcyn, M.J.; Mateus, O.; Jacobs, L.L.; Morais, M.L.; da Silva Tavares, T. New mosasaur material from the Maastrichtian of Angola, with notes on the phylogeny, distribution and palaeoecology of the genus Prognathodon. Publ. Van Het Nat. Genoot. Limbg. Reeks XLV Aflevering 2006, 1, 57–67. [Google Scholar]
- Schulp, A.S.; Polcyn, M.J.; Mateus, O.; Jacobs, L.L.; Morais, M.L. A new species of Prognathodon (Squamata, Mosasauridae) from the Maastrichtian of Angola, and the affinities of the mosasaur genus Liodon. In Proceedings of the Second Mosasaur Meeting, Fort Hays Studies Special Issue, Hays, KS, USA, 3–6 May 2007; pp. 1–12. [Google Scholar]
- O’Leary, M.A.; Kaufman, S.G. MorphoBank 3.0: Web Application for Morphological Phylogenetics and Taxonomy. 2012. Available online: http://morphobank.org/permalink/?3966 (accessed on 24 July 2022).
- Goloboff, P.; Catalano, S. T.N.T., version 1.5, with a full implementation of phylogenetic morphometrics [computer software]. Cladistics 2016, 32, 221–238. [Google Scholar] [CrossRef]
- Goloboff, P.A.; Farris, J.S.; Nixon, K.C. T.N.T., a free program for phylogenetic analysis. Cladistics 2008, 24, 774–786. [Google Scholar] [CrossRef]
- Longrich, N.R.; Martill, D.M.; Andres, B. Late Maastrichtian pterosaurs from North Africa and mass extinction of Pterosauria at the Cretaceous-Paleogene boundary. PLoS Biol. 2018, 16, e2001663. [Google Scholar]
- Andres, B.; Clark, J.; Xu, X. The earliest pterodactyloid and the origin of the group. Curr. Biol. 2014, 24, 1011–1016. [Google Scholar] [CrossRef] [PubMed]
- Broom, R. Note on Mesosuchus browni, Watson and on a new South African Triassic pseudosuchian (Euparkeria capensis). Rec. Albany Mus 1913, 2, 394–396. [Google Scholar]
- Frič, A. O zkamenĕlem pt aku Chocĕnskem. Vesmír 1881, 10, 232–233. [Google Scholar]
- Averianov, A.; Ekrt, B. Cretornis hlavaci Frič, 1881 from the Upper Cretaceous of Czech Republic (Pterosauria, Azhdarchoidea). Cretac. Res. 2015, 55, 164–175. [Google Scholar] [CrossRef]
- Varricchio, D.J. A new bird from the Upper Cretaceous Two Medicine Formation of Montana. Can. J. Earth Sci. 2002, 39, 19–26. [Google Scholar] [CrossRef]
- Lamm, E. Preparation and sectioning of specimens. In Bone Histology of Fossil Tetrapods; Padian, K., Lamm, E., Eds.; University of California Press: Berkeley, CA, USA, 2013; pp. 55–160. [Google Scholar]
- Sampson, S.D. Predatory Dinosaur Remains from Madagascar: Implications for the Cretaceous biogeography of Gondwana. Science 1998, 280, 1048–1051. [Google Scholar] [CrossRef]
- Benton, M.J.; Bouaziz, S.; Buffetaut, E.; Martill, D.; Ouaja, M.; Soussi, M.; Truema, C. Dinosaurs and other fossil vertebrates from fluvial deposits in the lower Cretaceous of southern Tunisia. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2000, 157, 227–246. [Google Scholar] [CrossRef]
- Strganac, C.; Salminen, J.; Jacobs, L.L.; Polcyn, M.J.; Ferguson, K.M.; Mateus, O.; Schulp, A.S.; Morais, M.L.; Tavares, T.S.; Gonçalves, A.O. Carbon isotope stratigraphy, magnetostratigraphy, and 40Ar/39Ar age of the Cretaceous South Atlantic coast, Namibe Basin, Angola. J. Afr. Earth Sci. 2014, 99, 452–462. [Google Scholar] [CrossRef]
- U.S. Geological Survey. Geological Map of Africa [Map on the Internet]. 2002. Available online: http://www.uni-koeln.de/sfb389/e/e1/ (accessed on 24 July 2022).
- Strganac, C.; Jacobs, L.L.; Polcyn, M.J.; Mateus, O.; Myers, T.S.; Salminen, J.; May, S.R.; Araújo, R.; Ferguson, K.M.; Gonçalves, A.O.; et al. Geological setting and paleoecology of the Upper Cretaceous Bench 19 marine vertebrate bonebed at Bentiaba, Angola. Neth. J. Geosci.—Geol. En Mijnb. 2015, 94, 121–136. [Google Scholar] [CrossRef]
- Strganac, C.; Jacobs, L.L.; Polcyn, M.J.; Ferguson, K.M.; Mateus, O.; Gonçalves, A.O.; Morais, M.-L.; da Silva Tavares, T. Stable oxygen isotope chemostratigraphy and paleotemperature regime of mosasaurs at Bentiaba, Angola. Neth. J. Geosci.—Geol. En Mijnb. 2015, 94, 137–143. [Google Scholar] [CrossRef]
- Marsh, O.C. Principal characters of American pterodactyls. Am. J. Sci. 1876, 3, 479–480. [Google Scholar] [CrossRef]
- Currey, J.D.; Alexander, R.M. The thickness of the walls of tubular bones. J. Zool. 2009, 206, 453–468. [Google Scholar] [CrossRef]
- Bennett, S.C. Morphological evolution of the pectoral girdle of pterosaurs: Myology and function. Geol. Soc. Lond. Spec. Publ. 2003, 217, 191–215. [Google Scholar] [CrossRef]
- Bennett, S.C. The osteology and functional morphology of the Late Cretaceous pterosaur Pteranodon. Part I. General description and osteology. Paleontographica A 2001, 260, 1–112. [Google Scholar] [CrossRef]
- Bennett, S.C. The ontogeny of Pteranodon and other pterosaurs. Paleobiol. Winter 1993, 19, 92–106. [Google Scholar] [CrossRef]
- Owen, R. Report on British Fossil Reptiles, Part II. In Proceedings of the 11th Meeting of the British Association for the Advancement of Science, Plymouth, UK, 24 July 1841; pp. 60–204. [Google Scholar]
- Andres, B.; Padian, K.; Pterosauria, R. Owen 1842. In Phylonyms; de Queiroz, K., Cantino, P.D., Gauthier, J.A., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 1201–1204. [Google Scholar]
- Plieninger, F. Beiträge Zur Kenntnis der Flugsaurier. Paläontographica 1901, 48, 65–90. [Google Scholar]
- Andres, B.; Padian, K.; Pterodactyloidea, F. Plieninger 1901. E. In Phylonyms; de Queiroz, K., Cantino, P.D., Gauthier, J.A., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 1205–1207. [Google Scholar]
- Unwin, D.M. On the Phylogeny and Evolutionary History of Pterosaurs. In Geological Society; Special Publications: London, UK, 2003; Volume 217, pp. 139–190. [Google Scholar]
- Padres missionarios da Congregação do Espírito Santo e do Sagrado Coração de Maria. In Diccionario Portuguez-Olunyaneka; Typographia da Missão: Huilla, Colombia, 1896.
- Jacobs, L.L.; Polcyn, M.J.; Mateus, O.; Schulp, A.S.; Gonçalves, A.O.; Morais, M.L. Post-Gondwana Africa and the vertebrate history of the Angolan Atlantic coast. Mem. Mus. Vic. 2016, 74, 343–362. [Google Scholar] [CrossRef]
- Averianov, A.O.; Arkhangelsky, M.S.; Pervushov, E.M. A new Late Cretaceous azhdarchid (Pterosauria, Azhdarchidae) from the Volga Region. Paleontol. J. 2008, 42, 634–642. [Google Scholar] [CrossRef]
- Andres, B.; Myers, T.S. Lone Star pterosaurs. Earth Environ. Sci. Trans. R. Soc. Edinb. 2013, 103, 383–398. [Google Scholar] [CrossRef]
- Frey, E.; Buchy, M.C.; Stinnesbeck, W.; González, A.G.; Stefano, A. Muzquizopteryx coahuilensis n.g., sp. nov., a nyctosaurid pterosaur with soft tissue preservation from the Coniacian (Late Cretaceous) of northeast Mexico (Coahuila). Oryctos 2006, 6, 19–39. [Google Scholar]
- Bennett, S.C. The osteology and functional morphology of the Late Cretaceous pterosaur Pteranodon. Part II. Size and functional morphology. Paleontographica A 2001, 260, 115–153. [Google Scholar] [CrossRef]
- Cox, R.M.; Butler, M.A.; John-Alder, H.B. The evolution of sexual size dimorphism in reptiles. In Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism; Fairbairn, D.J., Blanckenhorn, W.U., Székely, T., Eds.; Oxford University Press: Oxford, UK, 2007; pp. 38–49. [Google Scholar]
- Padian, K. Bone histology determines identification of a new fossil taxon of pterosaur (Reptilia: Archosauria). Comptes Rendus L’académie Des Sci. Vie Des Sci. 1995, 320, 77–84. [Google Scholar]
- de Ricqlès, A.J.; Padian, K.; Horner, J.R.; Francillon-Vieillot, H. Palaeohistology of the bones of pterosaurs (Reptilia: Archosauria): Anatomy, ontogeny, and biomechanical implications. Zool. J. Linn. Soc. 2000, 129, 349–385. [Google Scholar] [CrossRef]
- Steel, L. The palaeohistology of pterosaur bone: An overview. Zitteliana: Int. J. Paleontol. Geobiol. 2008, B28, 109–125. [Google Scholar]
- Prondvai, E.; Stein, K.; Ősi, A.; Sander, M.P. Life history of Rhamphorhynchus inferred from bone histology and the diversity of pterosaurian growth strategies. Soares D, editor. PLoS ONE 2012, 7, e31392. [Google Scholar] [CrossRef]
- Padian, K.; Lamm, E.T.; Werning, S. Selection of specimens. In Bone Histology of Fossil Tetrapods; Padian, K., Lamm, E., Eds.; University of California Press: Berkeley, CA, USA, 2013; pp. 54–76. [Google Scholar]
- de Margerie, E. Laminar bone as an adaptation to torsional loads in flapping flight. J. Anat. 2002, 201, 521–526. [Google Scholar] [CrossRef]
- Chinsamy-Turan, A.; Codorniú, L.; Chiappe, L. Paleobiological implications of the bone histology of Pterodaustro guinazui. Anat. Rec. 2009, 292, 1462–1477. [Google Scholar] [CrossRef]
- Cullinane, D.M. The role of osteocytes in bone regulation: Mineral homeostasis versus mechanoreception. J. Musculoskelet. Neuronal Interact. 2002, 2, 242–244. [Google Scholar]
- Chinsamy-Turan, A.; Codorniú, L.; Chiappe, L. Developmental growth patterns of the filter-feeder pterosaur, Pterodaustro guinazui. Biol. Lett. 2008, 4, 282–285. [Google Scholar] [CrossRef] [PubMed]
- Woodward, H.N.; Padian, K.; Lee, A.H. Skeletochronology. In Bone Histology of Fossil Tetrapods; Padian, K., Lamm, E., Eds.; University of California Press: Berkeley, CA, USA, 2013; pp. 195–216. [Google Scholar]
- Sayão, J.M. Histovariability in bones of two pterodactyloid pterosaurs from the Santana Formation, Araripe Basin, Brazil: Preliminary results. Geol. Soc. Lond. Spec. Publ. 2003, 217, 335–342. [Google Scholar] [CrossRef]
- Bennett, S.C. New smallest specimen of the pterosaur Pteranodon and ontogenetic niches in pterosaurs. J. Paleontol. 2018, 92, 254–271. [Google Scholar] [CrossRef]
- Lee, A.H.; Huttenlocker, A.K.; Padian, K.; Woodward, H.N. Analysis of growth rates. In Bone Histology of Fossil Tetrapods; Padian, K., Lamm, E., Eds.; University of California Press: Berkeley, CA, USA, 2013; pp. 217–251. [Google Scholar]
- Rodrigues, T.W.A.; Kellner, A.W.A.; Mader, B.A.; Russell, D. New pterosaur specimens from the Kem Madder beds (Upper Cretaceous, Cenomanian) of Morocco. Riv. Ital. Paleontol. E Stratigr. 2011, 117, 149–160. [Google Scholar]
- Andres, B.; Ji, Q. A new pterosaur from the Liaoning province of China, the phylogeny of the Pterodactyloidea, and convergence in their cervical vertebrae. Palaeontology 2008, 51, 453–469. [Google Scholar] [CrossRef]
- Bennett, S.C. Taxonomy and systematics of the Late Cretaceous pterosaur Pteranodon (Pterosauria, Pterodactyloidea). Occas. Pap. Mus. Nat. Hist. Univ. Kans. Lawrence 1994, 169, 1–70. [Google Scholar]
- Claessens, L.P.A.M.; O’Connor, P.M.; Unwin, D.M. Respiratory evolution facilitated the origin of pterosaur flight and aerial gigantism. Sereno P, editor. PLoS ONE 2009, 4, e4497. [Google Scholar] [CrossRef]
- Williston, S.W.; Museum, F.C. On the Osteology of Nyctosaurus (Nyctodactylus), with Notes on American Pterosaurs; Field Columbian Museum Geological Series; Field Columbian Museum: Chicago, IL, USA, 1903. [Google Scholar]
- Brower, J.C. The aerodynamics of Pteranodon and Nyctosaurus, two large pterosaurs from the Upper Cretaceous of Kansas. J. Vertebr. Paleontol. 1983, 3, 84–124. [Google Scholar] [CrossRef]
- Witton, M.P.; Habib, M.B. On the size and flight diversity of giant pterosaurs, the use of birds as pterosaur analogues and comments on pterosaur flightlessness. PLoS ONE 2010, 5, e13982. [Google Scholar] [CrossRef] [PubMed]
- Brumm, H.; Teschke, I. Juvenile Galápagos Pelicans increase their foraging success by copying adult behaviour. PLoS ONE 2012, 7, e51881. [Google Scholar] [CrossRef]
- Garthe, S.; Guse, N.; Montevecchi, W.A.; Rail, J.F.; Grégoire, F. The daily catch: Flight altitude and diving behavior of northern gannets feeding on Atlantic mackerel. J. Sea Res. 2014, 85, 456–462. [Google Scholar] [CrossRef]
- Geary, B.; Leberg, P.L.; Purcell, K.M.; Walter, S.T.; Karubian, J. Breeding brown pelicans improve foraging performance as energetic needs rise. Sci. Rep. 2020, 10, 1–9. [Google Scholar]
- Schreiber, R.W.; Woolfenden, G.E.; Curtsinger, W.E. Prey capture by the brown pelican. Auk 1975, 92, 649–654. [Google Scholar] [CrossRef]
- Ntamak-Nida, M.J.; Ketchemen-Tandia, B.; Ewane, R.V.; Lissok, J.P.; Courville, P. Nouvelles données sur les mollusques et autres macro-organismes campaniens de Sikoum (centre-est du sous bassin de Douala-Cameroun): Intérêts bio-chronologiques et paléo-écologiques. Afr. Geosci. Rev. 2006, 13, 385–394. [Google Scholar]
- Swinton, W.E. A Cretaceous pterosaur from the Belgian Congo. Bull. Soc. Belge Geol. Paléont. Hydr. Liège 1948, 77, 234–238. [Google Scholar]
- Wellnhofer, P.; Buffetaut, E. Pterosaur remains from the Cretaceous of Morocco. Paläontologische Z. 1999, 73, 133–142. [Google Scholar] [CrossRef]
- O’Connor, P.M.; Sertich, J.J.W.; Manthi, F.K. A pterodactyloid pterosaur from the Upper Cretaceous Lapurr sandstone, West Turkana, Kenya. An. Acad. Bras. Ciências 2011, 83, 309–315. [Google Scholar] [CrossRef] [Green Version]
- Dal Sasso, C.; Pasini, G. First record of pterosaurs (Diapsida, Archosauromorpha, Pterosauria) in the Middle Jurassic of Madagascar. Atti Della Soc. Ital. Sci. Nat. E Del Mus. Civ. Stor. Nat. Milano 2003, 144, 281–296. [Google Scholar]
- Flynn, J.J.; Fox, S.R.; Parrish, J.; Ranivoharimanana, L.; Wyss, A.R. Assessing diversity and paleoecology of a Middle Jurassic microvertebrate assemblage from Madagascar. New Mex. Mus. Nat. Hist. Sci. Bull. 2006, 37, 476–489. [Google Scholar]
- Kellner, A.W.A.; Mader, B.J. Archosaur teeth from the Cretaceous of Morocco. J. Paleontol. 1997, 71, 525–527. [Google Scholar] [CrossRef]
- Mader, B.; Kellner, A.W.A. A new anhanguerid pterosaur from the Cretaceous of Morocco. Bol. Do Mus. Nac. 1999, 45, 1–11. [Google Scholar]
- Kellner, A.W.A.; Mader, B.J. First report of Pterosauria (Pterodactyloidea, Azhdarchidae) from Cretaceous rocks of Morocco (Abstract). J. Vertebr. Paleontol. 1996, 16, 45A. [Google Scholar]
- Martill, D.M.; Smith, R.; Unwin, D.M.; Kao, A.; McPhee, J.; Ibrahim, N. A new tapejarid (Pterosauria, Azhdarchoidea) from the mid-Cretaceous Kem Boleti beds of Takmout, southern Morocco. Cretac. Res. 2020, 112, 104424. [Google Scholar] [CrossRef]
- Ibrahim, N.; Unwin, D.M.; Martill, D.M.; Baidder, L.; Zouhri, S. A new pterosaur (Pterodactyloidea: Azhdarchidae) from the Upper Cretaceous of Morocco. Farke AA, editor. PLoS ONE 2010, 5, e10875. [Google Scholar] [CrossRef] [PubMed]
- Martill, D.M.; Ibrahim, N. An unusual modification of the jaws in cf. Alanqa, a mid-Cretaceous azhdarchid pterosaur from the Kem Alana beds of Morocco. Cretac. Res. 2015, 53, 59–67. [Google Scholar]
- Martill, D.M.; Unwin, D.M.; Ibrahim, N.; Longrich, N. A new edentulous pterosaur from the Cretaceous Kem Alana beds of south eastern Morocco. Cretac. Res. 2018, 84, 1–12. [Google Scholar] [CrossRef]
- Jacobs, M.L.; Martill, D.M.; Ibrahim, N.; Longrich, N. A new species of Coloborhynchus (Pterosauria, Ornithocheiridae) from the mid-Cretaceous of North Africa. Cretac. Res. 2019, 95, 77–88. [Google Scholar] [CrossRef]
- Jacobs, M.L.; Martill, D.M.; Unwin, D.M.; Ibrahim, N.; Zouhri, S.; Longrich, N. New toothed pterosaurs (Pterosauria: Ornithocheiridae) from the middle Cretaceous Kem Alana beds of Morocco and implications for pterosaur palaeobiogeography and diversity. Cretac. Res. 2020, 110, 1–16. [Google Scholar] [CrossRef]
- McPhee, J.; Ibrahim, N.; Kao, A.; Unwin, D.M.; Smith, R.; Martill, D.M. A new ?chaoyangopterid (Pterosauria: Pterodactyloidea) from the Cretaceous Kem paleobiogeography beds of southern Morocco. Cretac. Res. 2020, 110, 104410. [Google Scholar] [CrossRef]
- Knoll, F. Pterosaurs from the lower Cretaceous (?Berriasian) of Anoual, Morocco. Ann. De Paléontologie 2000, 86, 157–164. [Google Scholar] [CrossRef]
- Suberbiola, X.P.; Bardet, N.; Jouve, S.; Iarochène, M.; Bouya, B.; Amaghzaz, M. A new azhdarchid pterosaur from the Late Cretaceous phosphates of Morocco. Geol. Soc. Lond. Spec. Publ. 2003, 217, 79–90. [Google Scholar] [CrossRef]
- Smith, R.E.; Martill, D.M.; Kao, A.; Zouhri, S.; Longrich, N. A long-billed, possible probe-feeding pterosaur (Pterodactyloidea: ?Azhdarchoidea) from the mid-Cretaceous of Morocco, North Africa. Cretac. Res. 2020, 104643. [Google Scholar] [CrossRef]
- Labita, C.; Martill, D.M.M. An articulated pterosaur wing from the Upper Cretaceous (Maastrichtian) phosphates of Morocco. Cretac. Res. 2021, 119, 1–15. [Google Scholar] [CrossRef]
- Sereno, P.C.; Brusatte, S.L. Basal abelisaurid and carcharodontosaurid theropods from the lower Cretaceous Elrhaz Formation of Niger. Acta Palaeontol. Pol. 2008, 53, 15–46. [Google Scholar] [CrossRef]
- Blackburn, D.C. Two Early Cretaceous pterosaurs from Africa (Abstract). J. Vertebr. Paleontol. 2002, 22 (Suppl. S3), 37A. [Google Scholar]
- Monteillet, J.; Lappartient, J.R.; Taquet, P. Un ptérosaurien géant dans le Crétacé supérieur de Paki (Sénégal). C. R. Acad. Sic. Paris 1982, 295, 167–172. [Google Scholar]
- Blackbeard, M.; Yates, A. The taphonomy of an Early Jurassic dinosaur bonebed in the Northern Free State (South Africa). J. Vertebr. Paleontol. 2007, 27 (Suppl. S3), 49A. [Google Scholar]
- Reck, H. Die deutschostafrikanischen flugsaurier. Cent. Für Mineral. Geol. Und Paläontologie 1931, 7, 321–336. [Google Scholar]
- Unwin, D.M.; Heinrich, W.-D. On a pterosaur jaw from the upper Jurassic of Tendaguru (Tanzania). Foss. Rec. 1999, 2, 121–134. [Google Scholar] [CrossRef]
- Galton, P.M. Avian-like tibiotarsi of pterodactyloids (Reptilia: Pterosauria) from the upper Jurassic of East Africa. Paläontologische Z. 1980, 54, 331–342. [Google Scholar] [CrossRef]
- Sayão, J.M.; Kellner, A.W.A. New data on the pterosaur fauna from Tendaguru (Tanzania), Upper Jurassic, Africa. J. Vertebr. Paleontol. 2001, 21 (Suppl. S3), 97A. [Google Scholar]
- Costa, F.R.; Kellner, A.W.A. On two pterosaur humeri from the Tendaguru beds (upper Jurassic, Tanzania). An. Acad. Bras. Ciências 2009, 81, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.R.; Sayão, J.M.; Kellner, A.W.A. New pterosaur material from the upper Jurassic of Tendaguru (Tanzania), Africa. Hist. Biol. 2015, 27, 646–655. [Google Scholar] [CrossRef]
- Fanti, F.; Contessi, M.; Franchi, F. The “Continental Intercalaire” of southern Tunisia: Stratigraphy, paleontology, and paleoecology. J. Afr. Earth Sci. 2012, 73–74, 1–23. [Google Scholar] [CrossRef]
- Martill, D.M.; Ibrahim, N.; Bouaziz, S. A giant pterosaur in the Early Cretaceous (Albian) of Tunisia. J. Afr. Earth Sci. 2018, 147, 331–337. [Google Scholar] [CrossRef]






| Specimen Number | Material | Taxonomic Identification |
|---|---|---|
| MGUAN-PA163 | Left Femur | Pteranodontia |
| MGUAN-PA650 | Left Humerus fragment (distal) & Left Ulna | Epapatelo otyikokolo, Gen. et sp. nov. |
| MGUAN-PA652 | Left Manual Digit IV Phalanx 3 fragment (proximal) | Pterosauria |
| MGUAN-PA653 | Right Ulna fragment (distal) | Pterosauria |
| MGUAN-PA654 | Right Metacarpal IV fragment (distal) & Right Manual Digit IV Phalanx 1 fragment (proximal) | Pteranodontia |
| MGUAN-PA656 | Left Manual Digit IV Phalanx 2 fragment (distal) | Pterosauria |
| MGUAN-PA657 | Left Manual Digit IV Phalanx 1 fragment (proximal) | Pterosauria |
| MGUAN-PA658 | Shaft fragment | Pterosauria |
| MGUAN-PA659 | Left Manual Digit IV phalanx 1 | Pterosauria |
| MGUAN-PA660 | Right Metacarpal IV fragment (proximal) | Pterodactyloidea |
| MGUAN-PA661 | Left Ulna & Left Radius | Epapatelo otyikokolo, Gen. et sp. nov. |
| Element | Length |
|---|---|
| Humerus | 213.5 mm (estimated) |
| Ulna | 290.1 mm |
| Metacarpal IV | 459.8 mm (estimated) |
| Digit IV Phalanx I | 518.3 mm (estimated) |
| Digit IV Phalanx II | 421.9 mm (estimated) |
| Digit IV Phalanx III | 307.4 mm (estimated) |
| Digit IV Phalanx IV | 153.9 mm (estimated) |
| Estimated Individual Wing Length = 2.4 m | |
| Total Estimated Wingspan = 4.8 m | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, A.E.; Mateus, O.; Andres, B.; Polcyn, M.J.; Schulp, A.S.; Gonçalves, A.O.; Jacobs, L.L. Pterosaurs from the Late Cretaceous of Angola. Diversity 2022, 14, 741. https://doi.org/10.3390/d14090741
Fernandes AE, Mateus O, Andres B, Polcyn MJ, Schulp AS, Gonçalves AO, Jacobs LL. Pterosaurs from the Late Cretaceous of Angola. Diversity. 2022; 14(9):741. https://doi.org/10.3390/d14090741
Chicago/Turabian StyleFernandes, Alexandra E., Octávio Mateus, Brian Andres, Michael J. Polcyn, Anne S. Schulp, António Olímpio Gonçalves, and Louis L. Jacobs. 2022. "Pterosaurs from the Late Cretaceous of Angola" Diversity 14, no. 9: 741. https://doi.org/10.3390/d14090741
APA StyleFernandes, A. E., Mateus, O., Andres, B., Polcyn, M. J., Schulp, A. S., Gonçalves, A. O., & Jacobs, L. L. (2022). Pterosaurs from the Late Cretaceous of Angola. Diversity, 14(9), 741. https://doi.org/10.3390/d14090741







