Rhizomaticola guizhouensis gen. et sp. nov. and Five Rosellinia Like Species Isolated from Decaying Wood
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection and Isolation
2.2. DNA Extraction, Polymerase Chain Reaction (PCR) Amplification and Sequencing
2.3. Sequence Alignment and Phylogenetic Analyses
| Species | Strains | GenBank Accession Number | References | |||
|---|---|---|---|---|---|---|
| ITS | LSU | rpb2 | tub2 | |||
| Amphirosellinia fushanensis | HAST 91111209HT | NR_153514 | N/A | GQ848339 | GQ495950 | [3] |
| A. nigrospora | HAST 91092308HT | NR_153513 | N/A | GQ848340 | GQ495951 | [3] |
| Annulohypoxylon annulatum | CBS 140775ET | NR_153579 | KY610418 | KY624263 | N/A | [49] |
| A. truncatum | CBS 140778ET | NR_153580 | KY610419 | KY624277 | N/A | [49] |
| Anthostomella thailandica | MFLUCC 15-0017HT | NR_153556 | KX533448 | KX599538 | KX600496 | [50] |
| Astrocystis bambusae | HAST 89021904 | GU322449 | N/A | GQ844836 | GQ495942 | [3] |
| A. mirabilis | HAST 94070803 | GU322448 | N/A | GQ844835 | GQ495941 | [3] |
| Barrmaelia rappazii | Cr2 = CBS 142771HT | MF488989 | MF488989 | MF488998 | MF489017 | [51] |
| B. rhamnicola | BR = CBS 142772ET | MF488990 | MF488990 | MF488999 | MF489018 | [51] |
| Biscogniauxia arima | 122 WSPIT | EF026150 | N/A | GQ304736 | AY951672 | [3] |
| Brunneiperidium gracilentum | MFLUCC:14-0011HT | KP297400 | KP340549 | KP340529 | KP406611 | [52] |
| Cainia anthoxanthis | MFLUCC 15-0539HT | NR_138407 | NG_070382 | N/A | N/A | [53] |
| C. globosa | MFLUCC 13-0663HT | NR_171724 | KX822123 | N/A | N/A | [50] |
| Camillea tinctor | YMJ 363 | JX507806 | N/A | JX507790 | JX507795 | [54] |
| Clypeosphaeria mamillana | WU 33598ET | NR_153909 | NG_067338 | MF489001 | MH704637 | [55] |
| Collodiscula japonica | CJ = CBS:124266 | JF440974 | MH874889 | KY624273 | KY624316 | [56] |
| C. leigongshanensis | GZ70 = GZUH0107HT | KP054281 | KP054282 | KR002588 | KR002587 | [29] |
| Coniocessia cruciformis | IRAN 1475CHT | NR_145220 | GU553347 | N/A | N/A | [57] |
| C. nodulisporioides | CBS 125778 | MH863756 | MH875224 | N/A | N/A | [57] |
| Coniolariella gamsii | IRAN 2506C | KY052004 | KY052005 | N/A | N/A | (submitted directly) |
| C. hispanica | CBS 124506T | MH863381 | MH874902 | N/A | N/A | [57] |
| Creosphaeria sassafras | CBS 127876 | MH864737 | MH876173 | N/A | N/A | [57] |
| Dematophora pepo | CBS:123592 | MN984620 | N/A | N/A | MN987246 | [32] |
| Diabolocovidia claustri | CPC37593HT | NR_170827 | NG_074445 | N/A | N/A | [58] |
| Diatrype lijiangensis | MFLU 19-0717HT | NR_165229 | MK810546 | N/A | MK852583 | [59] |
| Diatrypella heveae | MFLU:17-1216HT | MF959501 | NG_069531 | N/A | MG334557 | [60] |
| D. vulgaris | CBS 128327T | NR_159873 | NG_069986 | N/A | N/A | [57] |
| Durotheca comedens | YMJ 90071615 | EF026128 | N/A | JX507793 | EF025613 | [38] |
| D. guizhouensis | GMBC0065HT | MH645423 | MH645421 | MH645422 | MH645420 | [38] |
| Emarcea eucalyptigena | CBS 139908HT | MK762711 | NG_066346 | MK791286 | N/A | [61] |
| Engleromyces sinensis | BJTC 200803 | MZ622705 | MZ622702 | N/A | N/A | (submitted directly) |
| Entalbostroma erumpens | ICMP:21152HT | NR_154013 | N/A | KX258204 | KX258205 | [62] |
| Entoleuca mammata | JDR 100 | GU300072 | N/A | GQ844782 | GQ470230 | [3] |
| Eutypa lata | CBS 208.87NT | MH862066 | MH873755 | KF453595 | DQ006969 | [57] |
| Eutypella citricola | CBS 128332 | MH864883 | MH876331 | N/A | N/A | [57] |
| Furfurella nigrescens | CBS:143622HT | MK527844 | MK527844 | MK523275 | MK523333 | [51] |
| F. stromatica | CBS 144409HT | MK527846 | MK527846 | MK523277 | MK523334 | [51] |
| Graphostroma platystomum | CPC:37153 | MT223799 | MT223894 | MT223680 | MT223734 | [63] |
| Halorosellinia oceanica | BCC < THA > :60405 | MK606079 | MK629003 | N/A | N/A | (submitted directly) |
| H. xylocarpi | MFLU 18-0545HT | NR_166290 | NG_068301 | N/A | MN077076 | [40] |
| Hansfordia pulvinata | CBS 194.56 | KU683763 | MH869122 | KU684307 | N/A | [12] |
| Hansfordia pulvinata | CBS:144422 | MK442587 | MK442527 | N/A | N/A | [64] |
| Helicogermslita clypeata | MFLU 18-0852HT | NR_175685 | NG_081506 | MW658647 | MW775614 | [65] |
| Hypocopra rostrata | NRRL 66178 | KM067909 | KM067909 | N/A | N/A | (submitted directly) |
| Hypocreodendron sanguineum | 169 (JDR) | GU322433 | N/A | GQ844819 | GQ487710 | [3] |
| Hypoxylon rickii | MUCL 53309ET | NR_137115 | KY610416 | KY624281 | KC977288 | [49] |
| Idriella lunata | CBS:204.56T | MH857584 | MH869129 | N/A | N/A | [57] |
| Induratia thailandica | MFLU 18-0784HT | MK762707 | MK762714 | MK791283 | N/A | [61] |
| Jackrogersella multiformis | CBS 119016ET | NR_154784 | KY610473 | KY624290 | KX271262 | [49] |
| Kretzschmaria clavus | JDR 114 | EF026126 | N/A | GQ844789 | EF025611 | [3] |
| Kretzschmaria lucidula | JDR 112 | EF026125 | N/A | GQ844790 | EF025610 | [3] |
| Kretzschmariella culmorum | JDR 88 | KX430043 | N/A | KX430045 | KX430046 | (submitted directly) |
| Lopadostoma turgidum | CBS 133207ET | NR_132036 | KC774618 | KC774563 | MF489024 | [66] |
| Lunatiannulus irregularis | MFLUCC:14-0014HT | KP297398 | KP340540 | KP340526 | KP406609 | [52] |
| Microdochium phragmitis | CBS:285.71ET | MH860125 | KP858949 | KP859122 | MH704636 | [67] |
| Nemania bipapillata | HAST 90080610 | GU292818 | N/A | GQ844771 | GQ470221 | [3] |
| N. serpens | HAST 235 | GU292820 | N/A | GQ844773 | GQ470223 | [3] |
| N. sphaeriostoma | JDR 261 | GU292821 | N/A | GQ844774 | GQ470224 | [3] |
| Neoxylaria arengae | MFLUCC 15-0292HT | NR_171264 | N/A | MT502418 | N/A | [68] |
| Nigropunctata nigrocircularis | MFLU 19-2130HT | NR_175683 | NG_081504 | N/A | MW775612 | [65] |
| N. bambusicola | MFLU 19-2145HT | NR_175684 | NG_081505 | MW658646 | N/A | [65] |
| Podosordaria mexicana | 176 WSP | GU324762 | N/A | GQ853039 | GQ844840 | [3] |
| Podosordaria muli | 167 WSPHT | GU324761 | N/A | GQ853038 | GQ844839 | [3] |
| Poronia pileiformis | WSP 88113001ET | NR_158882 | N/A | GQ853037 | GQ502720 | [3] |
| Requienella fraxini | CBS 140475HT | NR_138415 | MH878686 | N/A | N/A | [57] |
| R. seminuda | CBS 140502ET | NR_154630 | MH878683 | MK523300 | N/A | [57] |
| Rhizomaticola guizhouensis | FJS12 = GZUH0101HT | ON815473 | 0N815474 | ON897692 | ON924997 | This study |
| Rh. guizhouensis | 2022FJS24 = GZUH0335 | OP177724 | OP177725 | OP184058 | OP184057 | This study |
| Rosellinia aquila | MUCL 51703 | KY610392 | KY610460 | KY624285 | KX271253 | [49] |
| Ro. corticium | STMA 13324 | MN984621 | MN984627 | MN987237 | MN987241 | [32] |
| Sarcoxylon compunctum | CBS:359.61 | KT281903 | KY610462 | KY624230 | KX271255 | [49] |
| Stilbohypoxylon elaeicola | 94082615 (HAST) | GU322440 | N/A | GQ844827 | GQ495933 | [68] |
| Stilbohypoxylon elaeidis | MFLUCC 15-0295aHT | MT496745 | NG_074460 | MT502416 | MT502420 | [68] |
| Stilbohypoxylon quisquiliarum | JDR 172 | EF026119 | N/A | GQ853020 | EF025605 | [3] |
| Vamsapriya bambusicola | MFLUCC11-0477 HT | KM462835 | NG_067527 | KM462834 | KM462833 | [58] |
| V. indica | MFLUCC 12-0544 | KM462839 | KM462840 | KM462841 | KM462838 | [58] |
| Wawelia regia | CBS:110.10 | MH854595 | MH866123 | N/A | N/A | [57] |
| Xylaria bambusicola | WSP 205HT | EF026123 | N/A | GQ844801 | AY951762 | [3] |
| X. feejeensis | HAST 92092013 | GU322454 | N/A | GQ848336 | GQ495947 | [3] |
| X. hypoxylon | HAST 95082001 | GU300095 | N/A | GQ844811 | GQ487703 | [3] |
| Zygosporium pseudomasonii | CBS 146059HT | MN562147 | MN567654 | MN556815 | N/A | (submitted directly) |
3. Results
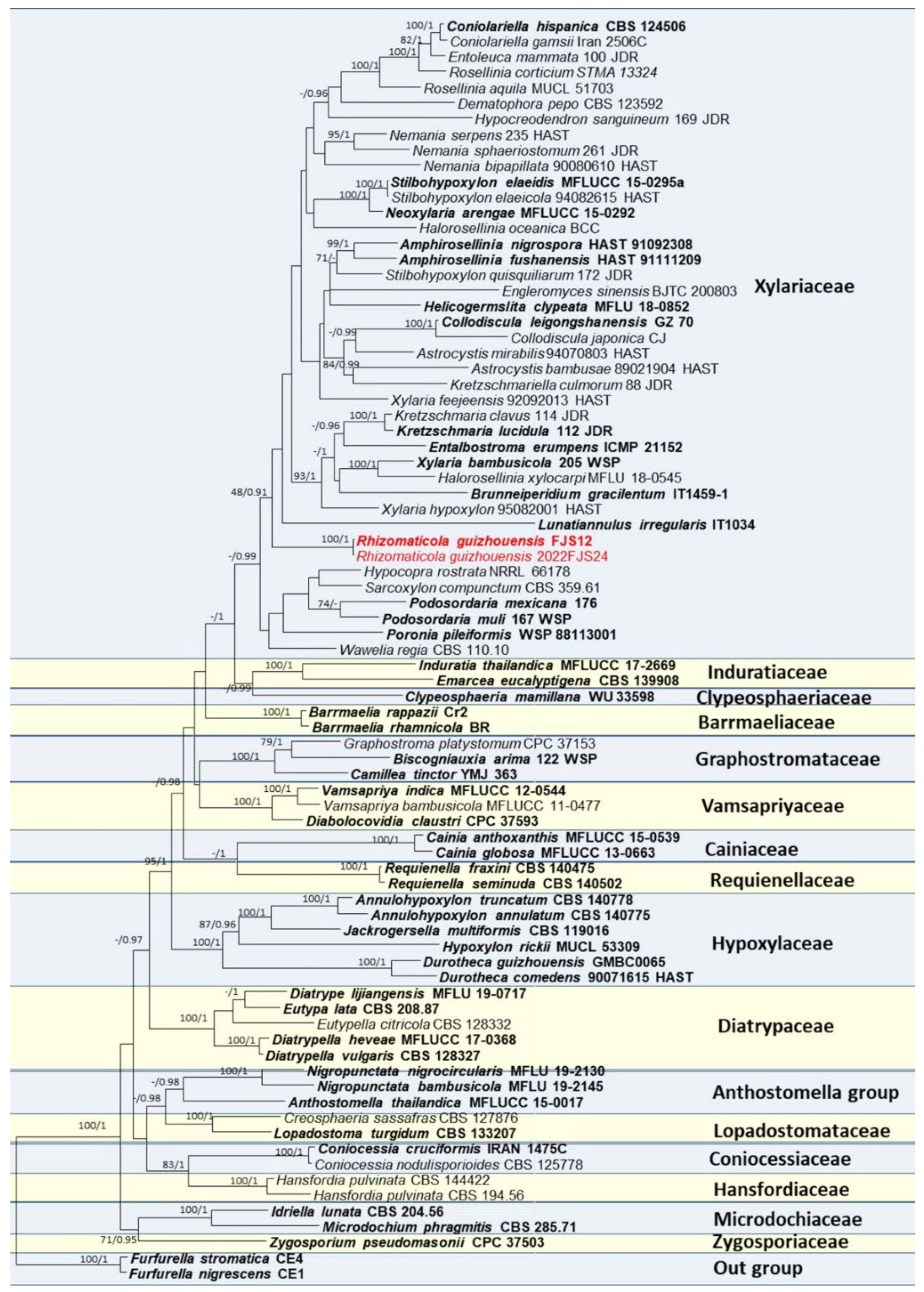
3.1. Phylogenetic Analyses
3.2. Taxonomy
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Peláez, F.; González, V.; Platas, G.; Sánchez-Ballesteros, J.; Rubio, V. Molecular phylogenetic studies within the Xylariaceae based on ribosomal DNA sequences. Fungal Divers. 2008, 31, 111–134. [Google Scholar]
- Tang, A.; Jeewon, R.; Hyde, K.D. A re-evaluation of the evolutionary relationships within the Xylariaceae based on ribosomal and protein-coding gene sequences. Fungal Divers. 2009, 34, 127–155. [Google Scholar]
- Hsieh, H.-M.; Lin, C.-R.; Fang, M.-J.; Rogers, J.D.; Fournier, J.; Lechat, C.; Ju, Y.M. Phylogenetic status of Xylaria subgenus Pseudoxylaria among taxa of the subfamily Xylarioideae (Xylariaceae) and phylogeny of the taxa involved in the subfamily. Mol. Phylogenet. Evol. 2010, 54, 957–969. [Google Scholar] [CrossRef] [PubMed]
- Fournier, J.; Lechat, C.; Courtecuisse, R. The genus Xylaria sensu lato (Xylariaceae) in Guadeloupe and Martinique (French West Indies) II. Taxa with robust upright stromata. Ascomycete.org 2019, 11, 77–115. [Google Scholar]
- Fournier, J.; Lechat, C.; Courtecuisse, R. The genus Xylaria sensu lato (Xylariaceae) in Guadeloupe and Martinique (French West Indies) III. Taxa with slender upright stromata. Ascomycete.org 2020, 12, 81–164. [Google Scholar]
- Carmona, A.; Fournier, J.; Williams, C.; Piepenbring, M. New records of Xylariaceae from Panama. N. Am. Fungi 2009, 4, 1–11. [Google Scholar] [CrossRef]
- Stadler, M.; Læssøe, T.; Fournier, J.; Decock, C.; Schmieschek, B.; Tichy, H.V.; Peršoh, D. A polyphasic taxonomy of Daldinia (Xylariaceae). Stud. Mycol. 2014, 77, 1–143. [Google Scholar] [CrossRef]
- Edwards, R.L.; Jonglaekha, N.; Kshirsagar, A.; Maitland, D.J.; Mekkamol, S.; Nugent, L.K.; Phosri, C.; Rodtong, S.; Ruchikachorn, N.; Sangvichien, E.; et al. The Xylariaceae as phytopathogens. Recent Res. Dev. Plant Sci. 2003, 1, 1–19. [Google Scholar]
- Visser, A.A.; Ros, V.I.D.; de Beer, Z.W.; Debets, A.J.M.; Hartog, E.; Kuyper, T.W.; Læssøe, T.; Slippers, B.; Aanen, D.K. Levels of specificity of Xylaria species associated with fungus-growing termites: A phylogenetic approach. Mol. Ecol. 2009, 18, 553–567. [Google Scholar] [CrossRef]
- Pažoutová, S.; Šrůtka, P.; Holuša, J.; Chudíčková, M.; Kolařík, M. Diversity of xylariaceous symbionts in Xiphydria woodwasps: Role of vector and a host tree. Fungal Ecol. 2010, 3, 392–401. [Google Scholar] [CrossRef]
- Okane, I.; Toyama, K.; Nakagiri, A.; Suzuki, K.I.; Srikitikulchai, P.; Sivichai, S.; Hywel-Jones, N.; Potacharoen, W.; Læssøe, T. Study of endophytic Xylariaceae in Thailand: Diversity and taxonomy inferred from rDNA sequence analyses with saprobes forming fruit bodies in the field. Mycoscience 2008, 49, 359–372. [Google Scholar] [CrossRef]
- U’Ren, J.M.; Miadlikowska, J.; Zimmerman, N.B.; Lutzoni, F.; Stajich, J.E.; Arnold, A.E. Contributions of North American endophytes to the phylogeny, ecology, and taxonomy of Xylariaceae (Sordariomycetes, Ascomycota). Mol. Phylogenet. Evol. 2016, 98, 210–232. [Google Scholar] [CrossRef]
- Stadler, M. Importance of secondary metabolites in the Xylariaceae as parameters for assessment of their taxonomy, phylogeny, and functional biodiversity. Curr. Res. Environ. Appl. Mycol. 2011, 1, 75–133. [Google Scholar] [CrossRef]
- Surup, F.; Wiebach, V.; Kuhnert, E.; Stadler, M. Truncaquinones A and B, asterriquinones from Annulohypoxylon truncatum. Tetrahedron Lett. 2016, 47, 2183–2185. [Google Scholar] [CrossRef]
- Wijayawardene, N.N.; Hyde, K.D.; Al-Ani, L.K.T.; Tedersoo, L.; Haelewaters, D.; Rajeshkumar, K.C.; Zhao, R.L.; Aptroot, A.; Leontyev, D.V.; Saxena, R.K.; et al. Outline of Fungi and fungi-like taxa. Mycosphere 2020, 11, 1060–1456. [Google Scholar] [CrossRef]
- Petrini, L.E. Rosellinia—A World Monograph; Gebrüder Borntraeger Verlagsbuchhandlung: Stuttgart, Germany, 2013; pp. 1–410. [Google Scholar]
- Teng, S.C. Fungi of China; Science Press: Beijing, China, 1963; pp. 1–808. [Google Scholar]
- Tai, F.L. Sylloge Fungorum Sinicorum; Science Press: Beijing, China, 1979; pp. 1–1527. [Google Scholar]
- Ju, Y.M.; Rogers, J.D. Astrocystis reconsidered. Mycologia 1990, 82, 342–349. [Google Scholar] [CrossRef]
- Ju, Y.M.; Rogers, J.D. The Xylariaceae of Taiwan (excluding Anthostomella). Mycotaxon 1999, 73, 343–440. [Google Scholar]
- Yuan, Z.Q.; Zhao, Z.Y. Studies on the genera Amphisphaerella, Coniochaeta and Rosellinia of XingJiang, China. Acta Mycol. Sin. 1993, 12, 180–186. [Google Scholar]
- Lu, B.S.; Hyde, K.D.; Ho, W.H.; Tsui, K.M.; Taylor, J.E.; Wong, K.M.; Zhou, D.Y. Checklist of Hong Kong Fungi; Fungal Diversity Press: Hongkong, China, 2000; pp. 1–207. [Google Scholar]
- Liu, C.F.; Lu, T.; Gao, J.M.; Wang, M.Q.; Lu, B.S. Two new Chinese records of Rosellinia. Mycosystema 2010, 29, 459–462. [Google Scholar]
- Petrini, L.E. Nomenclatural novelties [Rosellinia]. Index Fungorum. 2013, 25, 1–6. [Google Scholar]
- Li, W.; Guo, L. Rosellinia brunneola sp. nov. and R. beccariana new to China. Mycotaxon 2015, 130, 233–236. [Google Scholar] [CrossRef]
- Li, W.; Guo, L. Rosellinia hainanensis sp. nov. and three Rosellinia species new to China. Mycotaxon 2016, 131, 541–545. [Google Scholar] [CrossRef]
- Li, W.; Guo, L. Rosellinia jiangxiensis and R. yunnanensis spp. nov. and a new Rosellinia record from China. Mycotaxon 2018, 133, 31–35. [Google Scholar] [CrossRef]
- Li, W.; Guo, L. Rosellinia angusta and R. menglana spp. nov. and two new Rosellinia records from China. Mycotaxon 2018, 133, 591–595. [Google Scholar] [CrossRef]
- Li, Q.R.; Kang, J.C.; Hyde, K.D. Two new species of the genus Collodiscula (Xylariaceae) from China. Mycol. Prog. 2015, 205, 187–196. [Google Scholar] [CrossRef]
- Su, H.; Li, Q.R.; Kang, J.C.; Wen, T.C.; Hyde, K.D. Rosellinia convexa, sp. nov. (Xylariales, Pezizomycotina) from China. Mycoscience 2016, 57, 164–170. [Google Scholar] [CrossRef]
- Xie, X.; Liu, L.; Zhang, X.; Long, Q.D.; Sheng, X.C.; Boonmee, S.; Kang, J.C.; Li, Q.R. Contributions to species of Xylariales in China—2. Rosellinia pervariabilis and R. tetrastigmae spp. nov. and a new record of R. caudata. Mycotaxon 2019, 134, 183–196. [Google Scholar] [CrossRef]
- Wittstein, K.; Cordsmeier, A.; Lambert1, C.; Wendt, L.; Sir, E.B.; Weber, J.; Wurzler, N.; Petrini, L.E.; Stadler, M. Identification of Rosellinia species as producers of cyclodepsipeptide PF1022 A and resurrection of the genus Dematophora as inferred from polythetic taxonomy. Stud. Mycol. 2020, 96, 1–16. [Google Scholar] [CrossRef]
- Chomnunti, P.; Hongsanan, S.; Aguirre-Hudson, B.; Tian, Q.; Peršoh, D.; Dhami, M.K.; Alisa, A.S.; Xu, J.C.; Liu, X.Z.; Stadler, M.; et al. The sooty moulds. Fungal Divers. 2014, 66, 1–36. [Google Scholar] [CrossRef]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes–application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.J.W.T.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Liu, Y.J.; Whelen, S.; Hall, B.D. Phylogenetic relationships among ascomycetes: Evidence from an RNA polymerase II subunit. Mol. Biol. Evol. 1999, 16, 1799–1808. [Google Scholar] [CrossRef]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Long, Q.D.; Liu, L.L.; Zhang, X.; Wen, T.C.; Kang, J.C.; Hyde, K.D.; Shen, X.C.; Li, Q.R. Contributions to species of Xylariales in China-1. Durotheca species. Mycol. Prog. 2019, 18, 495. [Google Scholar] [CrossRef]
- Pi, Y.H.; Zhang, X.; Liu, L.L.; Long, Q.D.; Shen, X.C.; Kang, Y.Q.; Hyde, K.D.; Boonmee, S.; Kang, J.C.; Li, Q.R. Contributions to species of Xylariales in China–4. Hypoxylon wujiangensis sp. nov. Phytotaxa 2020, 455, 21–30. [Google Scholar] [CrossRef]
- Hyde, K.D.; Norphanphoun, C.; Maharachchikumbura, S.S.N.; Bhat, D.J.; Jones, E.B.G.; Bundhun, D.; Chen, Y.J.; Bao, D.F.; Boonmee, S.; Calabon, M.S.; et al. Refined families of Sordariomycetes. Mycosphere 2020, 11, 305–1059. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Nylander, J.A.A. MrModeltest v2.2. Program Distributed by the Author; Evolutionary Biology Centre, Uppsala University: Uppsala, Swden, 2004. [Google Scholar]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In Proceedings of the Gateway Computing Environments Workshop 2010 (GCE), New Orleans, LA, USA, 14–15 November 2010. [Google Scholar]
- Stamatakis, A.; Hoover, P.; Rougemont, J. A rapid bootstrap algorithm for the RAxML web servers. Syst. Biol. 2008, 57, 758–771. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Rannala, B.; Yang, Z. Probability distribution of molecular evolutionary trees: A new method of phylogenetic inference. J. Mol. Evol. 1996, 43, 304–311. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Liu, J.K.; Hyde, K.D.; Jeewon, R.; Kang, J.C.; Fan, C.; Boonmee, S.; Bhat, D.J.; Luo, Z.L.; Lin, C.G.; et al. A taxonomic reassessment of Tubeufiales based on multi-locus phylogeny and morphology. Fungal Divers. 2018, 92, 131–344. [Google Scholar] [CrossRef]
- Wendt, L.; Sir, E.B.; Kuhnert, E.; Heitkämper, S.; Lambert, C.; Hladki, A.I.; Romero, A.I.; Luangsa-ard, J.J.; Srikitikulchai, P.; Peršoh, D.; et al. Resurrection and emendation of the Hypoxylaceae, recognised from a multigene phylogeny of the Xylariales. Mycol. Prog. 2018, 17, 115–154. [Google Scholar] [CrossRef]
- Hyde, K.D.; Hongsanan, S.; Jeewon, R.; Bhat, D.J.; McKenzie, E.H.C.; Jones, E.B.G.; Phookamsak, R.; Ariyawansa, H.A.; Boonmee, S.; Zhao, Q.; et al. Fungal diversity notes 367–490: Taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers. 2016, 80, 1–270. [Google Scholar] [CrossRef]
- Voglmayr, H.; Aguirre-Hudson, M.B.; Wagner, H.G.; Tello, S.; Jaklitsch, W.M. Lichens or endophytes? The enigmatic genus Leptosillia in the Leptosilliaceae fam. nov. (Xylariales), and Furfurella gen. nov. (Delonicicolaceae). Persoonia 2019, 42, 228–260. [Google Scholar] [CrossRef]
- Daranagama, D.A.; Camporesi, E.; Tian, Q.; Liu, X.; Chamyuang, S.; Stadler, M.; Hyde, K.D. Anthostomella is polyphyletic comprising several genera in Xylariaceae. Fungal Divers. 2015, 73, 203–238. [Google Scholar] [CrossRef]
- Senanayake, I.C.; Maharachchikumbura, S.; Hyde, K.D.; Bhat, J.D.; Jones, E.B.G.; McKenzie, E.H.C.; Dai, D.Q.; Daranagama, D.A.; Dayarathne, M.C.; Goonasekara, I.D.; et al. Towards unraveling relationships in Xylariomycetidae (Sordariomycetes). Fungal Divers. 2015, 73, 73–144. [Google Scholar] [CrossRef]
- Mirabolfathy, M.; Ju, Y.M.; Hsieh, H.M.; Rogers, J.D. Obolarina persica sp. nov. associated with dying Quercus in Iran. Mycoscience 2013, 54, 315–320. [Google Scholar] [CrossRef]
- Liu, F.; Bonthond, G.; Groenewald, J.; Cai, L.; Crous, P. Sporocadaceae, a family of coelomycetous fungi with appendage-bearing conidia. Stud. Mycol. 2019, 92, 287–415. [Google Scholar] [CrossRef]
- Jaklitsch, W.M.; Voglmayr, H. Phylogenetic relationships of five genera of Xylariales and Rosasphaeria gen. nov. (Hypocreales). Fungal Divers. 2012, 52, 75–98. [Google Scholar] [CrossRef]
- Vu, D.; Groenewald, M.; de Vries, M.; Gehrmann, T.; Stielow, B.; Eberhardt, U.; Al-Hatmi, A.; Groenewald, J.Z.; Cardinali, G.; Houbraken, J.; et al. Large-scale generation and analysis of filamentous fungal DNAbarcodes boosts coverage for kingdom fungi and reveals thresholds for fungal species and higher taxon delimitation. Stud. Mycol. 2019, 92, 135–154. [Google Scholar] [CrossRef]
- Sun, Y.R.; Liu, N.G.; Samarakoon, M.C.; Jayawardena, R.S.; Hyde, K.D.; Wang, Y. Morphology and phylogeny reveal Vamsapriyaceae fam. nov. (Xylariales, Sordariomycetes) with two novel Vamsapriya species. J. Fungi 2021, 7, 891. [Google Scholar] [CrossRef]
- Thiyagaraja, V.; Senanayake, I.C.; Wanasinghe, D.N.; Karunarathna, S.C.; Worthy, F.R.; To-Anun, C. Phylogenetic and morphological appraisal of Diatrype lijiangensis sp. nov. (Diatrypaceae, Xylariales) from China. Asian J. Mycol. 2019, 2, 198–208. [Google Scholar] [CrossRef]
- Senwanna, I.C. Novel taxa of Diatrypaceae from Para rubber (Hevea brasiliensis) in northern Thailand; introducing a novel genus Allocryptovalsa. Mycosphere 2017, 8, 1835–1855. [Google Scholar] [CrossRef]
- Samarakoon, M.C.; Thongbai, B.; Hyde, K.D.; Brönstrup, M.; Beutling, U.; Lambert, C.; Miller, A.N.; Liu, J.K.; Promputtha, I.; Stadler, M. Elucidation of the life cycle of the endophytic genus Muscodor and its transfer to Induratia in Induratiaceae fam. nov. based on a polyphasic taxonomic approach. Fungal Divers. 2020, 101, 177–201. [Google Scholar] [CrossRef]
- Johnston, P.; Rogers, J.; Park, D.; Martin, N.A. Entalbostroma erumpens gen. et sp. nov. (Xylariaceae) from Phormium in New Zealand. Mycotaxon 2016, 131, 765–771. [Google Scholar] [CrossRef]
- Crous, P.W.; Wingfield, M.J.; Schumacher, R.K.; Akulov, A.; Bulgakov, T.S.; Carnegie, A.J.; Jurjević, Ž.; Decock, C.; Denman, S.; Lombard, L.; et al. New and interesting fungi. 3. Fungal Syst. Evol. 2020, 6, 157–231. [Google Scholar] [CrossRef]
- Crous, P.W.; Schumacher, R.K.; Akulov, A.; Thangavel, R.; Hernández-Restrepo, M.; Carnegie, A.; Cheewangkoo, R.; Wingfield, M.J.; Summerell, B.; Quaedvlieg, W.; et al. New and Interesting Fungi. 2. Fungal Syst. Evol. 2019, 3, 57–134. [Google Scholar] [CrossRef]
- Samarakoon, M.C.; Hyde, K.D.; Maharachchikumbura, S.S.N.; Stadler, M.; Jones, E.B.G.; Promputtha, I.; Suwannarach, N.; Camporesi, E.; Bulgakov, T.S.; Liu, J.K. Taxonomy, phylogeny, molecular dating and ancestral state reconstruction of Xylariomycetidae (Sordariomycetes). Fungal Divers. 2022, 112, 1–88. [Google Scholar] [CrossRef]
- Jaklitsch, W.M.; Fournier, J.; Rogers, J.D.; Voglmayr, H. Phylogenetic and taxonomic revision of Lopadostoma. Persoonia 2014, 32, 52–82. [Google Scholar] [CrossRef]
- Hernández-Restrepo, M.; Groenewald, J.; Crous, P. Taxonomic and phylogenetic re-evaluation of Microdochium, Monographella and Idriella. Persoonia Mol. Phylogeny Evol. Fungi 2016, 36, 57–82. [Google Scholar] [CrossRef] [Green Version]
- Konta, S.; Hyde, K.D.; Phookamsak, R.; Xu, J.C.; Maharachchikumbura, S.S.N.; Daranagama, D.A.; McKenzie, E.H.C.; Boonmee, S.; Tibpromma, S.; Eungwanichayapant, P.D.; et al. Polyphyletic genera in Xylariaceae (Xylariales): Neoxylaria gen. nov. and Stilbohypoxylon. Mycosphere 2020, 11, 2629–2651. [Google Scholar] [CrossRef]
- Rogers, J.D.; Ju, Y.M. The genus Stilbohypoxylon. Mycol. Res. 1997, 101, 135–138. [Google Scholar] [CrossRef]
- Petrini, L.E. A revision of the genus Stilbohypoxylon (Xylariaceae). Sydowia 2004, 56, 51–71. [Google Scholar]
- Takemoto, S.; Nakamura, H.; Sasaki, A.; Shimane, T. Species-specific PCRs differentiate Rosellinia necatrix from R. compacta as the prevalent cause of white root rot in Japan. J. Gen. Plant Pathol. 2011, 77, 107–111. [Google Scholar] [CrossRef]
- Sivanesan, A. New ascomycetes and some revisions. Trans. Br. Mycol. Soc. 1975, 65, 19. [Google Scholar] [CrossRef]
- Bahl, J.; Hyde, R. Phylogeny of Rosellinia capetribulensis sp. nov. and its allies (Xylariaceae). Mycologia 2005, 97, 1102–1110. [Google Scholar] [CrossRef]
- Petrini, L.E. Rosellinia and related genera in New Zealand. N. Z. J. Bot. 2003, 41, 71–138. [Google Scholar] [CrossRef]
- Læssøe, T.; Spooner, B.M. Rosellinia & Astrocystis (Xylariaceae): New Species and Generic Concepts. Kew Bull. 1993, 49, 1–70. [Google Scholar]
- Petrini, L.E.; Petrini, O. Morphological studies in Rosellinia (Xylariaceae): The first step towards a polyphasic taxonomy. Mycol. Res. 2005, 109, 569–580. [Google Scholar] [CrossRef]
- Rogers, J.D.; Miller, A.N.; Vasilyeva, L.N. Pyrenomycetes of the Great Smoky Mountains National Park. VI. Kretzschmaria, Nemania, Rosellinia and Xylaria (Xylariaceae). Fungal Divers. 2008, 29, 107–116. [Google Scholar]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Long, S.; Lin, Y.; Liu, L.; Shen, X.; Kang, J. Rhizomaticola guizhouensis gen. et sp. nov. and Five Rosellinia Like Species Isolated from Decaying Wood. Diversity 2022, 14, 703. https://doi.org/10.3390/d14090703
Li Q, Long S, Lin Y, Liu L, Shen X, Kang J. Rhizomaticola guizhouensis gen. et sp. nov. and Five Rosellinia Like Species Isolated from Decaying Wood. Diversity. 2022; 14(9):703. https://doi.org/10.3390/d14090703
Chicago/Turabian StyleLi, Qirui, Sihan Long, Yan Lin, Lili Liu, Xiangchun Shen, and Jichuan Kang. 2022. "Rhizomaticola guizhouensis gen. et sp. nov. and Five Rosellinia Like Species Isolated from Decaying Wood" Diversity 14, no. 9: 703. https://doi.org/10.3390/d14090703
APA StyleLi, Q., Long, S., Lin, Y., Liu, L., Shen, X., & Kang, J. (2022). Rhizomaticola guizhouensis gen. et sp. nov. and Five Rosellinia Like Species Isolated from Decaying Wood. Diversity, 14(9), 703. https://doi.org/10.3390/d14090703





