Carica papaya L.: A Tropical Fruit with Benefits beyond the Tropics
Abstract
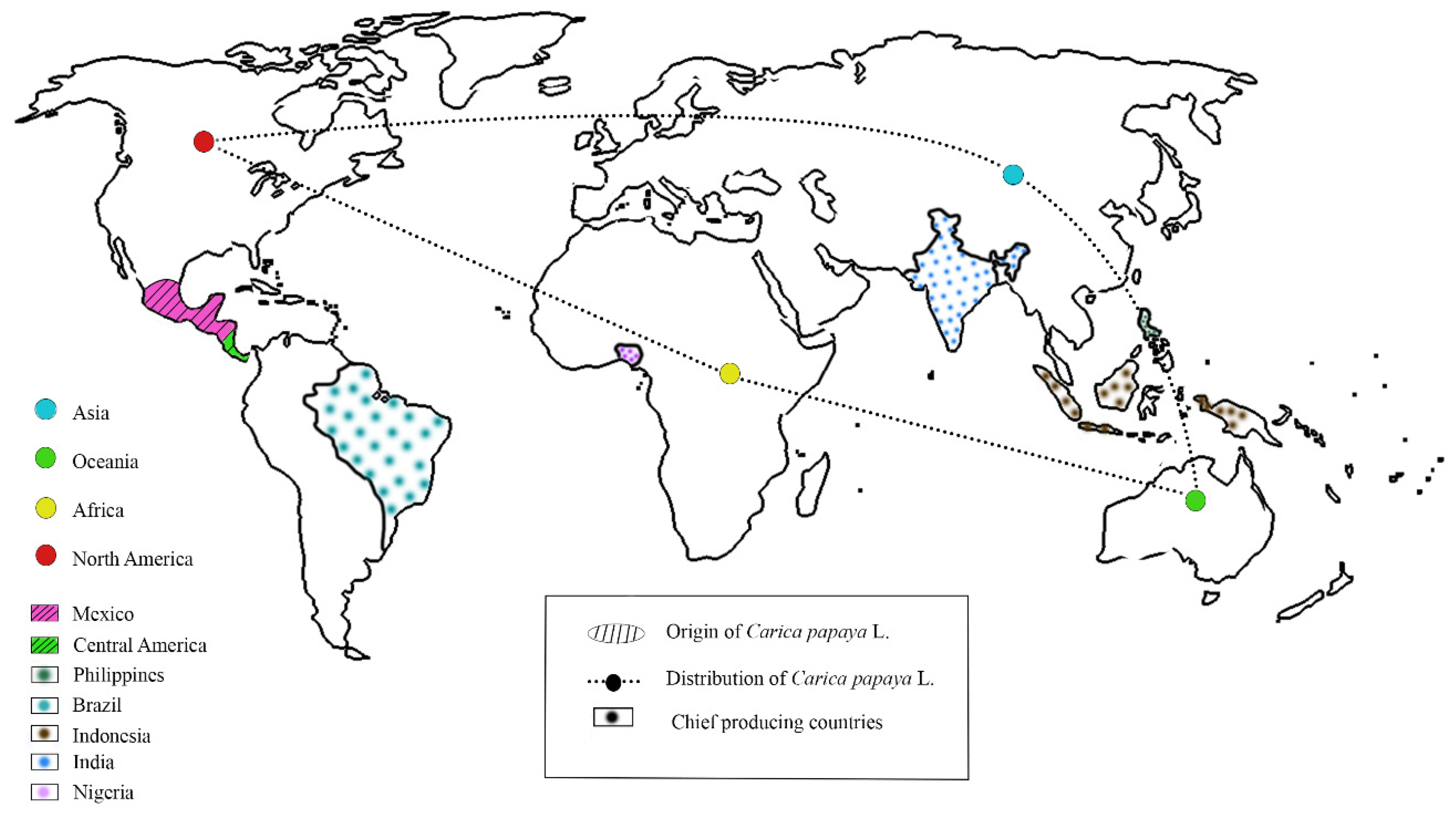
1. Introduction
2. Botanical Description
3. Nutritional Benefits
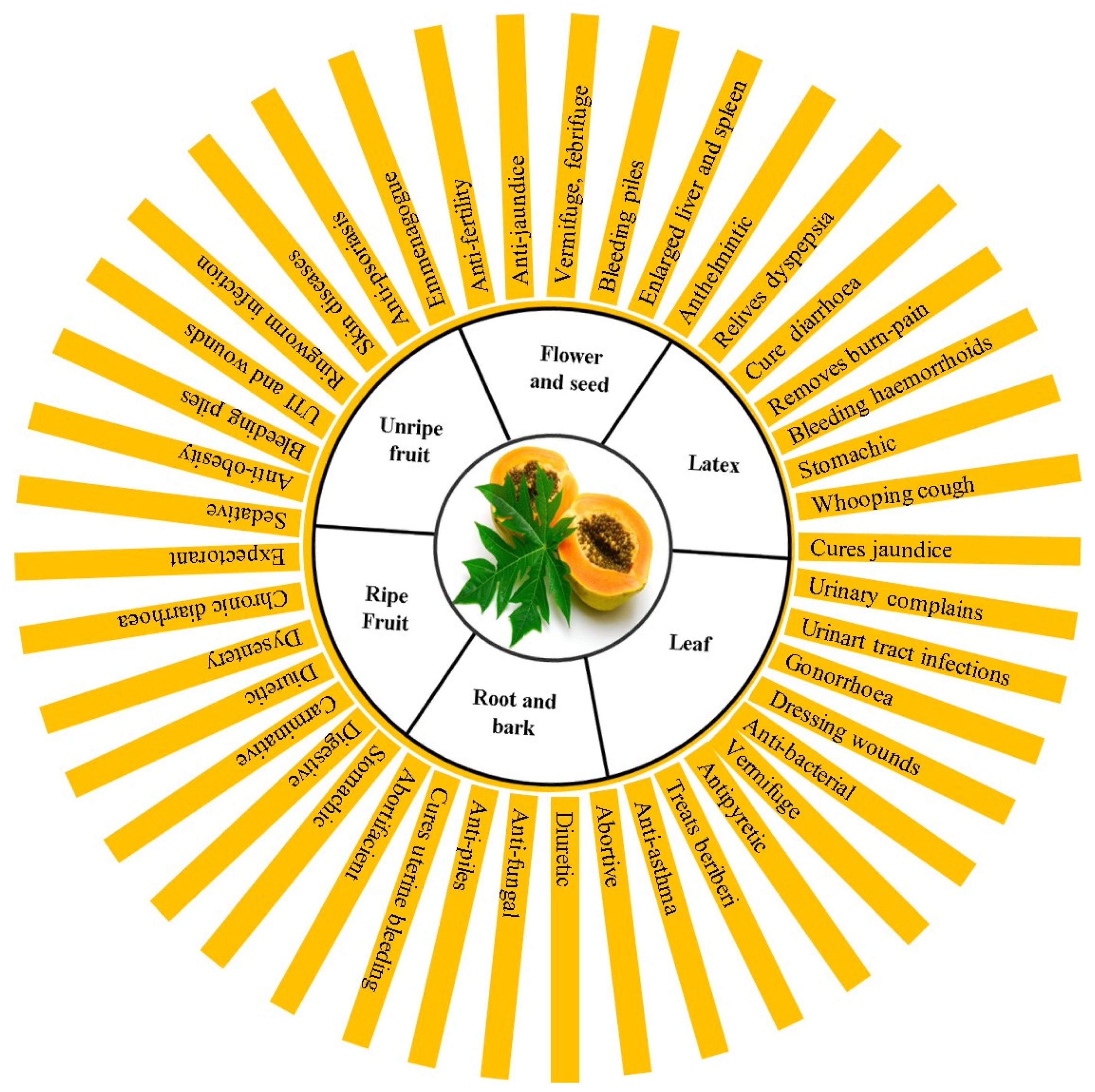
4. Traditional and Modern Medicinal Uses
5. Papaya Leaf Extract for Disease Management
6. Phytochemistry
7. Papaya Diseases and Etiology
7.1. Foot Rot/Stem Rot (Fungi: Pythium Aphanidermatum)
7.2. Collar Rot/Soft Fruit Rot (Fungi: Phytophthora Palmivora)
7.3. Powdery Mildew (Fungi: Oidium Caricae)
7.4. Papaya Anthrocnose (Fungi: Colletotrichum Gloeosporioides)
7.5. Papaya Ring Spot (Virus: Papaya Ring Spot Virus, PRSV)
7.6. Papaya Mosaic (Virus: Papaya Mosaic Virus, PMV)
7.7. Papaya Leaf Curl (PLCV)
8. Post-Harvest Diseases of Papaya
9. Papaya Genomics
10. Papaya Biotechnology—Crop Improvement
11. Value-Added Products of Papaya
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumar, L.; Srinivasan, V. Chromosome number of carica dodecaphylla vell fl. Flum. Curr. Sci. 1944, 13, 15. [Google Scholar]
- Badillo, V. Nota correctiva vasconcellea. St. Hill. Y No Vascon. (Caricaceae). Ernstia 2001, 11, 75–76. [Google Scholar]
- Gonsalves, C.; Lee, D.R.; Gonsalves, D. The adoption of genetically modified papaya in hawaii and its implications for developing countries. J. Dev. Stud. 2007, 43, 177–191. [Google Scholar] [CrossRef]
- Fuentes, G.; Santamaría, J.M. Papaya (Carica papaya L.): Origin, domestication, and production. In Genetics and Genomics of Papaya; Springer: Berlin/Heidelberg, Germany, 2014; pp. 3–15. [Google Scholar]
- Manshardt, R.M. Papaya. In Biotechnology of Perennial Fruit Crops; Hammerschlag, f.A., Litz, r.E., Eds.; Cambridge University Press: Oxford, UK, 1992; pp. 489–511. [Google Scholar]
- Silva, J.d.; Rashid, Z.; Nhut, D.T.; Sivakumar, D.; Gera, A.; Souza, M.T.; Tennant, P. Papaya (Carica papaya L.) biology and biotechnology. Tree For. Sci. Biotechnol. 2007, 1, 47–73. [Google Scholar]
- Chen, L.-F.; Bau, H.-J.; Yeh, S.-D. Identification of Viruses Capable of Breaking Transgenic Resistance of Papaya Conferred by the Coat Protein Gene of Papaya Ringspot Virus. Acta Hortic. 2002, 575, 465–474. [Google Scholar] [CrossRef]
- Morton, J.F.D.C.F. Fruits of Warm Climates; Morton, J.F., Ed.; Distributed by Creative Resources Systems: Miami, FL, USA; Winterville, NC, USA, 1987. [Google Scholar]
- Karunamoorthi, K.; Kim, H.-M.; Jegajeevanram, K.; Xavier, J.; Vijayalakshmi, J. Papaya: A gifted nutraceutical plant—A critical review of recent human health research. Cellmed 2014, 4, 2.1–2.17. [Google Scholar] [CrossRef][Green Version]
- Saeed, F.; Arshad, M.U.; Pasha, I.; Naz, R.; Batool, R.; Khan, A.A.; Nasir, M.A.; Shafique, B. Nutritional and phyto-therapeutic potential of papaya (Carica papaya Linn.): An overview. Int. J. Food Prop. 2014, 17, 1637–1653. [Google Scholar] [CrossRef]
- Alara, O.R.; Abdurahman, N.H.; Alara, J.A. Carica papaya: Comprehensive overview of the nutritional values, phytochemicals and pharmacological activities. Adv. Tradit. Med. 2020, 22, 1–31. [Google Scholar] [CrossRef]
- De Oliveira, J.G.; Vitória, A.P. Papaya: Nutritional and pharmacological characterization, and quality loss due to physiological disorders. An overview. Food Res. Int. 2011, 44, 1306–1313. [Google Scholar] [CrossRef]
- Starley, I.F.; Mohammed, P.; Schneider, G.; Bickler, S.W. The treatment of paediatric burns using topical papaya. Burns 1999, 25, 636–639. [Google Scholar] [CrossRef]
- Maciunas, R.J.; Onofrio, B.M. The long-term results of chymopapain. Ten-year follow-up of 268 patients after chemonucleolysis. Clin. Orthop. Relat. Res. 1986, 206, 37–41. [Google Scholar] [CrossRef]
- Begum, M. Phytochemical and Pharmacological Investigation of Carica Papaya Leaf; East west University: Dhaka, Bangladesh, 2014. [Google Scholar]
- Reed, C.F. Information summaries on 1000 economic plants. Typescripts Submitt. USDA 1976, 102–103. [Google Scholar]
- Elizabeth, K. Immense help from nature’s workshop. Edn 1994, 1, 207–209. [Google Scholar]
- Sharma, A.; Sharma, R.; Sharma, M.; Kumar, M.; Barbhai, M.D.; Lorenzo, J.M.; Sharma, S.; Samota, M.K.; Atanassova, M.; Caruso, G. Carica papaya L. Leaves: Deciphering its antioxidant bioactives, biological activities, innovative products, and safety aspects. Oxid. Med. Cell. Longev. 2022, 2022, 1–20. [Google Scholar] [CrossRef]
- Hu, T.; Guo, Y.Y.; Zhou, Q.F.; Zhong, X.K.; Zhu, L.; Piao, J.H.; Chen, J.; Jiang, J.G. Optimization of ultrasonic-assisted extraction of total saponins from eclipta prostrasta l. Using response surface methodology. J. Food Sci. 2012, 77, C975–C982. [Google Scholar] [CrossRef]
- Patil, T.; Patil, S.; Patil, A.; Patil, S. Carica papaya leaf extracts—An ethnomedicinal boon. Int. J. Pharmacogn. Phytochem. 2014, 6, 260–265. [Google Scholar]
- Rumiyati, S. Effect of the protein fraction of Carica papaya L. Leaves on the expressions of p53 and bcl-2 in breast cancer cells line. Maj. Farm. Indones 2006, 17, 170–176. [Google Scholar]
- Singh, S.P.; Kumar, S.; Mathan, S.V.; Tomar, M.S.; Singh, R.K.; Verma, P.K.; Kumar, A.; Kumar, S.; Singh, R.P.; Acharya, A. Therapeutic application of carica papaya leaf extract in the management of human diseases. DARU J. Pharm. Sci. 2020, 28, 735–744. [Google Scholar] [CrossRef]
- Otsuki, N.; Dang, N.H.; Kumagai, E.; Kondo, A.; Iwata, S.; Morimoto, C. Aqueous extract of carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J. Ethnopharmacol. 2010, 127, 760–767. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Parat, M.-O.; Hodson, M.P.; Pan, J.; Shaw, P.N.; Hewavitharana, A.K. Chemical characterization and in vitro cytotoxicity on squamous cell carcinoma cells of carica papaya leaf extracts. Toxins 2015, 8, 7. [Google Scholar] [CrossRef]
- Pandey, S.; Walpole, C.; Cabot, P.J.; Shaw, P.N.; Batra, J.; Hewavitharana, A.K. Selective anti-proliferative activities of carica papaya leaf juice extracts against prostate cancer. Biomed. Pharmacother. 2017, 89, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.; Fazal, H.; Ayaz, M.; Abbasi, B.H.; Mohammad, I.; Fazal, L. Dengue fever treatment with Carica papaya leaves extracts. Asian Pac. J. Trop Biomed. 2011, 1, 330–333. [Google Scholar] [CrossRef]
- Saotoing, P.; Tchuenguem, F.F.; Njan, N.A.; Messi, J. Medicinal plants used in traditional treatment of malaria in cameroon. J. Ecol. Nat. Environ. 2011, 3, 104–117. [Google Scholar]
- Kovendan, K.; Murugan, K.; Naresh Kumar, A.; Vincent, S.; Hwang, J.-S. Bioefficacy of larvicdial and pupicidal properties of carica papaya (caricaceae) leaf extract and bacterial insecticide, spinosad, against chikungunya vector, aedes aegypti (diptera: Culicidae). Parasitol. Res. 2012, 110, 669–678. [Google Scholar] [CrossRef]
- Morimoto, C.; Dang, N. Compositions for Cancer Prevention, Treatment, or Amelioration Comprising Papaya Extract. US Patent 20080069907, 20 March 2008. [Google Scholar]
- Fauziya, S.; Krishnamurthy, R. Papaya (carica papaya): Source material for anticancer. CIBTech. J. Pharm. Sci. 2013, 2, 25–34. [Google Scholar]
- Nguyen, T.T.; Shaw, P.N.; Parat, M.O.; Hewavitharana, A.K. Anticancer activity of c arica papaya: A review. Mol. Nutr. Food Res. 2013, 57, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Anjum, V.; Arora, P.; Ansari, S.H.; Najmi, A.K.; Ahmad, S. Antithrombocytopenic and immunomodulatory potential of metabolically characterized aqueous extract of carica papaya leaves. Pharm. Biol. 2017, 55, 2043–2056. [Google Scholar] [CrossRef] [PubMed]
- Imaga, N.A.; Gbenle, G.O.; Okochi, V.I.; Akanbi, S.; Edeoghon, S.O.; Oigbochie, V.E.; Kehinde, M.O.; Bamiro, S.B. Antisickling property of carica papaya leaf extract. Afr. J. Biochem. Res. 2009, 3, 102–106. [Google Scholar]
- Gurung, S.; Škalko-Basnet, N. Wound healing properties of carica papaya latex: In vivo evaluation in mice burn model. J. Ethnopharmacol. 2009, 121, 338–341. [Google Scholar] [CrossRef]
- Sagnia, B.; Fedeli, D.; Casetti, R.; Montesano, C.; Falcioni, G.; Colizzi, V. Antioxidant and anti-inflammatory activities of extracts from cassia alata, eleusine indica, eremomastax speciosa, carica papaya and polyscias fulva medicinal plants collected in cameroon. PLoS ONE 2014, 9, e103999. [Google Scholar] [CrossRef]
- Salim, E.; Kumolosasi, E.; Jantan, I. Inhibitory effect of selected medicinal plants on the release of pro-inflammatory cytokines in lipopolysaccharide-stimulated human peripheral blood mononuclear cells. J. Nat. Med. 2014, 68, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Sadek, K.M. Antioxidant and immunostimulant effect of carica papaya linn. Aqueous extract in acrylamide intoxicated rats. Acta Inform. Med. 2012, 20, 180. [Google Scholar] [CrossRef] [PubMed]
- Teh, B.P.; Ahmad, N.B.; Mohamad, S.B.; Tan, T.Y.C.; Mohd Abd Razak, M.R.B.; Afzan, A.B.; Syed Mohamed, A.F.B. Carica papaya Leaf Juice for Dengue: A Scoping Review. Nutrients 2022, 14, 1584. [Google Scholar] [CrossRef]
- Dharmarathna, S.L.C.A.; Wickramasinghe, S.; Waduge, R.N.; Rajapakse, R.P.V.J.; Kularatne, S.A.M. Does carica papaya leaf-extract increase the platelet count? An experimental study in a murine model. Asian Pac. J. Trop. Biomed. 2013, 3, 720–724. [Google Scholar] [CrossRef]
- Gammulle, A.; Ratnasooriya, W.; Jayakody, J.; Fernando, C.; Kanatiwela, C.; Udagama, P.V. Thrombocytosis and anti-inflammatory properties, and toxicological evaluation of carica papaya mature leaf concentrate in a murine model. Online Int. J. Med. Plant Res. 2012, 1, 21–30. [Google Scholar]
- Yunita, F.; Hanani, E.; Kristianto, J. The effect of Carica papaya L. Leaves extract capsules on platelets count and hematocrit level in dengue fever patient. Int. J. Med. Aromat. Plants 2012, 2, 573–578. [Google Scholar]
- Norahmad, N.A.; Mohd Abd Razak, M.R.; Mohmad Misnan, N.; Md Jelas, N.H.; Sastu, U.R.; Muhammad, A.; Ho, T.C.D.; Jusoh, B.; Zolkifli, N.A.; Thayan, R. Effect of freeze-dried carica papaya leaf juice on inflammatory cytokines production during dengue virus infection in ag129 mice. BMC Complement Altern. Med. 2019, 19, 44. [Google Scholar] [CrossRef]
- Ranasinghe, P.; Ranasinghe, P.; Abeysekera, W.K.M.; Premakumara, G.S.; Perera, Y.S.; Gurugama, P.; Gunatilake, S.B. In vitro erythrocyte membrane stabilization properties of Carica papaya L. Leaf extracts. Pharmacogn. Res. 2012, 4, 196. [Google Scholar] [CrossRef]
- Srikanth, B.K.; Reddy, L.; Biradar, S.; Shamanna, M.; Mariguddi, D.D.; Krishnakumar, M. An open-label, randomized prospective study to evaluate the efficacy and safety of carica papaya leaf extract for thrombocytopenia associated with dengue fever in pediatric subjects. Pediatric Health Med. Ther. 2019, 10, 5–11. [Google Scholar] [CrossRef]
- Isela, E.J.-R.; Juan, C.D.-Z.; Jorge, L.; Pedro, H.M.-O.; Andrés, E.C.-R.; Carlos, A.; Tovilla-Z, A.; Rodríguez-H, H.; Aguilar-M, T.; Ramón-F, D.Y.; et al. Hypoglycemic effect of carica papaya leaves in streptozotocin-induced diabetic rats. BMC Complement Alternat. Med. 2012, 12, 236. [Google Scholar]
- Aruoma, O.I.; Somanah, J.; Bourdon, E.; Rondeau, P.; Bahorun, T. Diabetes as a risk factor to cancer: Functional role of fermented papaya preparation as phytonutraceutical adjunct in the treatment of diabetes and cancer. Mutat. Res.-Fundam. Mol. Mech. Mutagen. 2014, 768, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Nurain, I.O.; Bewaji, C.O.; Johnson, J.S.; Davenport, R.D.; Zhang, Y. Potential of three ethnomedicinal plants as antisickling agents. Mol. Pharm. 2017, 14, 172–182. [Google Scholar] [CrossRef]
- Suresh, K. Antimicrobial and phytochemical investigation of the leaves of Carica papaya L., Cynodon dactylon (L.) pers., Euphorbia hirta L., Melia azedarach L. And Psidium guajava L. Ethnobot. Leafl. 2008, 2008, 157. [Google Scholar]
- Baskaran, C.; Ratha bai, V.; Velu, S.; Kumaran, K. The efficacy of carica papaya leaf extract on some bacterial and a fungal strain by well diffusion method. Asian Pac. J. Trop. Dis. 2012, 2, S658–S662. [Google Scholar] [CrossRef]
- Indran, M.; Mahmood, A.; Kuppusamy, U. Protective effect of carica papaya l leaf extract against alcohol induced acute gastric damage and blood oxidative stress in rats. West. Indian Med. J. 2008, 57, 323–326. [Google Scholar] [PubMed]
- Owoyele, B.V.; Adebukola, O.M.; Funmilayo, A.A.; Soladoye, A.O. Anti-inflammatory activities of ethanolic extract of carica papaya leaves. Inflammopharmacology 2008, 16, 168–173. [Google Scholar] [CrossRef]
- Fuggate, P.; Wongs-Aree, C.; Noichinda, S.; Kanlayanarat, S. Quality and volatile attributes of attached and detached ‘pluk mai lie’papaya during fruit ripening. Sci. Hortic. 2010, 126, 120–129. [Google Scholar] [CrossRef]
- Balbontín, C.; Gaete-Eastman, C.; Vergara, M.; Herrera, R.; Moya-León, M.A. Treatment with 1-mcp and the role of ethylene in aroma development of mountain papaya fruit. Postharvest Biol. Technol. 2007, 43, 67–77. [Google Scholar] [CrossRef]
- Flath, R.A.; Forrey, R.R. Volatile components of papaya (Carica papaya L., solo variety). J. Agric. Food Chem. 1977, 25, 103–109. [Google Scholar] [CrossRef]
- Almora, K.; Pino, J.A.; Hernández, M.; Duarte, C.; González, J.; Roncal, E. Evaluation of volatiles from ripening papaya (Carica papaya L., var. Maradol roja). Food Chem. 2004, 86, 127–130. [Google Scholar] [CrossRef]
- MacLeod, A.J.; Pieris, N.M. Volatile components of papaya (Carica papaya L.) with particular reference to glucosinolate products. J. Agric. Food Chem. 1983, 31, 1005–1008. [Google Scholar] [CrossRef]
- Zunjar, V.; Mammen, D.; Trivedi, B. Antioxidant activities and phenolics profiling of different parts of carica papaya by lcms-ms. Nat. Prod. Res. 2015, 29, 2097–2099. [Google Scholar] [CrossRef] [PubMed]
- Julianti, T.; De Mieri, M.; Zimmermann, S.; Ebrahimi, S.N.; Kaiser, M.; Neuburger, M.; Raith, M.; Brun, R.; Hamburger, M. Hplc-based activity profiling for antiplasmodial compounds in the traditional indonesian medicinal plant Carica papaya L. J. Ethnopharmacol. 2014, 155, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Esti, M.; Benucci, I.; Lombardelli, C.; Liburdi, K.; Garzillo, A.M.V. Papain from papaya (Carica papaya L.) fruit and latex: Preliminary characterization in alcoholic–acidic buffer for wine application. Food Bioprod. Process. 2013, 91, 595–598. [Google Scholar] [CrossRef]
- Burdick, E.M. Carpaine: An alkaloid of carica papaya: Its chemistry and pharmacology. Econ. Bot. 1971, 25, 363–365. [Google Scholar] [CrossRef]
- Krishna, K.; Paridhavi, M.; Patel, J.A. Review on nutritional, medicinal and pharmacological properties of papaya (carica papaya linn.). Nat. Prod. Rad. 2008, 7, 364–373. [Google Scholar]
- Yogiraj, V.; Goyal, P.K.; Chauhan, C.S.; Goyal, A.; Vyas, B. Carica papaya linn: An overview. Int. J. Herb. Med. 2014, 2, 1–8. [Google Scholar]
- Tang, C.-S. Localization of benzyl glucosinolate and thioglucosidase in carica papaya fruit. Phytochemistry 1973, 12, 769–773. [Google Scholar] [CrossRef]
- Barroso, P.T.; de Carvalho, P.P.; Rocha, T.B.; Pessoa, F.L.; Azevedo, D.A.; Mendes, M.F. Evaluation of the composition of Carica papaya L. Seed oil extracted with supercritical co2. Biotechnol. Rep. 2016, 11, 110–116. [Google Scholar] [CrossRef]
- Sancho, L.E.G.-G.; Yahia, E.M.; González-Aguilar, G.A. Identification and quantification of phenols, carotenoids, and vitamin c from papaya (Carica papaya L., cv. Maradol) fruit determined by hplc-dad-ms/ms-esi. Food Res. Int. 2011, 44, 1284–1291. [Google Scholar] [CrossRef]
- Oduola, T.; Adeniyi, F.; Ogunyemi, E.; Bello, I.; Idowu, T. Antisickling agent in an extract of unripe pawpaw (carica papaya): Is it real? Afr. J. Biotechnol. 2006, 5, 1947–1949. [Google Scholar]
- Canini, A.; Alesiani, D.; D’Arcangelo, G.; Tagliatesta, P. Gas chromatography–mass spectrometry analysis of phenolic compounds from Carica papaya L. Leaf. J. Food Compost. Anal. 2007, 20, 584–590. [Google Scholar] [CrossRef]
- FAOSTAT; FAO. Food and Agriculture Data. Crop Statistics. 2019. Available online: http://www.fao.org/faostat (accessed on 14 August 2020).
- Srivastava, J.; Singh, A. Diseases of Horticultural Crops: Diagnosis and Management: Volume 1: Fruit Crops; CRC Press: Boca Raton, FL, USA, 2022. [Google Scholar]
- Available online: http://agropedia.iitk.ac.in/content/papaya-diseases-its-control (accessed on 13 August 2012).
- Ventura, J.A.; Costa, H.; Tatagiba, J.d.S. Papaya Diseases and Integrated Control. In Diseases of Fruits and Vegetables: Volume II; Naqvi, S.A.M.H., Ed.; Springer: Dordrecht, Germany, 2004. [Google Scholar] [CrossRef]
- Narayanasamy, P. Crop Diseases Management: Principles and Practices; New India Publishing Agency: Delhi, India, 2011. [Google Scholar]
- Cannon, P.F.; Kirk, P.M. Fungal Families of the World; Cabi: Wallingford, UK, 2007. [Google Scholar]
- Hine, R.B.; Holtzmann, O.V.; Raabe, R.D. Diseases of Papaya (Carica papaya L.) in Hawaii; University of Hawaii: Honolulu, HI, USA, 1965; 26p, (Bulletin 136); Available online: http://hdl.handle.net/10125/14967 (accessed on 8 February 2010).
- Ooka, J. Oidium caricae. In Crop Knowledge Master; University of Hawai’i at Manoa, College of Tropical Agriculture and Human Resources: Honolulu, Hawaii, 1993. [Google Scholar]
- Snowdon, A.L. A Colour Atlas of Post-Harvest Diseases and Disorders of Fruits and Vegetables. Volume 1: General Introduction and Fruits; Wolfe Scientific Ltd.: London UK, 1990. [Google Scholar]
- Rana, S. Fungal Diseases of Papaya; Diseases of Fruit Crops; Oxford and IBH Publishing Co. Pvt. Ltd.: New Delhi, India, 2001. [Google Scholar]
- Umer, M.; Mubeen, M.; Iftikhar, Y.; Ali, H.; Zafar-ul-Hye, M.; Asghar, R.; Abbas, M.; Rehman, M.A.; Moya-Elizondo, E.A.; He, Y. Papaya Ring Spot Virus: An Understanding of a Severe Positive-Sense Single Stranded RNA Viral Disease and its Management. Phyton 2022, 91, 2099–2110. [Google Scholar] [CrossRef]
- Gonsalves, D. Control of papaya ringspot virus in papaya: A case study. Annu. Rev. Phytopathol. 1998, 36, 415–437. [Google Scholar] [CrossRef]
- Conover, R. Virus Diseases of Papaya in Florida; Amer Phytopathological Soc 3340 Pilot Knob Road, Phytopathology: St. Paul, MN, USA, 1962; p. 6. [Google Scholar]
- Bau, H.-J.; Kung, Y.-J.; Raja, J.; Chan, S.-J.; Chen, K.-C.; Chen, Y.-K.; Wu, H.-W.; Yeh, S.-D. Potential threat of a new pathotype of papaya leaf distortion mosaic virus infecting transgenic papaya resistant to papaya ringspot virus. Phytopathology 2008, 98, 848–856. [Google Scholar] [CrossRef]
- Shen, W.; Wang, Y.; Tuo, D.; Yan, P.; Yang, Y.; Li, X.; Zhou, P. Agroinoculation of carica papaya with infectious clones of papaya mosaic virus. Acta Virol. 2014, 58, 380–382. [Google Scholar] [CrossRef][Green Version]
- Thomas, K.; Krishnaswami, C. Leaf crinkle—A transmissible disease of papaya. Curr. Sci. 1939, 8, 316. [Google Scholar]
- Saxena, S.; Hallan, V.; Singh, B.; Sane, P. Evidence from nucleic acid hybridization tests for a geminivirus infectioncausing leaf curl disease of papaya in india. Indian J. Exp. Biol. 1998, 36, 229–232. [Google Scholar]
- Bananej, K.; Shafiq, M.; Shahid, M.S. Association of cotton leaf curl gezira virus with tomato leaf curl betasatellite infecting carica papaya in iran. Australas. Plant Dis. Notes 2021, 16, 1–4. [Google Scholar] [CrossRef]
- Fatima, S.; Javeed, Z.; Ade, A. Post-Harvest Rots of Fruits; Discovery Publishing House: Delhi, India, 2006. [Google Scholar]
- Rahman, M.; Mahmud, T.; Kadir, J.; Abdul Rahman, R.; Begum, M. Major postharvest fungal diseases of papaya cv.‘Sekaki’in selangor, malaysia. Pertanika J. Trop. Agric. Sci 2008, 31, 27–34. [Google Scholar]
- Hunter, J.E.; Buddenhagen, I. Incidence, epidemiology and control of fruit diseases of papaya in hawaii. Trop Agr. 1972, 49, 61–72. [Google Scholar]
- Gupta, G.K.; Sharma, S.K.; Ramteke, R. Biology, epidemiology and management of the pathogenic fungus macrophomina phaseolina (tassi) goid with special reference to charcoal rot of soybean (Glycine max (L.) merrill). J. Phytopathol. (1986) 2012, 160, 167–180. [Google Scholar] [CrossRef]
- Helal, R.; Hosen, S.; Shamsi, S. Mycoflora associated with post-harvest disease of papaya (Carica papaya L.) and their pathogenic potentiality. Bangladesh J. Bot. 2018, 47, 389–395. [Google Scholar] [CrossRef]
- Nishijima, K.; Miura, C.; Armstrong, J.; Brown, S.; Hu, B. Effect of Forced, Hot-Air Treatment of Papaya Fruit on Fruit Quality and Incidence of Postharvest Diseases. Plant Dis. 1992, 76, 723–727. [Google Scholar] [CrossRef]
- Amaral, D.D.; Monteiro, A.L.R.; Silva, E.I.D.; Lins, S.R.D.O.; OLIVEIRA, S. Frequency of quiescent fungi and post-harvest alternative management of stem end rot in papaya. Revista Caatinga 2017, 30, 786–793. [Google Scholar] [CrossRef]
- Palou, L.; Smilanick, J.L. Postharvest Pathology of Fresh Horticultural Produce; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Debysingh, N.; Wickham, L.D.; Mohammed, M.; Legall, G.; Paliyath, G.; Subramanian, J. Effects of pre-and post-harvest treatments with hexanal formulations on time to ripening and shelf life of papaya (Carica papaya L.) fruits. Trop. Agric. 2018, 95, 36–42. [Google Scholar]
- Bautista-Baños, S.; Sivakumar, D.; Bello-Pérez, A.; Villanueva-Arce, R.; Hernández-López, M. A review of the management alternatives for controlling fungi on papaya fruit during the postharvest supply chain. Crop Prot. 2013, 49, 8–20. [Google Scholar] [CrossRef]
- Suseela Bhai, R.; Ishwara Bhat, A.; Anandaraj, M. Premature yellowing and bean shedding in vanilla (vanilla planifolia andrews). In Proceedings of the Symposium on Resent Development in the Diagnosis and Management of Plant Diseases for Meeting Global Challenges, Dharwad, KA, India, 18–20 December 2003; pp. 88–89. [Google Scholar]
- Bosquez-Molina, E.; Ronquillo-de Jesús, E.; Bautista-Baños, S.; Verde-Calvo, J.; Morales-López, J. Inhibitory effect of essential oils against colletotrichum gloeosporioides and rhizopus stolonifer in stored papaya fruit and their possible application in coatings. Postharvest Biol. Technol. 2010, 57, 132–137. [Google Scholar] [CrossRef]
- Sarkhosh, A.; Schaffer, B.; Vargas, A.; Palmateer, A.; Lopez, P.; Soleymani, A.; Farzaneh, M. Antifungal activity of five plant-extracted essential oils against anthracnose in papaya fruit. Biol. Agric. Hortic. 2018, 34, 18–26. [Google Scholar] [CrossRef]
- Allong, R.; Wickham, L.; Majeed, M. Effect of cultivar, hot water treatment and storage conditions on quality of fresh-cut papaya (Carica papaya L.). J. Appl. Hortic. 2000, 2, 15–18. [Google Scholar] [CrossRef]
- Li, X.; Zhu, X.; Zhao, N.; Fu, D.; Li, J.; Chen, W.; Chen, W. Effects of hot water treatment on anthracnose disease in papaya fruit and its possible mechanism. Postharvest Biol. Technol. 2013, 86, 437–446. [Google Scholar] [CrossRef]
- Marpudi, S.; Abirami, L.S.S.; Pushkala, R.; Nagarajan, S. Enhancement of storage life and quality maintenance of papaya fruits using aloe vera based antimicrobial coating. Indian J. Biotechnol. 2011, 10, 83–89. [Google Scholar]
- Sivakumar, D.; Sultanbawa, Y.; Ranasingh, N.; Kumara, P.; Wijesundera, R. Effect of the combined application of chitosan and carbonate salts on the incidence of anthracnose and on the quality of papaya during storage. J. Hortic. Sci. Biotechnol. 2005, 80, 447–452. [Google Scholar] [CrossRef]
- Chau, K.; Alvarez, A.M. Postharvest fruit rot of papaya caused by stemphylium lycopersici. Plant Dis. 1983, 67, 1279–1281. [Google Scholar] [CrossRef]
- Ming, R.; van Droogenbroeck, B.; Moore, P.H.; Zee, F.T.; Kyndt, T.; Scheldeman, X.; Sekioka, T.; Gheysen, G. Molecular diversity of carica papaya and related species. Plant Genome Biodivers. Evol. 2005, 1, 229–254. [Google Scholar]
- Ming, R.; Hou, S.; Feng, Y.; Yu, Q.; Dionne-Laporte, A.; Saw, J.H.; Senin, P.; Wang, W.; Ly, B.V.; Lewis, K.L.; et al. The draft genome of the transgenic tropical fruit tree papaya (carica papaya linnaeus). Nature 2008, 452, 991–996. [Google Scholar] [CrossRef]
- Van Droogenbroeck, B.; Breyne, P.; Goetghebeur, P.; Romeijn-Peeters, E.; Kyndt, T.; Gheysen, G. Aflp analysis of genetic relationships among papaya and its wild relatives (caricaceae) from ecuador. Theor. Appl. Genet. 2002, 105, 289–297. [Google Scholar] [CrossRef]
- Wai, C.M.; Ming, R.; Moore, P.H.; Paull, R.E.; Yu, Q. Development of chromosome-specific cytogenetic markers and merging of linkage fragments in papaya. Trop. Plant Biol. 2010, 3, 171–181. [Google Scholar] [CrossRef]
- Zhang, W.; Wai, C.M.; Ming, R.; Yu, Q.; Jiang, J. Integration of genetic and cytological maps and development of a pachytene chromosome-based karyotype in papaya. Trop. Plant Biol. 2010, 3, 166–170. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, X.; Yu, Q.; Ming, R.; Jiang, J. DNA methylation and heterochromatinization in the male-specific region of the primitive y chromosome of papaya. Genome Res. 2008, 18, 1938–1943. [Google Scholar] [CrossRef]
- Arumuganathan, K.; Earle, E.D. Estimation of nuclear DNA content of plants by flow cytometry. Plant Mol. Biol. Rep. 1991, 9, 229–241. [Google Scholar] [CrossRef]
- Stokstad, E. Papaya Takes on Ringspot Virus and Wins; American Association for the Advancement of Science: Washington, DC, USA, 2008. [Google Scholar]
- Jaillon, O.; Aury, J.; Noel, B.; Policriti, A.; Clepet, C.; Casagrande, A.; Choisne, N.; Aubourg, S.; Vitulo, N.; Jubin, C. French-italian public the grapevine genome sequence suggests ancestral hexaploidization in major angiosperm phyla. Nature 2007, 449, 463. [Google Scholar]
- Sasaki, T. The map-based sequence of the rice genome. Nature 2005, 436, 793–800. [Google Scholar] [CrossRef]
- Tuskan, G.A.; Difazio, S.; Jansson, S.; Bohlmann, J.; Grigoriev, I.; Hellsten, U.; Putnam, N.; Ralph, S.; Rombauts, S.; Salamov, A. The genome of black cottonwood, populus trichocarpa (torr. & gray). Science 2006, 313, 1596–1604. [Google Scholar]
- Kanchana-udomkan, C.; Ford, R.; Drew, R. Molecular markers in papayas. In Genetics and Genomics of Papaya; Springer: Berlin/Heidelberg, Germany, 2014; pp. 355–375. [Google Scholar]
- Adawiah, Z.R.; Norliza, A.; Fairuz, Y.M.; Norzihan, A.; Kalsom, A.U. Sequence information on single nucleotide polymorphism (snp) through genome sequencing analysis of carica papaya variety eksotika and sekaki. J. Trop. Agric. Fd. Sci. 2016, 44, 219–228. [Google Scholar]
- Wu, M.; Lewis, J.; Moore, R.C. A wild origin of the loss-of-function lycopene beta cyclase (cyc-b) allele in cultivated, red-fleshed papaya (carica papaya). Am. J. Bot. 2017, 104, 116–126. [Google Scholar] [CrossRef]
- Chia, C.L.; Manshardt, R.M. Why some Papaya Plants Fail to Fruit; (Fruits and Nuts; F&N-5); University of Hawaii: Honolulu, HI, USA, 2001; 2p, Available online: http://hdl.handle.net/10125/12186 (accessed on 1 July 2022).
- Ming, R.; Yu, Q.; Moore, P.H. Sex determination in papaya. Semin. Cell Dev. Biol. 2007, 18, 401–408. [Google Scholar] [CrossRef]
- Liao, Z.; Dong, F.; Liu, J.; Xu, L.; Marshall-Colon, A.; Ming, R. Gene regulation network analyses of pistil development in papaya. BMC Genom. 2022, 23, 8. [Google Scholar] [CrossRef]
- Liu, Z.; Moore, P.H.; Ma, H.; Ackerman, C.M.; Ragiba, M.; Yu, Q.; Pearl, H.M.; Kim, M.S.; Charlton, J.W.; Stiles, J.I.; et al. A primitive y chromosome in papaya marks incipient sex chromosome evolution. Nature 2004, 427, 348–352. [Google Scholar] [CrossRef]
- Yu, Q.; Hou, S.; Feltus, F.A.; Jones, M.R.; Murray, J.E.; Veatch, O.; Lemke, C.; Saw, J.H.; Moore, R.C.; Thimmapuram, J. Low x/y divergence in four pairs of papaya sex-linked genes. Plant J. 2008, 53, 124–132. [Google Scholar] [CrossRef]
- Aradhya, M.K.; Manshardt, R.M.; Zee, F.; Morden, C.W. A phylogenetic analysis of the genus carica l.(caricaceae) based on restriction fragment length variation in a cpdna intergenic spacer region. Genet. Resour. Crop Evol. 1999, 46, 579–586. [Google Scholar] [CrossRef]
- Kim, M.; Moore, P.; Zee, F.; Fitch, M.M.; Steiger, D.; Manshardt, R.; Paull, R.; Drew, R.; Sekioka, T.; Ming, R. Genetic diversity of carica papaya as revealed by aflp markers. Genome 2002, 45, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Ming, R.; Yu, Q.; Moore, P. Papaya Genome and Genomics; Springer: New York, NY, USA, 2012; pp. 241–259. [Google Scholar]
- Yang, N.; Liu, J.; Gao, Q.; Gui, S.; Chen, L.; Yang, L.; Huang, J.; Deng, T.; Luo, J.; He, L. Genome assembly of a tropical maize inbred line provides insights into structural variation and crop improvement. Nat. Genet. 2019, 51, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Liao, Z.; Zhang, X.; Zhang, S.; Lin, Z.; Zhang, X.; Ming, R. Structural variations in papaya genomes. BMC Genom. 2021, 22, 335. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, S.; Castro, L.; Fermin, G.; Tennant, P. 10 chapter advances in papaya genomics. In Omics Technologies and Crop Improvement; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2014; pp. 219–252. [Google Scholar]
- Liao, Z.; Yu, Q.; Ming, R. Development of male-specific markers and identification of sex reversal mutants in papaya. Euphytica 2017, 213, 53. [Google Scholar] [CrossRef]
- Chávez-Pesqueira, M.; Núnez-Farfán, J. Genetic diversity and structure of wild populations of carica papaya in northern mesoamerica inferred by nuclear microsatellites and chloroplast markers. Ann. Bot. 2016, 118, 1293–1306. [Google Scholar] [CrossRef]
- Abberton, M.; Batley, J.; Bentley, A.; Bryant, J.; Cai, H.; Cockram, J.; Costa de Oliveira, A.; Cseke, L.J.; Dempewolf, H.; De Pace, C. Global agricultural intensification during climate change: A role for genomics. Plant Biotechnol. J. 2016, 14, 1095–1098. [Google Scholar] [CrossRef]
- Guerra-García, A.; Piñero, D. Current approaches and methods in plant domestication studies. Bot. Sci. 2017, 95, 345–362. [Google Scholar] [CrossRef][Green Version]
- Fitch, M.M.; Manshardt, R.M.; Gonsalves, D.; Slightom, J.L.; Sanford, J.C. Stable transformation of papaya via microprojectile bombardment. Plant Cell Rep. 1990, 9, 189–194. [Google Scholar] [CrossRef]
- Fitch, M.M.; Manshardt, R.M.; Gonsalves, D.; Slightom, J.L. Transgenic papaya plants from agrobacterium-mediated transformation of somatic embryos. Plant Cell Rep. 1993, 12, 245–249. [Google Scholar] [CrossRef]
- Manshardt, R. History and future of the solo papaya. In Genetics and Genomics of Papaya; Ming, R., Moore, P.H., Eds.; Springer: New York, NY, USA, 2014; pp. 95–113. [Google Scholar]
- Ye, C.; Li, H. 20 years of transgenic research in china for resistance to papaya ringspot virus. Transgenic Plant J. 2010, 4, 58–63. [Google Scholar]
- Randle, M.; Tennant, P. Transgenic papaya. In Genetically Modified Crops; Springer: Berlin/Heidelberg, Germany, 2021; pp. 129–160. [Google Scholar]
- Tecson Mendoza, E.M.; Antonio, C.L.; Botella, J.R. Recent advances in the development of transgenic papaya technology. Biotechnol. Annu. Rev. 2008, 14, 423–462. [Google Scholar] [CrossRef] [PubMed]
- Bau, H.-J.; Cheng, Y.-H.; Yu, T.-A.; Yang, J.-S.; Yeh, S.-D. Broad-spectrum resistance to different geographic strains of papaya ringspot virus in coat protein gene transgenic papaya. Phytopathology 2003, 93, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Ying, Z.; Yu, X.; Davis, M.J. New Method for Obtaining Transgenic Papaya Plants by Agrobacterium-Mediated Transformation of Somatic Embryos. Proc. Florida State Hortic Soc. 1999, 112, 201–205. [Google Scholar]
- Jefferson, R.A.; Kavanagh, T.A.; Bevan, M.W. Gus fusions: Beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J. 1987, 6, 3901–3907. [Google Scholar] [CrossRef]
- Stewart, C. The utility of green fluorescent protein in transgenic plants. Plant Cell Rep. 2001, 20, 376–382. [Google Scholar] [CrossRef]
- Zhu, Y.J.; Agbayani, R.; Jackson, M.C.; Tang, C.; Moore, P.H. Expression of the grapevine stilbene synthase gene vst1 in papaya provides increased resistance against diseases caused by phytophthora palmivora. Planta 2004, 220, 241–250. [Google Scholar] [CrossRef]
- McCafferty, H.R.K.; Moore, P.H.; Zhu, Y.J. Improved carica papaya tolerance to carmine spider mite by the expression of manduca sexta chitinase transgene. Transgenic Res. 2006, 15, 337–347. [Google Scholar] [CrossRef]
- McCafferty, H.R.K.; Moore, P.H.; Zhu, Y.J. Papaya transformed with the galanthus nivalis gna gene produces a biologically active lectin with spider mite control activity. Plant Sci. 2008, 175, 385–393. [Google Scholar] [CrossRef]
- Genglin, Z.; Peng, Z.; Anping, G.; Wentao, S.; Xiaoying, L. An initial study of transgenic carica papaya used as a kind of vaccine for anti tuberculosis. Acta Bot. Yunnanica 2003, 25, 223–229. [Google Scholar]
- CHEN, N.-M.; Paull, R. Development and prevention of chilling injury in papaya fruit. J. Am. Soc. Hortic. Sci. 1986, 111, 639–643. [Google Scholar] [CrossRef]
- Paull, R.E.; Nishijima, W.; Reyes, M.; Cavaletto, C. Postharvest handling and losses during marketing of papaya (Carica papaya L.). Postharvest Biol. Technol. 1997, 11, 165–179. [Google Scholar] [CrossRef]
- Lelièvre, J.M.; Latchè, A.; Jones, B.; Bouzayen, M.; Pech, J.C. Ethylene and fruit ripening. Physiol. Plant. 1997, 101, 727–739. [Google Scholar] [CrossRef]
- Trang, T.T.L.; Manshardt, R. A transgenic approach for determining sex of papaya seedlings. Acta Hortic. 2010, 851, 179–188. [Google Scholar] [CrossRef]
- Fitch, M.M.; Manshardt, R.M.; Gonsalves, D.; Slightom, J.L.; Sanford, J.C. Virus resistant papaya plants derived from tissues bombarded with the coat protein gene of papaya ringspot virus. Biotechnology 1992, 10, 1466–1472. [Google Scholar] [CrossRef]
- Yang, J.-S.; Yu, T.-A.; Cheng, Y.-H.; Yeh, S.-D. Transgenic papaya plants from agrobacterium-mediated transformation of petioles of in vitro propagated multishoots. Plant Cell Rep. 1996, 15, 459–464. [Google Scholar] [CrossRef]
- De la Fuente, J.M.; Ramírez-Rodríguez, V.; Cabrera-Ponce, J.L.; Herrera-Estrella, L. Aluminum tolerance in transgenic plants by alteration of citrate synthesis. Science 1997, 276, 1566–1568. [Google Scholar] [CrossRef]
- Zhu, Y.J.; Agbayani, R.; Moore, P.H. Ectopic expression of dahlia merckii defensin dmamp1 improves papaya resistance to phytophthora palmivora by reducing pathogen vigor. Planta 2007, 226, 87–97. [Google Scholar] [CrossRef]
- CS, D.; Samreen, F.; Prakash, J. A review on composition, processed products and medicinal uses of papaya (Carica papaya L.). Int. J. Food Sci. Nutr. 2015, 3, 99–117. [Google Scholar]
- Mahmood, A.; Sidik, K.; Salmah, I. Wound healing activity of Carica papaya L. Aqueous leaf extract in rats. Int. J. Mol. Med. Adv. Sci. 2005, 1, 398–401. [Google Scholar]
- Davison, J. Pickles: A Global History; Reaktion Books: London, UK, 2018. [Google Scholar]
- Saliasi, I.; Llodra, J.C.; Bravo, M.; Tramini, P.; Dussart, C.; Viennot, S.; Carrouel, F. Effect of a toothpaste/mouthwash containing carica papaya leaf extract on interdental gingival bleeding: A randomized controlled trial. Int. J. Environ. Res. Public Health 2018, 15, 2660. [Google Scholar] [CrossRef] [PubMed]


| Medicinal Property | Description | Treatment and Results | Reference |
|---|---|---|---|
| Anti-cancer | The aqueous extract increases the survival chances from cancers affecting the body’s vital parts like lungs, intestines, liver and blood. | Brewed extract of 50 times dilution with RPMI showed positive results for cell death in cancerous cell lines like AGS (stomach), Capan-1 (pancreas), Karpas (lymphoma), MCF-7 (breast). | [29] |
| Invitro treatments showed cytotoxic effects against benign, malignant cells of prostate origin. | Leaf extract (1–0.1 mg/mL) showed inhibitory and cytotoxic effects on prostate cell lines (RWPE-2, HPR-1, BPH-1, PC-3, DU-145 and LNCap) after 72 h of treatment. | [25] | |
| The component of leaf extract reduced adhesion and invasion of metastatic cancer cells in the extracellular matrix. | Leaf juice (3–30 µg/mL) was used for adhesion in PC-3 cells on wells with type 1 collagen and fibronectin. Dose dependant effect on anti-adhesion was observed. | [30,31] | |
| Suppression of DNA synthesis and inhibiting the proliferation of cancer cell lines in-vitro. AGS(stomach), DLD-1 (colon), MCF-7 (Breast), DOV-13 (Ovarian) | Suppressed DNA synthesis due to suppression in 3H-thymidine incorporation was observed by applying papaya extracts at (1.25–27 mg/mL) concentrations post 24 h exposure. | [31] | |
| Decrease in production of Interleukins-2,4 and Increased production of cytokines like TNF-α (tumour necrosis factor) | 0.125–0.5% papaya extract application to human peripheral blood mononuclear cells (PBMC) showed a decrease in interleukins and enhancement of cytokines. | [23] | |
| Immunomodulatory | Strong effects of immunomodulation effects are observed in the cancer cell lines. | Immunomodulatory effects were observed after treating the Wistar rat and albino mice through aqueous papaya extract (50–150 mg/kg). | [32,33,34] |
| Leaf extract mediated through ethanol had induced the TNF-α in lipopolysaccharide (LPS) induced dendritic cells | TNF-α in imDC (lipopolysaccharide (LPS) induced dendritic cells) was higher when treated along with papaya leaf extract (200 ng). | [35] | |
| Methanol-mediated papaya leaf extract had reported a decrease in the secretion of pro-inflammatory interleukins in human peripheral blood mononuclear cells (PBMC). | PBMCs stimulated by LPS at 0.1 µg/mL treated with the leaf extracts were analysed using ELISA and have shown positive inhibition of pro-inflammatory interleukins. | [36] | |
| The aqueous extract had increased the rate of Immunoglobulins IgG and IgM in acrylamide intoxicated mice trials. | Gastro gavaging fruit extract of 250 mg/kg orally in 0.05% acrylamide-induced Wistar albino mice showed an increase in immune functions (IgG and IgM). | [37] | |
| Anti-dengue virus | Oral administration of leaf extract significantly increased the count of platelets, RBC, and WBC in mice and humans. | Papaya leaf extract 0.2 mL or 2 g/mouse increased the platelet count from 3.4 × 105/µL (day 3) to 11.33 × 105/µL (day 21). RBC count increased from 6 × 106/µL to 9 × 106/µL. | [38,39,40,41] |
| The freeze-dried papaya leaf juice has proven to downregulate the inflammatory cytokines in mice infected with (Den-2) dengue virus. | The oral fed (500–2000 mg/kg body wt.) treatment for 3 days increased plasma CCL2/MCP-1 during the viremia peak and downregulation of 8 inflammatory cytokine genes. | [42] | |
| Controlled trials conducted in patients reported an improvement of mean blood platelets to count with the administration of leaf extract, which had a characteristic of membrane stabilization preventing platelet lysis. | 285 subjects with dengue fever were evaluated for treatment with Caripill syrup and showed a significant increase in mean platelet count from day 3 (89,739.31, p = 0.030) to day 5 (168,992, p = 0.023). | [40,43,44] | |
| Anti-diabetes | The papaya leaf extract has exhibited hypolipidemic and anti-hyperglycemic effects in diabetic rats. Similarly, the aqueous extract released excess insulin from the beta cells and reduced glucose and triacylglycerol concentrations in the blood. | Aqueous papaya extract (0.75 g–3 g/100 mL) has reduced the blood glucose levels in diabetic rats. Islet cells of the pancreas were regenerated and significant positive differences were observed in rats treated with extract compared to control rats. | [45] |
| The potential of fermented papaya extract to regulate blood glucose, inflammation, and free radical-induced oxidative damage, which cause liver, bladder, breast, and prostate cancer in type 2 diabetics, may ameliorate detrimental therapeutic effects. | FPP, a certified dietary supplement ISO 9002 produced by yeast fermentation, has antioxidants like superoxide, dimutase, catalase, glutathione peroxidase, therodoxine, and xanthine oxidase, helping in boosting immunity. These prevent the damage (oxidative stress, carcinogenic protein modifications, suppress DNA repair) from existing free radicals. | [46] | |
| Anti-anaemia | Papaya extracts exhibit Antisickling and membrane stabilizing properties. Fragiliograms revealed that leaf extract reduced hemolysis and preserved the integrity of the erythrocyte membrane. | Under high hypoxia, pretreatment of SS cell suspensions with leaf extract reduced sickle cell formation to 0–5% after 40 min compared to untreated SS cell suspensions with >60% sickle cells. These findings point to papaya leaf extract as a potential therapeutic against sickle cell disease. | [33] |
| The papaya leaf extract has reduced haemoglobin polymerization and osmotic fragility in human crescent RBCs which could be a potentially viable alternative to the hydroxyurea which is used in the therapy of sickle cell disease | RBCs were induced to sickling by 2% sodium metabisulfite. The percentage of sickle cells dropped from 91.6% to 47.6%, and the polymerization rate was 6 × 10−2 in the presence of hydroxyurea and papaya extract. | [47] | |
| Antibacterial | The most potent antibacterial action was found in papaya leaf extract, which significantly inhibited the growth of gram-positive bacteria (Pseudomonas aeruginosa, Bacillus subtilis, and Staphylococcus aureus). | 60% inhibition activity against bacterial growth has been observed through papaya extract application on Petri dishes. The average zone of inhibition (3.8 to 4 mm) for 75 µL concentration was reported. | [48] |
| Papaya leaves extracted with ethanol, methanol, ethyl acetate, acetone, and chloroform were strongly bactericidal against gram-negative bacteria (Bacillus cereus, Klebsiella pneumonia, Micrococcus luteus, Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus). | Phytochemical screening and antimicrobial activity for various solvent extractions of papaya leaf extract were investigated. The chloroform-mediated extract showed more activity against M. luteus (15.17 ± 0.29 mm), and acetone extraction showed (11.23 ± 0.25 mm) against C. albicans. | [49] | |
| Gastroprotective | Aqueous extract of Carica papaya leaf (CPL) decreased alcohol-induced acute stomach damage and blood oxidative stress in rats. The extract-treated rats had a lower stomach ulcer index than alcohol-treated controls, which could serve as an efficient therapeutic agent for treating gastritis and oxidative stress. | 500 mg/kg of aqueous CPL extract induced mucous production and HCO3- in the stomach and has successfully protected the gastric mucosal lining of mice against haemorrhages caused by alcohol. | [50] |
| The treatment of rats with experimental stomach ulcers with papaya leaf extract mediated by ethanol resulted in a considerable diminution in ulcers. The ulceration, volume of stomach acids, and pH of rats with aspirin-induced stomach ulcers increased. | The experiment involved rats induced by artificial ulcers and oedema and pellet granuloma were orally fed with 25–200 mg/kg of papaya leaf extract. Significant (p < 0.05) reduction in the symptoms was observed. | [51] |
| S.No. | Plant Parts | Chemical Constituent | Chemical Structure | Reference(s) |
|---|---|---|---|---|
| 1. | Leaves | Carpaine | 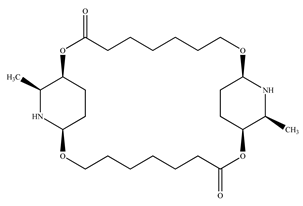 | [33,57,60,61,62] |
| Anthraquinone |  | |||
| Ferulic acid | 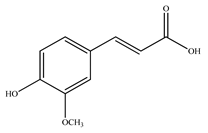 | |||
| Caffeic acid |  | |||
| Nicotinic acid | 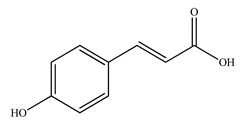 | |||
| n–Hexadecanoic acid (palmitic acid) |  | |||
| 2. | Fruit | Benzyl–β–D–glucoside | 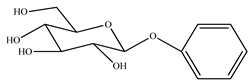 | [61,62] |
| Benzyl isothiocyanate (BITC) | 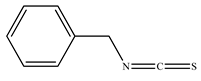 | |||
| β–Cryptoxanthin | 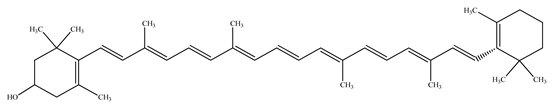 | |||
| β–Carotene | 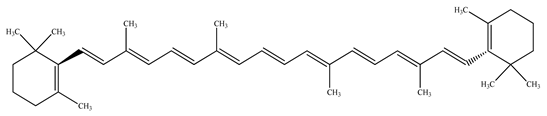 | |||
| Butanoic acid (butyric acid) | 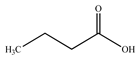 | |||
| Cis–Linalool oxide | 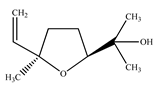 | |||
| Trans–Linalool oxide |  | |||
| Caricapinoside |  | |||
| 3. | Seeds | Benzyl isothiocyanate | 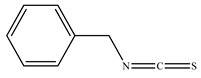 | [60,61,62,63,64] |
| Linoleic acid | 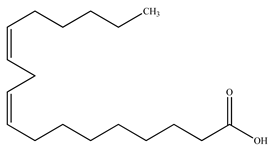 | |||
| Oleic acid | 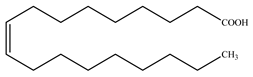 | |||
| Palmitic acid | 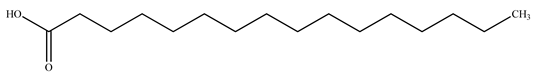 | |||
| Carpaine | 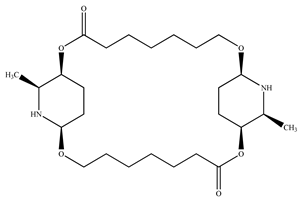 | |||
| Benzyl glucosinolate |  | |||
| Benzylthiourea |  | |||
| Hentriacontane |  | |||
| β–Sitosterol | 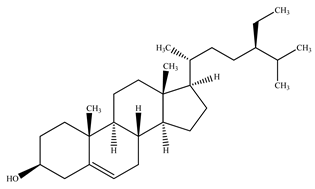 | |||
| 1,2,3,4–Tetrahydropyridin–3–yl–octanoate |  | |||
| 4. | Bark | β–Sitosterol |  | [61] |
| 5. | Juice | n–Butyric acid | 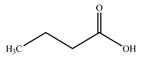 | [61,62] |
| n–Octanoic acid | 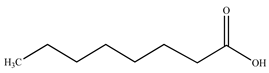 | |||
| Linoleic acid | 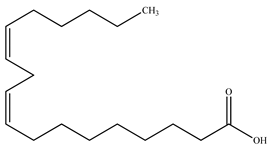 | |||
| Myristic acid | 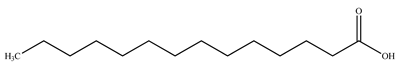 | |||
| Palmitic acid | 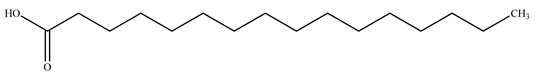 | |||
| Stearic acid | 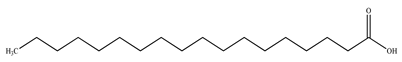 | |||
| Oleic acid | 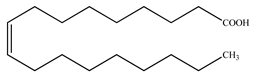 | |||
| Vaccenic acid | 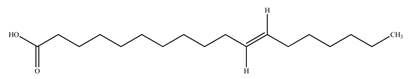 | |||
| n–Hexanoic acid | 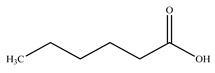 |
| Source | Gene(s) | Type of Transformation | Useful Trait Introduced | Reference |
|---|---|---|---|---|
| E. coli; E. coli K12 | uidA; nptII gene | Microprojectile-mediated transformation of zygotic/somatic embryos/freshly explanted hypocotyl sections | GUS expression; Kanamycin resistance | [133] |
| PRSV; E. coli; E. coli K12 | PRV cp gene; uidA; nptII gene | Microprojectile-mediated transformation of immature zygotic embryos | Resistance against PRSV | [151] |
| PRSV; E. coli; E. coli K12 | PRV cp gene; uidA; nptII gene | Agrobacterium-mediated transformation of somatic embryos | Resistance against PRSV | [134] |
| E. coli; E. coli K12 | uidA; nptII gene | Agrobacterium-mediated transformation of petioles | GUS expression; Kanamycin resistance | [152] |
| P. aeruginosa | CSb gene | Particle bombardment-mediated transformation | Al tolerance in tobacco and papaya | [153] |
| PRSV | Coat protein gene | Particle bombardment-mediated transformation of embryogenic tissues derived from immature zygotic embryos | Resistance against PRSV | [139] |
| M. tuberculosis | esat-6 gene | Agrobacterium-mediated transformation | Vaccine against tuberculosis in papaya | [146] |
| A. victoria | gpfp | Particle bombardment -mediated transformation of hypocotyl-derived embryogenic calli | GFP -mediated selection of transformants | [121] |
| Grapevine (V. vinifera L.) | Stilbene synthase gene (Vst 1) | Particle bombardment-mediated transformation of papaya embryogenic callus | Resistance against Phytophthora palmivora | [143] |
| M. sexta | Chitinase gene | Microprojectile bombardment of hypocotyl-derived embryogenic calli | Resistance to spider mites | [144] |
| D. pinnata | Defensin (DmAMP1) | Particle bombardment -mediated transformation of hypocotyl-derived embryogenic calli | Resistance to Phytophthora palmivora | [154] |
| G. nivalis | G. nivalis agglutin | Biolistic gene gun-mediated transformation of embryogenic calli | Resistance to spider mites | [145] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koul, B.; Pudhuvai, B.; Sharma, C.; Kumar, A.; Sharma, V.; Yadav, D.; Jin, J.-O. Carica papaya L.: A Tropical Fruit with Benefits beyond the Tropics. Diversity 2022, 14, 683. https://doi.org/10.3390/d14080683
Koul B, Pudhuvai B, Sharma C, Kumar A, Sharma V, Yadav D, Jin J-O. Carica papaya L.: A Tropical Fruit with Benefits beyond the Tropics. Diversity. 2022; 14(8):683. https://doi.org/10.3390/d14080683
Chicago/Turabian StyleKoul, Bhupendra, Baveesh Pudhuvai, Chelsi Sharma, Arvind Kumar, Vinay Sharma, Dhananjay Yadav, and Jun-O Jin. 2022. "Carica papaya L.: A Tropical Fruit with Benefits beyond the Tropics" Diversity 14, no. 8: 683. https://doi.org/10.3390/d14080683
APA StyleKoul, B., Pudhuvai, B., Sharma, C., Kumar, A., Sharma, V., Yadav, D., & Jin, J.-O. (2022). Carica papaya L.: A Tropical Fruit with Benefits beyond the Tropics. Diversity, 14(8), 683. https://doi.org/10.3390/d14080683









