Population Genetic Differentiation and Structure of Maruca vitrata (Lepidoptera: Crambidae) in India
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and DNA Extraction
2.2. Amplification and Sequencing of COI Gene Fragment
2.3. Sequence Data Analyses
3. Results
3.1. Variability in the Mitochondrial COI Gene
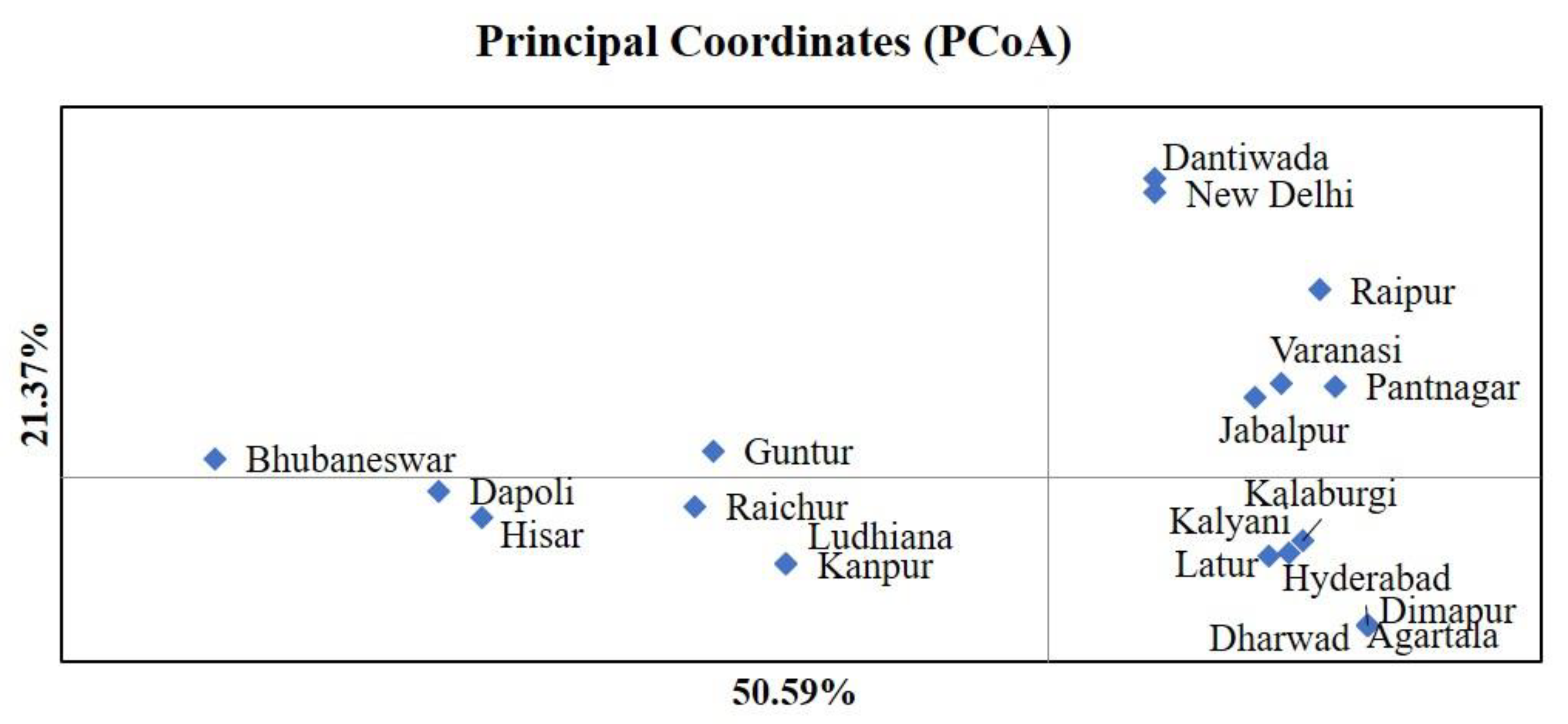
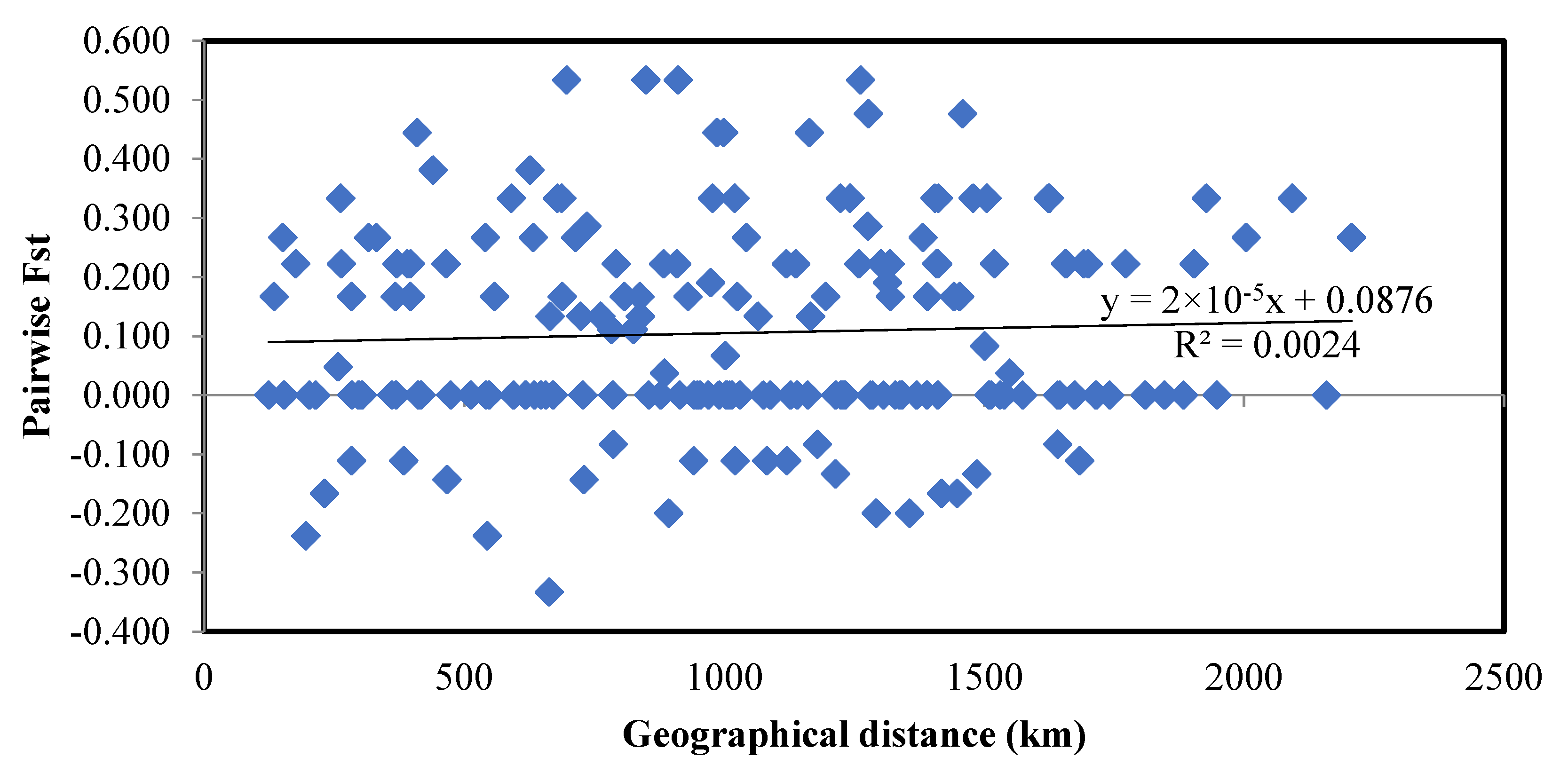
3.2. Demographic Inference and Population Structure
3.3. Haplotype Distribution and Phylogenetic Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Agunbiade, T.A.; Coates, B.S.; Sun, W.L.; Tsai, M.R.; Valero, M.C.; Tamò, M.; Pittendrigh, B.R. Comparison of the mitochondrial genomes of the old and new world strains of the legume pod borer, Maruca vitrata (Lepidoptera: Crambidae). Int. J. Trop. Insect Sci. 2017, 37, 125–136. [Google Scholar] [CrossRef]
- Margam, V.M.; Coates, B.S.; Ba, M.N.; Sun, W.; Binso-Dabire, C.L.; Baoua, I.; Ishiyaku, M.F.; Shukle, J.T.; Hellmich, R.L.; Covas, F.G.; et al. Geographic distribution of phylogenetically distinct Legume Pod Borer, Maruca vitrata (Lepidoptera: Pyraloidea: Crambidae). Mol. Biol. Rep. 2011, 38, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Agunbiade, T.A.; Coates, B.S.; Datinon, B.; Djouaka, R.; Sun, W.; Tamò, M.; Pittendrigh, B.R. Genetic Differentiation among Maruca vitrata F. (Lepidoptera: Crambidae) populations on cultivated cowpea and wild host plants: Implications for insect resistance management and biological control strategies. PLoS ONE 2014, 9, e92072. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.C. Bionomics, host plant resistance, and management of the legume pod borer, Maruca vitrata—A review. Crop Prot. 1998, 17, 373–386. [Google Scholar] [CrossRef]
- Chakravarty, S.; Agnihotri, M.; Jagdish, J. Seasonal abundance of predatory bugs, Eocanthecona furcellata (Wolff.) and Rhynocoris fuscipes (F.) and its olfactory responses towards plant and pest mediated semiochemical cues in pigeonpea ecosystem. Legum. Res. 2017, 40, 351–357. [Google Scholar] [CrossRef]
- Chatterjee, M.; Yadav, J.; Vennila, S.; Shashank, P.R.; Jaiswal, N.; Sreevathsa, R.; Rao, U. Diversity analysis reveals genetic homogeneity among Indian populations of legume pod borer, Maruca vitrata (F.). 3 Biotech 2019, 9, 319. [Google Scholar] [CrossRef]
- Srinivasan, R.; Tamò, M.; Malini, P. Emergence of Maruca vitrata as a major pest of food legumes and evolution of management practices in Asia and Africa. Annu. Rev. Entomol. 2021, 66, 141–161. [Google Scholar] [CrossRef]
- Mahalle, R.; Taggar, G. Insecticides against Maruca vitrata (Fabricius) (Lepidoptera: Crambidae) on pigeonpea. Pestic. Res. J. 2018, 30, 235. [Google Scholar] [CrossRef]
- Sambathkumar, S.; Durairaj, C.; Mohankumar, S.; Preetha, B.; Aravintharaj, R.; Ganapathy, N. Spatial genetic vriation in south Indian larval populations of legume pod borer, Maruca vitrata Geyer (Lepidoptera: Crambidae). Indian J. Exp. Biol. 2019, 57, 923–930. [Google Scholar]
- Periasamy, M.; Schafleitner, R.; Muthukalingan, K.; Ramasamy, S. Phylogeographical structure in mitochondrial DNA of legume pod borer (Maruca vitrata) population in tropical Asia and Sub-Saharan Africa. PLoS ONE 2015, 10, e0124057. [Google Scholar] [CrossRef]
- Malini, P.; Ramasamy, S.; Schafleitner, R.; Muthukalingan, K. Pheromone-binding proteins based phylogenetics and phylogeography of Maruca spp. from Asia, Africa, Oceania, and South America. Ecol. Evol. 2019, 9, 9239–9272. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, S.; Padwal, K.P.; Srivastava, C.P. Molecular Characterization of intraspecific variations in Helicoverpa armigera (Hübner) populations across India. J. Environ. Biol. 2021, 42, 1320–1329. [Google Scholar] [CrossRef]
- Karthika, P.; Vadivalagan, C.; Thirumurugan, D.; Murugan, K. Intra-species variation and geographic differentiation among the populations of the quarantine agricultural pest Leucinoides orbonalis (Lepidoptera: Crambidae) in the global assemblage—A prospective of DNA barcoding. Mitochondrial DNA Part A 2019, 30, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; deWaard, J.R. Biological identifications through DNA barcodes. Proc. Biol. Sci. 2003, 270, 313–321. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA Primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Hebert, P.D.N.; deWaard, J.R.; Landry, J.F. DNA Barcodes for 1/1000 of the animal kingdom. Biol. Lett. 2010, 6, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Shashank, P.R.; Thomas, A.; Ramamurthy, V.V. DNA Barcoding and Phylogenetic Relationships of Spodoptera litura and S. exigua (Lepidoptera: Noctuidae). Fla. Entomol. 2015, 98, 223–228. [Google Scholar] [CrossRef]
- Padwal, K.G.; Chakravarty, S.; Srivastava, C.P. Genetic variability and population structure of Leucinodes orbonalis (Guenée), a severe insect pest of brinjal in India. J. Environ. Biol. 2022, 43, 59–65. [Google Scholar] [CrossRef]
- Murray, M.G.; Thompson, W.F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 1980, 8, 4312–4325. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Librado, P.; Rozas, J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 2009, 25, 1451–1452. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Excoffier, L.; Lischer, H.E.L. Arlequin Suite Ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in excel. Population genetic software for teaching and research—An update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef] [PubMed]
- Bandelt, H.J.; Forster, P.; Röhl, A. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 1999, 16, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, J.; Chinna Babu Naik, V.; Ghodke, A.; Kranthi, S.; Kranthi, K.R.; Singh, B.P.; Choudhary, J.S.; Krishna, M.S.R. Population genetic structure of cotton pink bollworm, Pectinophora gossypiella (Saunders) (Lepidoptera: Gelechiidae) using mitochondrial cytochrome oxidase I (COI) gene sequences from India. Mitochondrial DNA Part A 2017, 28, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.C.; Sheppard, W.S. Phylogenetic relationships of honey bees (Hymenoptera: Apinae: Apini) inferred from nuclear and mitochondrial DNA sequence data. Mol. Phylogenet. Evol. 2005, 37, 25–35. [Google Scholar] [CrossRef]
- Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 1989, 123, 585–595. [Google Scholar] [CrossRef]
- Fu, Y.X. Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 1997, 147, 915–925. [Google Scholar] [CrossRef]
- Fu, Y.X. Statistical properties of segregating sites. Theor. Popul. Biol. 1995, 48, 172–197. [Google Scholar] [CrossRef]
- Hudson, R.R. Gene genealogies and the coalescent process. In Oxford Surveys in Evolutionary Biology; Futuyuma, D., Antonouics, J., Eds.; Oxford University Press: New York, NY, USA, 1990; Volume 7, pp. 1–44. [Google Scholar]
- Srivastava, C.P.; Pimbert, M.P.; Jadhav, D.R. Monitoring adult populations of Maruca testulalis (Geyer) with light traps at Patancheru and Hisar in India. Int. Pigeonpea Newsl. 1992, 15, 27–28. [Google Scholar]
- Taggar, G.K.; Singh, R.; Cheema, H.K.; Singh, P. Relative abundance, population dynamics and damage potential of spotted pod borer, Maruca vitrata (Fabricius) on early pigeonpea in Punjab. Int. J. Trop. Insect Sci. 2019, 39, 229–234. [Google Scholar] [CrossRef]
- Chaitanya, T.; Sreedevi, K.; Navatha, L.; Krishna, T.M.; Prasanti, L. Bionomics and population dynamics of legume pod borer, Maruca vitrata (Geyer) in Cajanus cajan (L.) Millsp. Curr. Biot. 2012, 5, 446–453. [Google Scholar]
- Sreekanth, M.; Ratnam, M.; Seshamahalakshmi, M.; Rao, Y.K.; Narayana, E. Population build-up and seasonal abundance of spotted pod borer, Maruca vitrata (Geyer) on pigeonpea (Cajanus cajan (L) Millsp.). J. App. Biol. Biotechnol. 2015, 3, 43–45. [Google Scholar] [CrossRef]
- Sampathkumar, S.; Durairaj, C. Relative abundance of legume pod borer, Maruca vitrata Geyer (Lepidoptera: Crambidae) on pigeonpea and its relationship with weather parameters. Madras Agric. J. 2015, 102, 67–70. [Google Scholar]
- Sambathkumar, S.; Durairaj, C.; Mohankumar, S.; Preetha, B.; Aravintharaj, R.; Ganapathy, N.; Surendran, R. Host induced genetic variation in legume pod borer, Maruca Vitrata. J. Environ. Biol. 2017, 38, 1281–1291. [Google Scholar] [CrossRef]
- Subramanian, S.; Mohankumar, S. Genetic variability of the bollworm, Helicoverpa armigera, occurring on different host plants. J. Insect Sci. 2006, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Emelianov, I.; Mallet, J.; Baltensweiler, W. Genetic differentiation in Zeiraphera diniana (Lepidoptera: Tortricidae, the Larch budmoth): Polymorphism, host races or sibling species? Heredity 1995, 75, 416–424. [Google Scholar] [CrossRef]
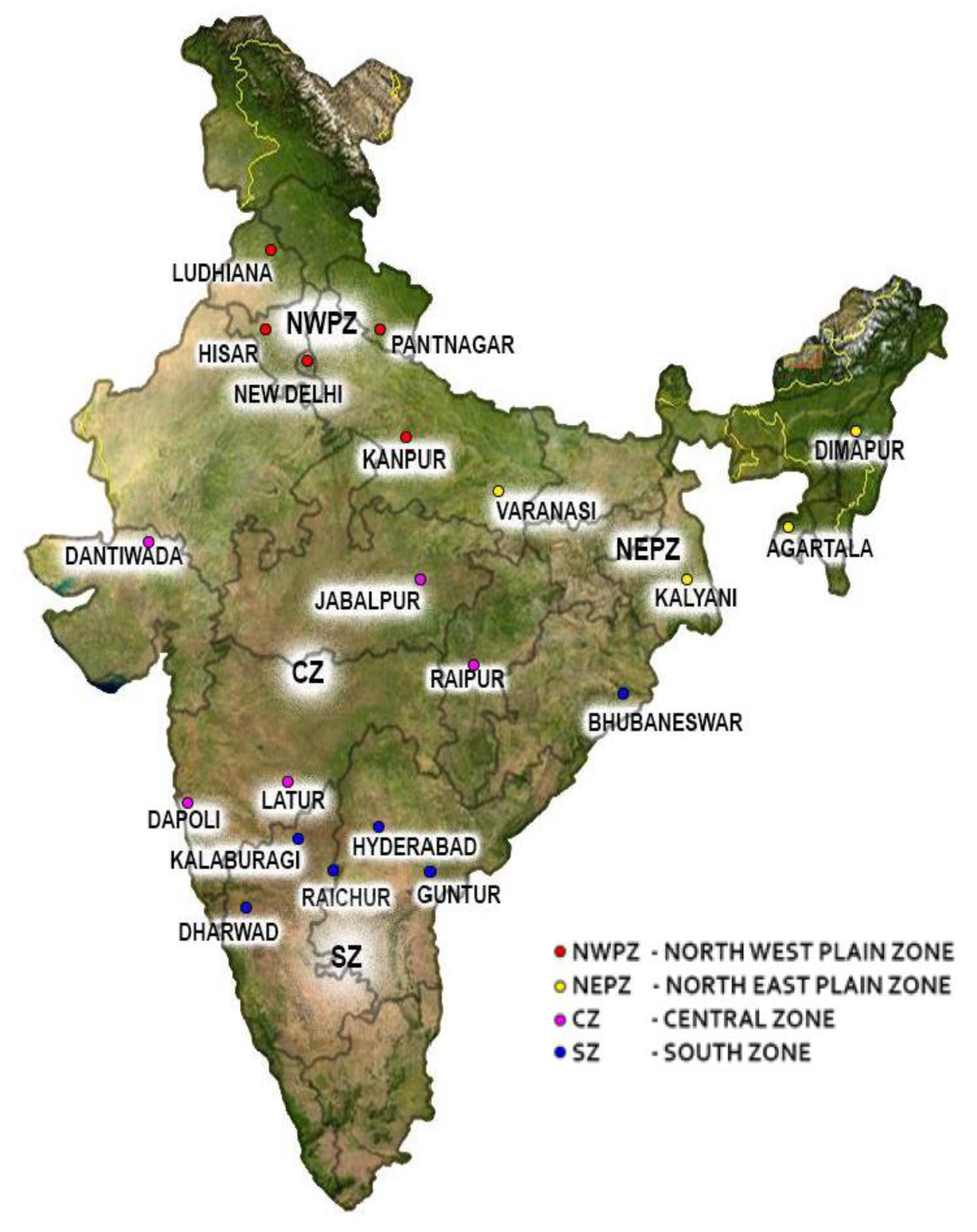



| Zones | Sample Code | Sampling Location (State) | Geographic Co-Ordinates | GenBank Accession Number(s) * |
|---|---|---|---|---|
| NWPZ (n = 20) | LDH | Ludhiana (Punjab) | 30.90° N, 75.81° E | MW417880, MW417881, MW417882, MW417883 |
| HSR | Hissar (Haryana) | 29.14° N, 75.71° E | MW417884, MW417885, MW417886, MW417887 | |
| NDLS | New Delhi | 28.64° N, 77.16° E | MW417888, MW417889, MW417890, MW417891 | |
| PBW | Pantnagar (Uttarakhand) | 29.02° N, 79.49° E | MW417892, MW417893, MW417894, MW417895 | |
| CNB | Kanpur (Uttar Pradesh) | 26.44° N, 80.33° E | MW417868, MW417869, MW417870, MW417871 | |
| NEPZ (n = 16) | BSB | Varanasi (Uttar Pradesh) | 25.27° N, 82.99° E | MW417872, MW417873, MW417874, MW417875 |
| KYI | Kalyani (West Bengal) | 22.94° N, 88.53° E | MW417876, MW417877, MW417878, MW417879 | |
| AGTL | Agartala (Tripura) | 23.91° N, 91.32° E | MW417864, MW417865, MW417866, MW417867 | |
| DMV | Dimapur (Nagaland) | 25.05° N, 93.03° E | MW417860, MW417861, MW417862, MW417863 | |
| CZ (n = 20) | DWZ | Dantiwada (Gujarat) | 24.32° N, 72.32° E | MW417912, MW417913, MW417914, MW417915 |
| JBP | Jabalpur (Madhya Pradesh) | 23.21° N, 79.95° E | MW417896, MW417897, MW417898, MW417899 | |
| R | Raipur (Chattisgarh) | 21.24° N, 81.70° E | MW417900, MW417901, MW417902, MW417903 | |
| LUR | Latur (Maharashtra) | 18.42° N, 76.61° E | MW417908, MW417909, MW417910, MW417911 | |
| DPLI | Dapoli (Maharashtra) | 17.75° N, 73.18° E | MW417904, MW417905, MW417906, MW417907 | |
| SZ (n = 24) | BBS | Bhubaneswar (Odisha) | 20.27° N, 85.81° E | MW417916, MW417917, MW417918, MW417919 |
| HYB | Hyderabad (Telangana) | 17.31° N, 78.16° E | MW417920, MW417921, MW417922, MW417923 | |
| GNT | Guntur (Andhra Pradesh) | 16.36° N, 80.43° E | MW417924, MW417925, MW417926, MW417927 | |
| KLBG | Kalaburagi (Karnataka) | 17.32° N, 76.84° E | MW417936, MW417937, MW417938, MW417939 | |
| RC | Raichur (Karnataka) | 16.20° N, 77.33° E | MW417928, MW417929, MW417930, MW417931 | |
| DWR | Dharwad (Karnataka) | 15.49° N, 74.98° E | MW417932, MW417933, MW417934, MW417935 |
| Index | M. vitrata Population | ||||
|---|---|---|---|---|---|
| NWPZ | NEPZ | CZ | SZ | Pooled | |
| N | 20 | 16 | 20 | 24 | 80 |
| Hn | 4 | 3 | 8 | 6 | 13 |
| Hd | 0.558 | 0.242 | 0.732 | 0.550 | 0.554 |
| Π | 0.00139 | 0.00060 | 0.00193 | 0.00127 | 0.00136 |
| K | 0.874 | 0.375 | 1.216 | 0.801 | 0.856 |
| S | 3 | 3 | 8 | 4 | 10 |
| Nucleotide composition (Relative values) | |||||
| A (%) | 30.95 | 30.94 | 30.92 | 30.92 | 30.93 |
| C (%) | 15.05 | 15.08 | 15.06 | 15.05 | 15.06 |
| G (%) | 14.60 | 14.61 | 14.63 | 14.63 | 14.62 |
| T (%) | 39.40 | 39.37 | 39.39 | 39.40 | 39.39 |
| A + T (%) | 70.35 | 70.31 | 70.31 | 70.32 | 70.32 |
| C + G (%) | 29.65 | 29.69 | 29.69 | 29.68 | 29.68 |
| Neutrality tests | |||||
| Fu’s Fs | −0.3174 | −0.8982 | −4.1487 | −2.6631 | −9.6793 |
| Tajima’s D | −0.0087 | −1.6965 | −1.5532 | −0.6905 | −1.5224 |
| Fu and Li’s D | −1.0065 | −2.2045 * | −1.4854 | −0.8560 | −1.3226 |
| Fu and Li’s F | −0.8685 | −2.3662 * | −1.7409 | −0.9355 | −1.6481 |
| Source of Variation | df | Sum of Squares | Variance Components | Percentage Variation | Fixation Indices |
|---|---|---|---|---|---|
| Among groups (zones) | 3 | 1.942 | 0.00231 | 0.54 | FCT: 0.00537 (p > 0.10) |
| Among populations within groups | 16 | 11.562 | 0.05762 | 13.37 | FSC: 0.13448 (p > 0.05) |
| Within populations | 60 | 22.250 | 0.37083 | 86.09 | FST: 0.13912 (p > 0.05) |
| Total | 79 | 33.812 | 0.43026 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahalle, R.M.; Chakravarty, S.; Srivastava, C.P. Population Genetic Differentiation and Structure of Maruca vitrata (Lepidoptera: Crambidae) in India. Diversity 2022, 14, 546. https://doi.org/10.3390/d14070546
Mahalle RM, Chakravarty S, Srivastava CP. Population Genetic Differentiation and Structure of Maruca vitrata (Lepidoptera: Crambidae) in India. Diversity. 2022; 14(7):546. https://doi.org/10.3390/d14070546
Chicago/Turabian StyleMahalle, Rashmi Manohar, Snehel Chakravarty, and Chandra Prakash Srivastava. 2022. "Population Genetic Differentiation and Structure of Maruca vitrata (Lepidoptera: Crambidae) in India" Diversity 14, no. 7: 546. https://doi.org/10.3390/d14070546
APA StyleMahalle, R. M., Chakravarty, S., & Srivastava, C. P. (2022). Population Genetic Differentiation and Structure of Maruca vitrata (Lepidoptera: Crambidae) in India. Diversity, 14(7), 546. https://doi.org/10.3390/d14070546





