Abstract
The increasing availability of field data presents an opportunity to understand the fundamental ecological relationships and functions of large trees in tropical forests at regional and global scales. However, it is not always clear what the relationships or patterns of diversity and structure are among sites in different biogeographic regions. We evaluated the relationship of the biomass and diameter of the largest trees with a diversity of species and compared, between the sites, the attributes of structure, diversity, and the influence of the 50 hyperdominant species in each site, aiming at the potential formation of groups by sites with characteristics and patterns of similar diversity within biogeographic regions. The average wood density together with the diversity of genera and families are the most important attributes to discriminate biogeographic regions when considering all forest information. Large trees play a fundamental role in forest ecology and seem to express regional environmental characteristics. The upper canopy of tropical forests remains one of the least studied environments in all terrestrial biomes, and is often referred to as “the last biotic frontier” or a “black box,” and large trees are also part of this mysterious frontier.
1. Introduction
The mega-biota has massive importance for biosphere function [1,2] and human living [3]. Big-sized trees in forests regulate resources such as light, water, and soil nutrients, hence the growth and performance of medium-sized and small-sized trees. Big trees are usually associated with areas with high conservation value, since they provide habitat and food for many organisms [4] and store a large amount of biomass [1]. Big trees also influence forest recovery, since they are linked with forest structural attributes [5] and act as nuclei of forest regeneration. Big trees help to kick-start succession by improving microclimate and soil conditions, attracting frugivorous seed dispersers, and favoring the regeneration of old-growth species [5].
Due to buttresses and other trunk anomalies, big trees are much harder to measure accurately than smaller ones [3]. The challenges persist with other attributes, such as height and crown diameter. However, the relative rarity of big trees in the forest, compared to smaller trees, also brings advantages. Species identification is more accessible than that for smaller trees [6], due to the restricted species pool. It is much easier to memorize specific trunk and bark characteristics for big trees.
The sheer size of the Amazon, its environmental heterogeneity and species diversity pose challenges and practical difficulties in understanding general ecological relationships and biogeographic patterns [7]. The Amazon already has well-documented gradients affecting forest composition and structure: one from the Guiana Shield to the Southwestern Amazon, related to variation in soil fertility, and another gradient from Colombia to the Southeastern Amazon, related to the length of the dry season [8,9,10]. However, for big trees, simple questions, e.g., “How many species become big trees?”, “What species of big trees are more abundant?” and “What is governing the distribution of the big trees?”, are still open and intriguing to scientists. Previous studies, for example, have documented that big trees seem to be more abundant in Africa [11], and they seem to contribute more to the total biomass than the same class of trees in Asia and South America [12].
From the perspective of biodiversity conservation, the communities and structures of large trees are fundamental, mainly for the maintenance and functioning of the ecosystem (by conserving and increasing carbon storage and productivity), and, above all, by providing insights for a better understanding of the role of each large species in biogeochemical cycles [13]. In this study, we analyzed the primary relationships between large trees’ diversity and structure attributes. We identified which patterns and dominant large species correspond to the biogeographic regions proposed by Morrone [14]. We used individual tree characteristics (diameter, biomass, and wood density) and a diversity index at the plot and site levels. Since the complementarity of the values of these characteristics can inform us about a positive influence on biogeographic patterns [15] and are convincing hypotheses that deserve to be tested, we evaluated how the attributes of structure, diversity, and dominance of the leading large species are related to representing which sites have the most similar ecological patterns and characteristics [16,17], and how these patterns are essential in explaining the defined biogeographic regions. Using this relational assessment, we investigated the following research questions: (a) What is the relationship between species diversity and richness and the size of large trees? With large species being more susceptible to biodiversity loss, more significant insights into the functional importance of these species in local biodiversity and biomass are imperative. Recent studies in a tropical forest in Congo [18] and temperate forests in North America [19] showed how large species contribute disproportionately to an ecosystem’s functional diversity, structural heterogeneity, and ecological importance. Investigating whether these results are valid in the Amazon, regarding species composition, forest structure, and biogeographic conditions, is particularly important to provide more information about the need for biodiversity conservation for ecosystem functioning. (b) What is the relationship between the proportion of large species richness and its contribution to community biomass storage? In terms of planning for forest carbon mitigation and biodiversity conservation, identifying whether a potential relationship exists will allow for integrated forest management planning, or will highlight the need for similar tactics in terms of management for carbon mitigation and management for biodiversity conservation [18]. (c) How do the attributes of structure and diversity potentially explain sites with similar patterns and biogeographic regions in Amazonia? We discuss which locations with large trees present similar patterns of diversity and structure and how these locations are distributed in biogeographic regions. This study focused on the ForestPlots network data available for the Brazilian Amazon (availability data on 10 October 2021) and categorized individuals over 70 cm in diameter with a breast height more than 1.3 m as large trees.
2. Materials and Methods
2.1. Study Area
This study was based on the ForestPlots network (Figure 1), including recent plots where giant trees have been reported in the Brazilian Amazon [20] (https://www.forestplots.net/, (accessed on 10 October 2021)). The plots were monitored by different research teams (Supplementary Materials Appendix S3). Within the plots, each tree had its diameter (d) recorded. We filtered out plots where less than 90% of the trees had been identified, at least at the genus level. Botanical identification was harmonized using the Taxonomic Name Resolution Service (https://tnrs.iplantcollaborative.org (accessed on 10 October 2021)), Missouri Botanical Garden (https://www.missouribotanicalgarden.org/ (accessed on 10 October 2021)), and Flora package in R.
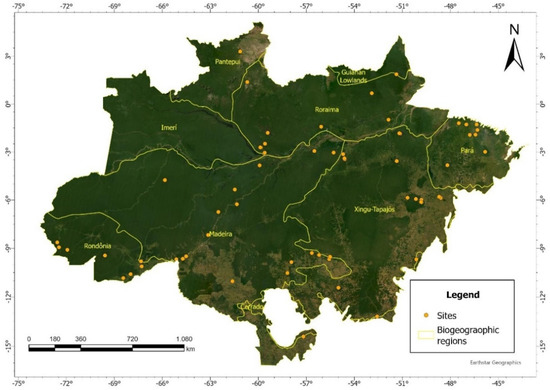
Figure 1.
Distribution of plots in the Brazilian Amazon and delimitation of the biogeographic regions proposed by Morrone [14].
2.2. Data Analysis
Each tree in the dataset was associated with a basic wood density using the lowest available taxonomic level of botanical identifications (i.e., species or genus) and the corresponding average wood density recorded in the Global Wood Density Database [21,22]. The plots were totalized in terms of structure and diversity. The diversity was computed using the Shannon index for species, genus, and family levels. The structural attributes were computed as mean, maximum, and standard deviation of diameter, and mean and standard deviation of basic wood density. The individual tree-level biomass for each site was obtained using the pan-tropical allometric model developed by [23]:
where D is in cm, H is in m, and ρ is the basic density of wood in g cm3. This model performed well in all forest types and bioclimatic conditions [23].
AGBest = 0.673 × (ρD2H)0.976; σ = 0.357, AIC = 3130, df = 4002
The analysis considered two contexts for the recorded trees: (1) all trees with diameter greater than 10 cm, and (2) only the big trees (diameter greater than 70 cm) (Figure 2). The threshold of 70 cm for diameter was suggested by Harris et al. [3] to define big trees in tropical forests. For both contexts (all trees and big trees), we computed the average of the diversity, richness, and structural attributes for each site. We estimated the forest diversity, characterizing the communities in terms of species, genus, and family composition [24]. Diversity was calculated using the Shannon index (H’). A species accumulation curve was used to compare the theoretical and actual numbers of recorded species [25]. The top 50 abundant species were also retained to describe the sites in terms of relative density. In total, we computed 116 variables (100 of species density, 6 of diversity, and 10 of structure).
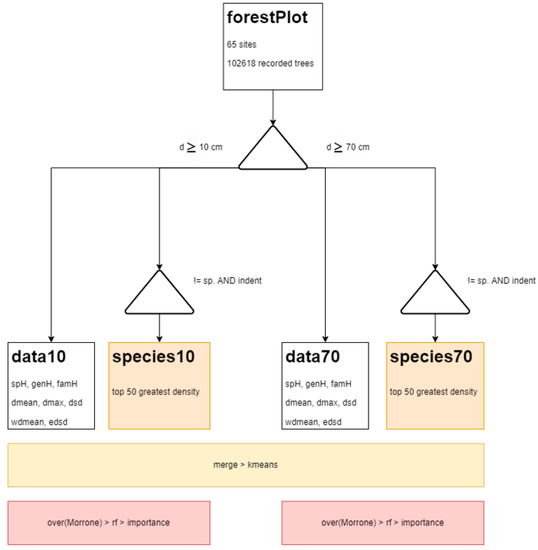
Figure 2.
Processing flowchart. dmean, dmax, and dsd are the mean, maximum, and standard deviation of the diameters, respectively.
We implemented a k-means analysis with increasing numbers of clusters to groups based on the attributes of structure (dbh and wood density) and diversity (species, genus, and family diversity), and the relative density of top 50 species for all trees (dbh ≥ 10 cm) and big trees (dbh ≥ 70 cm). The algorithm compared the source dataset and synthetic datasets with similar distribution properties to determine the interrelationships between the attributes through their dimensions (components).
Based on its geolocation, each site was associated with one biogeographic region, as proposed by Morrone [14]. Morrone’s regions considered terrestrial plant and animal taxa to divide the Brazilian Amazon into eight regions. We excluded three biogeographic regions (i.e., Pantepui, Guianan Lowlands, and Cerrado) due to single-site occurrence. We used the biogeographic regions to extract structural and diversity patters based on the big tree dataset. The attributes were used to classify the regions using a random forest algorithm, followed by analysis of importance of the variables. We compared the patterns extracted from big trees (d ≥ 70 cm) to patterns derived from all recorded trees (d ≥ 10 cm).
We computed variable importance by permuting data. For each tree, the prediction error on the out-of-bag portion of the data is recorded (error rate for classification). Then, the same is conducted after permuting each predictor variable. The difference between the two are then averaged over all trees and normalized by the standard deviation of the differences. We considered both variable importance indicators: decrease in average accuracy and decrease in Gini importance (mean decrease in impurity). The analyses were performed using R software with the packages randomForest and terra.
3. Results
The plot network recorded 102618 trees, divided into 239 plots, representing 65 sites distributed in the Brazilian Amazon. Big trees (DBH ≥ 70 cm) identified at the species level were present in 222 of the plots, covering 62 sites. The 3900 big trees represented 3.8% of the trees measured and were distributed across 344 species. The number of trees reaching the big-sized classification ranged from 0 to 46 individuals inside a single plot. The highest density of big trees was found in the State Forest of Amapá (FEA) (Appendix S1).
The most abundant species were Goupia glabra (6.9%, n = 267), Dinizia excelsa (2.56%, n = 99), Aspidosperma excelsum (2.4%, n = 94), Nectandra rubra (2.3%, n = 89), Minquartia guianensis (2.25%, n = 87), Couratari guianensis (2.14%, n = 83), Conceveiba guianensis (2.07%, n = 80), Manilkara huberi (1.88%, n = 73), Eperua rubiginosa (1.65%, n = 64), Micropholis venulosa (1.63%, n = 63), Dipteryx odorata (1.47%, n = 57), Tabebuia serratifolia (1.44%, n = 56), Qualea albiflora (1.42%, n = 55), Bertholletia excelsa (1.39%, n = 54), Caryocar villosum (1.37%, n = 53), Sloanea grandiflora (1.32%, n = 51), and Eschweilera coriacea (1.29%, n = 50). The other 82 records (2.12%) were associated with the genus Eschweilera, not identified at the species level.
The species accumulation curve, considering all plots, was not stabilized (Figure 3A), indicating that expanding the sampling area would potentially include new species. There was a negative relationship between the diversity of big trees and the maximum diameter (r = −0.36, p < 0.001; Figure 3B). Many species can have individuals reaching 70 cm in diameter. However, with the higher diameter, fewer species were recorded. Only 20% of the big tree species reached classes above 115 cm (Figure 3C). A stronger positive relationship was observed between species richness and the proportion of biomass in big trees (r = 0.5 p < 0.001; Figure 3D), and 75% of the biomass was concentrated in less than 10% of big tree species. The increase in the occurrence of big tree species resulted in a significant increase in total biomass.
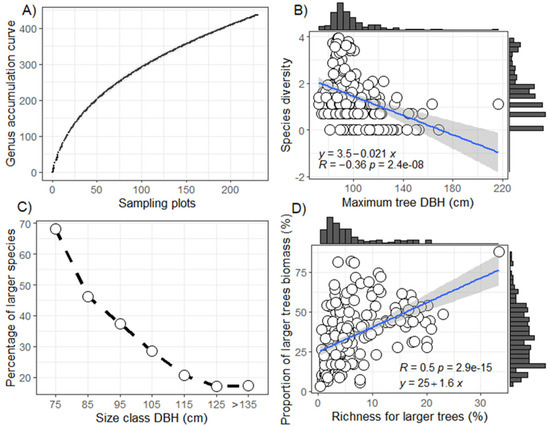
Figure 3.
Richness and diversity estimates and their relationship with dendrometric attributes of the big trees. Panel (A) shows the species accumulation curve by sampled area. Panel (B) shows the relationship of species diversity with the largest diameters per plot. Panel (C) indicates the percentage of species by diameter class. Panel (D) shows the proportion of biomass of the big trees in relation to the percentage of species richness of big trees per plot. Marginal histograms indicate frequency.
The 116 attributes were computed for each site, considering trees equal to or greater than 10 cm, and a subset of trees equal to or greater than 70 cm. The k-means algorithm and the optimal gap statistic indicated the formation of four groups (Figure 4). The k-means algorithm produced two significant axes that explained 75% of the total variance, separating the sites into four distinct groups. The FEA site formed a single group, strongly influenced by the high diversity and abundance of big trees. An intermediary group had the largest number of sites (n = 59) and split into two other groups on a gradient related to diversity and biomass.
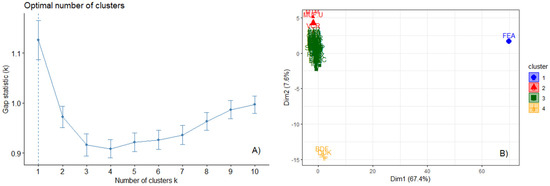
Figure 4.
(A) Distinction and formation of groups by sites with similar characteristics (structure and diversity attributes) generated by k-means. (B) The percentage of variation explained by the eigenvalues of the formed groups was 75%.
The first k-means component was dominated by information related to big tree strata of the forest. A total of 49 of the 50 most abundant big tree species contributed with high loadings, together with 30 of the 50 most abundant trees and the mean diameter of all trees above 10 cm in diameter. The second component was related to the diversity and biomass. This second component highlighted the influence of 10 species that were not registered as big trees in the database.
The random forest plot indicated that average wood biomass was the most important attribute to characterize the biogeographic regions proposed by Morrone [14]. The second most important attribute was the relative abundance of Minquartia guianensis. The diversity of species, genera, and families showed intermediary importance, as well as structural attributes. Besides Minquartia guianensis, two other species were highly important to biogeographic determination: Eschweilera coriacea and Goupia glabra (Figures S2 and S3).
4. Discussion
Populations of big trees are not homogeneous within the Brazilian Amazon, and their abundance (or absence) may express environmental and ecological differences in biogeographic regions. Exploring patterns of big trees is very important to forest ecology. Identifying big trees is usually easier than identifying smaller trees [6], and on the other side, the importance of big trees has been subject to recent discoveries [1,5,26].
Of the 102618 trees recorded in the database used in this study, 3.8% represented big trees (3900 trees). Our proportion was superior to values observed in Africa, where Harris et al. [3] registered 2.6% (118 out of 4560 trees). We recorded 2374 species of tree above 10 cm dbh and 344 species (14%) of trees that could reach ≥70 cm. In a similar study in Africa, Harris et al. [3] recorded 400 species, and 92 species (~25%) that could reach dbh ≥ 80 cm. The number of species and maximum diameter were inversely proportional, reinforcing the idea that the big tree species pool is more restricted [3]. The range of big trees per hectare in Africa varies between 5 and 11.7, while we observed a more comprehensive range in Brazil—0 to 46.
Larger individuals are often considered the strongest competitor [27], and they are the main contributor to woody biomass production, highlighting the importance of big trees to biogeochemical cycling. A previous study correlated the above-ground biomass to the size of big trees [1]. However, we expanded the importance of big trees, noticing the dominance of some species and a positive relationship between big tree diversity and above-ground total biomass.
We found four distinct patterns driven by the structure and diversity attributes derived from big trees (Figure 4 and Appendix S4). However, those four patterns did not match the boundaries of the biogeographic region. Our results showed a big average group formed by 49 out of 65 sites (Group 3—centered in both k-means dimensions), while three other groups occupied the extremes of the biplot dimensions (Figure 4B). Groups 2 and 4 dominated the opposite extremes of the second-dimension axis (diversity and biomass), and group 1 was located at the extreme of the first-dimension axis (big trees stratum).
The Northeastern Amazon site exhibits patterns of diversity and structure that are more evident than in other regions (Figure 4B, Group 1). It is not clear why this region has such strong patterns of dominance in biomass, diversity, and structure of big trees. It may be related to the better conservation status, as this sub-region has been less exposed to intense anthropogenic activities than other regions [28]. However, this may also be partly due to the lower environmental heterogeneity in this region, which contains relatively few variations in altitude, precipitation, soil, and temperature [29]. Recognizing and documenting those differences is important, because several studies have focused on dominance patterns in Southeastern Amazonia [30,31,32], which may not represent the Amazon [33].
Although the natural patterns extracted by k-means did not match the boundaries of the biogeographic regions proposed by Morrone, it was possible to extract unique characteristics in terms of big tree populations. The Madeira region is governed by Alexa grandiflora, Hymenolobium petraeum, and Minquartia guianensis. The Pará region has the most diverse dominance, composed mainly of Bertholletia excelsa, Carapa guianensis, Caryocar glabrum, Eschweilera coriacea, Qualea albiflora, and Symphonia globulifera. The Xingu-Tapajos region is dominated by Alexa grandiflora, Caryocar glabrum, Goupia glabra, and Hymenaea courbaril. Rondônia is dominated by Caryocar glabrum, Goupia glabra, Protium decandrum, Symphonia globulifera, and Tabebuia serratifolia. Roraima province is characterized by Dinizia excelsa, Minquartia guianensis, and Osteophloeum platyspermum (Figure S3). Those biogeographic regions have already been related to other characteristics, such as forest height [34,35], structural trends for biomass [36,37], and diversity [17,38,39,40]. Related diversity patterns and species ecology to already known patterns could improve forest ecology comprehension.
Several species of trees coexist around big trees, and this is intrinsically associated with their three-dimensional shape (biomass, diameter, and height), producing direct and indirect effects on the structure and diversity of tropical forests [26,41]. In general, the tree biomass, height, and shape vary greatly over large ecological gradients [42], and due to competition for light and nutrients [43,44]. Big trees act by filtering sunlight and direct precipitation above the forest canopy [45], regulating the space and spatial distribution of several other species’ floristic composition and richness [46]. Big trees are long-term survivors, suggesting that they may be more adapted to local factors [46]. Consequently, big trees themselves may be considered natural indicators, expressing the differences between regions and reflecting forest ecology singularities.
In addition to supporting biodiversity, species with large trees may be inextricably linked to essential ecosystem services that tropical forests provide locally and globally. For example, these larger trees contain the highest proportion of carbon in forests, even though the highest densities of trees within a given ecosystem type are often associated with young or recovering forests, characterized by many small trees. The loss of such fundamental resources—for example, through selective logging—can lead to the functional collapse of entire forest ecosystems due to impacts at various trophic levels.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/d14070503/s1: Figure S1: Loadings for the attributes forming the two main k-means components. Figure S2: Importance of the attributes based on the random forest modeling for the big tree subset (only trees above 70 cm in diameter). Figure S3: Importance of the attributes based on the random forest modeling for the forest (all trees above 10 cm in diameter).
Author Contributions
Conceptualization, data curation, and formal analysis: R.B.d.L. and E.B.G.; funding acquisition: E.B.G., D.A.S.d.S. and R.B.d.L.; investigation, methodology, validation, visualization, and project administration: R.B.d.L., E.B.G., A.P.B.B., D.A.S.d.S., C.P.d.O. and C.S.C.d.S.; writing—original draft: R.B.d.L. and E.B.G.; writing—review and editing: R.B.d.L., E.B.G. and A.P.B.B. All authors have read and agreed to the published version of the manuscript.
Funding
This project was funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior Brasil (CAPES; Finance Code 001); Conselho Nacional de Desenvolvimento Científico e Tecnológico (Processes 403297/2016-8 and 301661/2019-7) and (Processes 550467/2010-6); Amazon Fund (grant 14.2.0929.1); Fundo Iratapuru; and Universidade do Estado do Amapá (Processes 0022.0279.1202.0018/2021).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are available at https://doi.org/10.5061/dryad.4xgxd25cg (accessed on 10 October 2021).
Acknowledgments
We thank ForestPlots.net (https://forestplots.net/secure/plots) and the Principal Investigators listed below for making their plot data available for analysis: Marcelo Fragomeni Simon, Marcelo Brilhante de Medeiros, Ted Feldpausch, Luiz Aragão, Laura Vedovato, Paulo Morandi, Beatriz Marimon, Ben Hur Marimon Junior, Marco Leandro Garcia , Leandro S Charão, Ima Célia Guimares Vieira, Aretha Franklin Guimarães, Clarissa Alves Rosa, Wagner Tadeu Vieira Santiago, William Laurance, Susan Laurance, Ana Andrade, José Luís Camargo, Niro Higuchi, Fernando Elias, Joice Ferreira, Jos Barlow, Oliver Phillips, Ângelo Gilberto Manzatto, Samuel de Padua Chaves e Carvalho, Lilian Blanc, Plinio Sist, Lucas Jose Mazzei de Freitas, Estelle Dugachard, Alan Bernardes da Silveira, Domingos de Jesus Rodrigues, Rainiellen de Sá Carpanedo, Larissa Cavalheiro, Flávia Rodrigues Barbosa, Ana Paula Zopeletto Massing, Gabriel de Oliveira de Almeida, Fernanda Coelho, Carolina Castilho, William E. Magnusson, Raimunda Oliveira de Araújo, Karina Melgaço, Flávia Costa, Flávia Delgado Santana, Reinaldo Barbosa, Marcelino Carneiro Guedes, Perseu da Silva Aparício, Wegliane Campelo da Silva, Jadson Coelho de Abreu, Thaiane Rodrigues de Sousa, Juliana Schietti, Adriane Esquivel Muelbert, Maryane Bento Trindade de Andrade, Philip Fearnside, Antonio S Lima, Pedro Lisboa, Natalino Silva, Marcos Silveira, Wendeson Castro, Rafael Salomão, Angélica Faria de Resende, Bruce Walker Nelson, Danilo Roberti Alves de Almeida, Douglas C Daly, Ghillean T. Prance, Edmar Almeida de Oliveira, Erika Berenguer, Toby Gardner, Ana Albernaz, Letícia Fernandes, William Balee, Yadvinder Malhi, Leandro Ferreira, Iêda Leão do Amaral, Celice Alexandre Silva, Robson Dos Santos Alves da Silva, Maíra Benchimol, Carlos Peres, Danielle Storck-Tonon, Marcos Salgado Vital, Ricardo de Oliveira Perdiz.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Enquist, B.J.; Abraham, A.J.; Harfoot, M.B.J.; Malhi, Y.; Doughty, C.E. The Megabiota Are Disproportionately Important for Biosphere Functioning. Nat. Commun. 2020, 11, 699. [Google Scholar] [CrossRef] [PubMed]
- Schweiger, A.H.; Svenning, J.-C. Analogous Losses of Large Animals and Trees, Socio-Ecological Consequences, and an Integrative Framework for Rewilding-Based Megabiota Restoration. People Nat. 2020, 2, 29–41. [Google Scholar] [CrossRef]
- Harris, D.J.; Ndolo Ebika, S.T.; Sanz, C.M.; Madingou, M.P.N.; Morgan, D.B. Large Trees in Tropical Rain Forests Require Big Plots. Plants People Planet 2021, 3, 282–294. [Google Scholar] [CrossRef]
- Ali, A.; Wang, L.-Q. Big-Sized Trees and Forest Functioning: Current Knowledge and Future Perspectives. Ecol. Indic. 2021, 127, 107760. [Google Scholar] [CrossRef]
- Poorter, L.; Craven, D.; Jakovac, C.; van der Sande, M.; Amissah, L.; Bongers, F.; Chazdon, R.; Farrior, C.; Kambach, S.; Meave, J.; et al. Multidimensional Tropical Forest Recovery. Science 2021, 374, 1370–1376. [Google Scholar] [CrossRef]
- Réjou-Méchain, M.; Fayolle, A.; Nasi, R.; Gourlet-Fleury, S.; Doucet, J.-L.; Gallye, M.; Hubertb, D.; Pasquier, A.; Billandb, A. Detecting Large-Scale Diversity Patterns in Tropical Trees Can We Trust Commercial Forest Inventories? For. Ecol. Manag. 2011, 261, 9. [Google Scholar] [CrossRef]
- Tuomisto, H.; Van doninck, J.; Ruokolainen, K.; Moulatlet, G.M.; Figueiredo, F.O.G.; Sirén, A.; Cárdenas, G.; Lehtonen, S.; Zuquim, G. Discovering Floristic and Geoecological Gradients across Amazonia. J. Biogeogr. 2019, 46, 1734–1748. [Google Scholar] [CrossRef]
- Baker, T.R.; Phillips, O.L.; Malhi, Y.; Almeida, S.; Arroyo, L.; Fiore, A.D.; Erwin, T.; Higuchi, N.; Killeen, T.J.; Laurance, S.G.; et al. Increasing Biomass in Amazonian Forest Plots. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2004, 359, 353–365. [Google Scholar] [CrossRef]
- ter Steege, H.; Pitman, N.; Phillips, O.; Chave, J.; Sabatier, D.; Duque, A.; Molino, J.-F.; Prévost, M.-F.; Spichiger, R.; Castellanos, H.; et al. Continental-Scale Patterns of Canopy Tree Composition and Function Across Amazonia. Nature 2006, 443, 444–447. [Google Scholar] [CrossRef]
- Malhi, Y.; Aragão, L.E.O.C.; Galbraith, D.; Huntingford, C.; Fisher, R.; Zelazowski, P.; Sitch, S.; McSweeney, C.; Meir, P. Exploring the Likelihood and Mechanism of a Climate-Change-Induced Dieback of the Amazon Rainforest. Proc. Natl. Acad. Sci. USA 2009, 106, 20610–20615. [Google Scholar] [CrossRef] [Green Version]
- Lewis, S.L.; Sonké, B.; Sunderland, T.; Begne, S.K.; Lopez-Gonzalez, G.; van der Heijden, G.M.F.; Phillips, O.L.; Affum-Baffoe, K.; Baker, T.R.; Banin, L.; et al. Above-Ground Biomass and Structure of 260 African Tropical Forests. Philos. Trans. R. Soc. B 2013, 368, 20120295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bastin, J.; Rutishauser, E.; Kellner, J.R.; Saatchi, S.; Pélissier, R.; Hérault, B.; Slik, F.; Bogaert, J.; De Cannière, C.; Marshall, A.R.; et al. Pan-tropical Prediction of Forest Structure from the Largest Trees. Glob. Ecol. Biogeogr. 2018, 27, 1366–1383. [Google Scholar] [CrossRef]
- Ter Steege, H.; Pitman, N.; Sabatier, D.; Baraloto, C.; Salomão, R.; Guevara Andino, J.; Phillips, O.; Castilho, C.; Magnusson, W.; Molino, J.-F.; et al. Hyperdominance in the Amazonian Tree Flora. Science 2013, 342, 1243092. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morrone, J. Biogeographical Regionalisation of the Neotropical Region. Zootaxa 2014, 3782, 1–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Souza, L.A.S.; Eisenlohr, P.V. Drivers of Floristic Variation in Biogeographic Transitions: Insights from the Ecotone between the Largest Biogeographic Domains of South America. Acta Bot. Bras. 2020, 34, 155–166. [Google Scholar] [CrossRef]
- Pérez-Luque, A.J.; Benito, B.M.; Bonet-García, F.J.; Zamora, R. Ecological Diversity within Rear-Edge: A Case Study from Mediterranean Quercus Pyrenaica Willd. Forests 2020, 12, 10. [Google Scholar] [CrossRef]
- Draper, F.C.; Costa, F.R.C.; Arellano, G.; Phillips, O.L.; Duque, A.; Macía, M.J.; ter Steege, H.; Asner, G.P.; Berenguer, E.; Schietti, J.; et al. Amazon Tree Dominance across Forest Strata. Nat. Ecol. Evol. 2021, 5, 757–767. [Google Scholar] [CrossRef]
- Kearsley, E.; Hufkens, K.; Verbeeck, H.; Bauters, M.; Beeckman, H.; Boeckx, P.; Huygens, D. Large-sized Rare Tree Species Contribute Disproportionately to Functional Diversity in Resource Acquisition in African Tropical Forest. Ecol. Evol. 2019, 9, 4349–4361. [Google Scholar] [CrossRef]
- Lutz, J.A.; Furniss, T.J.; Johnson, D.J.; Davies, S.J.; Allen, D.; Alonso, A.; Anderson-Teixeira, K.J.; Andrade, A.; Baltzer, J.; Becker, K.M.L.; et al. Global Importance of Large-diameter Trees. Glob. Ecol. Biogeogr. 2018, 27, 849–864. [Google Scholar] [CrossRef] [Green Version]
- ForestPlots.net; Blundo, C.; Carilla, J.; Grau, R.; Malizia, A.; Malizia, L.; Osinaga-Acosta, O.; Bird, M.; Bradford, M.; Catchpole, D.; et al. Taking the Pulse of Earth’s Tropical Forests Using Networks of Highly Distributed Plots. Biol. Conserv. 2021, 260, 108849. [Google Scholar] [CrossRef]
- Chave, J.; Coomes, D.; Jansen, S.; Lewis, S.L.; Swenson, N.G.; Zanne, A.E. Towards a Worldwide Wood Economics Spectrum. Ecol. Lett. 2009, 12, 351–366. [Google Scholar] [CrossRef]
- Zanne, A.E.; Lopez-Gonzalez, G.; Coomes, D.A.; Ilic, J.; Jansen, S.; Lewis, S.L.; Miller, R.B.; Swenson, N.G.; Wiemann, M.C.; Chave, J. Towards a Worldwide Wood Economics Spectrum 2009; 2047488 bytes; Dryad: Lewis County, WA, USA, 2009. [Google Scholar]
- Chave, J.; Réjou-Méchain, M.; Búrquez, A.; Chidumayo, E.; Colgan, M.S.; Delitti, W.B.C.; Duque, A.; Eid, T.; Fearnside, P.M.; Goodman, R.C.; et al. Improved Allometric Models to Estimate the Aboveground Biomass of Tropical Trees. Glob. Chang. Biol. 2014, 20, 3177–3190. [Google Scholar] [CrossRef] [PubMed]
- McElhinny, C.; Gibbons, P.; Brack, C.; Bauhus, J. Forest and Woodland Stand Structural Complexity: Its Definition and Measurement. For. Ecol. Manag. 2005, 218, 1–24. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.C.; Sander, E.L.; Ma, K.H.; Colwell, R.K.; Ellison, A.M. Rarefaction and Extrapolation with Hill Numbers: A Framework for Sampling and Estimation in Species Diversity Studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef] [Green Version]
- Pinho, B.X.; Peres, C.A.; Leal, I.R.; Tabarelli, M. Critical role and collapse of tropical mega-trees: A key global resource. In Advances in Ecological Research; Elsevier: Amsterdam, The Netherlands, 2020; Volume 62, pp. 253–294. ISBN 978-0-12-821134-2. [Google Scholar]
- Li, S.; Su, J.; Lang, X.; Liu, W.; Ou, G. Positive Relationship between Species Richness and Aboveground Biomass across Forest Strata in a Primary Pinus Kesiya Forest. Sci. Rep. 2018, 8, 2227. [Google Scholar] [CrossRef]
- Mustin, K.; Carvalho, W.D.; Hilário, R.R.; Costa-Neto, S.V.; Silva, C.; Vasconcelos, I.M.; Castro, I.J.; Eilers, V.; Kauano, É.E.; Mendes-Junior, R.N.G.; et al. Biodiversity, Threats and Conservation Challenges in the Cerrado of Amapá, an Amazonian Savanna. Nat. Conserv. 2017, 22, 107–127. [Google Scholar] [CrossRef]
- De Lima, R.B.; Rutishauser, E.; da Silva, J.A.A.; Guedes, M.C.; Herault, B.; de Oliveira, C.P.; da Silva Aparício, P.; Sotta, E.D.; da Silva, D.A.S.; Ferreira, R.L.C. Accurate Estimation of Commercial Volume in Tropical Forests. For. Sci. 2021, 67, 14–21. [Google Scholar] [CrossRef]
- Pitman, N.C.A.; Terborgh, J.W.; Silman, M.R.; Núñez, P.; Neill, D.A.; Cerón, C.E.; Palacios, W.A.; Aulestia, M. Dominance and Distribution of Tree Species in Upper Amazonian Terra Firme Forests. Ecology 2001, 82, 2101–2117. [Google Scholar] [CrossRef]
- Pitman, N.C.A.; Silman, M.R.; Terborgh, J.W. Oligarchies in Amazonian Tree Communities: A Ten-Year Review. Ecography 2013, 36, 114–123. [Google Scholar] [CrossRef]
- Arellano, G.; Jørgensen, P.M.; Fuentes, A.F.; Loza, M.I.; Torrez, V.; Macía, M.J. Oligarchic Patterns in Tropical Forests: Role of the Spatial Extent, Environmental Heterogeneity and Diversity. J. Biogeogr. 2016, 43, 616–626. [Google Scholar] [CrossRef]
- Tuomisto, H.; Ruokolainen, K.; Yli-Halla, M. Dispersal, Environment, and Floristic Variation of Western Amazonian Forests. Science 2003, 299, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Gorgens, E.B.; Motta, A.Z.; Assis, M.; Nunes, M.H.; Jackson, T.; Coomes, D.; Rosette, J.; Aragão, L.E.O.C.; Ometto, J.P. The Giant Trees of the Amazon Basin. Front. Ecol. Environ. 2019, 17, 373–374. [Google Scholar] [CrossRef] [Green Version]
- Gorgens, E.B.; Nunes, M.H.; Jackson, T.; Coomes, D.; Keller, M.; Reis, C.R.; Valbuena, R.; Rosette, J.; Almeida, D.R.A.; Gimenez, B.; et al. Resource Availability and Disturbance Shape Maximum Tree Height across the Amazon. Glob. Chang. Biol. 2021, 27, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Aragão, L.E.O.C.; Poulter, B.; Barlow, J.B.; Anderson, L.O.; Malhi, Y.; Saatchi, S.; Phillips, O.L.; Gloor, E. Environmental Change and the Carbon Balance of Amazonian Forests: Environmental Change in Amazonia. Biol. Rev. 2014, 89, 913–931. [Google Scholar] [CrossRef] [PubMed]
- Marvin, D.C.; Asner, G.P.; Knapp, D.E.; Anderson, C.B.; Martin, R.E.; Sinca, F.; Tupayachi, R. Amazonian Landscapes and the Bias in Field Studies of Forest Structure and Biomass. Proc. Natl. Acad. Sci. USA 2014, 111, E5224–E5232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fauset, S.; Johnson, M.O.; Gloor, M.; Baker, T.R.; Monteagudo, M.A.; Brienen, R.J.W.; Feldpausch, T.R.; Lopez-Gonzalez, G.; Malhi, Y.; ter Steege, H.; et al. Hyperdominance in Amazonian Forest Carbon Cycling. Nat. Commun. 2015, 6, 6857. [Google Scholar] [CrossRef] [PubMed]
- Duque, A.; Muller-Landau, H.; Valencia, R.; Cardenas, D.; Davies, S.; Oliveira, A.; Pérez, Á.; Romero-Saltos, H.; Vicentini, A. Insights into Regional Patterns of Amazonian Forest Structure, Diversity, and Dominance from Three Large Terra-Firme Forest Dynamics Plots. Biodivers. Conserv. 2017, 26, 669–686. [Google Scholar] [CrossRef]
- Draper, F.C.; Asner, G.P.; Honorio Coronado, E.N.; Baker, T.R.; García-Villacorta, R.; Pitman, N.C.A.; Fine, P.V.A.; Phillips, O.L.; Zárate Gómez, R.; Amasifuén Guerra, C.A.; et al. Dominant Tree Species Drive Beta Diversity Patterns in Western Amazonia. Ecology 2019, 100, e02636. [Google Scholar] [CrossRef] [Green Version]
- Crowther, T.W.; Glick, H.B.; Covey, K.R.; Bettigole, C.; Maynard, D.S.; Thomas, S.M.; Smith, J.R.; Hintler, G.; Duguid, M.C.; Amatulli, G.; et al. Mapping Tree Density at a Global Scale. Nature 2015, 525, 201–205. [Google Scholar] [CrossRef]
- Lines, E.R.; Zavala, M.A.; Purves, D.W.; Coomes, D.A. Predictable Changes in Aboveground Allometry of Trees along Gradients of Temperature, Aridity and Competition: Predictable Variation in Tree Aboveground Allometry. Glob. Ecol. Biogeogr. 2012, 21, 1017–1028. [Google Scholar] [CrossRef]
- Iida, Y.; Kohyama, T.S.; Kubo, T.; Kassim, A.R.; Poorter, L.; Sterck, F.; Potts, M.D. Tree Architecture and Life-History Strategies across 200 Co-Occurring Tropical Tree Species: Tree Architecture and Life-History Strategies across 200 Species. Funct. Ecol. 2011, 25, 1260–1268. [Google Scholar] [CrossRef]
- Rutishauser, E.; Hérault, B.; Petronelli, P.; Sist, P. Tree Height Reduction After Selective Logging in a Tropical Forest. Biotropica 2016, 48, 285–289. [Google Scholar] [CrossRef]
- Giardina, F.; Konings, A.G.; Kennedy, D.; Alemohammad, S.H.; Oliveira, R.S.; Uriarte, M.; Gentine, P. Tall Amazonian Forests Are Less Sensitive to Precipitation Variability. Nat. Geosci. 2018, 11, 405–409. [Google Scholar] [CrossRef]
- Jucker, T.; Hardwick, S.R.; Both, S.; Elias, D.M.O.; Ewers, R.M.; Milodowski, D.T.; Swinfield, T.; Coomes, D.A. Canopy Structure and Topography Jointly Constrain the Microclimate of Human-modified Tropical Landscapes. Glob. Chang. Biol. 2018, 24, 5243–5258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).