Three-Dimensional Quantification of Copepods Predictive Distributions in the Ross Sea: First Data Based on a Machine Learning Model Approach and Open Access (FAIR) Data
Abstract
1. Introduction
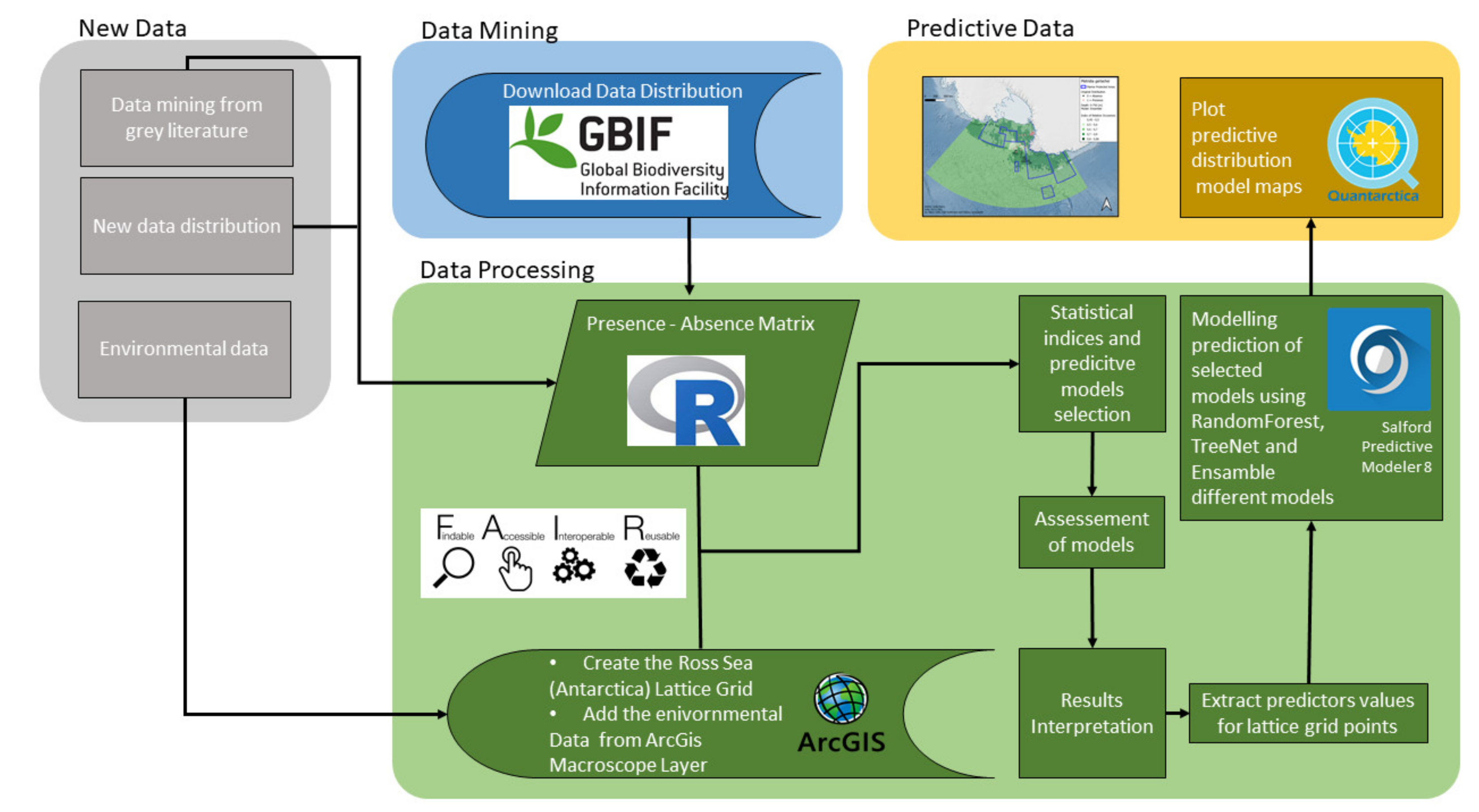
2. Materials and Methods
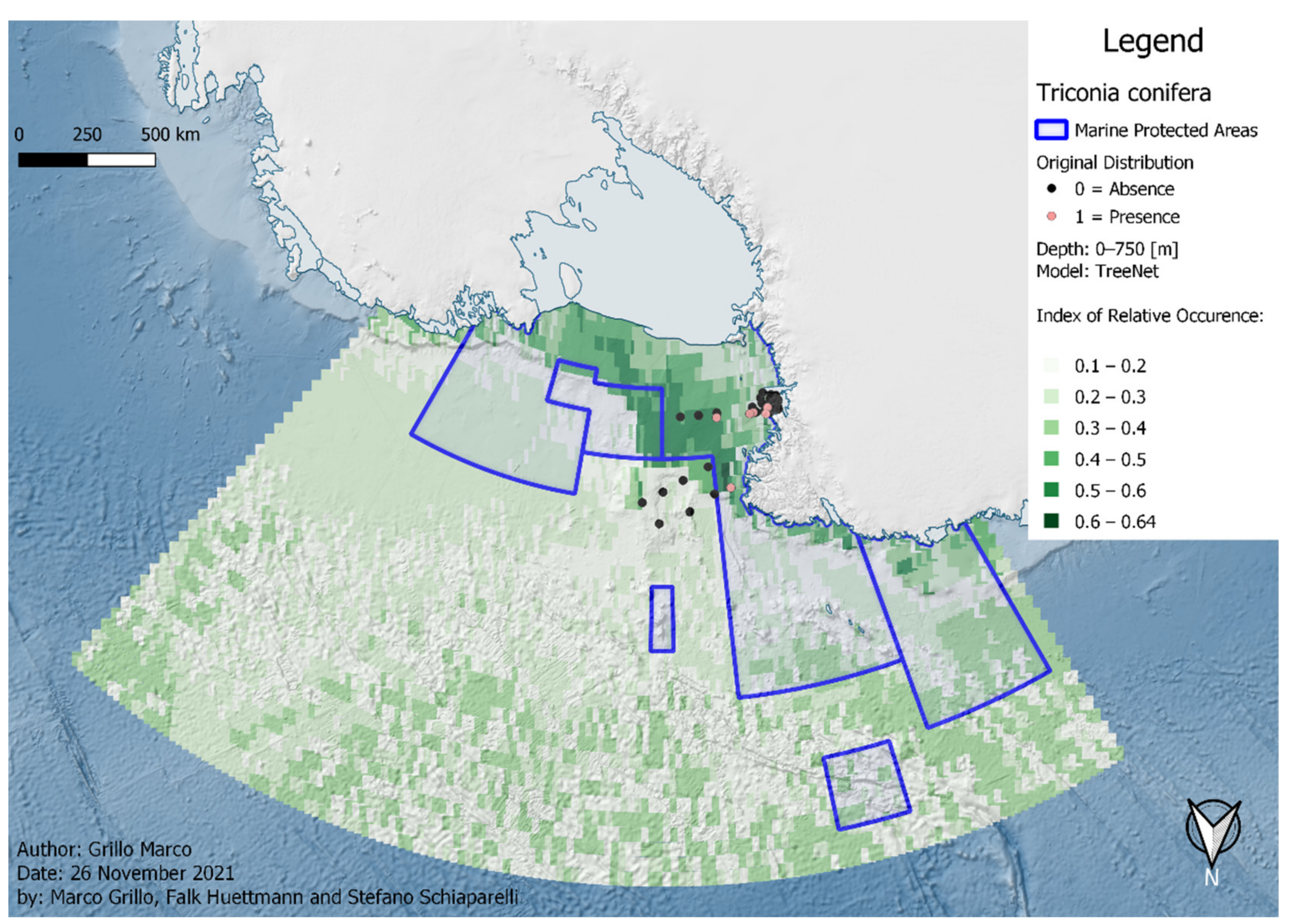
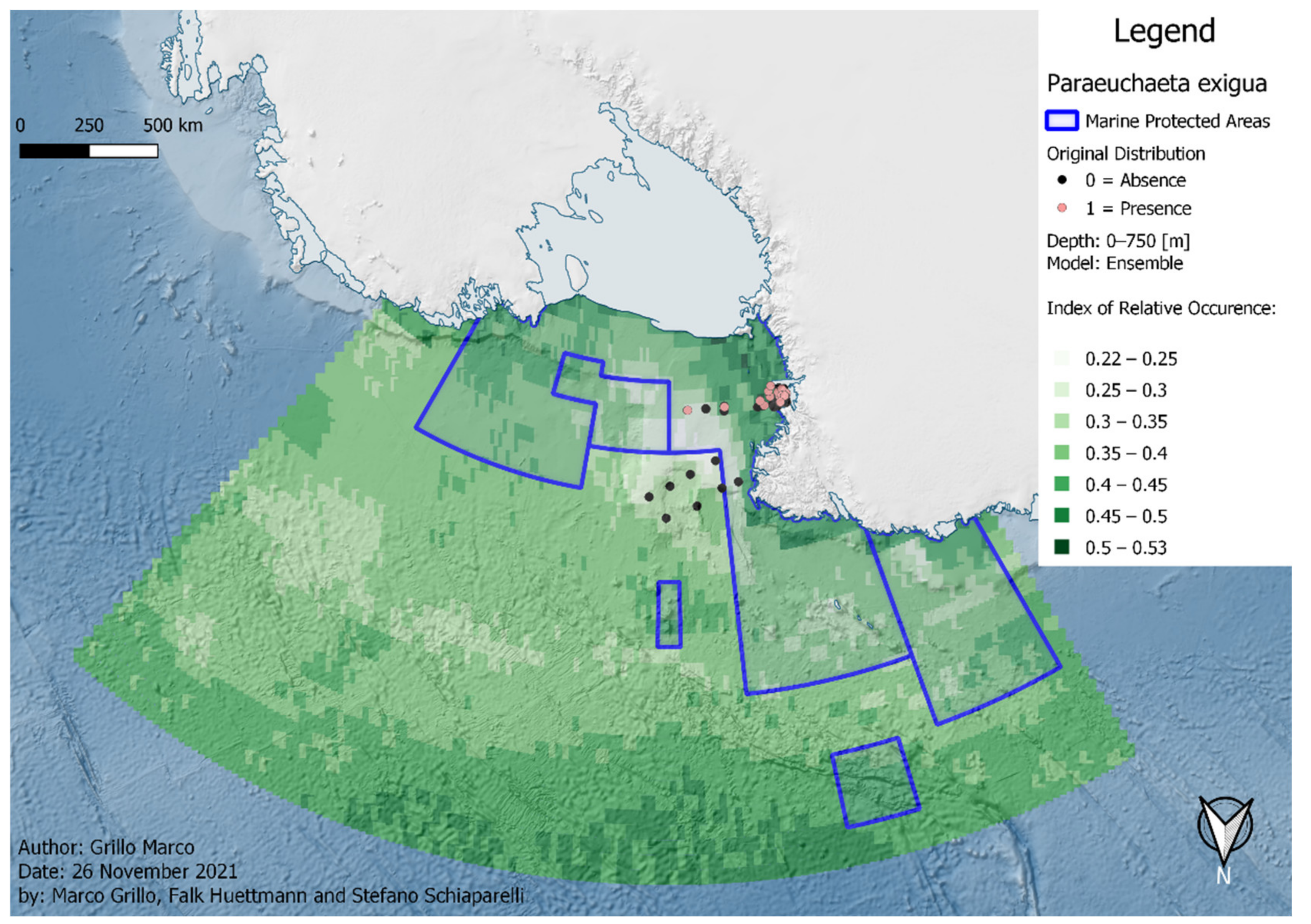
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ainley, D.G.; Brooks, C.M.; Eastman, J.T.; Massaro, M. Unnatural Selection of Antarctic Toothfish in the Ross Sea, Antarctica. In Protection of the Three Poles; Springer: Berlin/Heidelberg, Germany, 2012; pp. 53–75. [Google Scholar]
- Food and Agriculture Organization of the United Nations. The State of World Fisheries and Aquaculture 2016. In Contributing to Food Security and Nutrition for All; Food and Agriculture Organization of the United Nations: Rome, Italy, 2016. [Google Scholar]
- Estes, J.A.; Terborgh, J.; Brashares, J.S.; Power, M.E.; Berger, J.; Bond, W.J.; Carpenter, S.R.; Essington, T.E.; Holt, R.D.; Jackson, J.B. Trophic Downgrading of Planet Earth. Science 2011, 333, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Brooks, C.M.; Crowder, L.B.; Österblom, H.; Strong, A.L. Reaching Consensus for Conserving the Global Commons: The Case of the Ross Sea, Antarctica. Conserv. Lett. 2020, 13, e12676. [Google Scholar] [CrossRef]
- Ainley, D.G.; Ballard, G.; Jones, R.M.; Jongsomjit, D.; Pierce, S.D.; Smith, W.O.J.; Veloz, S. Trophic Cascades in the Western Ross Sea, Antarctica: Revisited. Mar. Ecol. Prog. Ser. 2015, 534, 1–16. [Google Scholar] [CrossRef]
- Huettmann, F.; Schmid, M.S.; Humphries, G.R.W. A First Overview of Open Access Digital Data for the Ross Sea: Complexities, Ethics, and Management Opportunities. Hydrobiologia 2015, 761, 97–119. [Google Scholar] [CrossRef]
- Mormede, S.; Dunn, A.; Parker, S.; Hanchet, S. Using Spatial Population Models to Investigate the Potential Effects of the Ross Sea Region Marine Protected Area on the Antarctic Toothfish Population. Fish. Res. 2017, 190, 164–174. [Google Scholar] [CrossRef]
- Behrens, E.; Pinkerton, M.; Parker, S.; Rickard, G.; Collins, C. The Impact of Sea-Ice Drift and Ocean Circulation on Dispersal of Toothfish Eggs and Juveniles in the Ross Gyre and Amundsen Sea. J. Geophys. Res. Ocean. 2021, 126, e2021JC017329. [Google Scholar] [CrossRef]
- Parker, S.J.; Sundby, S.; Stevens, D.; Di Blasi, D.; Schiaparelli, S.; Ghigliotti, L. Buoyancy of Post-Fertilised Dissostichus Mawsoni Eggs and Implications for Early Life History. Fish. Oceanogr. 2021, 30, 697–706. [Google Scholar] [CrossRef]
- Atkinson, A. Life Cycle Strategies of Epipelagic Copepods in the Southern Ocean. J. Mar. Syst. 1998, 15, 289–311. [Google Scholar] [CrossRef]
- Turner, J.T. The Importance of Small Planktonic Copepods and Their Roles in Pelagic Marine Food Webs. Zool. Stud. 2004, 43, 255–266. [Google Scholar]
- Edwards, M.; Richardson, A.J. Impact of Climate Change on Marine Pelagic Phenology and Trophic Mismatch. Nature 2004, 430, 881–884. [Google Scholar] [CrossRef]
- Hays, G.C.; Richardson, A.J.; Robinson, C. Climate Change and Marine Plankton. Trends Ecol. Evol. 2005, 20, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.; Marshall, G.J. Climate Change in the Polar Regions; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- IPCC (Intergovernmental Panel on Climate Change). Climate Change 2013: Executive Summary; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2014. [Google Scholar]
- Atkinson, A. Subantarctic Copepods in an Oceanic, Low Chlorophyll Environment: Ciliate Predation, Food Selectivity and Impact on Prey Populations. Mar. Ecol. Prog. Ser. 1996, 130, 85–96. [Google Scholar] [CrossRef]
- Razouls, S.; Desreumaux, N.; Kouwenberg, J.; de Bovée, F. Diversity and Geographic Distribution of Marine Planktonic Copepods. Marine Planktonic Copepods. 2022. Available online: http://copepods.obs-banyuls.fr/en (accessed on 1 March 2022).
- Broyer, C.; de Koubbi, P. Biogeographic Atlas of the Southern Ocean 2014; Scientific Committee on Antarctic Research: Cambridge, UK, 2014; pp. 290–296. [Google Scholar]
- Barton, A.D.; Pershing, A.J.; Litchman, E.; Record, N.R.; Edwards, K.F.; Finkel, Z.V.; Kiørboe, T.; Ward, B.A. The Biogeography of Marine Plankton Traits. Ecol. Lett. 2013, 16, 522–534. [Google Scholar] [CrossRef] [PubMed]
- Poloczanska, E.S.; Brown, C.J.; Sydeman, W.J.; Kiessling, W.; Schoeman, D.S.; Moore, P.J.; Brander, K.; Bruno, J.F.; Buckley, L.B.; Burrows, M.T. Global Imprint of Climate Change on Marine Life. Nat. Clim. Chang. 2013, 3, 919–925. [Google Scholar] [CrossRef]
- Humphries, G.R.; Magness, D.R.; Huettmann, F. Machine Learning for Ecology and Sustainable Natural Resource Management; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Guisan, A.; Zimmermann, N.E. Predictive Habitat Distribution Models in Ecology. Ecol. Model. 2000, 135, 147–186. [Google Scholar] [CrossRef]
- Moilanen, A.; Leathwick, J.R.; Quinn, J.M. Spatial Prioritization of Conservation Management. Conserv. Lett. 2011, 4, 383–393. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J.R. Species Distribution Models: Ecological Explanation and Prediction across Space and Time. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 677–697. [Google Scholar] [CrossRef]
- Bonello, G.; Grillo, M.; Cecchetto, M.; Giallain, M.; Granata, A.; Guglielmo, L.; Pane, L.; Schiaparelli, S. Distributional Records of Ross Sea (Antarctica) Planktic Copepoda from Bibliographic Data and Samples Curated at the Italian National Antarctic Museum (MNA): Checklist of Species Collected in the Ross Sea Sector from 1987 to 1995. ZooKeys 2020, 969, 1. [Google Scholar] [CrossRef]
- Carli, A.; Feletti, M.; Pane, L. Zooplankton Biomass and Copepod Abundance of Terra Nova Bay, Ross Sea Antarctic Campaign 1994/1995). J. Mar. Sci. Eng. 2002, 8, 51–55. [Google Scholar]
- Carli, A.; Mariottini, G.L.; Pane, L. Contribution to the Study of Copepods Collected in Terra Nova Bay (Ross Sea). Nat. Sc. Com. Ant. Ocean. Camp. 1990, 88, 129–167. [Google Scholar]
- Carli, A.; Pane, L.; Stocchino, C. Planktonic Copepods in Terra Nova Bay (Ross Sea): Distribution and Relationship with Environmental Factors. In Ross Sea Ecology; Springer: Berlin/Heidelberg, Germany, 2000; pp. 309–321. [Google Scholar]
- Carli, A.; Feletti, M.; Mariottini, G.L.; Pane, L. Contribution to the Study of Copepods Collected during the Italian Oceanographic Campaign in Antarctica 1989–90. 1992, pp. 179–210. Available online: https://www.coldregions.org/vufind/Record/193046 (accessed on 1 March 2022).
- Pane, L.; Feletti, M.; Francomacaro, B.; Mariottini, G.L. Summer Coastal Zooplankton Biomass and Copepod Community Structure near the Italian Terra Nova Base (Terra Nova Bay, Ross Sea, Antarctica). J. Plankton Res. 2004, 26, 1479–1488. [Google Scholar] [CrossRef]
- Sertorio, T.Z.; Licandro, P.; Ossola, C.; Artegiani, A. Copepod Communities in the Pacific Sector of the Southern Ocean in Early Summer. In Ross Sea Ecology; Springer: Berlin/Heidelberg, Germany, 2000; pp. 291–307. [Google Scholar]
- Zunini Sertorio, T.; Salemi Picone, P.; Bernat, P.; Cattini, E.; Ossola, C. Copepods Collected in Sixteen Stations during the Italian Antarctic Expedition 1987–1988. Natl. Sci. Com. Antarct. Ocean. Camp. 1990, 1988, 67–125. [Google Scholar]
- Zunini Sertorio, T.; Licandro, P.; Ricci, F.; Giallain, M.; Antarctica, N.S.C. for A Study on Ross Sea Copepods. Nat. Sc. Com. Ant. Ocean. Camp. 1992, 88, 217–246. [Google Scholar]
- Hoshiai, T.; Tanimura, A.; Watanabe, K. Ice Algae as Food of an Antarctic Ice-Associated Copepod, Paralabidocera Antarctica (IC Thompson). In Proceedings of the NIPR Symposium on Polar Biology, Tokyo, Japan, 24–26 November 1987; Volume 1, p. 3. [Google Scholar]
- Arashkevich, Y.G. The Food and Feeding of Copepods in the Northwestern. Pacific. Oceanol. 1969, 9, 695–709. [Google Scholar]
- Boxshall, G.A.; Halsey, S.H. An Introduction to Copepod Diversity; Ray Society: London, UK, 2004. [Google Scholar]
- Michels, J.; Schnack-Schiel, S.B. Feeding in Dominant Antarctic Copepods—Does the Morphology of the Mandibular Gnathobases Relate to Diet? Mar. Biol. 2005, 146, 483–495. [Google Scholar] [CrossRef]
- Yu, J.; Tian, J.; Yang, G. Ingestion, Fecundity and Population Growth of Harpacticus Sp.(Harpacticoida, Copepod) Fed on Five Species of Algae. Aquac. Res. 2017, 48, 2209–2220. [Google Scholar] [CrossRef]
- Nishida, S.; Ohtsuka, S. Specialized Feeding Mechanism in the Pelagic Copepod Genus Heterorhabdus (Calanoida: Heterorhabdidae), with Special Reference to the Mandibular Tooth and Labral Glands. Mar. Biol. 1996, 126, 619–632. [Google Scholar] [CrossRef]
- Ohtsuka, S.; Soh, H.Y.; Nishida, S. Evolutionary Switching from Suspension Feeding to Carnivory in the Calanoid Family Heterorhabdidae (Copepoda). J. Crustacean Biol. 1997, 17, 577–595. [Google Scholar] [CrossRef]
- Tseng, L.-C.; Dahms, H.-U.; Chen, Q.-C.; Hwang, J.-S. Copepod Feeding Study in the Upper Layer of the Tropical South China Sea. Helgol. Mar. Res. 2009, 63, 327–337. [Google Scholar] [CrossRef]
- Spinelli, M.L.; Pájaro, M.; Martos, P.; Esnal, G.B.; Sabatini, M.; Capitanio, F.L. Potential Zooplankton Preys (Copepoda and Appendicularia) for Engraulis Anchoita in Relation to Early Larval and Spawning Distributions in the Patagonian Frontal System (SW Atlantic Ocean). Sci. Mar. 2012, 76, 39–47. [Google Scholar] [CrossRef]
- Sameoto, D.D.; Jaroszynski, L.O.; Fraser, W.B. BIONESS, a New Design in Multiple Net Zooplankton Samplers. Can. J. Fish. Aquat. Sci. 1980, 37, 722–724. [Google Scholar] [CrossRef]
- Guglielmo, L.; Costanzo, G.; Zagami, G.; Manganaro, A.; Arena, G. Zooplankton Ecology in the Southern Ocean. Data Rep. Part II Natl. Sci. Comm. Antarct. Genova 1992, 969, 30–468. [Google Scholar]
- Guglielmo, L.; Costanzo, G.; Manganaro, A.; Zagami, G. Spatial and Vertical Distribution of Zooplanktonic Communities in the Terra Nova Bay (Ross Sea). Nat. Sci. Antarct. Ocean. Campaign. 1990, 1988, 257–398. [Google Scholar]
- Team: QGIS Geographic Information System; Open Source Geospat. Found Proj. 2020. Available online: https://qgis.org/en/site/ (accessed on 1 March 2022).
- R Core Team. Rf. R: A Language and Environment for Statistical Computing; R Foundation: Vienna, Austria, 2013. [Google Scholar]
- Roth, G.; Matsuoka, K.; Skoglund, A.; Melvær, Y.; Tronstad, S. Quantarctica: A Unique, Open, Standalone GIS Package for Antarctic Research and Education. In Proceedings of the EGU General Assembly Conference Abstracts, Vienna, Austria, 8–13 April 2017; p. 1973. [Google Scholar]
- Brown, M. Ocean Data View 4.0. Oceanography 1998, 11, 19–21. [Google Scholar] [CrossRef]
- Hardy, S.M.; Lindgren, M.; Konakanchi, H.; Huettmann, F. Predicting the Distribution and Ecological Niche of Unexploited Snow Crab (Chionoecetes opilio) Populations in Alaskan Waters: A First Open-Access Ensemble Model. Integr. Comp. Biol. 2011, 51, 608–622. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meißner, K.; Fiorentino, D.; Schnurr, S.; Arbizu, P.M.; Huettmann, F.; Holst, S.; Brix, S.; Svavarsson, J. Distribution of Benthic Marine Invertebrates at Northern Latitudes―An Evaluation Applying Multi-Algorithm Species Distribution Models. J. Sea Res. 2013, 85, 241–254. [Google Scholar] [CrossRef]
- Huettmann, F.; Schmid, M. 9.1. Climate Change and Predictions of Pelagic Biodiversity Components. Biogeogr. Atlas South. Ocean. Sci. Comm. Antarct. Res. Camb. 2014, 16, 470–475. [Google Scholar]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Mi, C.; Huettmann, F.; Sun, R.; Guo, Y. Combining Occurrence and Abundance Distribution Models for the Conservation of the Great Bustard. PeerJ 2017, 5, e4160. [Google Scholar] [CrossRef]
- Pearce, J.; Ferrier, S. Evaluating the Predictive Performance of Habitat Models Developed Using Logistic Regression. Ecol. Model. 2000, 133, 225–245. [Google Scholar] [CrossRef]
- Swets, J.A. Measuring the Accuracy of Diagnostic Systems. Science 1988, 240, 1285–1293. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, H.S. CCAMLR Fisheries: Challenges to Effective Conservation and Management. J. Int. Wildl. Law Policy 2009, 12, 180–189. [Google Scholar] [CrossRef]
- C-CAMLR-XXXV, Report of the thirty-fifth meeting of the Scientific Committee, Hobart, Australia, 17–21 October, Annex 6, 3.2, 3.7-3.9. 2016. CCAMLR CONSERVATION MEASURE 91-05 (2016) for the Ross Sea Region Marine Protected Area, Specifically, Addressing the Priorities of Annex 91-05/C. 2016. Available online: https://www.ccamlr.org/en/system/files/e-sc-xxxv.pdf (accessed on 1 March 2022).
- Senô, J.; Komaki, Y.; Takeda, A. Reports on the Biology of the” Umitaka-Maru” Expedition Plankton Collected by the” Umitaka-Maru” in the Antarctic and Adjacent Waters by Larva Net, with Special References to Copepoda. Mem. Natl. Inst. Polar Res. Spec. 1963, 40, 212–220. [Google Scholar]
- Hopkins, T.L. Zooplankton Standing Crop in the Pacific Sector of the Antarctic. Biol. Antarct. Seas 1971, 4, 347–362. [Google Scholar]
- Maruyama, T.; Toyoda, H.; Suzuki, S. Preliminary Report on the Biomass of Macroplankton and Micronekton Collected with a Bongo Net during the Umitaka Maru FIBEX Cruise. Trans. Tokyo Univ. Fish. Tokyo Suisan Daigaku Tokubetsu Kenkyu Hokoku 1982, 5, 125. [Google Scholar]
- Hutchinson, G.E. Concluding Remarks Cold Spring Harbor Symposia on Quantitative Biology. GS Search 1957, 22, 415–427. [Google Scholar]
- Drew, C.A.; Wiersma, Y.F.; Huettmann, F. Predictive Species and Habitat Modeling in Landscape Ecology: Concepts and Applications; Springer Science & Business Media: Berlin, Germany, 2010. [Google Scholar]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A. Novel Methods Improve Prediction of Species’ Distributions from Occurrence Data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Leathwick, J.R.; Elith, J.; Francis, M.P.; Hastie, T.; Taylor, P. Variation in Demersal Fish Species Richness in the Oceans Surrounding New Zealand: An Analysis Using Boosted Regression Trees. Mar. Ecol. Prog. Ser. 2006, 321, 267–281. [Google Scholar] [CrossRef]
- Huettmann, F.; Schmid, M. Publicly Available Open Access Data and Machine Learning Model-Predictions Applied with Open Source Gis for the Entire Antarctic Ocean: A first Meta-Analysis and Synthesis from 53 Charismatic Species. Horiz. Earth Sci. Res. 2014, 23, 23–33. [Google Scholar]
- Pinkerton, M.H.; Smith, A.N.; Raymond, B.; Hosie, G.W.; Sharp, B.; Leathwick, J.R.; Bradford-Grieve, J.M. Spatial and Seasonal Distribution of Adult Oithona Similis in the Southern Ocean: Predictions Using Boosted Regression Trees. Deep. Sea Res. Part I Oceanogr. Res. Pap. 2010, 57, 469–485. [Google Scholar] [CrossRef]
- Voronina, N.; Kolosova, E.; Melnikov, I. Zooplankton Life under the Perennial Antarctic Sea Ice. Polar Biol. 2001, 24, 401–407. [Google Scholar]
- Hureau, J.-C. The Significance of Fish in the Marine Antarctic Ecosystems. Polar Biol. 1994, 14, 307–313. [Google Scholar] [CrossRef]
- Pinkerton, M.H.; Cummings, V.; Forman, J.; Brown, J.; Bury, S.J. Trophic Connections in the Ross Sea: Information from Stomach Contents Analysis and Stable Isotopes of Carbon and Nitrogen; Final Research Report for Ministry of Fisheries, Project IPY200701 Obj10; NIWA, Wellington, 2011, 18.
- Stevens, D.W.; Dunn, M.R.; Pinkerton, M.H.; Forman, J.S. Diet of Antarctic Toothfish (Dissostichus Mawsoni) from the Continental Slope and Oceanic Features of the Ross Sea Region, Antarctica. Antarct. Sci. 2014, 26, 502–512. [Google Scholar] [CrossRef]
- Granata, A.; Zagami, G.; Vacchi, M.; Guglielmo, L. Summer and Spring Trophic Niche of Larval and Juvenile Pleuragramma Antarcticum in the Western Ross Sea, Antarctica. Polar Biol. 2009, 32, 369–382. [Google Scholar] [CrossRef]
- Williams, R. Trophic Relationships between Pelagic Fish and Euphausiids in Antarctic Waters. In Antarctic Nutrient Cycles and Food Webs; Springer: Berlin/Heidelberg, Germany, 1985; pp. 452–459. [Google Scholar]
- Culina, A.; Baglioni, M.; Crowther, T.W.; Visser, M.E.; Woutersen-Windhouwer, S.; Manghi, P. Navigating the Unfolding Open Data Landscape in Ecology and Evolution. Nat. Ecol. Evol. 2018, 2, 420–426. [Google Scholar] [CrossRef]


| Family | Species | Worms Aphia ID | Trophic Guild |
|---|---|---|---|
| Acartiidae | Paralabidocera antarctica (Thompson I.C., 1898) | 345234 | Phytoplankton feeder [34] |
| Aetideidae | Aetideopsis antarctica (Wolfenden, 1908) | 341236 | Phyto- and zooplankton feeder [35] |
| Aetideopsis minor (Wolfenden, 1911) | 254600 | Phyto- and zooplankton feeder [35] | |
| Gaetanus tenuispinus (Sars G.O., 1900) | 237965 | Phyto- and zooplankton feeder [35] | |
| Augaptilidae | Haloptilus ocellatus Wolfenden, 1905 | 342624 | Zooplankton feeder [36] |
| Calanidae | Calanoides acutus (Giesbrecht, 1902) | 342434 | Phytoplankton feeder [18,37] |
| Calanus propinquus Brady, 1883 | 342435 | Phytoplankton feeder [18,37] | |
| Clausocalanidae | Microcalanus pygmaeus (Sars G.O., 1900) | 104513 | Phyto- and zooplankton feeder [37] |
| Ctenocalanus vanus Giesbrecht, 1888 | 104510 | Phytoplankton feeder [37] | |
| Euchaetidae | Paraeuchaeta antarctica (Giesbrecht, 1902) | 344974 | Zooplankton feeder [37] |
| Paraeuchaeta exigua (Wolfenden, 1911) | 345051 | Zooplankton feeder [36] | |
| Paraeuchaeta similis (Wolfenden, 1908) | 345055 | Zooplankton feeder [36] | |
| Harpacticidae | Harpacticus furcifer Giesbrecht, 1902 | 293136 | Phytoplankton feeder [38] |
| Heterorhabdidae | Heterorhabdus austrinus Giesbrecht, 1902 | 343728 | Zooplankton feeder [39,40] |
| Lucicutiidae | Lucicutia ovalis (Giesbrecht, 1889) | 104609 | Phytoplankton feeder [41] |
| Metridinidae | Metridia gerlachei Giesbrecht, 1902 | 344689 | Phyto-, zooplankton [37] and suspension feeder [36] |
| Metridia curticauda Giesbrecht, 1889 | 104628 | Suspension feeder [36] | |
| Oithonidae | Oithona frigida Giesbrecht, 1902 | 344712 | Phytoplankton feeder [42] |
| Oithona similis Claus, 1866 | 106656 | Phytoplankton feeder [42] | |
| Oncaeidae | Oncaea curvata Giesbrecht, 1902 | 344719 | Zooplankton feeder [36] |
| Triconia conifera (Giesbrecht, 1891) | 128957 | Zooplankton feeder [36] | |
| Rhincalanidae | Rhincalanus gigas Brady, 1883 | 220837 | Phyto- and zooplankton feeder [37] |
| Scolecitrichidae | Scolecithricella minor (Brady, 1883) | 104811 | Phyto- and zooplankton feeder [36] |
| Racovitzanus antarcticus Giesbrecht, 1902 | 104784 | Phyto- and zooplankton feeder [36] | |
| Scaphocalanus subbrevicornis (Wolfenden, 1911) | 104802 | Phyto- and zooplankton feeder [36] | |
| Stephidae | Stephos longipes Giesbrecht, 1902 | 345365 | Phytoplankton feeder [37] |
| Algorithm | ROC (%) Range | Class |
|---|---|---|
| TreeNet | 0–49 | Poor |
| RandomForest | 50–70 | Good |
| Ensemble | 71–100 | Very good |
| Sample Size (# of Occurrences) | TreeNet ROC (%) | RandomForest ROC (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species | 0–750 [m] | 0–10 [m] | 11–70 [m] | 71–750 [m] | 0–750 [m] | 0–10 [m] | 11–70 [m] | 71–750 [m] | 0–750 [m] | 0–10 [m] | 11–70 [m] | 71–750 [m] |
| A. antarctica | 30 | 0 | 0 | 30 | 83 | 47 | ||||||
| A. minor | 1 | 0 | 0 | 1 | ||||||||
| C. acutus | 534 | 71 | 144 | 319 | 66 | 70 | 69 | 67 | 64 | 60 | 64 | 59 |
| C. propinquus | 242 | 28 | 55 | 159 | 66 | 73 | 73 | 60 | 63 | 70 | 69 | 55 |
| C. vanus | 125 | 8 | 22 | 95 | 82 | 83 | 84 | 76 | 75 | 58 | 76 | 71 |
| G. tenuispinus | 2 | 0 | 0 | 2 | ||||||||
| H. ocellatus | 5 | 0 | 2 | 3 | 73 | 87 | ||||||
| H. furcifer | 1 | 1 | 0 | 0 | ||||||||
| H. austrinus | 6 | 3 | 0 | 3 | 78 | 88 | ||||||
| L. ovalis | 3 | 2 | 0 | 1 | 49 | 38 | ||||||
| M. curticauda | 7 | 1 | 1 | 5 | 83 | 94 | ||||||
| M. gerlachei | 579 | 58 | 104 | 417 | 67 | 65 | 58 | 66 | 66 | 60 | 53 | 65 |
| M. pygmaeus | 1 | 0 | 0 | 1 | ||||||||
| O. frigida | 68 | 1 | 3 | 64 | 81 | 79 | ||||||
| O. similis | 231 | 55 | 52 | 124 | 84 | 84 | 87 | 79 | 79 | 78 | 80 | 73 |
| O. curvata | 214 | 55 | 43 | 116 | 83 | 84 | 85 | 76 | 77 | 82 | 83 | 68 |
| P. antarctica | 185 | 8 | 11 | 166 | 72 | 52 | 89 | 63 | 65 | 8 | 52 | 59 |
| P. exigua | 119 | 8 | 5 | 106 | 80 | 75 | 73 | 68 | 53 | 66 | ||
| P. similis | 3 | 0 | 0 | 3 | 60 | 0 | ||||||
| P. antarctica | 24 | 19 | 1 | 4 | 91 | 62 | ||||||
| R. antarcticus | 53 | 13 | 1 | 39 | 93 | 91 | ||||||
| R. gigas | 32 | 8 | 12 | 12 | 94 | 81 | 89 | 96 | 93 | 82 | 84 | 95 |
| S. subbrevicornis | 9 | 4 | 0 | 5 | 91 | 93 | ||||||
| S. minor | 4 | 0 | 0 | 4 | ||||||||
| S. longipes | 37 | 3 | 1 | 33 | 85 | 77 | ||||||
| T. conifera | 19 | 0 | 0 | 19 | 96 | 80 | ||||||
| Other copepods | 9 | 1 | 0 | 8 | 84 | 42 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grillo, M.; Huettmann, F.; Guglielmo, L.; Schiaparelli, S. Three-Dimensional Quantification of Copepods Predictive Distributions in the Ross Sea: First Data Based on a Machine Learning Model Approach and Open Access (FAIR) Data. Diversity 2022, 14, 355. https://doi.org/10.3390/d14050355
Grillo M, Huettmann F, Guglielmo L, Schiaparelli S. Three-Dimensional Quantification of Copepods Predictive Distributions in the Ross Sea: First Data Based on a Machine Learning Model Approach and Open Access (FAIR) Data. Diversity. 2022; 14(5):355. https://doi.org/10.3390/d14050355
Chicago/Turabian StyleGrillo, Marco, Falk Huettmann, Letterio Guglielmo, and Stefano Schiaparelli. 2022. "Three-Dimensional Quantification of Copepods Predictive Distributions in the Ross Sea: First Data Based on a Machine Learning Model Approach and Open Access (FAIR) Data" Diversity 14, no. 5: 355. https://doi.org/10.3390/d14050355
APA StyleGrillo, M., Huettmann, F., Guglielmo, L., & Schiaparelli, S. (2022). Three-Dimensional Quantification of Copepods Predictive Distributions in the Ross Sea: First Data Based on a Machine Learning Model Approach and Open Access (FAIR) Data. Diversity, 14(5), 355. https://doi.org/10.3390/d14050355







