Abstract
The study of banana herbs and fruits is rarely conducted using multidisciplinary approaches. However, a multidisciplinary approach could be useful for gaining information on many aspects, including remote sensing, biodiversity and biogeography, owing to the uniqueness of bananas. The present article reviews a study on banana herbs from an Indonesian perspective. It starts by obtaining information related to firmness, peel’s colour change, water content and sugar content corresponding to Brix and Starch values. It then proceeds to find the relation between remote sensing (RS) technologies of all these biophysical characteristics and genomics, transcriptomic and metabolomics. Besides this, geospatial sciences, such as geographic information systems (GIS), may help visualise biogeographical factors that help analyse a land’s suitability for growing bananas. Furthermore, the plant canopy, health and plant disease, and the herbs’ water content, analysed through satellite images and aerial photos of drones, helps describe the banana distribution in Indonesia, at both the local and regional levels. Similar techniques may be applied to explore and analyse the characteristics of the fruit. In the end, the integration of these methods can foster advanced studies on bananas, even making it possible for its scope to extend to industries, food technology, post-harvest, and eco-tourism.
Keywords:
banana; biogeography; biodiversity; GIS; remote sensing; spectral analysis; Brix; multi-omics 1. Introduction
Research on bananas can be divided into two major streams. The first one explores banana herbs and the second focuses on their fruits. The banana originated from South-East Asia and the western region of the Pacific 7000 years ago [1,2]. Further to this, the banana’s origins can be traced to several countries in South-East Asia, making them hotspots for these herbs. Bananas are also predominant in Eastern Indonesia and the Pacific region, especially Australimusa (Philippines, Moluccas and Papua New Guinea), where the tallest herbs and the biggest fruits (Fe’i banana) can be found. Calimusa has the smallest fruits and herbs that spread in the western regions of Indonesia, Malaysia, and Thailand. Both belong to the groups named Eumusa-Rhodoclamps [3].
The terminology relating to ‘banana’ is always interchangeable with banana herbs. In this case, it might be related to the name of a fruit called the banana. Some of these can be consumed directly and are also called bananas. The others need to be cooked and are known as plantains. The ancestors of these two groups are the Eumusa, a major species of banana [4]. Most of the edible bananas today came from the hybridisation of the Musa acuminata and Musa balbisiana, which produced diploid genome bananas (such as AA and AB). They are also capable of producing triploid genome bananas (AAA, AAB, and ABB) [5,6]. In recent times, the production of edible bananas increased rapidly, reaching its highest global production at 22.2 million tonnes in 2020, i.e., a growth of 1.7% compared to 2019. This is partially due to high supply growth from Ecuador, Costa Rica and Colombia (three of the five leading exporters of bananas) [7].
In general, the study of bananas using a geospatial perspective, including remote sensing (RS) technology, is still limited, but growing intensively. In the last three years, many studies reported implementing RS technologies and methods to study bananas. For instance, two recently published papers showed the benefits of using aerial photographs from an unmanned aerial vehicle (UAV) for detecting banana herbs, and especially ones inflicted with disease-causing fusarium [8,9]. In other studies, similar methods have been used to generate satellite images for detecting diseased banana plants [10]. Besides that, spectral reflectance acquired from a handheld spectroradiometer also offers potential benefits for water content and Brix detection in banana fruits [11,12]. The aforementioned banana genotypes can be further classified using advanced methods, such as a hyperspectral spectroradiometer [13]. More recently, a machine learning and a deep learning algorithm was developed for using red, green, and blue (RGB) images from a UAV to identify and measure the population of banana plants [14]. The explanation below will focus on the specific use of RS data and technologies for detection, identification and classification of banana herbs, based on their biophysical characteristics, disease status, essential minerals (vitamins, beta-carotene), and banana field suitability assessment.
Challenges to Detecting, Identifying and Classifying Banana Herbs
Even though banana herbs generally have small dimensions, they are still easy to identify. It is possible to distinguish them from other types of plants. A single banana herb has a height range of 0–5 m and a canopy with a width range of 1–3 m from the centre. Groups like Musa acuminata, Musa balbisiana, and Musa paradisiaca are smaller, compared to a Fe’i banana, such as Musa troglodytarum. Satellite data helps to identify aspects like this more clearly. The common names for satellite data include Moderate Resolution Imaging Spectroradiometer (MODIS) for both Terra and Aqua, Landsat 5, 7, 8, and 9, Sentinel 2 for free access data and Worldview and Quickbird for commercial data sources. All these devices have a similar spectral band wavelength but a different pixel size. It is important that the research on bananas using satellite RS consider these two parameters, including their pixel size and spectral bands. Although some of them are free of charge and available to public registered users, a major obstacle when it comes to obtaining optical data are atmospheric factors, like clouds, which make observation difficult. Moreover, these factors can also affect classification [15,16].
MODIS satellite images have a coarser spatial resolution, compared to other RS data products. MODIS has a pixel size range of 250 m (bands 1–2), 500 m (bands 3–7) and 1000 m (bands 8–36). It has two key instruments aboard the Terra and Aqua satellites. The Terra passes the earth in a north to south direction, and passes the equator in the morning. Aqua, on the other hand, travels south to north and passes the equator in the afternoon. Its 36 spectral bands act as the richest multispectral source of optical RS data. It also considers satellite sensors that visit the same place every one to two days. The complete configuration of MODIS’ satellite data is provided in Table 1. Given the range of wavelengths and its application field, a suitable application for banana monitoring would be to use bands with a spatial resolution of about 250 to 500 m (bands 1–7). This condition facilitates the monitoring of banana herbs on a plantation scale. This type of MODIS is found in Terra file names beginning with MOD and Aqua file names beginning with MYD.

Table 1.
The characteristics and functions of the 36 bands of MODIS data satellite products.
The satellite imagery of MODIS does not provide a promising result in observing banana herbs individually. This data only works for observing the same objects in plantation scales. It is similar to Sentinel 2 satellite images, even though Sentinel 2 has better spatial resolution compared to MODIS at a range of 10–60 m pixels. Its blue (B2), green (B3), red (B4) and near-infrared (B8) channels have a 10 m resolution. The red edge (B5), near-infrared NIR (B6, B7 and B8A) and shortwave infrared SWIR (B11 and B12) have a ground sampling distance of 20 m. Finally, its coastal aerosol (B1), water vapour (B9), and cirrus band (B10) have a 60 m pixel size. The other advantage offered by this data is a red-edge band. This is because this band is helpful for observing the leaf area index (LAI) and chlorophyll content [17,18] and nitrogen content [19], as well as for doing a suitability analysis [20]. In addition to this, based on the implementation of the red-edge of Sentinel-2, the same band makes it possible to detect the Banana bunchy top disease (BBTD) and Xanthomonas Wilt of Banana (BXW) [10], and even banana borers [21]. However, the documentation of Landsat satellite images for banana herbs is still limited. The study conducted by [22], which elaborated on evapotranspiration, based on the surface energy balance for land (SEBAL) algorithm, may help in facilitating better explanation of conditions over banana plantations.
The use of high-resolution satellite imagery, namely SPOT-5, was studied as well [23]. The previous, and latest, version of SPOT-7 was decommissioned on 31 March 2015. Even with a spatial resolution of 2.5–10 m, and a similar spectral range as Worldview and Quickbird, better visualisation and analysis results were still achieved using SPOT 7. In this regard, the review of which satellite data to consider when analysing banana herbs and plantation types is provided in Table 2. Besides this, Figure 1a–d explain the capability of satellite images of varying spatial resolutions, derived from MODIS at 250 m × 250 m (Figure 1a), and Sentinel-2 data with three-pixel sizes of about 10 m × 10 m, 20 m × 20 m, and 60 m × 60 m, (Figure 1b–d). Figure 1g visualises the same areas of the banana plantation at 30 m × 30 m. A better visualisation is provided by using an aerial photograph (Figure 1h), Google Earth and Google Street view (Figure 1e,f), which can show the near real-time conditions of these plantations. Similar (but better) findings can be yielded by SPOT-5, helping to better identify and differentiate the quality of an individual banana herb, or a group of banana herbs, in a plantation (Figure 1(i1–i4)).

Table 2.
A comparison of the characteristics and functions of MODIS with other satellite data products*.

Figure 1.
A comparison of the spatial resolution of MODIS (a), Sentinel-2 data with pixel size at about 10 m × 10 m, 20 m × 20 m, and 60 m × 60 m, (b–d), field condition and coverage area using Google Earth (e,f), Landsat 8 (g) of a banana plantation in Lampung Tengah regency, Lampung-Indonesia, and an aerial photograph (h) of individual banana herbs in the Gadingrejo village, Pringsewu-Lampung, and SPOT-5′s observation in Northern Australia (i1–i4).
2. Materials and Methods
2.1. Use of Aerial Photographs to Observe Individual Banana Herbs
The banana is an herbaceous plant equipped with a pseudostem, and does not have woody tissue. It is a monocot, originating from the tropics of Indo-Malesia, Asia, and Australia. It has a stem-like structure called suckers. After being harvested and cut down, this part will replace the old body of the herb by growing into a new banana herb. Banana herbs also have many leaves that grow and overlap with the apparent trunks of their pseudostems. A mature herb has seven to ten leaves and can stretch to more than 3 m, forming a canopy over 6 m in diameter (Figure 2).

Figure 2.
The structure of a banana herb.
The actual size of the banana herbs makes it an interesting area of study. The size of banana herbs is small compared to the pixel size of all public satellite imagery data. This condition makes it difficult to observe. However, certain alternatives are available for providing a more detailed visualisation of these herbs. A single scene of an aerial photograph can be recorded by standard RGB camera sensors and modified cameras of near-infrared, red, green, and blue (NRGB) sensors. Attached to a drone, named Dji Phantom, flying at 100 m above the surface, with a ground surface distance (GSD) of 5 cm, the sensors provided beautiful imagery distinguishing banana herbs from other surrounding herbs. As a result, distribution of the plants over the surface can be easily detected. Research conducted by Harto et al. [8], for instance, successfully implemented object image analysis (OBIA) to distinguish banana herbs from other types of vegetation.
For this, multi-resolution segmentation, Lee sigma edge detection, and multi-threshold segmentation method were applied, by considering the shape, scale, and compactness parameters at about 10, 0.2, and 0.5 pixels, respectively. These parameters were derived from a normalised difference vegetation index (NDVI) (Equation (1)), a green ratio (Equation (2)), mean Green, mean digital surface model (DSM), and mean digital terrain model (DTM). The results show producer and user accuracy of 89% and 80%, respectively (Figure 3). Groups of banana herbs were successfully detected and differentiated from other plants. It is evident that the OBIA method is capable of detecting and classifying banana herbs, by providing an aerial image that is sharper and more optimised.
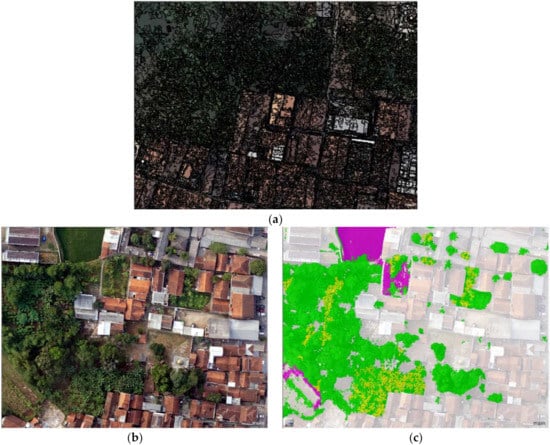
Figure 3.
An implementation of OBIA to classify the distribution of banana herbs. Left to right: Aerial imagery before segmentation (b), after segmentation (a) and classification results of banana plants displayed in yellow (c).
2.2. Observing the Banana Herbs, a Perspective of Biogeography and Biodiversity
The study conducted by Harto et al. [8] successfully described how banana herbs are distributed in the local community. It also showed that conditions of the local environment are suitable for these herbs, implying that they can be planted anywhere. However, land suitability is an important factor, as it helps identify those areas that can yield the healthiest bananas. This factor is quite essential, due to the economic value of bananas. The study conducted by Nuarsa et al. [24], and further enhanced by Wikantika et al. [25], explained the possibility of employing some geographical parameters and agro-climate parameters, including elevation (area above sea level), rainfall (in millimetres), duration of drought season and slope (%), to locate the most suitable location for growing banana herbs (Table 3). For this purpose, geographic information systems (GIS) were chosen to derive four suitable regions. These systems used the aforementioned parameters to map the land suitability of Bali (Figure 4) in general and Bukti village (Figure 5). However, it is worth noting that these four physical parameters are not the sole criteria. Other parameters may include soil type and pH, soil moisture and a soil temperature range of 24.4–30 °C. Using some other microclimate parameters, such as air temperature (°C), air humidity (%) and sunlight intensity (lx), can also yield unique results.

Table 3.
Criteria used to map banana suitability.

Figure 4.
Distribution map of suitable levels of banana cultivation over Bali.
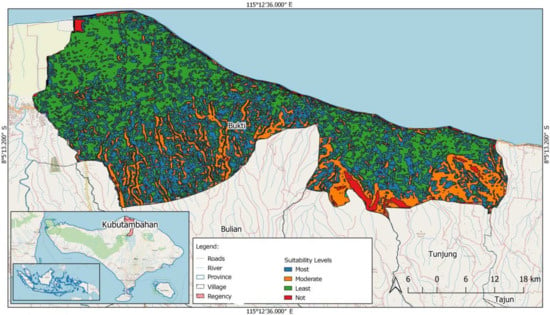
Figure 5.
Distribution map of land suitability for banana cultivation) in Bukti village.
According to the map, northern Bali is larger than southern Bali, although it has less land suitability (S3), compared to that of the latter (S1). Rainfall and elevation seem to be influencing these results quite significantly. However, it is worth noting that these differences are just to do with the type of scale used in the map. For a smaller area, Wikantika et al. [6] proved that a similar method can produce more details and successfully locate the most suitable (S1) places for banana cultivation (Figure 5). This result became a scientific foundation for Banana Smart villages in Bukti village. On the other hand, the GIS helps the community know the distribution of land suitability in Bukti village. The most suitable (S1) location was found to have the potential for other agricultural activities, especially horticulture. The success of this study indicates that it can be replicated in other regions of Indonesia.
Figure 4 and Figure 5 illustrate geographical variations (as demonstrated by the GIS analysis) in land suitability levels for banana cultivation. The same method and data were applied to analyse this stage resulting in two different results. There is a least suitable area improved to moderate and most suitable areas. The critical point of this lesson is that it is necessary to consider coverage area as the unit of analysis, before conducting suitability analysis. In a smaller area, such as villages, limiting the area may help to improve the suitability level for banana cultivation.
2.3. The Journey to Discover the Musa Troglodytarum
Musa troglodytarum was discovered in two regions: near the Galunggung mountain (the Padakembang villages and the Tasikmalaya regency) and Seram Island, Maluku. The first location is more prominent when it comes to biodiversity of bananas. The origin of this banana can be traced to Maluku, Papua, Australia and many other islands in Oceania [26]. This banana (cultivar) is popularly known as the Fe’i banana, Tongka Langit and Ranggap. The Fe’i banana’s finger is larger than the other cultivars, with a brilliant orange to red peel and yellow-orange pulp (Figure 6). It is also known for its high beta-carotene levels [27].

Figure 6.
The observers (left to right: local people of Padakembang village, Carolin, Giashinta Stefani, Ketut Wikantika, Agus Sutanto, Intan Fatmawati) with the Musa troglodytarum fruits found in the Padakembang villages near the Galunggung mountains, Tasikmalaya regency.
Besides Fe’i bananas, there are many banana cultivars in Seram Island. Relevant edaphic factors, including soil pH, soil moisture (%), temperature (°C), and alluvial soil, might be why the other 18 banana cultivars are found in this area and contribute to the rich diversity of bananas in Maluku. In general, soil pH ranges from 6.1–7.3, while soil moisture ranges from 25–79%. Soil temperature was recorded in range 24.4–30 °C, explaining the other two values. Meanwhile, microclimate conditions, such as air temperature (°C), air humidity (%) and sunlight intensity (lx), were observed in ranges of 25–29.4 °C, 68–93% and 5.770–82,100 (lx). The complete list of banana cultivars is shown in Table 4.

Table 4.
The richness of banana cultivars in Maluku.
Similar to the conditions in Maluku, the volcanic soil of the Padakembang villages Tasikmalaya regency was a major edaphic factor that helped foster the healthy growth of Ranggap. The soil’s pH ranged from 5–7, while soil moisture ranged from 40–85%, with a soil temperature of 23–24.32 °C. As for microclimatic conditions, air temperature (°C), humidity (%) and sunlight intensity (lx) were recorded in ranges of 25–26 °C, 82–85% and 52,000–56,000 (lx). These conditions enabled the healthy growth of the other 14 cultivars, including Rajabulu (AAB), Gancan (AA), Rejang (AA), Susu (AAB), Nangka (AAA), Klutuk (BB), Ambon lumut (AAA), Udang (AAA), Mas (AA), Limpiang (AAA), Yangambi (AAA), Klutuk wulung (BB), and Longong (AAB).
2.4. Use of Bananas for Community Developments and the Role of Ethnography and Domestication in Improving Banana Biodiversity
A banana is not merely a fruit or herb; it carries significant socio-cultural values. Especially for the Hindu people in Bali, for whom bananas and their leaves are sacred and used in prayers [28,29]. Besides this, the banana has the potential to enable community development. We decided to put this in practice using our research experience by transforming a traditional Bukti village into a smart village, calling it the Banana Smart Village (BSV). We then requested stakeholders to invest in the BSV community development programme in 2018. The programme aimed to develop the community of Bukti village, especially by equipping them with agricultural practices that can better cultivate bananas. This activity involved tissue culture and zero-waste technology. Geospatial science, UAV-based RS technology, and participative mapping for increasing economic values was introduced and incorporated through an Android e-commerce app called ‘Antar-Antar Pisang’ for marketing bananas [30].
The local distribution of these fruits reflects the economic value [31], ethnobotany and culture of the local community. Ethnobotany is defined as the science that connects the wisdom of the local community with the utilisation of vegetable resources, so that they can be developed and used efficiently [32]. A study conducted by Hapsari et al. [33] shows that understanding bananas locally can enrich the domestication of several banana cultivars, as well as develop understanding of the needs of the local communities. From the language perspective, in many places in Indonesia, the local community often uses its language to describe banana fruits. The name of banana has similar meaning to gedang (Javanese). It is also considered pitedahe gesang, which means lesson or guidance in Indonesian. An overview of the archaeobotany, various uses, and popularity of bananas, can also play a role in ethnography [33]. Ethnographic studies are essential to domesticating bananas and understanding these herbs from diverse viewpoints. Langhe et al. [2] mention using a multidisciplinary approach to auxiliary sciences, such as archaeobotany, genetics, linguistics (local languages), and phytogeography, to study a community’s socio-cultural relations and environment on the ground. [33] even tried to prepare an inventory of the various benefits and uses of the local banana cultivars grown in the regions of East Java (Figure 7).

Figure 7.
Banana Klutuk (Musa balbisana colla) with leaves used for wrapping food and Ensete glaucum, and Musa velutina for decorating ornaments ].
As shown by [32], various species of banana (e.g., Kepok kuning, Ambon Lumut, and Mas) are a source of food, medicinal ingredients, animal feed and agricultural materials. Meanwhile, in other areas, such as Campakamulya village and Cimaung-Bandung, the dominant banana cultivar is Ambon [35]. Its prevalence occurred quite accidently, as its cultivation in these areas was done just to utilise vacant land around houses [31]. Intentional forms of cultivar domestication can be found in banana plantations. For example, in the Lampung area, there is a monoculture of two cultivars, namely Cavendish and Mas.
3. Results and Discussion
3.1. Observing Banana Fruits through Spectral Reflectance
There are two methods for monitoring bananas: disturbed and undisturbed. The undisturbed method employs the response of energy transmitted from sun radiation for a multispectral radiometer, while the hyperspectral method uses synthetic energy to observe bananas, and then light is reflected to the sensor on the radiometer. It helps observe the changes occurring in various parts of the fruit, such as the colourisation of the banana peel during the ripening period. The disturbed method is more useful in observing biochemical changes in the flesh, including variations in water and sugar content, and for studying genetic characteristics that play a role in the growth of the fruit. An example of this is observing the dominant enzyme that controls the ripening time of the banana.
These two studies are clear examples of utilising spectral reflectance acquired from a multispectral and hyperspectral spectroradiometer, in addition to Brix detection, for studying banana fruits [11,12]. Although both are preliminary studies, the methods used here show the potential of RS technologies in delivering bio-physical information about fruits through undisturbed methods. One of the biophysical parameters observed was the estimated water content for Rajabulu or Musa paradisiaca. The RS technology was used to observe this fruit for twelve days, i.e., it required ripening time.
Several methods can be used to detect the water content of Rajabulu or Musa paradisiaca. The first method involves either GNDWI, which requires the average reflectance value mandated for the near-infrared (NIR) and shortwave infrared (SWIR) regions (Equation (3)), or the MNDWI, which needs the average reflectance value that is mandated for the green and NIR regions (Equation (4)). Both indices are modified versions of the normalised difference water index (NDWI), previously proposed by [36,37]. The second method uses a combination of the spectral band with a peak (highest value) and a valley (lowest value), including the water content index (WCI) (Equation (5)), a regression based on soil moisture and green band (RWC) (Equation (6)), and another combination of three bands in the fifth-day spectral reflectance (5th WCI) (Equation (7)). The input data for these proposed formulae were spectral reflectance values acquired from a multispectral radiometer, especially the MSR16R produced by Cropscan. Inc at Rochester, Minessota, The United States of America.
However, this preliminary study was not quite accurate, highlighting the need for further studies. The proposed method for estimating the water content includes two modified NDWI fruits and three water content indices, given the varying result of water content, but it is still in an acceptable range. Based on the RMSE value, the indices generated slightly varying values (WCI: 0.50; RWC: 0.51; 5th WCI: 0.92). Moreover, sufficient light sources (i.e., sunlight) were necessary for running the spectroradiometer to improve the observation results. Even the value calculations had to be supported by laboratory data of the water content. It was essential for enhancing the predicted quality, precision and accuracy. Figure 8 shows trends in the Rajabulu’s colourisation, especially with regards to its water content.
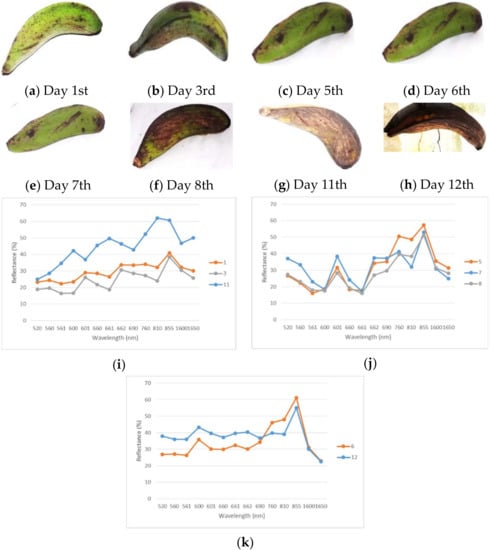
Figure 8.
The ripening process of the Rajabulu in the 1st, 3rd, 5th, 6th, 7th, 8th, 11th, and 12th day (a–h) and classification of spectral reflectance, based on its repetition and similar shape (i–k).
However, more studies are needed to formulate a standard operating procedure for acquiring data from banana fruits by considering aspects like the ripening time for the treated and untreated samples. Other studies that focus especially on the production and disease management of bananas can also be used to produce maps for yield estimation and disease distribution on a plantation scale. A controlled study by Koesyani et al. [12] demonstrated the steps for observing Cavendish banana fingers via an ASD hyperspectral spectroradiometer to obtain information related to sugar and starch content (Figure 9). It showed that Brix values increased during the observation time. The value of TSS was higher during the fruit ripening process. In general, the TSS value of bananas from the 1st day until the 7th showed an increasing trend, indicating that the sugar content increased during the ripening stage. On the 1st day, the TSS value was the lowest, which might correlate with low sugar content. On the 3rd day, it became higher.
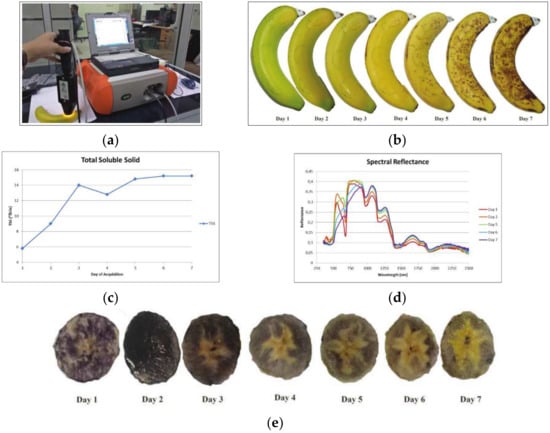
Figure 9.
Spectral reflectance acquisition of Cavendish banana using ASD Hyperspectral spectroradiometer (a) in seven different day observations (b), and its relationship to Brix and Starch content change (c–e).
However, there was a decrease in the TSS value on day 4 (12.8°Brix). This was followed by another spike, with the highest TSS value recorded on days 6 and 7, where the banana had a TSS value of 15.2°Brix. The anomaly on day four may be due to variation in the physiological age of the banana fingers used. The starch analysis was done by transversely cutting the banana fruit into pieces of 3–5 cm thickness, followed by dipping them in iodine solution for about 5 s every seven consecutive days of the observation. The presence of starch was recorded by the black patterns forming on the banana’s flesh as a result of its reaction with iodine. Dwivany et al. [38] reported a similar Cavendish banana starch content pattern. The conversion of starch to sugar has a consistent pattern in most banana pulps, starting from the middle part of the pulp and then spreading towards the edges.
3.2. Observing the Diseases Inflicting Banana Herbs through Spectral Reflectance
Banana plants can be susceptible to diseases through Fusarium bacteria, rhizome and even via the pseudostem [39,40]. The Fusarium oxysporum sp. may have a significant role in causing Panama diseases. This disease can be observed through discolouration of the leaves. The colour changes from green to yellowish-green or brownish [41,42,43]. It can be transmitted via water, air and even farmers and their equipment. It enters the plant through its pseudostem [35] and causes the herbs to lose their productivity [44]. As a response to this issue, research conducted by [10] has demonstrated the benefits of using RS data integration. UAV images and three different satellite devices, the WorldView2 (WV-2), PlanetScope (PS), and Sentinel 2(S2), provide low to high spatial resolution reflectance images that can be used to detect diseases like BBTD, and the Xanthomonas wilt of the banana (BXW) caused by Xanthomonas campestris.
Apart from these explanations, the most basic implementation of spectral values lies in how they help in understanding each stage of the banana herb’s growth. This is the reason why a phenological approach, that uses NDVI, is implemented here. It helps to provide data related to bio-physical parameters, such as chlorophyll, nitrogen and water content. Incorporating Equation (1), chlorophyll can be calculated using modified chlorophyll absorption in the reflectance index (MCARI) spectrometer (Equation (8)), nitrogen content can be calculated using the modified vegetation ratio index (VRI) spectrometer (Equation (10)) [45], while water content can be measured by using a modification of the normalised difference infrared index (NDII) by Hardisky et al. [46] (Equation (8)). The spectral value of the canopy of the Klutuk or Musa balbisiana colla (Figure 10) was used as one of the samples (Table 5), along with the index values stated below (Equations (9) and (10)) (Table 6).

Figure 10.
(a) The MSR16R used for the spectral reflectance data acquisition of (b) Klutuk based on its size, from the shortest to the tallest.

Table 5.
Spectral reflectance of Klutuk, based on its size, from shortest to tallest.

Table 6.
The characteristic of vegetation indices of Pisang Klutuk using NDVI, VRI, MCARI, and NDII, respectively.
The four banana Klutuk plants have NDVI values of 0.333, 0.385, 0.390 and 0.396, respectively (Table 5). The increasing NDVI values indicate the plant’s excellent physical growth, development, and healthy condition. This is further indicated by the greenness level of the NDVI, which also represents the highest chlorophyll content of each plant (leaves) (Figure 11a). The MCARI spectrometer estimated the chlorophyll content of each plant, also showing an increasing range from 10.54–17.02 g. The relationship between NDVI values and the MCARI spectrometer is strongly positive (R2 = 0.92) (Figure 11a). NDVI also shows a strongly positive relationship with the VRI spectrometer (R2 = 0.94), and a medium positive relationship with the NDII spectrometer (R2 = 0.42) (Figure 11b).
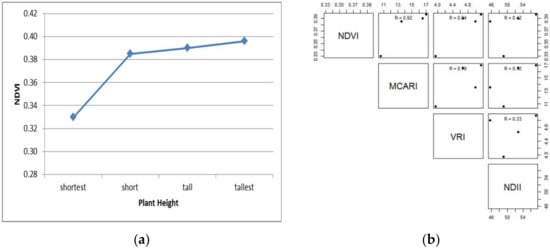
Figure 11.
The relationship between NDVI trends and other indices of Klutuk, based on its size, from shortest to tallest.The relationship between NDVI values and the MCARI spectrometer is strongly positive (R2 = 0.92) (Figure 11a). NDVI also shows a strongly positive relationship with the VRI spectrometer (R2 = 0.94), and a medium positive relationship with the NDII spectrometer (R2 = 0.42) (Figure 11b).
The MCARI spectrometer’s relationships with the VRI and NDII spectrometers also showed a strong relationship with R2 values of 0.8 and 0.72, respectively. Only the relationship between the VRI spectrometer and NDII spectrometer showed a weak positive relationship (R2 values ~ 0.33). It is also worth noting that the increasing NDVI values correlate with increasing chlorophyll, nitrogen and water content of the Klutuk banana herbs. However, the amount of nitrogen needed does not always accompany an increase in water demand. It shows the low relationship between the VRI spectrometer and the NDII spectrometer (Figure 11b).
3.3. Future Research
Scientists in the Center for Remote Sensing (CRS) and the School of Life Sciences and Technology of the Bandung Institute of Technology currently study bananas’ genomics, transcriptomics, and metabolomics. The studies regarding banana ripening via transcriptomics have been published as a transcriptome dataset of chitosan-treated Musa acuminata cv. Cavendish (AAA group) to study RNA profile during delayed ripening [47] and ethylene-treated Musa balbisiana cv. Pisang Klutuk Wulung (BB group) to study RNA profile of this late ripening banana [48]. For the metabolomics study, the metabolite profiles of chitosan and low temperature-treated banana was used to study the delay of the ripening mechanism on M. acuminata cv. Cavendish fruit [49]. A study on comparative genomics was also conducted to reveal variations among some ripening-related genes between M. acuminata and M. balbisiana, namely pectin methylesterase (PME) and pectin methylesterase inhibitor (PMEI) which are responsible for cell wall remodelling [50]. It also looked at beta-amylase (BAM), which plays a role in carbohydrate metabolism [51]. These findings extend the scope for future research on the ripening mechanism of the banana fruit by integrating its spectral reflectance with banana fruit, since omics study gives deep insight into this mechanism. Along with this, future study can focus on building the relationship between the spectral reflectance of banana and its genome, transcriptome, and metabolome characteristics during its ripening stage, using artificial intelligence (AI) for analysis. Remote sensing technology and data will become a leading and alternative approach to understand the function of banana herbs and fruits as candidates of functional food sources, due to their being rich in sugar, carbohydrate, and water. The findings of these studies could provide insights for other scientific fields, such as industry, food technology, post-harvest and eco-tourism.
4. Conclusions
Currently, the use of RS technology for studying the growth of banana herbs is gaining prominence due to its ability to incorporate data, such as spectral reflectance, aerial photographs and satellite images, yielding more accurate results. These advancements emphasise the need for a multidisciplinary study on bananas that will involve RS, biology, biodiversity, and biogeography perspectives. This could extend the scope of this study further, opening possibilities for innovations in various industries.
Author Contributions
The first author, K.W. is the principal investigator for the present study, while the co-authors contributed to implementing the proposed method. Certain parts of the research are still in progress; M.F.G. is conducting the remote sensing aspect of it while the laboratory analysis on the biology and biodiversity of bananas is being conducted by F.M.D. and C.N. In addition to this, L.F.Y. has assisted in preparation of this manuscript, while A.S. has provided guidance for the study of the Fe’i bananas. All authors have read and agreed to the published version of the manuscript.
Funding
This article review was a part of the research supported by Institut Teknologi Bandung under Riset Unggulan Program No. 159/IT1.B07.1/TA.00/2021 led by Ketut Wikantika, the Institute of Research and Community Service (LPPM-ITB) Institut Teknologi Bandung, and the Indonesia Research Foundation of Biogeography and Biodiversity (INABIG) in 2021.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| No | Abbreviation | Expansion |
| 1 | BAM | Beta-amylase |
| 2 | BBTD | Bunchy top disease |
| 3 | DSM | Digital surface model |
| 4 | DTM | Digital terrain model |
| 5 | GIS | Geographic information systems |
| 6 | GNDWI | Green normalized difference water index |
| 7 | GSD | Ground surface distance |
| 8 | OBIA | Implemented object image analysis |
| 9 | LAI | Leaf area index |
| 10 | MODIS | Moderate resolution imaging spectroradiometer |
| 11 | MCARI | Modified chlorophyll absorption in reflectance index |
| 12 | NIR | Near-infrared |
| 13 | NDII | Normalized difference infrared index |
| 14 | NDVI | Normalized difference vegetation index |
| 15 | NDWI | Normalized difference water index |
| 16 | PME | Pectin methylesterase |
| 17 | PMEI | Pectin methylesterase inhibitor |
| 18 | RGB | Red, green, and blue |
| 19 | RWC | Regression water content |
| 20 | RS | Remote sensing |
| 21 | SEBAL | Surface energy balance algorithm for land |
| 22 | TIRS | Thermal infrared sensors |
| 23 | UAV | Unmanned aerial vehicle |
| 24 | VRI | Vegetation ratio index |
| 25 | WCI | Water content index |
| 26 | BXW | Xanthomonas wilt of banana |
References
- D’hont, A.; Denoeud, F.; Aury, J.M.; Baurens, F.C.; Carreel, F.; Garsmeur, O.; Noel, B.; Bocs, S.; Droc, G.; Rouard, M.; et al. The banana (Musa acuminata) genome and the evolution of monocotyledonous plants. Nature 2012, 488, 213–217. [Google Scholar] [CrossRef] [Green Version]
- De Langhe, E.; Vrydaghs, L.; De, P.; Perrier, X.; Denham, T. Why Bananas Matter: An introduction to the history of banana domestication. Ethnobot. Res. Appl. 2009, 7, 165–177. [Google Scholar] [CrossRef] [Green Version]
- Simmonds, N.W. The Evolution of the Bananas; Longmans: London, UK, 1962. [Google Scholar]
- Bakry, F.; Carreel, F.; Jenny, C.; Horry, J.-P. Genetic Improvement of Banana. In Breeding Plantation Tree Crops; Jain, S.M., Priyadarshan, P.M., Eds.; Springer: New York, NY, USA, 2009; ISBN 978-0-387-71199-7. [Google Scholar]
- Perrier, X.; De Langhe, E.; Donohue, M.; Lentfer, C.; Vrydaghs, L.; Bakry, F.; Carreel, F.; Hippolyte, I.; Horry, J.P.; Jenny, C.; et al. Multidisciplinary perspectives on (Musa spp.) domestication. Proc. Natl. Acad. Sci. USA 2011, 108, 11311–11318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wikantika, K.; Dwivanny, F.M.; Ghazali, M.F.; Sutanto, A.; Kamalesha, G. Pisang Indonesia, 1st ed.; ITB Press: Bandung, Indonesia, 2021. [Google Scholar]
- FAO. Banana Market Review–Preliminary Results 2020; FAO: Rome, Italy, 2021; Volume 9. [Google Scholar]
- Harto, A.B.; Prastiwi, P.A.D.; Ariadji, F.N.; Suwardhi, D.; Dwivany, F.M.; Nuarsa, I.W.; Wikantika, K. Identification of banana plants from unmanned aerial vehicles (UAV) photos using object based image analysis (OBIA) method (a case study in Sayang Village, Jatinangor District, West Java). HAYATI J. Biosci. 2019, 26, 7–14. [Google Scholar] [CrossRef]
- Ye, H.; Huang, W.; Huang, S.; Cui, B.; Dong, Y.; Guo, A.; Ren, Y.; Jin, Y. Recognition of banana Fusarium wilt based on UAV remote sensing. Remote Sens. 2020, 12, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Gomez Selvaraj, M.; Vergara, A.; Montenegro, F.; Alonso Ruiz, H.; Safari, N.; Raymaekers, D.; Ocimati, W.; Ntamwira, J.; Tits, L.; Omondi, A.B.; et al. Detection of banana plants and their major diseases through aerial images and machine learning methods: A case study in DR Congo and Republic of Benin. ISPRS J. Photogramm. Remote Sens. 2020, 169, 110–124. [Google Scholar] [CrossRef]
- Ghazali, M.F.; Wikantika, K.; Dwivany, F.M. A preliminary result of monitoring banana (Musa sp) ripening process and its relationship with water content based on remote sensing analysis. Hayati J. Biosci. 2018, 22. [Google Scholar]
- Koesyani, S.F.D.; Dwivany, F.M.; Wikantika, K. Spectral reflectance analysis of banana in fruit ripening process. In Proceedings of the Proceedings Asian Conference on Remote Sensing, Kuala Lumpur, Malaysia, 15–19 October 2018; 2018; pp. 724–729. [Google Scholar]
- Sinha, P.; Robson, A.; Schneider, D.; Kilic, T.; Mugera, H.K.; Ilukor, J.; Tindamanyire, J.M. The potential of in-situ hyperspectral remote sensing for differentiating 12 banana genotypes grown in Uganda. ISPRS J. Photogramm. Remote Sens. 2020, 167, 85–103. [Google Scholar] [CrossRef]
- Neupane, B.; Horanont, T.; Hung, N.D. Deep learning based banana plant detection and counting using high-resolution red-green-blue (RGB) images collected from unmanned aerial vehicle (UAV). PLoS One 2019, 14, 1–22. [Google Scholar] [CrossRef]
- Dare, P.M. Shadow analysis in high-resolution satellite imagery of urban areas. Photogramm. Eng. Remote Sensing 2005, 71, 169–177. [Google Scholar] [CrossRef] [Green Version]
- Filippi, A.M.; Güneralp, İ. Influence of shadow removal on image classification in riverine environments. Opt. Lett. 2013, 38, 1676. [Google Scholar] [CrossRef] [PubMed]
- Delegido, J.; Verrelst, J.; Alonso, L.; Moreno, J. Evaluation of sentinel-2 red-edge bands for empirical estimation of green LAI and chlorophyll content. Sensors 2011, 11, 7063–7081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bramich, J.; Bolch, C.J.S.; Fischer, A. Improved red-edge chlorophyll-a detection for Sentinel 2. Ecol. Indic. 2021, 120, 106876. [Google Scholar] [CrossRef]
- Clevers, J.G.P.W.; Gitelson, A.A. Remote estimation of crop and grass chlorophyll and nitrogen content using red-edge bands on sentinel-2 and-3. Int. J. Appl. Earth Obs. Geoinf. 2013, 23, 344–351. [Google Scholar] [CrossRef]
- Xiao, C.; Li, P.; Feng, Z.; Liu, Y.; Zhang, X. Sentinel-2 red-edge spectral indices (RESI) suitability for mapping rubber boom in Luang Namtha Province, northern Lao PDR. Int. J. Appl. Earth Obs. Geoinf. 2020, 93, 102176. [Google Scholar] [CrossRef]
- Choosumrong, S.; Hataitara, R.; Mekarun, P. Application of UAV multi - spectral camera for estimating bananas disease infestations in complex farming in Phitsanulok Province. In Proceedings of the International Symposium on Geoinformatics for Spatial Infrastructure Development in Earth and Allied Sciences, Phitsanulok, Thailand, 2–4 September 2021; Japan Vietnam Geoinformation Concorcium: Pithsanulok, Thailand, 2021; p. 6. [Google Scholar]
- Silva, T.T.S.; Guerra, H.O.C.; Silva, B.B.; Santos, C.L.M.; Guimarães, J.P.; Santos, J.S. dos Evapotranspiration of banana using the SEBAL algorithm in an irrigated perimeter from the Northeastern Brazil. Brazilian J. Agric. Environ. Eng. 2021, 25, 149–155. [Google Scholar]
- Johansen, K.; Phinn, S. Mapping banana plantations in North Australia from objectoriented classification of SPOT-5 data. In Proceedings of the 28th Asian Conference on Remote Sensing: (ACRS 2007), Kuala Lumpur, Malaysia, 12–16 November 2007; 2007; 1, pp. 139–144. [Google Scholar]
- Nuarsa, I.W.; Dibia, I.N.; Wikantika, K.; Suwardhi, D.; Rai, I.N. Gis based analysis of agroclimate land suitability for Banana plants in Bali Province, Indonesia. HAYATI J. Biosci. 2018, 25, 11–17. [Google Scholar] [CrossRef]
- Wikantika, K.; Ghazali, M.F.; Dwivanny, F.M.; Yayusman, L.F. Desa Bukti: Desa Cerdas Berbasis Pisang Pertama di dunia, 1st ed.; ITB Press: Bandung, Indonesia, 2021. [Google Scholar]
- Hiariej, A.; Laras Arumingtyas, E.; Widoretno, W.; Azrianingsih, R. Phenotypic Variation of Fei Banana (Musa troglodytarum L.) Originated Maluku Islands. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 652–658. [Google Scholar]
- Dwivany, F.M.; Stefani, G.; Sutanto, A.; Nugrahapraja, H.; Wikantika, K.; Hiariej, A.; Hidayat, T.; Rai, I.N.; Sukriandi, N. Genetic relationship between tongka langit bananas (Musa troglodytarum l.) from galunggung and maluku, indonesia, based on its2. HAYATI J. Biosci. 2020, 27, 258–265. [Google Scholar] [CrossRef]
- Ernatip Upacara ‘Ngaben’ Di Desa Rama Agung–Bengkulu Utara. J. Penelit. Sej. Dan Budaya 2019, 4, 1115–1133. [CrossRef]
- Sunariani, N.N.; Sukarsa, M.; Budhi, M.K.S.; Marhaen, A. Kontribusi Pelaksanaan Ritual Hindu Terhadap Kesempatan Kerja Dan Kesejahteraan Masyarakat Di Kabupaten Badung Provinsi Bali (Studi Kasus Mlaspas Dan Ngenteg Linggih Di Pura Pasek Preteka Desa Abiansemal). J. Ekon. Kuantitatif Terap. 2015, 7, 145–154. [Google Scholar] [CrossRef]
- Banana Smart Village Biodiversity for Better Society. Available online: https://bananasmartvillages-gisitb.opendata.arcgis.com/ (accessed on 14 December 2021).
- Ismail, A.; Rachmadi, M.; Bana, N. Eksplorasi jenis-jenis pisang plantain lokal asal desa Sukaharja dan desa Sukamulih Tasikmalaya, Jawa barat sebagai sumber bibit unggul. J. Apl. Ipteks untuk Masy. 2014, 3, 92–97. [Google Scholar]
- Kasrina, K.; Zulaikha, A. Pisang Buah (Musa Spp): Keragaman Dan Etnobotaninya Pada Masyarakat Di Desa Sri Kuncoro Kecamatan Pondok Kelapa Kabupaten Bengkulu Tengah. In Proceedings of the Prosiding Semirata FMIPA Universitas Lampung, Iguazu, Brazil, 10–12 May 2013; pp. 33–40. [Google Scholar]
- Hapsari, L.; Kennedy, J.; Lestari, D.A.; Masrum, A.; Lestarini, W. Ethnobotanical survey of bananas (Musaceae) in Six districts of East Java, Indonesia. Biodiversitas 2017, 18, 160–174. [Google Scholar] [CrossRef]
- Cheryl, N. Pendataan kesesuaian lahan berupa edafik dan mikroklimat kultivar pisang di pulau Ambon dan pulau Seram, Provinsi Maluku; ITB: Bandung, Indonesia, 2018. [Google Scholar]
- Dwivany, F.M.; Ghazali, M.F.; Suwardhi, D.; Sutanto, A.; Hiariej, A.; Puturuhu, F.; Meitha, K.; Nugrahapraja, H.; Yayusman, L.F.; Kamalesha, G.; et al. Karakterisasi Pisang Berbasis GeoBioSpektral; ITB: Bandung, Indonesia, 2019; ISBN 978-623-7165-62-0. [Google Scholar]
- Gao, B.C. NDWI-A Normalized Difference Water Index for Remote Sensing of Vegetation Liquid Water from Space. Remote Sens. Environ. 1996, 58, 257–266. [Google Scholar] [CrossRef]
- McFeeters, S.K. The Use of The Normalized Difference Water Index (NDWI) in The Delineation of Water Feature. Int. J. Remote Sens. 1996, 17, 425–1432. [Google Scholar] [CrossRef]
- Dwivany, F.; Esyanti, R.R.; Robertlee, J.; Paramaputra, I.C.; Permatadewi, R.K.; Tambun, D.H.; Handayani, R.U.; Sa Pratiwi, A.; Zaskia, H. Environment effect on fruit ripening related gene to develop a new post harvest technology. AIP Conf. Proc. 2014, 1589, 285–287. [Google Scholar] [CrossRef]
- Ploetz, R.C.; Thomas, J.E.; Slabaugh, W.R. Diseases of Banana and Plantain. In Diseases of Tropical Fruit Crops; Ploetz, R.C., Ed.; CABI Publishing: Oxfordshire, UK, 2003; ISBN 0851993907. [Google Scholar]
- Jones, D.R.; Daniells, J.W. Introduction to Banana, Abacá and Enset. In Handbook of Diseases of Banana, Abacá and Enset; Jones, D.R., Ed.; CABI: Oxfordshire, UK, 2018; ISBN 9781780647197. [Google Scholar]
- Soesanto, L.; Mugiastuti, E.; Ahmad, F. Diagnosis Lima Penyakit Utama Karena Jamur Pada 100 Kultivar Bibit Pisang. J. Hama dan Penyakit Tumbuh. Trop. 2013, 12, 36–45. [Google Scholar] [CrossRef]
- Bioversity International. Screening for Resistance to Fusarium Wilt; Bioversity International: Rome, Italy, 2014; Volume 4, Available online: https://www.bioversityinternational.org/e-library/publications/detail/use-of-banana-diversity-for-nutritious-diets/ (accessed on 14 December 2021).
- Huda, M. Pengendalian layu fusarium pada tanaman pisang (Musa paradisiaca l.) secara kultur teknis dan hayati; IPB (Bogor Agricultural University): Bogor Regency, Indonesia, 2010; Available online: https://repository.ipb.ac.id/handle/123456789/27524?show=full (accessed on 14 December 2021).
- Loeillet, D. Close-up banana. Fruitrop Magazine, Montpellier, France, April 2016; 66–124. [Google Scholar]
- Xue, L.; Cao, W.; Luo, W.; Dai, T.; Zhu, Y. Monitoring Leaf Nitrogen Status in Rice with Canopy Spectral Reflectance. Agron. J. 2004, 96, 135–142. [Google Scholar] [CrossRef]
- Hardisky, M.A.; Klemas, V.; Smart, R.M. The Influence of Soil Salinity, Growth Form, and Leaf Moisture on-the Spectral Radiance of ~ partina alterniflora Canopies. Photogramm. Eng. Remote Sens. 1983, 49, 77–83. [Google Scholar]
- Dwivany, F.M.; Nugrahapraja, H.; Fukusaki, E.; Putri, S.P.; Novianti, C.; Radjasa, S.K.; Fauziah, T.; Nirmala Sari, L.D. Dataset of Cavendish banana transcriptome in response to chitosan coating application. Data Br. 2020, 29. [Google Scholar] [CrossRef] [PubMed]
- Martha, F.; Nugrahapraja, H. Transcriptome dataset of ethylene-treated Klutuk Wulung banana. Data Br. 2021, 38, 107376. [Google Scholar] [CrossRef]
- Aulia, A.; Parijadi, R.; Yamamoto, K.; Maulana, M.; Ikram, M. Metabolome Analysis of Banana (Musa acuminata) Treated With Chitosan Coating and Low Temperature Reveals Different Mechanisms Modulating Delayed Ripening. Front. Sustain. Food Syst. 2022, 6, 1–11. [Google Scholar] [CrossRef]
- Nugrahapraja, H.; Putri, A.E.; Martha, D.F. Genome-wide Identification and Characterization of the Pectin Methylesterase (PME) and Pectin Methylesterase Inhibitor (PMEI) Gene Family in the Banana A-genome (Musa acuminata) and B-genome (Musa balbisiana). Res. J. Biotechnol. 2021, 16, 179–191. [Google Scholar]
- Setiabudi, E.; Meitha, K.; Dwivany, F.M. In silico characterization and comparison of the fruit ripening related beta-amylase (BAM) gene family in banana genome A and B. Indones. J. Biotechnol. 2021, 26, 175–182. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).