Underwater Photogrammetry Captures the Initial Recovery of a Coral Reef at Lalo Atoll
(This article belongs to the Section Marine Diversity)
Abstract
:1. Introduction
2. Materials and Methods
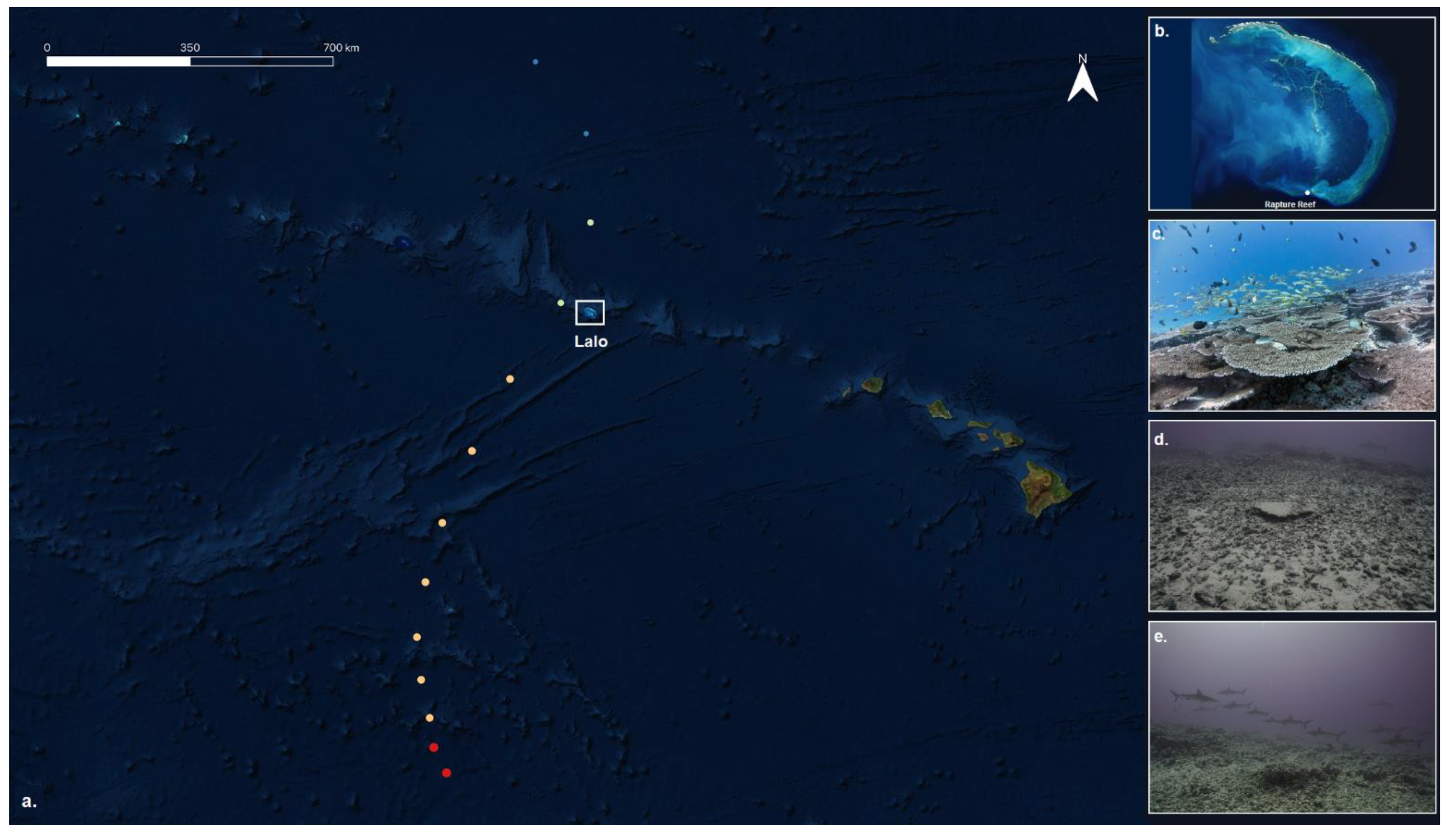
2.1. Fish and Benthic Photogrammetry Surveys
2.2. Generation and Analysis of 3D Models
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Species | 4 August 2021 | 17 August 2021 | Size × Count |
|---|---|---|---|
| Acanthuridae | |||
| Acanthurus olivaceus (H) | x | x | |
| Naso lituratus (H) | x | ||
| Balistidae | |||
| Melichthys niger (P) | x | x | |
| Melichthys vidua (H) | x | ||
| Sufflamen bursa (I) | x | x | |
| Sufflamen fraenatum (I) | x | ||
| Chaetodontidae | |||
| Chaetodon fremblii (I) | x | x | 8 cm × 4 |
| Chaetodon kleinii (P) | x | x | 7 cm × 3 |
| Heniochus diphreutes (P) | |||
| Cirrhitidae | |||
| Cirrhitops fasciatus (I) | x | x | |
| Fistulariidae | |||
| Fistularia commersonii (Pis) | x | ||
| Labridae | |||
| Anampses chrysocephalus (I) | x | ||
| Bodianus albotaeniatus (I) | x | x | 31 cm × 1 |
| Coris ballieui (I) | x | x | |
| Coris gaimard (I) | x | ||
| Coris venusta (I) | x | ||
| Labroides phthirophagus (I) | x | x | |
| Novaculichthys taeniourus (I) | x | ||
| Pseudocheilinus evanidus (I) | x | x | 6 cm × 1 |
| Stethojulis balteata (I) | x | ||
| Thalassoma duperrey (I) | x | ||
| Thalassoma duperrey/quinquevittatum (I) | x | 11 cm × 6, 16 cm × 1 | |
| Lutjanidae | |||
| Aprion virescens (Pis) | x | 54 cm × 1 | |
| Lutjanus kasmira (I) | x | x | |
| Monacanthidae | |||
| Cantherhines verecundus (H) | x | ||
| Pervagor spilosoma (O) | x | ||
| Mullidae | |||
| Mulloidichthys flavolineatus (I) | x | ||
| Parupeneus multifasciatus (I) | x | 17 cm × 1 | |
| Muraenidae | |||
| Gymnothorax meleagris (Pis) | x | ||
| Pomacanthidae | |||
| Apolemichthys arcuatus (I) | x | ||
| Centropyge fisheri (H) | x | x | |
| Centropyge potteri (H) | x | x | |
| Paracentropyge multifasciata (O) | |||
| Pomacentridae | |||
| Chromis hanui (P) | x | x | |
| Chromis vanderbilti (P) | x | x | 4 cm × 6 |
References
- Pearson, R.G. Recovery and recolonization of coral reefs. Mar. Ecol. Prog. Ser. 1981, 4, 105–122. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dollar, S.J.; Tribble, G.W. Recurrent storm disturbance and recovery: A long-term study of coral communities in Hawaii. Coral Reefs 1993, 12, 223–233. [Google Scholar] [CrossRef]
- Rogers, C.S.; Miller, J. Permanent ’phase shifts’ or reversible declines in coral cover? Lack of recovery of two coral reefs in St. John, US Virgin Islands. Mar. Ecol. Prog. Ser. 2006, 306, 103–114. [Google Scholar] [CrossRef]
- Houston, S.; Birchard, T. Tropical Cyclone Report: Hurricane Walaka (29 September–6 October 2018); Cnetral Pacific Hurricane Center: Honolulu, HI, USA, 2020.
- Pascoe, K.H.; Fukunaga, A.; Kosaki, R.K.; Burns, J.H.R. 3D assessment of a coral reef at Lalo Atoll reveals varying responses of habitat metrics following a catastrophic hurricane. Sci. Rep. 2021, 11, 12050. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, A.; Kosaki, R.K.; Hauk, B.B. Distribution and abundance of the introduced snapper Lutjanus kasmira (Forsskål, 1775) on shallow and mesophotic reefs of the Northwestern Hawaiian Islands. Bioinvasions Rec. 2017, 6, 259–268. [Google Scholar] [CrossRef]
- Meyer, C.G.; Holland, K.N.; Papastamatiou, Y.P. Seasonal and diel movements of giant trevally Caranx ignobilis at remote Hawaiian atolls: Implications for the design of Marine Protected Areas. Mar. Ecol. Prog. Ser. 2007, 333, 13–25. [Google Scholar] [CrossRef]
- Hiatt, R.W.; Strasburg, D.W. Ecological relationships of the fish fauna on coral reefs of the Marshall Islands. Ecol. Monogr. 1960, 30, 65–127. [Google Scholar] [CrossRef]
- Hobson, E.S. Feeding relationships of teleostean fishes on coral reefs in Kona, Hawaii. Fish. Bull. 1974, 72, 915–1031. [Google Scholar]
- Hoover, J.P. Hawaii’s Fish; Mutual Publishing: Honolulu, HI, USA, 1993; p. 183. [Google Scholar]
- Burns, J.H.R.; Delparte, D.; Gates, R.D.; Takabayashi, M. Integrating structure-from-motion photogrammetry with geospatial software as a novel technique for quantifying 3D ecological characteristics of coral reefs. PeerJ 2015, 3, e1077. [Google Scholar] [CrossRef] [PubMed]
- Swanson, D.; Bailey, H.; Schumacher, B.; Ferguson, M.; Vargas-Ángel, B. Ecosystem Sciences Division Standard Operating Procedures: Data Collection for Rapid Ecological Assessment Benthic Surveys; NOAA Technical Memorandum NMFS-PIFSC-71: Honolulu, HI, USA, 2018; p. 63.
- Jenness, J.S. Calculating landscape surface area from digital elevation models. Wildl. Soc. Bull. 2004, 32, 829–839. [Google Scholar] [CrossRef]
- Rogers, C.S. Responses of coral reefs and reef organisms to sedimentation. Mar. Ecol. Prog. Ser. 1990, 62, 185–202. [Google Scholar] [CrossRef]
- Smith, L.W. Status Review Report: Pocillopora Meandrina; National Marine Fisheries Service, Pacific Islands Regional Office: Honolulu, HI, USA, 2019.
- Fukunaga, A.; Kosaki, R.K.; Pascoe, K.H.; Burns, J.H.R. Fish assemblage structure in the Northwestern Hawaiian Islands is associated with the architectural complexity of coral-reef habitats. Diversity 2020, 12, 430. [Google Scholar] [CrossRef]
- Richardson, L.E.; Graham, N.A.J.; Pratchett, M.S.; Eurich, J.G.; Hoey, A.S. Mass coral bleaching causes biotic homogenization of reef fish assemblages. Globa. Chang. Biol. 2018, 24, 3117–3129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolfe, K.; Kenyon, T.M.; Mumby, P.J. The biology and ecology of coral rubble and implications for the future of coral reefs. Coral Reefs 2021, 40, 1769–1806. [Google Scholar] [CrossRef]
- Grigg, R.W.; Maragos, J.E. Recolonization of hermatypic corals on submerged lava flows in Hawaii. Ecology 1974, 55, 387–395. [Google Scholar] [CrossRef]
- Grigg, R.W. Community structure, succession and development of coral reefs in Hawaii. Mar. Ecol. Prog. Ser. 1983, 11, 1–14. [Google Scholar] [CrossRef]
- Ceccarelli, D.M.; McLeod, I.M.; Boström-Einarsson, L.; Bryan, S.E.; Chartrand, K.M.; Emslie, M.J.; Gibbs, M.T.; Gonzalez Rivero, M.; Hein, M.Y.; Heyward, A.; et al. Substrate stabilisation and small structures in coral restoration: State of knowledge, and considerations for management and implementation. PLoS ONE 2020, 15, e0240846. [Google Scholar] [CrossRef] [PubMed]
- Grigg, R.W. Acropora in Hawaii. Part 2. Zoogeography. Pac. Sci. 1981, 35, 15–24. [Google Scholar]
- Aeby, G.S. Outbreak of coral disease in the Northwestern Hawaiian Islands. Coral Reefs 2005, 24, 481. [Google Scholar] [CrossRef]
| 2017 1 | 2019 1 | 2021 | |
|---|---|---|---|
| Tabulate Acropora | 69.7 | - | - |
| Encrusting Porites | 0.8 | - | 0.04 |
| Encrusting Montipora | 0.1 | - | - |
| Branching Pocillopora | - | - | 0.31 |
| Crustose coralline algae | 1.7 | - | - |
| Macro and filamentous algae | - | - | 0.35 |
| Sponges | - | - | 0.66 |
| Hard substrata | 27.0 | 0.3 | - |
| Rubble | - | 67.9 | 80.66 |
| Sand | 0.7 | 31.8 | 17.70 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukunaga, A.; Pascoe, K.H.; Pugh, A.R.; Kosaki, R.K.; Burns, J.H.R. Underwater Photogrammetry Captures the Initial Recovery of a Coral Reef at Lalo Atoll. Diversity 2022, 14, 39. https://doi.org/10.3390/d14010039
Fukunaga A, Pascoe KH, Pugh AR, Kosaki RK, Burns JHR. Underwater Photogrammetry Captures the Initial Recovery of a Coral Reef at Lalo Atoll. Diversity. 2022; 14(1):39. https://doi.org/10.3390/d14010039
Chicago/Turabian StyleFukunaga, Atsuko, Kailey H. Pascoe, Ashley R. Pugh, Randall K. Kosaki, and John H. R. Burns. 2022. "Underwater Photogrammetry Captures the Initial Recovery of a Coral Reef at Lalo Atoll" Diversity 14, no. 1: 39. https://doi.org/10.3390/d14010039
APA StyleFukunaga, A., Pascoe, K. H., Pugh, A. R., Kosaki, R. K., & Burns, J. H. R. (2022). Underwater Photogrammetry Captures the Initial Recovery of a Coral Reef at Lalo Atoll. Diversity, 14(1), 39. https://doi.org/10.3390/d14010039








