Diversity Loss in Coralligenous Structuring Species Impacted by Fishing Gear and Marine Litter
Abstract
:1. Introduction
2. Materials and Methods
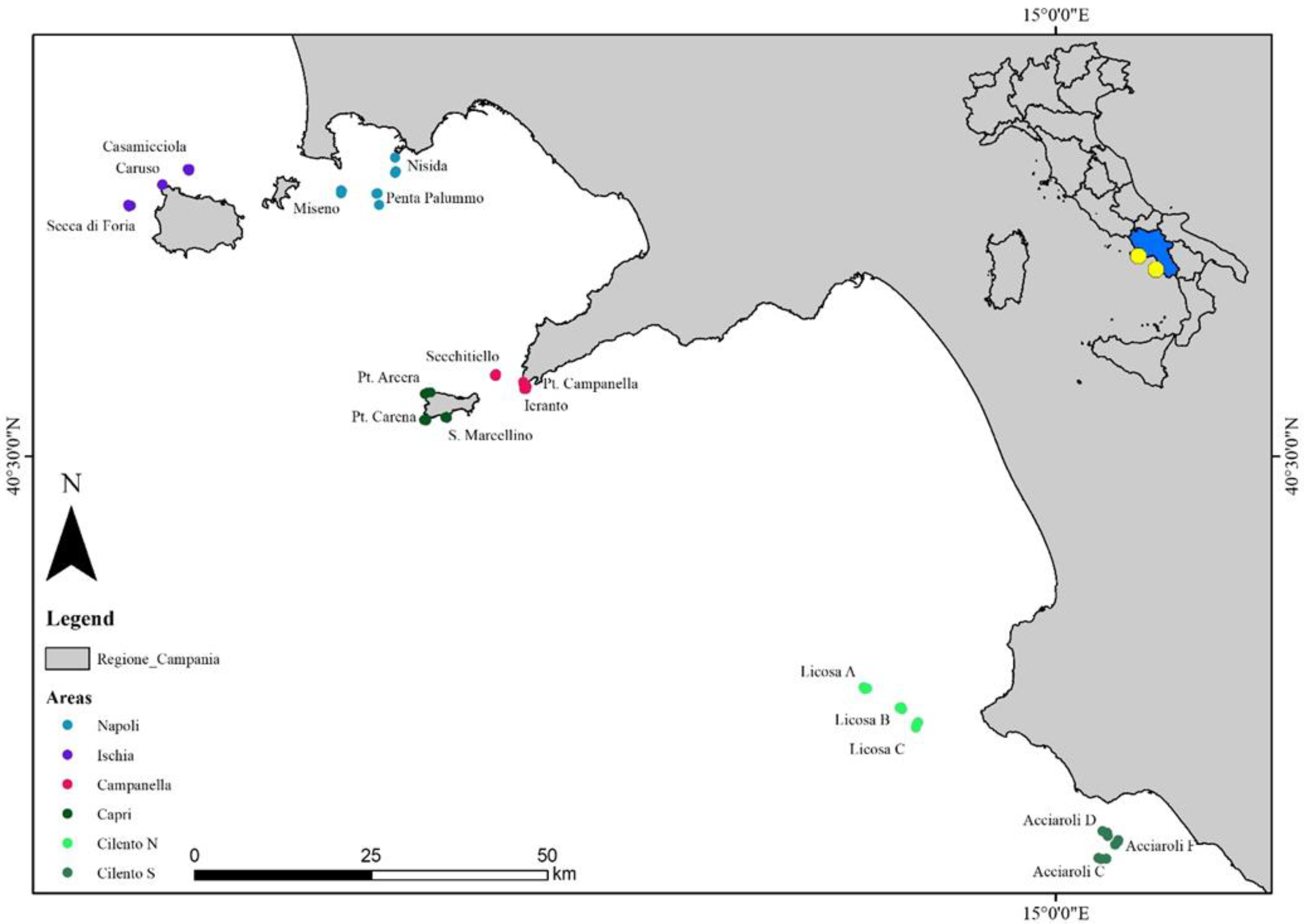
2.1. Study Area
2.2. Field Activities
2.3. Data Management
2.4. Statistical Analyses
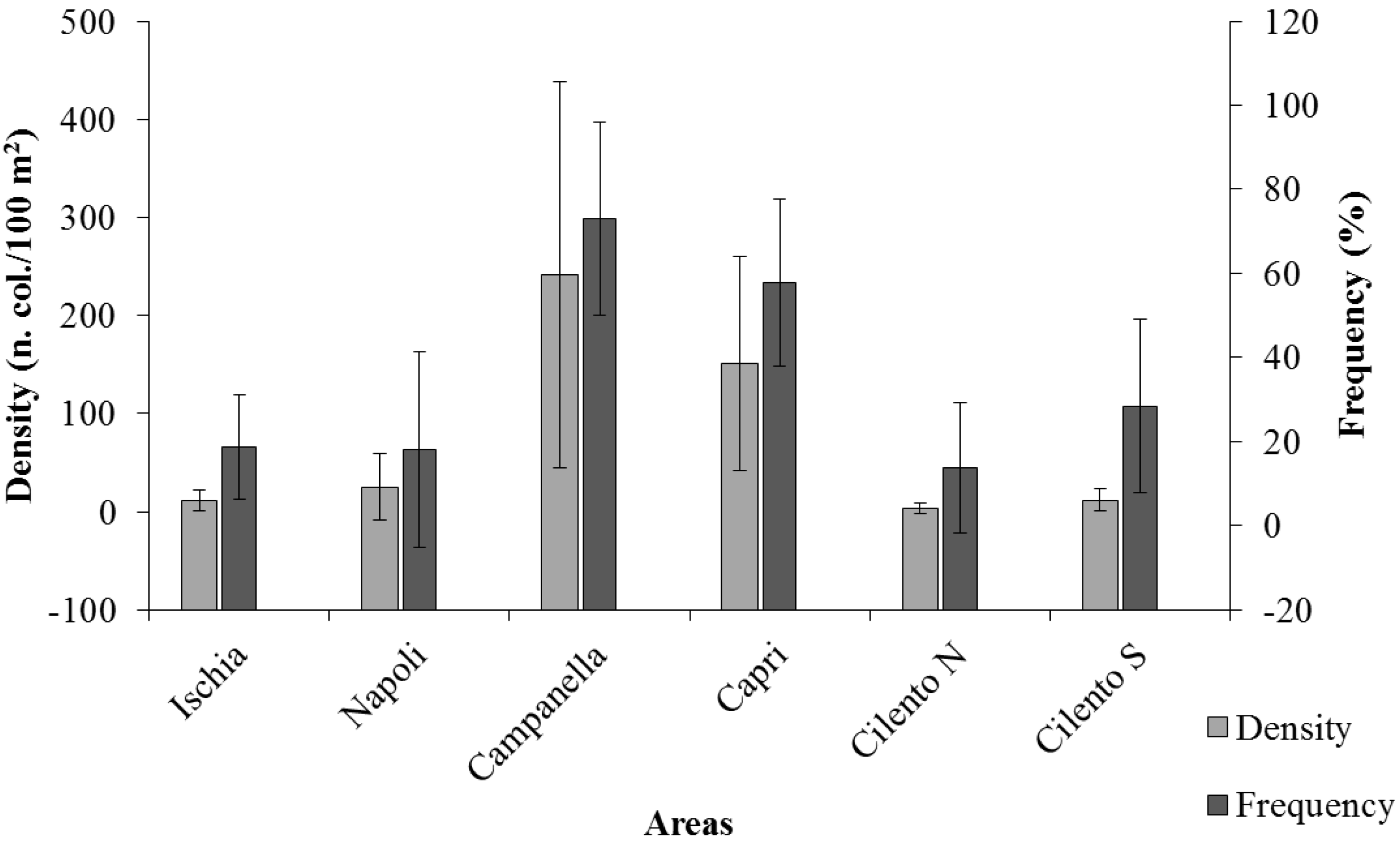
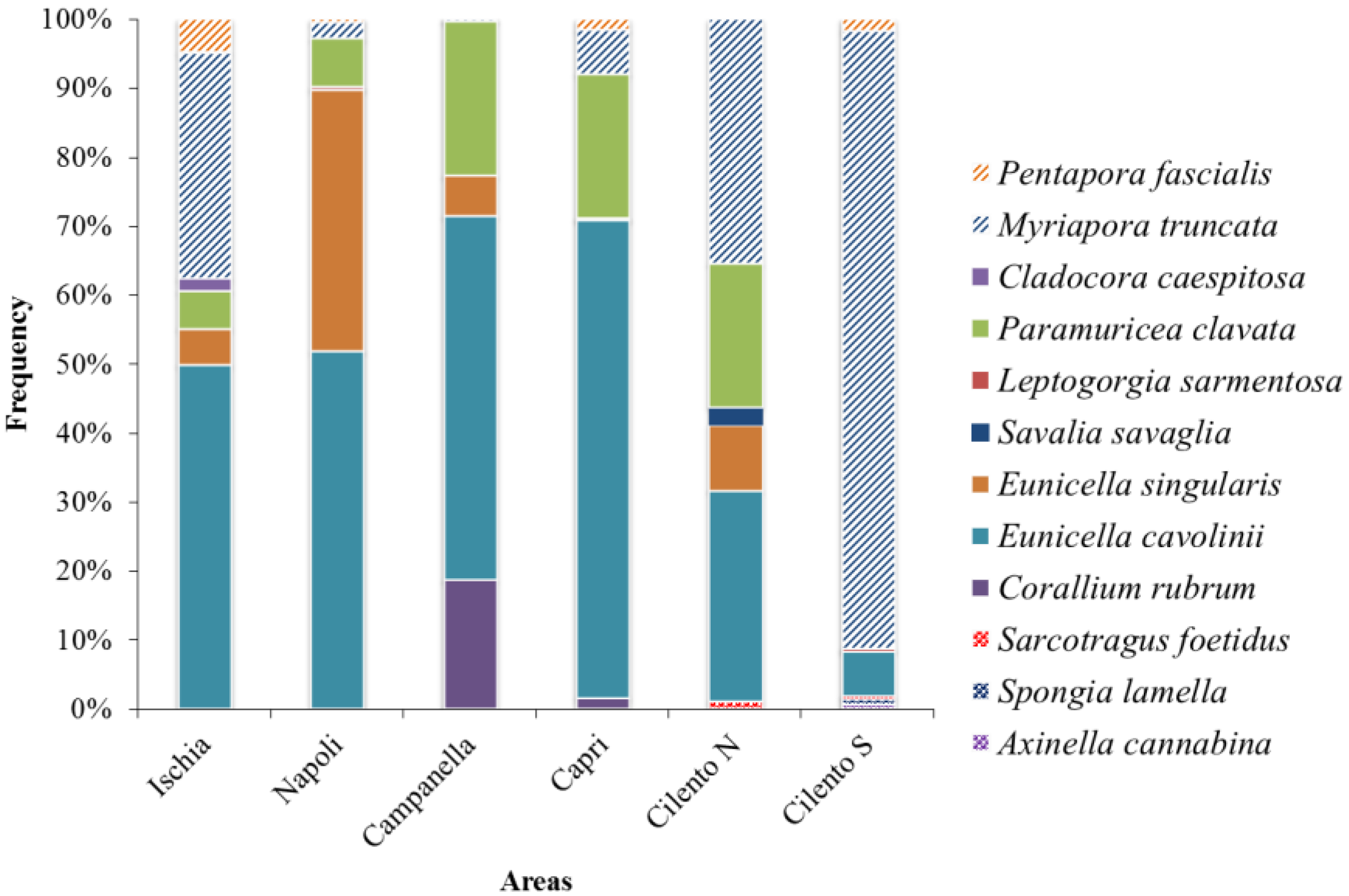
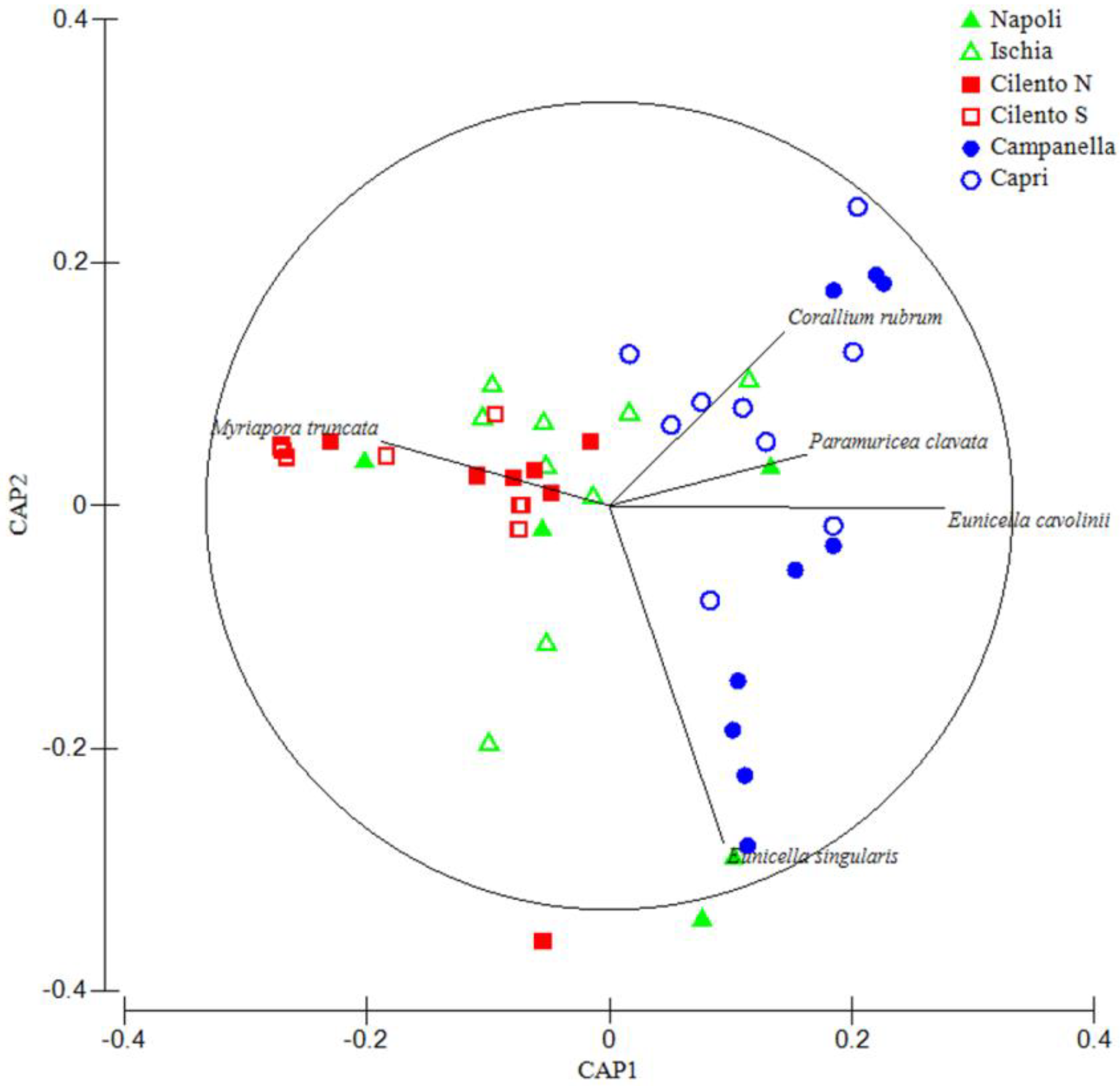
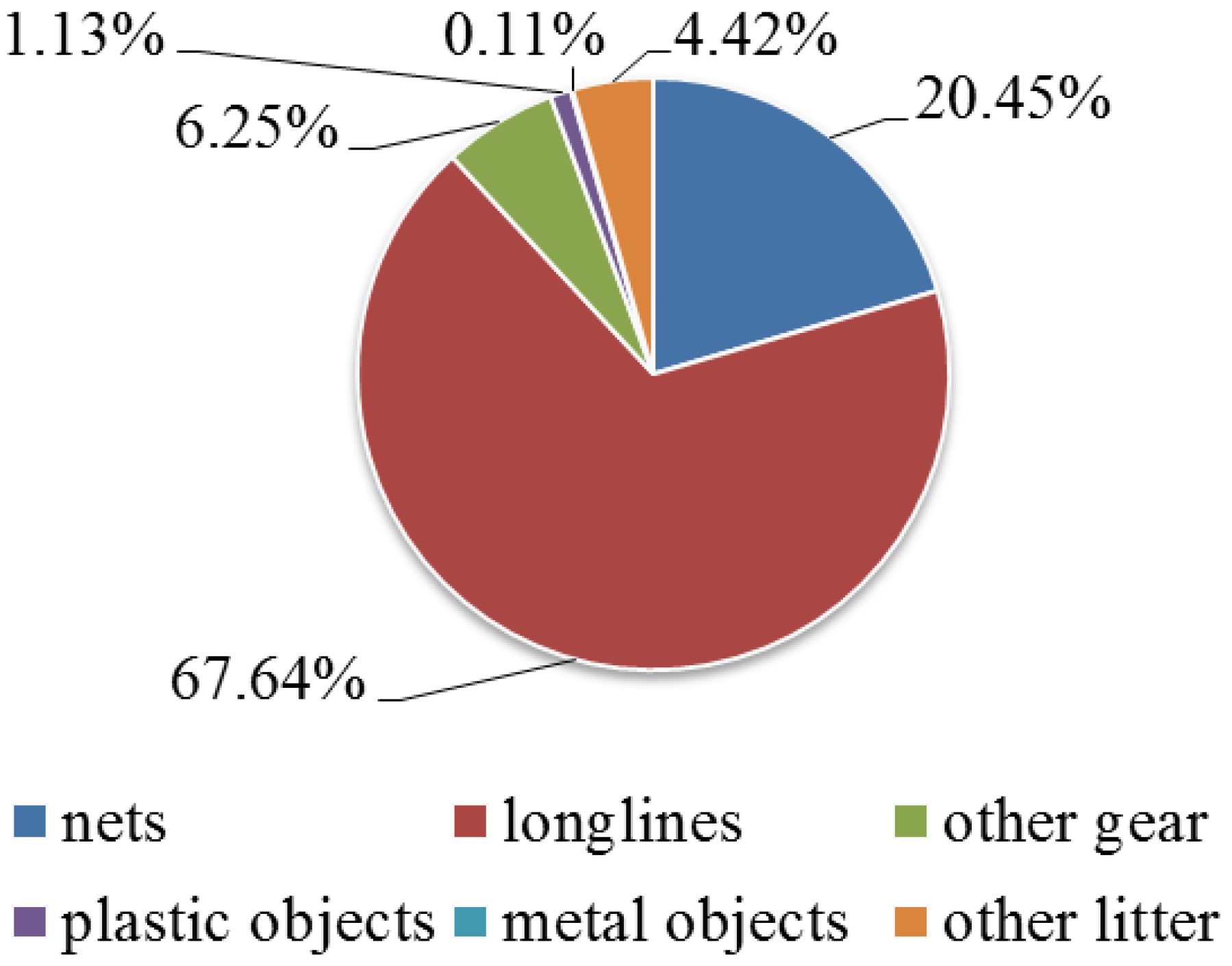
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ballesteros, E. Mediterranean coralligenous assemblages: A synthesis of present knowledge. Oceanogr. Mar. Biol. Annu. Rev. 2006, 44, 123–195. [Google Scholar]
- Sheehan, P.M.; Fagerstrom, J.A. The evolution of reef communities. Palaios 1988, 3, 251. [Google Scholar] [CrossRef]
- Baker, E.K.; Puglise, K.A.; Harris, P.T. (Eds.) Mesophotic Coral Ecosystems—A Lifeboat for Coral Reefs? The United Nations Environment Programme: Nairobi, Kenya; GRID-Arendal: Arendal, Norway, 2016; p. 98. [Google Scholar]
- Ferrigno, F.; Russo, G.F.; Semprucci, F.; Sandulli, R. Unveiling the state of some underexplored deep coralligenous banks in the Gulf of Naples (Mediterranean Sea, Italy). Reg. Stud. Mar. Sci. 2018, 22, 82–92. [Google Scholar] [CrossRef]
- Corriero, G.; Pierri, C.; Mercurio, M.; Marzano, C.N.; Tarantini, S.O.; Gravina, M.F.; Lisco, S.; De Giosa, F.; Valenzano, E.; Giangrande, A.; et al. Mediterranean mesophotic coral reef built by non-symbiotic scleractinians. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Basso, D. Deep rhodolith distribution in the Pontian Islands, Italy: A model for the paleoecology of a temperate sea. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1998, 137, 173–187. [Google Scholar] [CrossRef]
- Rendina, F.; Kaleb, S.; Caragnano, A.; Ferrigno, F.; Appolloni, L.; Donnarumma, L.; Russo, G.F.; Sandulli, R.; Roviello, V.; Falace, A. Distribution and characterization of deep rhodolith beds off the Campania coast (SW Italy, Mediterranean Sea). Plants 2020, 9, 985. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.G.; Lawton, J.H.; Shachak, M. Organisms as ecosystems engineers. Nord. Soc. Oikos 1994, 69, 373–386. [Google Scholar] [CrossRef]
- Sartoretto, S. Vitesse de Croissance et Bioérosion des Concrétionnements “Coralligènes” de Méditerranée Nord-Occidentale. Rapport avec les Variations Holocènes du Niveau Marin. Ph.D. Thesis, Université d’Aix-Marseille, Marseille, France, 1996; p. 194. [Google Scholar]
- Boudouresque, C.F. Marine biodiversity in the Mediterranean: Status of species, populations and communities. Sci. Rep. Port-Cros Natl. Park 2004, 20, 97–146. [Google Scholar]
- Coll, M.; Piroddi, C.; Steenbeek, J.; Kaschner, K.; Ben Rais Lasram, F.; Aguzzi, J.; Ballesteros, E.; Bianchi, C.N.; Corbera, J.; Dailianis, T.; et al. The biodiversity of the Mediterranean Sea: Estimates, patterns, and threats. PLoS ONE 2010, 5, e11842. [Google Scholar] [CrossRef] [Green Version]
- Ferrigno, F.; Appolloni, L.; Rendina, F.; Donnarumma, L.; Russo, G.F.; Sandulli, R. Red coral (Corallium rubrum) populations and coralligenous characterization within “Regno di Nettuno MPA” (Tyrrhenian Sea, Italy). Eur. Zool. J. 2020, 87, 203–213. [Google Scholar] [CrossRef] [Green Version]
- European Commission. Commission Decision (EU) 2017/848 of 17 May 2017 laying down criteria and methodological standards on good environmental status of marine waters and specifications and standardized methods for monitoring and assessment, and repealing Decision 2010/477/EU. Off. J. Eur. Union L 2017, 125, 43. [Google Scholar]
- Tonin, S. Economic value of marine biodiversity improvement in coralligenous habitats. Ecol. Indic. 2018, 85, 1121–1132. [Google Scholar] [CrossRef]
- Krieger, K.J.; Wing, B.L. Megafauna associations with deep water corals (Primnoa spp.) in the Gulf of Alaska. Hydrobiologia 2002, 471, 83–90. [Google Scholar] [CrossRef]
- Henry, L.A.; Roberts, J.M. Biodiversity and ecological composition of macrobenthos on cold-water coral mounds and adjacent off-mound habitat in the bathyal Porcupine Seabight, NE Atlantic. Deep-Sea Res. Part I Oceanogr. Res. Pap. 2007, 54, 654–672. [Google Scholar] [CrossRef]
- Lloret, J. Human health benefits supplied by Mediterranean marine biodiversity. Mar. Pollut. Bull. 2010, 60, 1640–1646. [Google Scholar] [CrossRef]
- Salomidi, M.; Katsanevakis, S.; Borja, A.; Braeckman, U.; Damalas, D.; Galpasoro, I.; Mifsud, R.; Mirto, S.; Pascual, M.; Pipitone, C.; et al. Assessment of goods and services, vulnerability, and conservation status of European seabed biotopes: A stepping stone towards ecosystem-based marine spatial management. Mediterr. Mar. Sci. 2012, 13, 49–88. [Google Scholar] [CrossRef] [Green Version]
- Buonocore, E.; Donnarumma, L.; Appolloni, L.; Miccio, A.; Russo, G.F.; Franzese, P.P. Marine natural capital and ecosystem services: An environmental accounting model. Ecol. Model. 2020, 424, 109029. [Google Scholar] [CrossRef]
- Walther, G.R.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.C.; Fromentin, J.-M.; Hoegh-Guldberg, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Rendina, F.; Bouchet, P.J.; Appolloni, L.; Russo, G.F.; Sandulli, R.; Kolzenburg, R.; Putra, A.; Ragazzola, F. Physiological response of the coralline alga Corallina officinalis L. to both predicted long-term increases in temperature and short-term heatwave events. Mar. Environ. Res. 2019, 150, 104764. [Google Scholar] [CrossRef] [PubMed]
- Halpern, B.S.; Walbridge, S.; Selkoe, K.A.; Kappel, C.V.; Micheli, F.; D’Agrosa, C.; Bruno, J.F.; Casey, K.S.; Ebert, C.; Fox, H.E.; et al. A global map of human impact on marine ecosystems. Science 2008, 319, 948–952. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rizzo, L.; Musco, L.; Crocetta, F. Cohabiting with litter: Fish and benthic assemblages in coastal habitats of a heavily urbanized area. Mar. Pollut. Bull. 2021, 164, 112077. [Google Scholar] [CrossRef]
- Clark, M.R.; Koslow, J.A. Impacts of fisheries on seamounts. In Seamounts: Ecology, Fisheries and Conservation. Blackwell Publishing; Pitcher, T.J., Morato, T., Hart, P.J.B., Clark, M.R., Haggan, N., Santos, R.S., Eds.; Blackwell Publishing: Oxford, UK, 2007; pp. 413–441. [Google Scholar]
- Macfadyen, G.; Huntington, T.; Cappell, R. Abandoned, lost or Otherwise Discarded Fishing Gear UNEP Regional Seas Reports and Studies. FAO Fish. Aquac. Tech. Pap. 2009, 523, 115. [Google Scholar]
- Althaus, F.; Williams, A.; Schlacher, T.A.; Kloser, R.J.; Green, M.A.; Barker, B.A.; Bax, N.J.; Brodie, P.; Schlacher-Hoenlinger, M.A. Impacts of bottom trawling on deep-coral ecosystems of seamounts are long-lasting. Mar. Ecol. Prog. Ser. 2009, 397, 279–294. [Google Scholar] [CrossRef]
- Buhl-Mortensen, P.; Buhl-Mortensen, L. Impacts of bottom trawling and litter on the seabed in Norwegian waters. Front. Mar. Sci. 2018, 5, 42. [Google Scholar] [CrossRef] [Green Version]
- Betti, F.; Bavestrello, G.; Bo, M.; Ravanetti, G.; Enrichetti, F.; Coppari, M.; Cappanera, V.; Venturini, S.; Cattaneo-Vietti, R. Evidences of fishing impact on the coastal gorgonian forests inside the Portofino MPA (NW Mediterranean Sea). Ocean. Coast. Manag. 2020, 187, 105105. [Google Scholar] [CrossRef]
- Ballesteros, L.V.; Matthews, J.L.; Hoeksema, B.W. Pollution and coral damage caused by derelict fishing gear on coral reefs around Koh Tao, Gulf of Thailand. Mar. Pollut. Bull. 2018, 135, 1107–1116. [Google Scholar] [CrossRef]
- Figueroa-Pico, J.; Tortosa, F.S.; Carpio, A.J. Coral fracture by derelict fishing gear affects the sustainability of the marginal reefs of Ecuador. Coral Reefs 2020, 39, 819–827. [Google Scholar] [CrossRef]
- Angiolillo, M.; di Lorenzo, B.; Farcomeni, A.; Bo, M. Distribution and assessment of marine debris in the deep Tyrrhenian Sea (NW Mediterranean Sea, Italy). Mar. Pollut. Bull. 2015, 92, 149–159. [Google Scholar] [CrossRef]
- Ferrigno, F.; Appolloni, L.; Russo, G.F.; Sandulli, R. Impact of fishing activities on different coralligenous assemblages of Gulf of Naples (Italy). J. Mar. Biol. Assoc. UK 2018, 98, 41–50. [Google Scholar] [CrossRef]
- Giusti, M.; Canese, S.; Fourt, M.; Bo, M.; Innocenti, C.; Goujard, A.; Daniel, B.; Angeletti, L.; Taviani, M.; Aquilina, L.; et al. Coral forests and Derelict Fishing Gears in submarine canyon systems of the Ligurian Sea. Prog. Oceanogr. 2019, 178, 102186. [Google Scholar] [CrossRef]
- Cerrano, C.; Arillo, A.; Azzini, F.; Calcinai, B.; Castellano, L.; Muti, C.; Valisano, L.; Zega, G.; Bavestrello, G. Gorgonian population recovery after a mass mortality event. Aquat. Conserv. Mar. Freshw. Ecosyst. 2005, 15, 147–157. [Google Scholar] [CrossRef]
- Piazzi, L.; Gennaro, P.; Balata, D. Threats to macroalgal coralligenous assemblages in the Mediterranean Sea. Mar. Pollut. Bull. 2012, 64, 2623–2629. [Google Scholar] [CrossRef] [PubMed]
- Gregory, M.R. Environmental implications of plastic debris in marine settings-entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2013–2025. [Google Scholar] [CrossRef] [PubMed]
- Rochman, C.M. The Complex Mixture, Fate and Toxicity of Chemicals Associated With Plastic Debris in the Marine Environment. In Marine Anthropogenic Litter; Springer: Berlin/Heidelberg, Germany, 2015; pp. 117–140. [Google Scholar]
- Caruso, G. Microplastics as vectors of contaminants. Mar. Pollut. Bull. 2019, 146, 921–924. [Google Scholar] [CrossRef] [PubMed]
- Romeo, T.; Battaglia, P.; Pedà, C.; Consoli, P.; Andaloro, F.; Fossi, M.C. First evidence of presence of plastic debris in stomach of large pelagic fish in the Mediterranean Sea. Mar. Pollut. Bull. 2015, 95, 358–361. [Google Scholar] [CrossRef]
- Rummel, D.C.; Löder, G.J.M.; Fricke, F.N.; Lang, T.; Griebeler, E.M.; Janke, M.; Gerdts, G. Plastic ingestion by pelagic and demersal fish from the North Sea and Baltic Sea. Mar. Pollut. Bull. 2016, 102, 134–141. [Google Scholar] [CrossRef]
- Markic, A.; Niemand, C.; Bridson, J.H.; Mazouni-Gaertner, N.; Gaertner, J.C.; Eriksen, M.; Bowen, M. Double trouble in the South Pacific subtropical gyre: Increased plastic ingestion by fish in the oceanic accumulation zone. Mar. Pollut. Bull. 2018, 136, 547–564. [Google Scholar] [CrossRef]
- Gall, S.C.; Thompson, R.C. The impact of debris on marine life. Mar. Pollut. Bull. 2015, 92, 170–179. [Google Scholar] [CrossRef]
- Consoli, P.; Falautano, M.; Sinopoli, M.; Perzia, P.; Canese, S.; Esposito, V.; Battaglia, P.; Romeo, T.; Andaloro, F.; Galgani, F.; et al. Composition and abundance of benthic marine litter in a coastal area of the central Mediterranean Sea. Mar. Pollut. Bull. 2018, 136, 243–247. [Google Scholar] [CrossRef]
- Consoli, P.; Scotti, G.; Romeo, T.; Fossi, M.C.; Esposito, V.; D’Alessandro, M.; Battaglia, P.; Galgani, F.; Figurella, F.; Pragnell-Raasch, H.; et al. Characterization of seafloor litter on Mediterranean shallow coastal waters: Evidence from Dive Against Debris®, a citizen science monitoring approach. Mar. Pollut. Bull. 2020, 150, 110763. [Google Scholar] [CrossRef]
- Rendina, F.; Ferrigno, F.; Appolloni, L.; Donnarumma, L.; Sandulli, R.; Fulvio, G.F. Anthropic pressure due to lost fishing gears and marine litter on different rhodolith beds off the Campania Coast (Tyrrhenian Sea, Italy). Ecol. Quest. 2020, 31, 1–17. [Google Scholar] [CrossRef]
- Scotti, G.; Esposito, V.; D’Alessandro, M.; Panti, C.; Vivona, P.; Consoli, P.; Figurella, F.; Romeo, T. Seafloor litter along the Italian coastal zone: An integrated approach to identify sources of marine litter. Waste Manag. 2021, 124, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Appolloni, L.; Bevilacqua, S.; Sbrescia, L.; Sandulli, R.; Terlizzi, A.; Russo, G.F. Does full protection count for the maintenance of beta/diversity patterns in marine communities? Evidence from Mediterranean fish assemblages. Aquatic conservation. Mar. Freshw. Ecosyst. 2017, 27, 828–838. [Google Scholar] [CrossRef] [Green Version]
- Appolloni, L.; Zeppilli, D.; Donnarumma, L.; Baldrighi, E.; Chianese, E.; Russo, G.F.; Sandulli, R. Seawater Acidification Affects Beta-Diversity of Benthic Communities at a Shallow Hydrothermal Vent in a Mediterranean Marine Protected Area (Underwater Archaeological Park of Baia, Naples, Italy). Diversity 2020, 12, 464. [Google Scholar] [CrossRef]
- Fabri, M.C.; Vinha, B.; Allais, A.G.; Bouhier, M.E.; Dugornay, O.; Gaillot, A.; Arnaubec, A. Evaluating the ecological status of cold-water coral habitats using noninvasive methods: An example from Cassidaigne canyon, northwestern Mediterranean Sea. Prog. Oceanogr. 2019, 178, 102172. [Google Scholar] [CrossRef] [Green Version]
- Cataudella, S.; Spagnolo, M. Lo Stato Della Pesca e Dell’acquacoltura nei Mari Italiani; Ministero delle Politiche Agricole Alimentari e Forestali: Rome, Italy, 2011; 877p.
- Appolloni, L.; Ferrigno, F.; Russo, G.F.; Sandulli, R. β-Diversity of morphological groups as indicator of coralligenous community quality status. Ecol. Indic. 2020, 109, 105840. [Google Scholar] [CrossRef]
- Ferrigno, F.; Russo, G.F.; Sandulli, R. Coralligenous Bioconstructions Quality Index (CBQI): A synthetic indicator to assess the status of different types of coralligenous habitats. Ecol. Indic. 2017, 82, 271–279. [Google Scholar] [CrossRef]
- Russo, G.F. Il banco di Santa Croce. Ambient. e Mass Media mare e le coste 96, 1992.
- Russo, G.F. I fondali marini del Golfo di Napoli e del litorale ischitano: Particolarità e paradossi. Scheria 1995, 10, 58–72. [Google Scholar]
- Appolloni, L.; Russo, G.F. Underwater landscape in the Bay of Naples for maritime spatial planning. In La Baia Di Napoli—Strategie Integrate per La Conservazione e La Fruizione Del Paesaggio Culturale; Aveta, A., Marino, B.G., Amore, R., Eds.; Artstudio: Napoli, Italy, 2017; pp. 71–74. [Google Scholar]
- Available online: https://www.minambiente.it/sites/default/files/archivio/allegati/strategia_marina/ARPA/SM_ARPA_metodMOD_7_REV.pdf (accessed on 10 January 2018).
- Gori, A.; Rossi, S.; Berganzo, E.; Pretus, J.L.; Dale, M.R.; Gili, J.M. Spatial distribution patterns of the gorgonians Eunicella singularis, Paramuricea clavata, and Leptogorgia sarmentosa (Cape of Creus, Northwestern Mediterranean Sea). Mar. Biol. 2011, 158, 143–158. [Google Scholar] [CrossRef]
- Terlizzi, A.; Anderson, M.J.; Fraschetti, S.; Benedetti-Cecchi, L. Scales of spatial variation in Mediterranean subtidal sessile assemblages at different depths. Mar. Ecol. Prog. Ser. 2007, 332, 25–39. [Google Scholar] [CrossRef] [Green Version]
- Anderson, M.J. Permutation tests for univariate or multivariate analysis of variance and regression. Can. J. Fish. Aquat. Sci. 2001, 58, 626–639. [Google Scholar] [CrossRef]
- Anderson, M.J.; Ter Braak, C.J.F. Permutation tests for multi-factorial analysis of variance. J. Stat. Comput. Simul. 2003. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. Primer: User Manual/Tutorial; Prim. Ltd.: Plymouth, UK, 2015; p. 93. [Google Scholar]
- Anderson, M.J.; Willis, T.J. Canonical Analysis of Principal Coordinates: A Useful Method of Constrained Ordination for Ecology. Ecology 2003, 84, 511–525. [Google Scholar] [CrossRef]
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.J. DISTLM v. 5: A FORTRAN Computer Program to Calculate a Distance-Based Multivariate Analysis for a Linear Model; Department of Statistics, University: Auckland, UK, 2004; Volume 10, p. 2016. [Google Scholar]
- Hammer, Ø.; Harper, D.A.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Guisan, A.; Thuiller, W. Predicting species distribution: Offering more than simple habitat models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef]
- Di Camillo, C.G.; Ponti, M.; Bavestrello, G.; Krzelj, M.; Cerrano, C. Building a baseline for habitat-forming corals by a multi-source approach, including Web Ecological Knowledge. Biodivers. Conserv. 2018, 27, 1257–1276. [Google Scholar] [CrossRef]
- Donnarumma, L.; Sandulli, R.; Appolloni, L.; Ferrigno, F.; Rendina, F.; Di Stefano, F.; Russo, G.F. Bathymetrical and temporal variations in soft-bottom molluscan assemblages in the coastal area facing the Sarno River mouth (Mediterranean Sea, Gulf of Naples). Ecol. Quest. 2020, 31, 1–20. [Google Scholar] [CrossRef]
- Sini, M.; Garrabou, J.; Trygonis, V.; Koutsoubas, D. Coralligenous formations dominated by Eunicella cavolini (Koch, 1887) in the NE Mediterranean: Biodiversity and structure. Mediterr. Mar. Sci. 2019, 20, 174–188. [Google Scholar] [CrossRef] [Green Version]
- Irving, A.D.; Connell, S.D. Sedimentation and light penetration interact to maintain heterogeneity of subtidal habitats: Algal versus invertebrate dominated assemblages. Mar. Ecol. Prog. Ser. 2002, 245, 83–91. [Google Scholar] [CrossRef] [Green Version]
- Balata, D.; Piazzi, L.; Cecchi, E.; Cinelli, F. Variability of Mediterranean coralligenous assemblages subject to local variation in sediment deposition. Mar. Environ. Res. 2005, 60, 403–421. [Google Scholar] [CrossRef]
- De Ruggiero, P.; Napolitano, E.; Iacono, R.; Pierini, S. A high-resolution modelling study of the circulation along the Campania coastal system, with a special focus on the Gulf of Naples. Cont. Shelf Res. 2016, 122, 85–101. [Google Scholar] [CrossRef]
- De Ruggiero, P.; Esposito, G.; Napolitano, E.; Iacono, R.; Pierini, S.; Zambianchi, E. Modelling the marine circulation of the Campania coastal system (Tyrrhenian Sea) for the year 2016: Analysis of the dynamics. J. Mar. Syst. 2020, 210, 103388. [Google Scholar] [CrossRef]
- Yang, H.Y.; Chen, B.; Piersma, T.; Zhang, Z.; Ding, C. Molluscs of an intertidal soft-sediment area in China: Does overfishing explain a high density but low diversity community that benefits staging shorebirds? J. Sea Res. 2016, 109, 20–28. [Google Scholar]
- Sedano, F.; Florido, M.; Rallis, I.; Espinosa, F.; Gerovasileiou, V. Comparing sessile benthos on shallow artificial versus natural hard substrates in the Eastern Mediterranean Sea. Mediterr. Mar. Sci. 2019, 20, 688–702. [Google Scholar] [CrossRef] [Green Version]
- Soininen, J. Species turnover along abiotic and biotic gradients: Patterns in space equal patterns in time? Bioscience 2010, 60, 433–439. [Google Scholar] [CrossRef] [Green Version]
- Ferrigno, F.; Bianchi, C.N.; Lasagna, R.; Morri, C.; Russo, G.F.; Sandulli, R. Corals in high diversity reefs resist human impact. Ecol. Indic. 2016, 70, 106–113. [Google Scholar] [CrossRef]
- Marengo, M.; Culioli, J.M.; Santoni, M.C.; Marchand, B.; Durieux, E.D.H. Comparative analysis of artisanal and recreational fisheries for Dentex dentex in a Marine Protected Area. Fish. Manag. Ecol. 2015, 22, 249–260. [Google Scholar] [CrossRef] [Green Version]
- Good, T.P.; June, J.A.; Etnier, M.A.; Broadhurst, G. Derelict fishing nets in Puget Sound and the Northwest Straits: Patterns and threats to marine fauna. Mar. Pollut. Bull. 2010, 60, 39–50. [Google Scholar] [CrossRef]
- Bo, M.; Bava, S.; Canese, S.; Angiolillo, M.; Cattaneo-Vietti, R.; Bavestrello, G. Fishing impact on deep Mediterranean rocky habitats as revealed by ROV investigation. Biol. Conserv. 2014, 171, 167–176. [Google Scholar] [CrossRef]
- Portman, M.E.; Brennan, R.E. Marine litter from beach-based sources: Case study of an Eastern Mediterranean coastal town. Waste Manag. 2017, 69, 535–544. [Google Scholar] [CrossRef] [Green Version]
- Kipson, S.; Linares, C.; Čižmek, H.; Cebrián, E.; Ballesteros, E.; Bakran-Petricioli, T.; Garrabou, J. Population structure and conservation status of the red gorgonian Paramuricea clavata (Risso, 1826) in the Eastern Adriatic Sea. Mar. Ecol. 2015, 36, 982–993. [Google Scholar] [CrossRef] [Green Version]
- Karvonen, A.; Rintamäki, P.; Jokela, J.; Valtonen, E.T. Increasing water temperature and disease risks in aquatic systems: Climate change increases the risk of some, but not all, diseases. Int. J. Parasitol. 2010, 40, 1483–1488. [Google Scholar] [CrossRef] [PubMed]
- Garrabou, J.; Coma, R.; Bensoussan, N.; Bally, M.; Chevaldonné, P.; Cigliano, M.; Diaz, D.; Harmelin, J.G.; Gambi, M.C.; Kersting, D.K.; et al. Mass mortality in Northwestern Mediterranean rocky benthic communities: Effects of the 2003 heat wave. Glob. Chang. Biol. 2009, 15, 1090–1103. [Google Scholar] [CrossRef]
- Turicchia, E.; Abbiati, M.; Sweet, M.; Ponti, M. Mass mortality hits gorgonian forests at Montecristo Island. Dis. Aquat. Org. 2018, 131, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Ponti, M.; Perlini, R.A.; Ventra, V.; Grech, D.; Abbiati, M.; Cerrano, C. Ecological shifts in Mediterranean coralligenous assemblages related to gorgonian forest loss. PLoS ONE 2014, 9, e102782. [Google Scholar] [CrossRef]
- Verdura, J.; Linares, C.; Ballesteros, E.; Coma, R.; Uriz, M.J.; Bensoussan, N.; Cebrian, E. Biodiversity loss in a Mediterranean ecosystem due to an extreme warming event unveils the role of an engineering gorgonian species. Sci. Rep. 2019, 9, 1–11. [Google Scholar]
- Martín, J.; Puig, P.; Palanques, A.; Masqué, P.; García-Orellana, J. Effect of commercial trawling on the deep sedimentation in a Mediterranean submarine canyon. Mar. Geol. 2008, 252, 150–155. [Google Scholar] [CrossRef] [Green Version]
- Tseng, L.C.; Dahms, H.U.; Hsu, N.J.; Hwang, J.S. Effects of sedimentation on the gorgonian Subergorgia suberosa (Pallas, 1766). Mar. Biol. 2011, 158, 1301–1310. [Google Scholar] [CrossRef]
- Clark, M.R.; Althaus, F.; Schlacher, T.A.; Williams, A.; Bowden, D.A.; Rowden, A.A. The impacts of deep-sea fisheries on benthic communities: A review. ICES J. Mar. Sci. 2016, 73 (Suppl. 1), i51–i69. [Google Scholar] [CrossRef]
- Lambert, G.I.; Murray, L.G.; Hiddink, J.G.; Hinz, H.; Lincoln, H.; Hold, N.; Cambiè, G.; Kaiser, M.J. Defining thresholds of sustainable impact on benthic communities in relation to fishing disturbance. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Casoli, E.; Piazzi, L.; Nicoletti, L.; Jona-Lasinio, G.; Cecchi, E.; Mancini, G.; Belluscio, A.; Ardizzone, G. Ecology, distribution and demography of erect bryozoans in Mediterranean coralligenous reefs. Estuar. Coast. Shelf Sci. 2020, 235, 106573. [Google Scholar] [CrossRef]
- Agardy, T. Effects of fisheries on marine ecosystems: A conservationist’s perspective. ICES J. Mar. Sci. 2000, 57, 761–765. [Google Scholar] [CrossRef] [Green Version]
- Pratchett, M.S.; Hoey, A.S.; Wilson, S.K.; Messmer, V.; Graham, N.A. Changes in biodiversity and functioning of reef fish assemblages following coral bleaching and coral loss. Diversity 2011, 3, 424–452. [Google Scholar] [CrossRef] [Green Version]
- Lenzen, M.; Moran, D.; Kanemoto, K.; Foran, B.; Lobefaro, L.; Geschke, A. International trade drives biodiversity threats in developing nations. Nature 2012, 486, 109–112. [Google Scholar] [CrossRef]
- Picone, F.; Buonocore, E.; Chemello, R.; Russo, G.F.; Franzese, P.P. Exploring the development of scientific research on Marine Protected Areas: From conservation to global ocean sustainability. Ecol. Inform. 2021, 61, 101200. [Google Scholar] [CrossRef]



| CSS | Ischia | Napoli | Campanella | Capri | Cilento N | Cilento S |
|---|---|---|---|---|---|---|
| Porifera | ||||||
| Axinella cannabina | 0.25 ± 0.25 | |||||
| Spongia lamella | 0.10 ± 0.29 | |||||
| Sarcotragus foetidus | 0.31 ± 0.46 | 0.04 ± 0.11 | 0.05 ± 0.14 | |||
| Cnidaria | ||||||
| Corallium rubrum | 44.94 ± 103.86 | 2.56 ± 7.68 | ||||
| Eunicella cavolinii | 5.80 ± 5.67 | 12.98 ± 26.18 | 127.39 ± 100.06 | 104.43 ± 71.09 | 0.98 ± 2.22 | 0.77 ± 2.31 |
| Eunicella singularis | 0.61 ± 1.30 | 9.45 ± 21.06 | 14.14 ± 26.95 | 0.37 ± 0.82 | 0.30 ± 0.91 | |
| Savalia savaglia | 0.08 ± 0.25 | |||||
| Leptogorgia sarmentosa | 0.11 ± 0.33 | 0.04 ± 0.12 | ||||
| Paramuricea clavata | 0.63 ± 1.60 | 1.75 ± 5.09 | 54.12 ± 56.72 | 31.64 ± 43.45 | 0.67 ± 2.00 | |
| Cladocora caespitosa | 0.21 ± 0.63 | |||||
| Bryozoa | ||||||
| Myriapora truncata | 3.81 ± 4.78 | 0.58 ± 1.08 | 0.56 ± 0.72 | 9.59 ± 22.17 | 1.14 ± 1.54 | 10.58 ± 10.84 |
| Pentapora fascialis | 0.57 ± 1.17 | 0.12 ± 0.25 | 0.10 ± 0.29 | 2.32 ± 5.74 | 0.20 ± 0.40 |
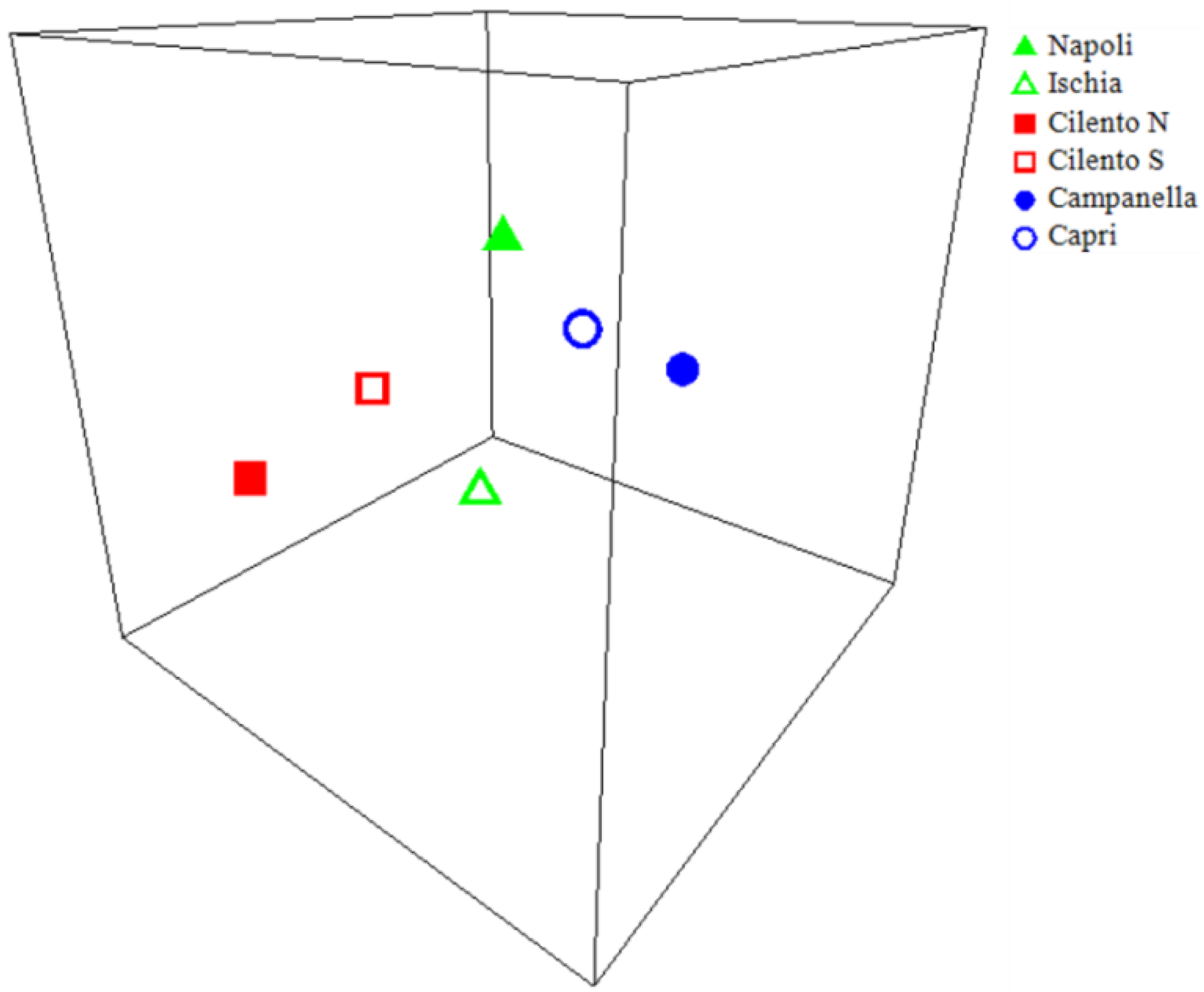
| Ischia | Napoli | Campanella | Capri | Cilento N | Cilento S | |
|---|---|---|---|---|---|---|
| Ischia | ||||||
| Napoli | 0.68 | |||||
| Campanella | 0.04 | 0.02 | ||||
| Capri | 0.06 | 0.03 | 0.43 | |||
| Cilento N | 0.21 | 0.24 | 0.01 | 0.01 | ||
| Cilento S | 0.50 | 0.70 | 0.02 | 0.02 | 0.50 |
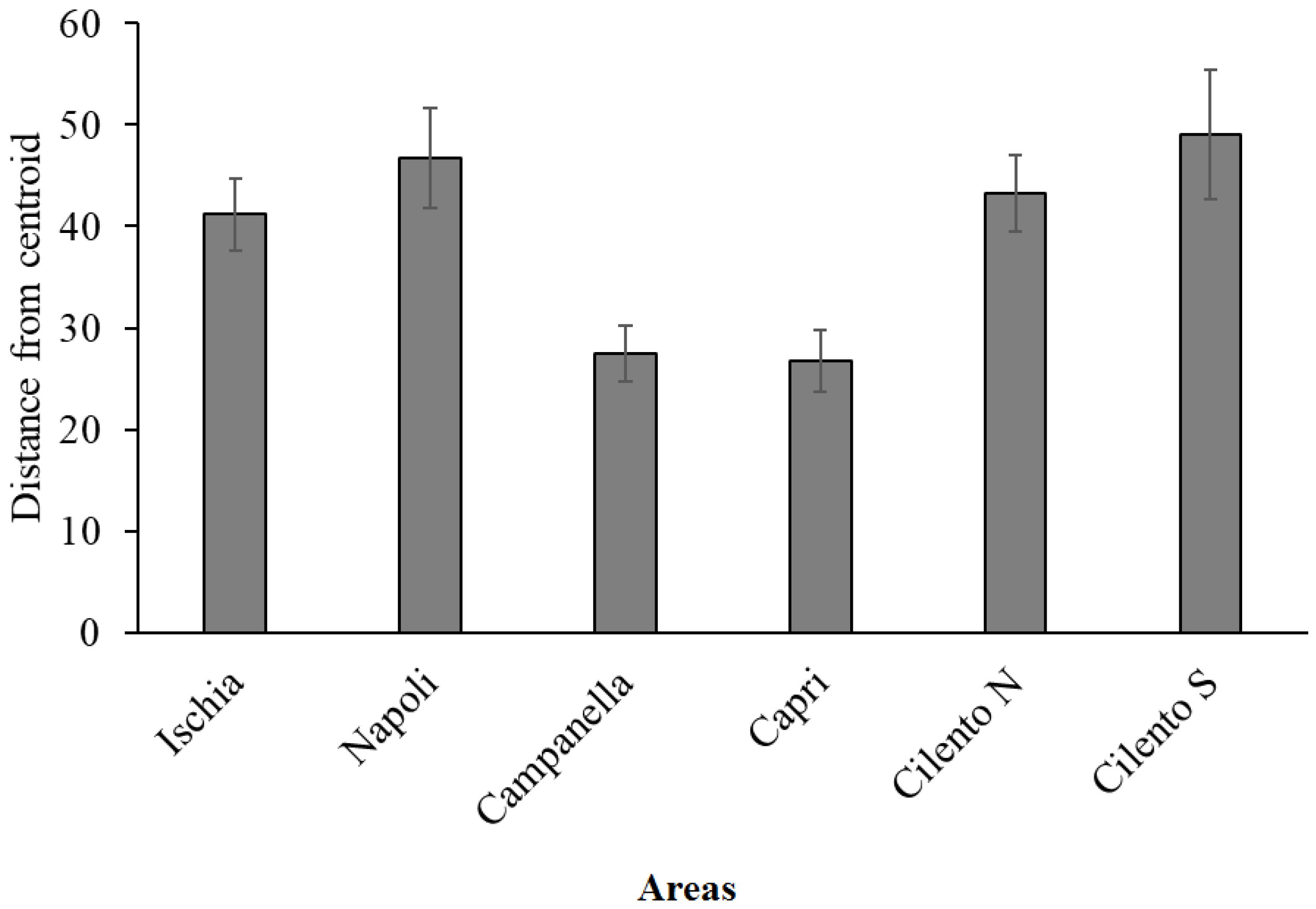
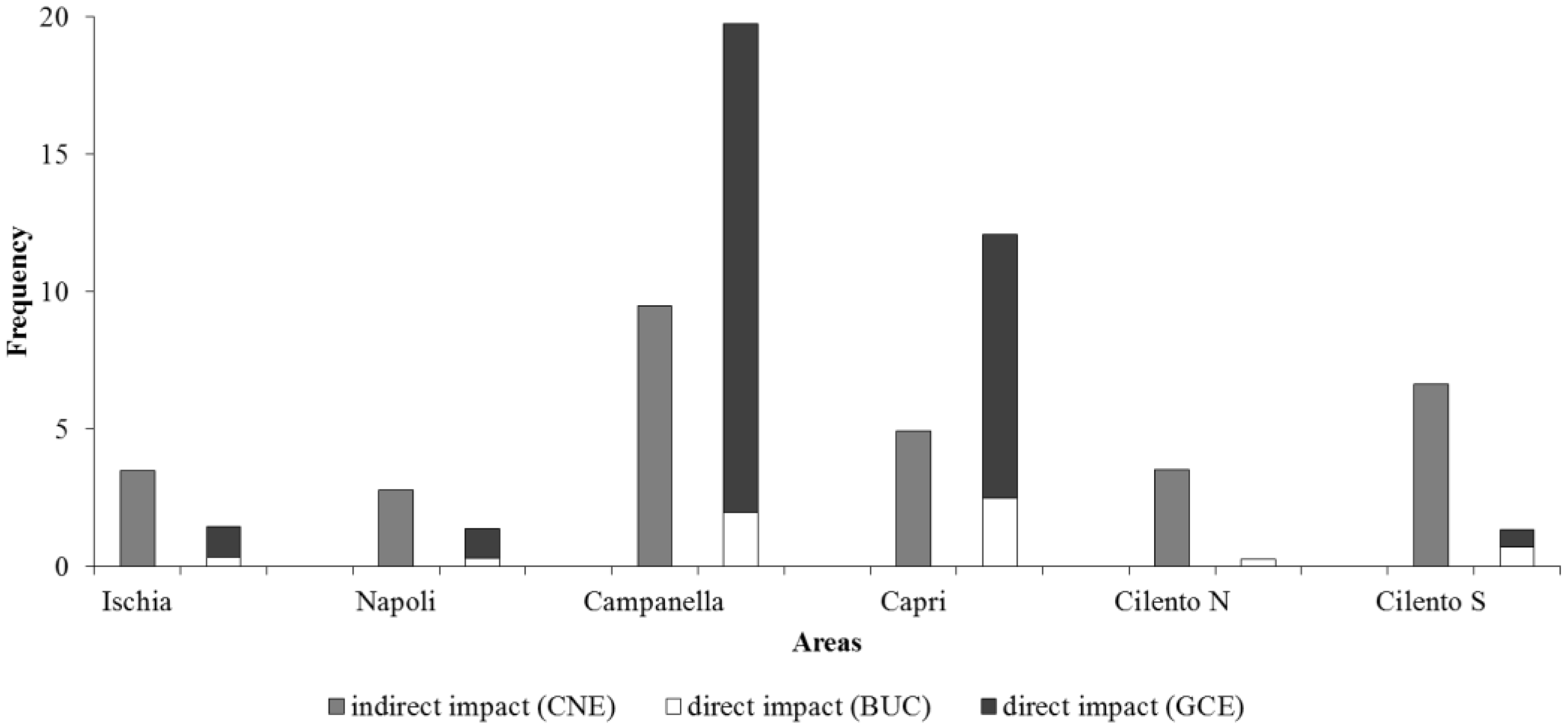
| Longlines | Indirect Impact (CNE) | Direct Impact (BUC + GCE) | Heterogeneity | |
|---|---|---|---|---|
| Napoli | 12.76 ± 13.13 | 2.77 ± 4.68 | 1.36 ± 1.73 | 46.78 ± 4.91 |
| Ischia | 11.67 ± 8.46 | 2.51 ± 4.77 | 1.44 ± 2.92 | 41.2 ± 3.57 |
| Cilento N | 3.35 ± 4.25 | 2.51 ± 4.77 | 0.26 ± 0.78 | 43.23 ± 3.78 |
| Cilento S | 2.93 ± 2.49 | 2.73 ± 4.69 | 1.33 ± 2.13 | 49.07 ± 6.41 |
| Campanella | 23.64 ± 18.51 | 3.39 ± 4.68 | 19.74 ± 18.19 | 27.52 ± 2.74 |
| Capri | 20.61 ± 14.11 | 4.27 ± 4.7 | 12.07 ± 9.07 | 26.74 ± 3.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrigno, F.; Appolloni, L.; Donnarumma, L.; Di Stefano, F.; Rendina, F.; Sandulli, R.; Russo, G.F. Diversity Loss in Coralligenous Structuring Species Impacted by Fishing Gear and Marine Litter. Diversity 2021, 13, 331. https://doi.org/10.3390/d13070331
Ferrigno F, Appolloni L, Donnarumma L, Di Stefano F, Rendina F, Sandulli R, Russo GF. Diversity Loss in Coralligenous Structuring Species Impacted by Fishing Gear and Marine Litter. Diversity. 2021; 13(7):331. https://doi.org/10.3390/d13070331
Chicago/Turabian StyleFerrigno, Federica, Luca Appolloni, Luigia Donnarumma, Floriana Di Stefano, Francesco Rendina, Roberto Sandulli, and Giovanni Fulvio Russo. 2021. "Diversity Loss in Coralligenous Structuring Species Impacted by Fishing Gear and Marine Litter" Diversity 13, no. 7: 331. https://doi.org/10.3390/d13070331
APA StyleFerrigno, F., Appolloni, L., Donnarumma, L., Di Stefano, F., Rendina, F., Sandulli, R., & Russo, G. F. (2021). Diversity Loss in Coralligenous Structuring Species Impacted by Fishing Gear and Marine Litter. Diversity, 13(7), 331. https://doi.org/10.3390/d13070331









