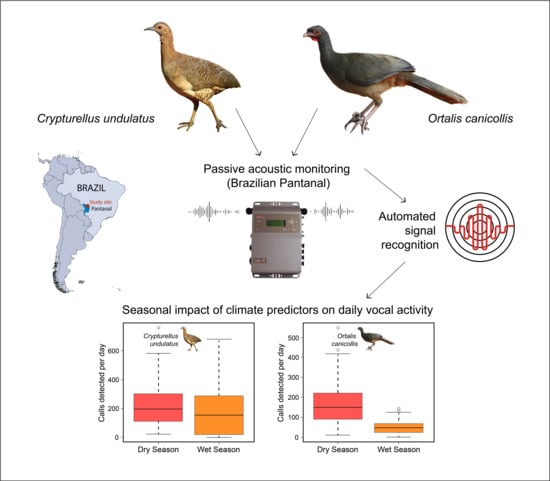
Seasonal Climate Impacts on Vocal Activity in Two Neotropical Nonpasserines
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Species
2.2. Study Area
2.3. Acoustic Monitoring
2.4. Acoustic Data Analyses
- Undulated Tinamou: minimum and maximum frequency range: 1150 and 1350 Hz, respectively; minimum and maximum detection length: 1.3 and 3 s, respectively; maximum intersyllable gap: 0.2 s; distance from the cluster center: 2.0.
- Chaco Chachalaca: minimum and maximum frequency range: 300 and 2500 Hz, respectively; minimum and maximum detection length: 0.5 and 20 s, respectively; maximum intersyllable gap: 0.1 s; distance from the cluster center: 2.0.
2.5. Environmental Variables
2.6. Statistical Analyses
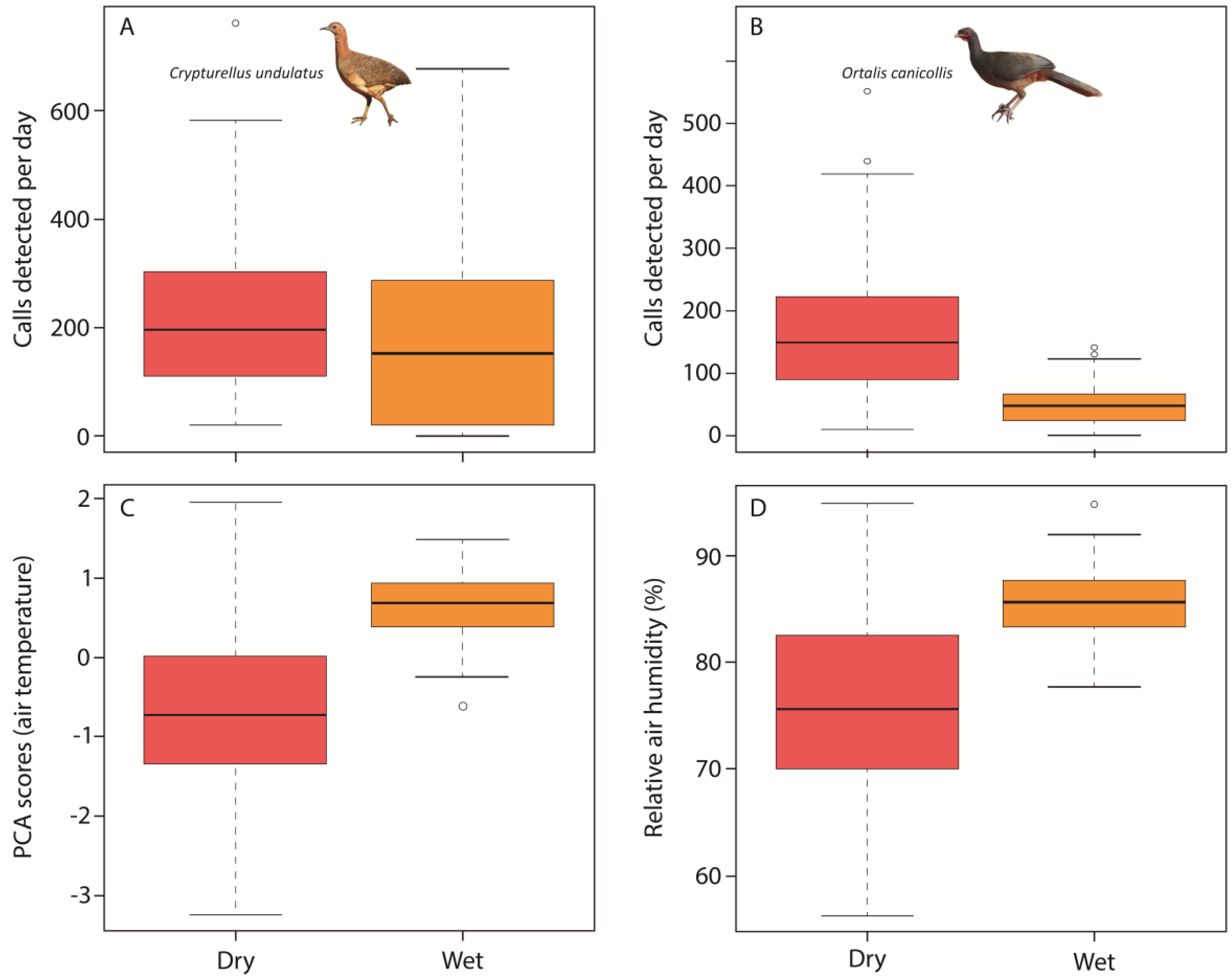
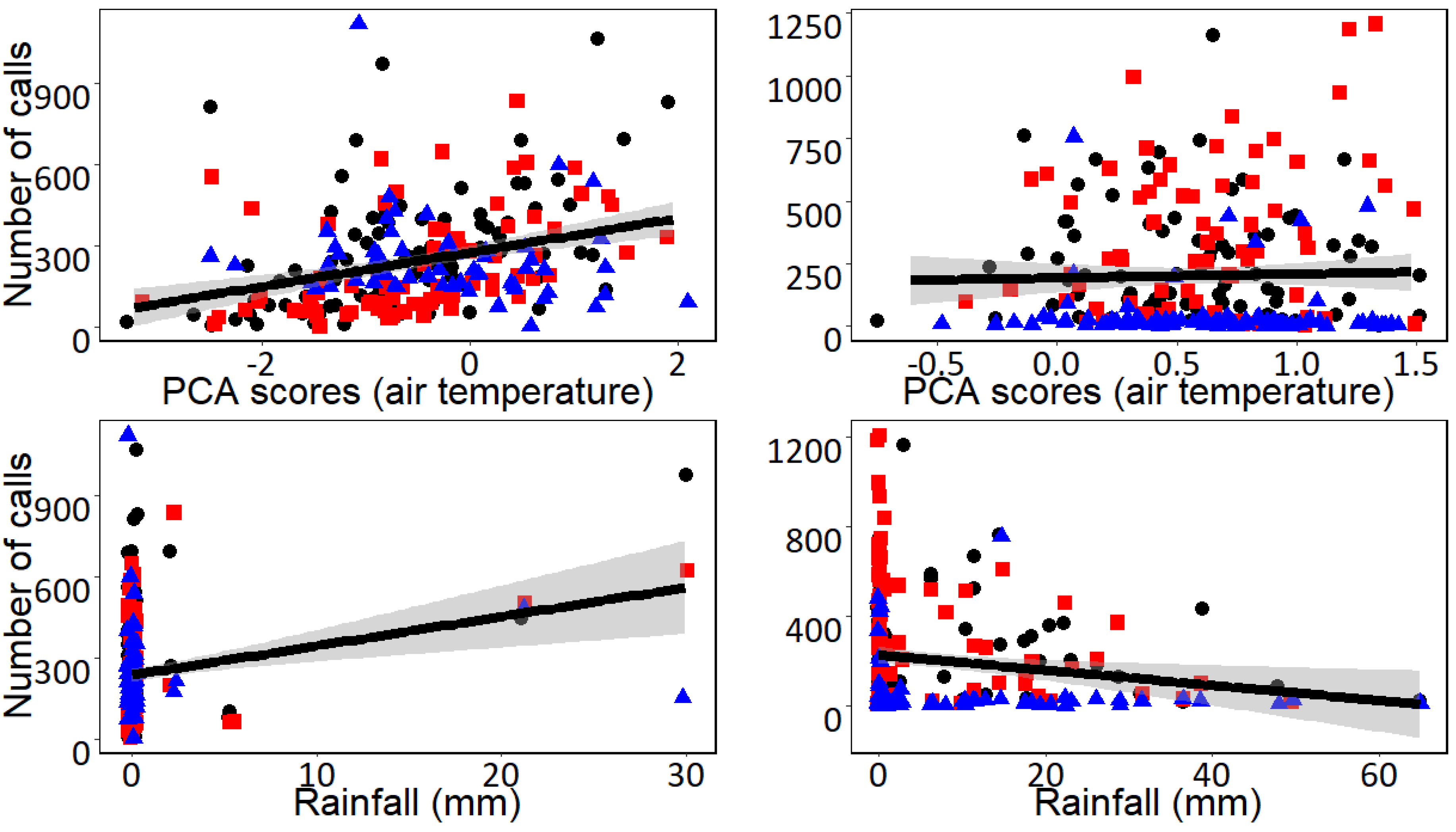
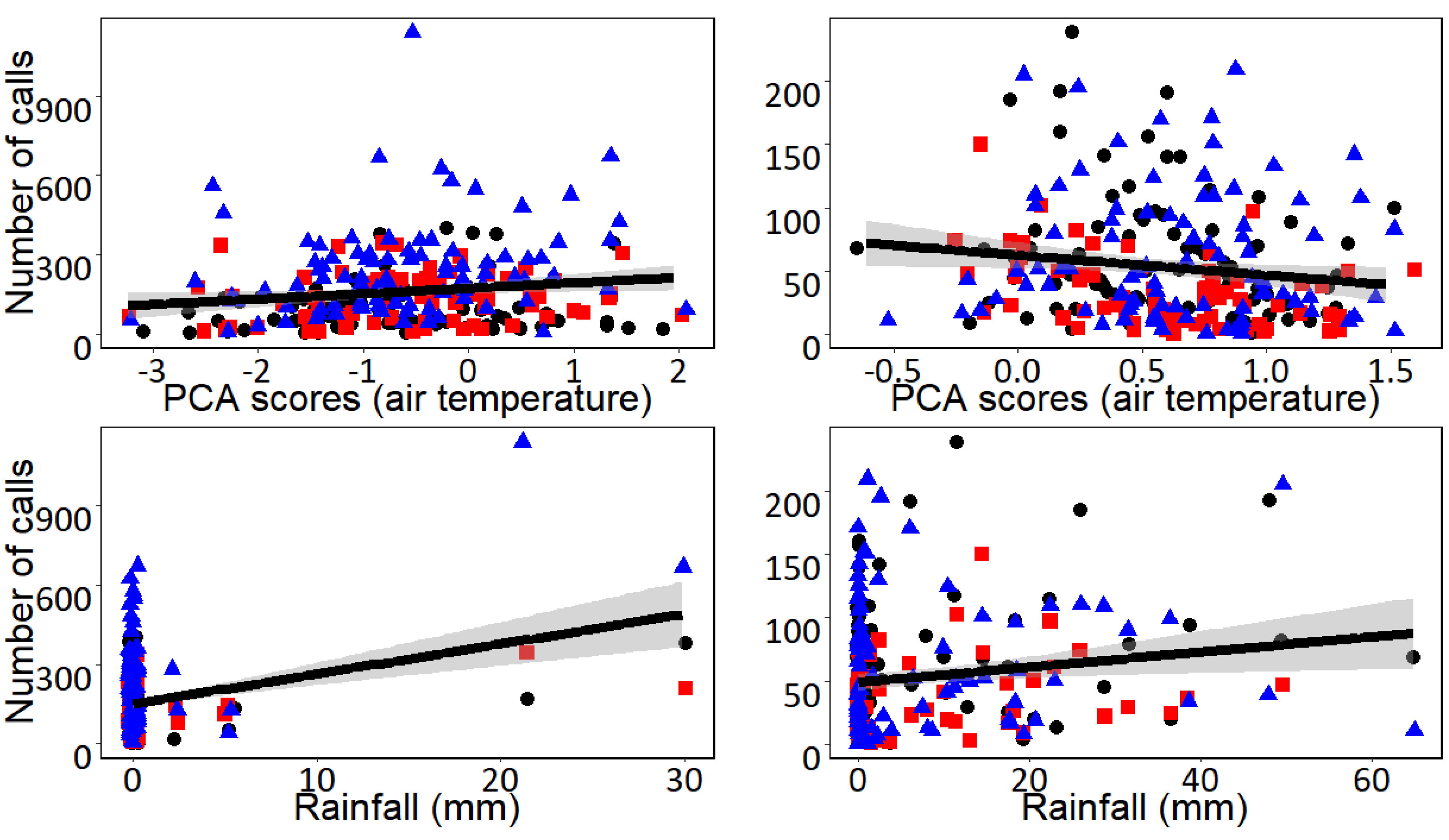
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marler, P. Bird calls: Their potential for behavioral neurobiology. Ann. N. Y. Acad. Sci. 2004, 1016, 31–44. [Google Scholar] [CrossRef]
- Farnsworth, A. Flight calls and their value for future ornithological studies and conservation research. Auk 2005, 122, 733–746. [Google Scholar] [CrossRef]
- Catchpole, C.K.; Slater, P.J. Bird Song: Biological Themes and Variations, 2nd ed.; Cambridge University Press: Cambridge, UK, 2008; p. 335. ISBN 978-0-521-87242-3. [Google Scholar]
- Sandoval, L.; Méndez, C.; Mennill, D.J. Vocal behaviour of White-eared Ground-sparrows (Melozone leucotis) during the breeding season: Repertoires, diel variation, behavioural contexts, and individual distinctiveness. J. Ornithol. 2016, 157, 1–12. [Google Scholar] [CrossRef]
- Staicer, C.A.; Spector, D.A.; Horn, A.G. The dawn chorus and other diel patterns in acoustic signaling. In Ecology and Evolution of Acoustic Communication in Birds; Kroodsma, D.E., Miller, E.H., Eds.; Cornell University Press: New York, NY, USA, 1996; pp. 426–453. [Google Scholar]
- Staicer, C.A. Honest advertisement of pairing status: Evidence from a tropical resident wood-warbler. Anim. Behav. 1996, 51, 375–390. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Osiejuk, T.S.; López-Iborra, G.M. Dawn chorus interpretation differs when using songs or calls: The Dupont’s Lark Chersophilus duponti case. PeerJ 2018, 6, e5241. [Google Scholar] [CrossRef] [PubMed]
- Amrhein, V.; Korner, P.; Naguib, M. Nocturnal and diurnal singing activity in the nightingale: Correlations with mating status and breeding cycle. Anim. Behav. 2002, 64, 939–944. [Google Scholar] [CrossRef]
- Rowan, W. Experiments in bird migration, I. Manipulation of the reproductive cycle: Seasonal histological changes in the gonads. In Proceedings of the Boston Society of Natural History, Boston, MA, USA, 1 January 1929; Volume 39, pp. 115–208. [Google Scholar]
- Ball, G.F.; Hulse, S.H. Birdsong. Am. Psychol. 1998, 53, 37–58. [Google Scholar] [CrossRef] [PubMed]
- Robbins, C.S. Effect of time and day on bird activity. Stud. Avian Biol. 1981, 6, 275–282. [Google Scholar]
- Boncoraglio, G.; Saino, N. Habitat structure and the evolution of bird song: A meta-analysis of the evidence for the acoustic adaptation hypothesis. Funct. Ecol. 2007, 21, 134–142. [Google Scholar] [CrossRef]
- York, J.E.; Young, A.J.; Radford, A.N. Singing in the moonlight: Dawn song performance of a diurnal bird varies with lunar phase. Biol. Lett. 2014, 10, 20130970. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Schuchmann, K.-L. Monitoring the annual vocal activity of two enigmatic nocturnal Neotropical birds: The Common Potoo (Nyctibius griseus) and the Great Potoo (Nyctibius grandis). J. Ornithol. 2020, 161, 1129–1141. [Google Scholar] [CrossRef]
- Potvin, D.A.; Parris, K.M.; Mulder, R.A. Geographically pervasive effects of urban noise on frequency and syllable rate of songs and calls in silvereyes (Zosterops lateralis). Proc. R. Soc. Lond. B Biol. Sci. 2011, 278, 2464–2469. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.J.; Wilson, D.R.; Mennill, D.J. Anthropogenic light is associated with increased vocal activity by nocturnally migrating birds. Condor 2016, 118, 338–344. [Google Scholar] [CrossRef]
- Møller, A.P. When climate change affects where birds sing. Behav. Ecol. 2010, 22, 212–217. [Google Scholar] [CrossRef]
- Bas, Y.; Devictor, V.; Moussus, J.P.; Jiguet, F. Accounting for weather and time-of-day parameters when analysing count data from monitoring programs. Biodivers. Conserv. 2008, 17, 3403–3416. [Google Scholar] [CrossRef]
- Keast, A. Temporal vocalisation patterns in members of a Eucalypt forest bird community: The effects of weather on song production. Emu 1994, 94, 172–180. [Google Scholar] [CrossRef]
- Zuberogoitia, I.; Burgos, G.; González-Oreja, J.A.; Morant, J.; Martínez, J.E.; Zabala Albizua, J. Factors affecting spontaneous vocal activity of Tawny Owls Strix aluco and implications for surveying large areas. Ibis 2019, 161, 495–503. [Google Scholar] [CrossRef]
- Ryan, M.J.; Brenowitz, E.A. The role of body size, phylogeny, and ambient noise in the evolution of bird song. Am. Nat. 1985, 126, 87–100. [Google Scholar] [CrossRef]
- Lengagne, T.; Slater, P.J. The effects of rain on acoustic communication: Tawny owls have good reason for calling less in wet weather. Proc. R. Soc. Lond. B Biol. Sci. 2002, 269, 2121–2125. [Google Scholar] [CrossRef]
- Digby, A.; Towsey, M.; Bell, B.D.; Teal, P.D. Temporal and environmental influences on the vocal behaviour of a nocturnal bird. J. Avian Biol. 2014, 45, 591–599. [Google Scholar] [CrossRef]
- Curio, E. Beiträge zur Populationsökologie des Trauerschnäppers (Ficedula hypoleuca Pallas). Zool. Jarhbücher 1959, 87, 185–230. [Google Scholar]
- Garson, P.J.; Hunter, J.R.M.L. Effects of temperature and time of year on the singing behaviour of wrens Troglodytes and great tits Parus major. Ibis 1979, 121, 481–487. [Google Scholar] [CrossRef]
- Strain, J.G.; Mumme, R.L. Effects of food supplementation, song playback, and temperature on vocal territorial behaviour of Carolina Wrens. Auk 1988, 105, 11–16. [Google Scholar] [CrossRef]
- Riebel, K.; Odom, K.J.; Langmore, N.E.; Hall, M.L. New insights from female bird song: Towards an integrated approach to studying male and female communication roles. Biol. Lett. 2019, 15, 20190059. [Google Scholar] [CrossRef]
- Topp, S.M.; Mennill, D.J. Seasonal variation in the duetting behaviour of rufous-and-white wrens (Thryothorus rufalbus). Behav. Ecol. Sociobiol. 2008, 62, 1107–1117. [Google Scholar] [CrossRef]
- Odom, K.J.; Omland, K.E.; McCaffrey, D.R.; Monroe, M.K.; Christhilf, J.L.; Roberts, N.S.; Logue, D.M. Typical males and unconventional females: Songs and singing behaviors of a tropical, duetting oriole in the breeding and non-breeding season. Front. Ecol. Evol. 2016, 4, 14. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Schuchmann, K.-L.; Marques, M.I. Vocal behavior of the Undulated Tinamou (Crypturellus undulatus) over an annual cycle in the Brazilian Pantanal: New ecological information. Biotropica 2020, 52, 165–171. [Google Scholar] [CrossRef]
- Hau, M.; Perfito, N.; Moore, I.T. Timing of breeding in tropical birds: Mechanisms and evolutionary implications. Ornitol. Neotrop. 2008, 19, 39–59. [Google Scholar]
- Demko, A.D.; Mennill, D.J. Rufous-capped Warblers Basileuterus rufifrons show seasonal, temporal and annual variation in song use. Ibis 2019, 161, 481–494. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Schuchmann, K.-L. Diel and Seasonal Variations of Vocal Behavior of the Neotropical White-Tipped Dove (Leptotila verreauxi). Diversity 2020, 12, 402. [Google Scholar] [CrossRef]
- Mennill, D.J. Variation in the vocal behavior of common loons (Gavia immer): Insights from landscape-level recordings. Waterbirds 2014, 37, 26–36. [Google Scholar] [CrossRef][Green Version]
- Pérez-Granados, C.; Schuchmann, K.-L.; Marques, M.I. Vocal activity of the Ferruginous Pygmy-Owl (Glaucidium brasilianum) is strongly correlated with moon phase and nocturnal temperature. Ethol. Ecol. Evol. 2020, 33, 62–72. [Google Scholar] [CrossRef]
- Cabot, J.; Christie, D.A.; Jutglar, F.; Sharpe, C.J. Undulated Tinamou (Crypturellus undulatus), version 1.0. In Birds of the World; del Hoyo, J., Elliott, A., Sargatal, J., Christie, D.A., de Juana, E., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar] [CrossRef]
- Cabot, J. Family Tinamidae (Tinamous). In Handbook of the Birds of the World. Volume 1: Ostrich to Duck; del Hoyo, J., Elliott, A., Sargatal, J., Eds.; Lynx Edicions: Barcelona, Spain, 1992; pp. 112–138. [Google Scholar]
- Davies, S.J.J.F. Ratites and Tinamous; Oxford University Press: New York, NY, USA, 2002. [Google Scholar]
- del Hoyo, J.; Kirwan, G.M. Chaco Chachalaca (Ortalis canicollis), version 1.0. In Birds of the World; del Hoyo, J., Elliott, A., Sargatal, J., Christie, D.A., Juana, E., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar] [CrossRef]
- Junk, W.J.; Da Cunha, C.N.; Wantzen, K.M.; Petermann, P.; Strüssmann, C.; Marques, M.I.; Adis, J. Biodiversity and its conservation in the Pantanal of Mato Grosso, Brazil. Aquat. Sci. 2006, 68, 278–309. [Google Scholar] [CrossRef]
- Heckman, C.W. The Pantanal of Poconé; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998. [Google Scholar]
- de Deus, F.F.; Arieira, J.; Schuchmann, K.-L.; Tissiani, A.S.O.; Marques, M.I. Avian beta-diversity in a Neotropical wetland: Effects of flooding and vegetation. Wetlands 2020, 40, 1513–1527. [Google Scholar] [CrossRef]
- Rempel, R.S.; Francis, C.M.; Robinson, J.N.; Campbell, M. Comparison of audio recording system performance for detecting and monitoring songbirds. J. Field Ornithol. 2013, 84, 86–97. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Bota, G.; Giralt, D.; Albarracín, J.; Traba, J. Cost-effective assessment of five audio recording systems for wildlife monitoring: Differences between recording distances and singing direction. Ardeola 2019, 66, 311–325. [Google Scholar] [CrossRef]
- Bioacoustics Research Program. Raven Pro: Interactive Sound Analysis Software (Version 1.5). [Computer software]; The Cornell Lab of Ornithology: Ithaca, NY, USA, 2014; Available online: https://www.birds.cornell.edu/raven (accessed on 18 May 2020).
- Knight, E.; Hannah, K.; Foley, G.; Scott, C.; Brigham, R.; Bayne, E. Recommendations for acoustic recognizer performance assessment with application to five common automated signal recognition programs. Avian Conserv. Ecol. 2017, 12, 14. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Schuchmann, K.-L. Nocturnal vocal behavior of the diurnal Undulated Tinamou Crypturellus undulatus is associated with temperature and moon phase. Ibis 2020, 163, 684–694. [Google Scholar] [CrossRef]
- Dormann, C.F.; Elith, J.; Bacher, S.; Buchmann, C.; Carl, G.; Carré, G.; Marquéz, J.R.G.; Gruber, B.; Lafourcade, B.; Leitão, P.J.; et al. Collinearity: A review of methods to deal with it and a simulation study evaluating their performance. Ecography 2013, 36, 27–46. [Google Scholar] [CrossRef]
- Harrison, X.A. A comparison of observation-level random effect and Beta-Binomial models for modelling overdispersion in Binomial data in ecology & evolution. PeerJ 2015, 3, e1114. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: http://www.R-project.org (accessed on 12 July 2021).
- Lancaster, D.A. Life history of the Boucard tinamou in British Honduras. Part I: Distribution and general behavior. Condor 1964, 66, 165–181. [Google Scholar] [CrossRef]
- Lancaster, D.A. Biology of the Brushland Tinamou, Nothoprocta cinerascens. Bull. Am. Mus. Nat. Hist. 1964, 127, 269–314. [Google Scholar]
- Wolda, H. Seasonal fluctuations in rainfall, food and abundance of tropical insects. J. Anim. Ecol. 1978, 47, 369–381. [Google Scholar] [CrossRef]
- Van Schaik, C.P.; Terborgh, J.W.; Wright, S.J. The phenology of tropical forests, adaptative significante, and consequences for primary consumers. Ann. Rev. Ecol. Syst. 1993, 24, 353–377. [Google Scholar] [CrossRef]
- Pérez-Granados, C.; Traba, J. Estimating bird density using passive acoustic monitoring: A review of methods and suggestions for further research. Ibis 2021, 163, 765–783. [Google Scholar] [CrossRef]
- Ehnes, M.; Foote, J.R. Comparison of autonomous and manual recording methods for discrimination of individually distinctive ovenbird songs. Bioacoustics 2015, 24, 111–121. [Google Scholar] [CrossRef]
- Dent, J.M.; Molles, L.E. Call-based identification as a potential tool for monitoring Great spotted kiwi. Emu 2016, 116, 315–322. [Google Scholar] [CrossRef]
| Dry Season | Wet Season | ||||||||
| Estimate | Std. Error | z-Value | p | Estimate | Std. Error | z-Value | p | ||
| Undulated Tinamou | (Intercept) | 0.162 | 0.095 | 1.697 | 0.091 | 0.209 | 0.099 | 2.114 | 0.036 |
| Axis I (Air temperature) | 0.350 | 0.060 | 5.800 | <0.001 | −0.030 | 0.063 | −0.486 | 0.627 | |
| Daily rainfall | 0.254 | 0.059 | 4.255 | <0.001 | −0.174 | 0.063 | −2.764 | 0.006 | |
| Station B | −0.280 | 0.139 | −2.007 | 0.046 | 0.320 | 0.141 | 2.261 | 0.025 | |
| Station C | −0.249 | 0.151 | −1.648 | 0.100 | −0.814 | 0.135 | −6.055 | <0.001 | |
| Chaco Chachalaca | (Intercept) | −0.428 | 0.091 | −4.723 | <0.001 | 0.218 | 0.108 | 2.011 | 0.046 |
| Axis I (Air temperature) | 0.167 | 0.053 | 3.127 | 0.002 | −0.089 | 0.069 | −1.284 | 0.200 | |
| Daily rainfall | 0.334 | 0.053 | 6.270 | <0.001 | −0.106 | 0.069 | 1.542 | 0.124 | |
| Station B | 0.211 | 0.136 | 1.555 | 0.121 | −0.649 | 0.155 | −4.194 | <0.001 | |
| Station C | 0.980 | 0.125 | 7.816 | <0.001 | −0.048 | 0.148 | −0.323 | 0.747 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Granados, C.; Schuchmann, K.-L. Seasonal Climate Impacts on Vocal Activity in Two Neotropical Nonpasserines. Diversity 2021, 13, 319. https://doi.org/10.3390/d13070319
Pérez-Granados C, Schuchmann K-L. Seasonal Climate Impacts on Vocal Activity in Two Neotropical Nonpasserines. Diversity. 2021; 13(7):319. https://doi.org/10.3390/d13070319
Chicago/Turabian StylePérez-Granados, Cristian, and Karl-L. Schuchmann. 2021. "Seasonal Climate Impacts on Vocal Activity in Two Neotropical Nonpasserines" Diversity 13, no. 7: 319. https://doi.org/10.3390/d13070319
APA StylePérez-Granados, C., & Schuchmann, K.-L. (2021). Seasonal Climate Impacts on Vocal Activity in Two Neotropical Nonpasserines. Diversity, 13(7), 319. https://doi.org/10.3390/d13070319