Annelid Diversity: Historical Overview and Future Perspectives
Abstract
1. Introduction
2. Results and Discussion
2.1. Systematics
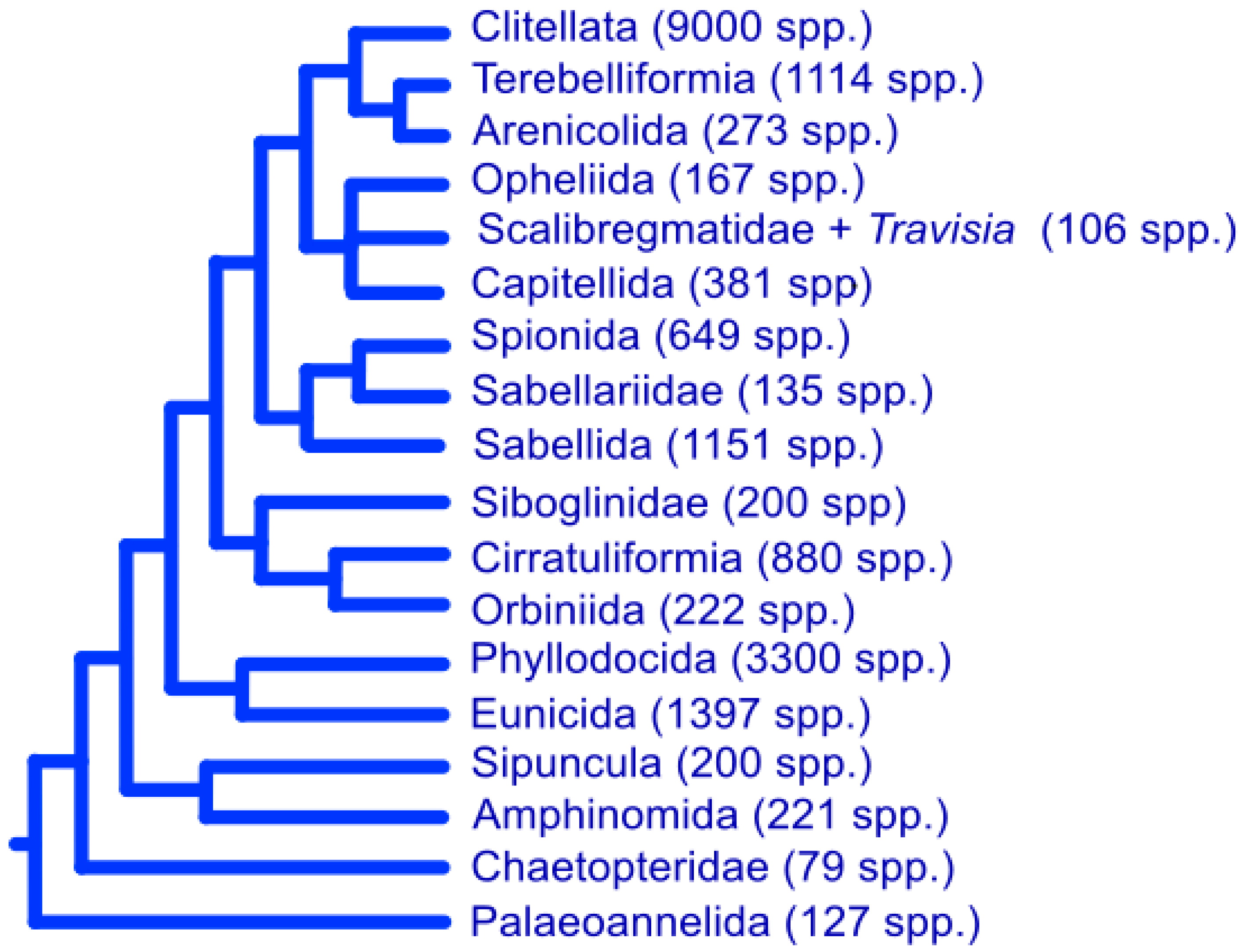
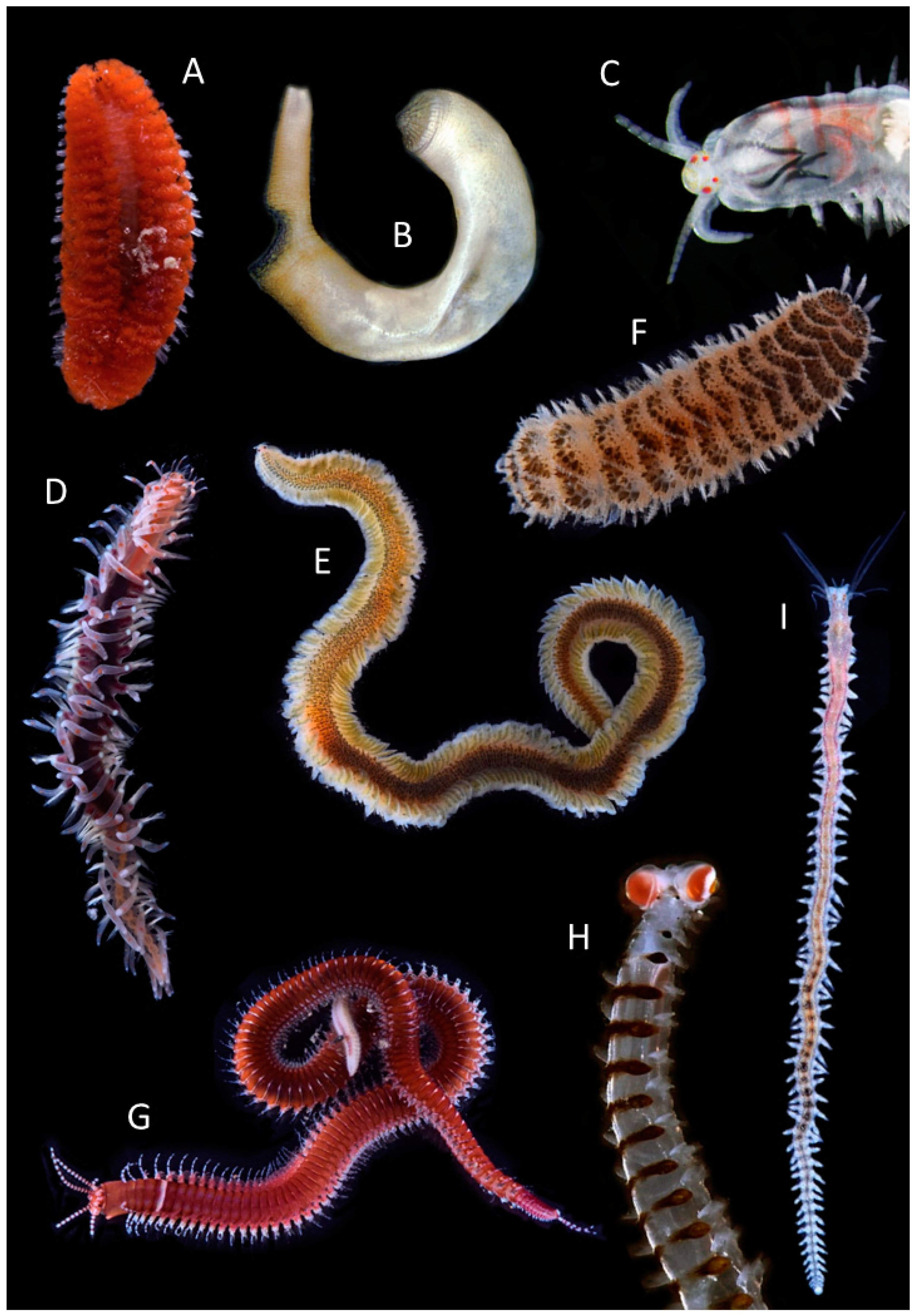
2.2. Annelid Diversity
- Literature thorough scrutiny, including old and obscure publications. It is not uncommon to see how some taxonomic and nomenclatural mistakes have been passed on to most recent publications. Many annelid genera and families require a thorough follow up of synonyms (species that may have been synonymized with others or moved to a different genus or taxonomic group), and also way to rescue some old names that have been lost in most recent taxonomic lists. Of course, databases such as WoRMS are of invaluable help, but as much as they try to keep updated with taxonomic progress there is often a lag [109].
- Some groups require an exhaustive taxonomic revision that includes the examination of type material. This has become more difficult lately as shipping of preserved specimens has become an extremely regulated process, and unaffordable (for economic or time-related reasons) for some museums. Travelling to museums where the type specimens of a genus are housed is also challenging as these may be spread around the globe.
- Taxonomists too often have a partial understanding of their group because we tend to specialize in particular families, genera, environments or geographic areas. This may lead to a non-confident or tentative perception of the overall diversity of larger groups.
- When referring to the total number of a species we generally mean that the currently accepted binomial names are based on morphologically recognizable entities. Taxonomists often hold knowledge of a wider number of morphospecies that are new but not formerly described. In fact, the average time it takes from the first collection of a specimen of a new species to its formal naming and description in a scientific paper is 21 years [110]. The reason for this may be the need for more specimens to account for intraspecific variability or to be preserved in a specific manner, revisions/examination of similar or related species are needed, the group requires a revision, a lack of funding or the need for collaboration with other (often lacking) experts in the group, e.g., taxonomic impediment, etc. [110,111]. Moreover, when the lines of evidence for delineating species are molecular (e.g., DNA sequences), these newly recognized lineages are not often accompanied by formal descriptions [112]. In these situations (no formal binomials), species will be missing from species lists in most cases.
- Cryptic species. The number of complexes of annelid species accounted for in recent years has vastly increased due to the use of molecular data. This has an impact in the real overall diversity present in the group. Although this is not necessary reflected in the total number of species accounted for, and this is because species delineated by molecular mean are often not accompanied by a formal species description (as discussed by Goldstein et al. [112] and exemplified by [61,113,114,115,116]).
2.3. Gaps of Knowledge and Future Perspectives
- An increase in field work in the areas that have been poorly surveyed. There is also a large amount of material, specially coming from deep sea expeditions, that is awaiting to be studied in natural history museums. Therefore, only diving in poorly studied geographic areas, in extreme environments and in museum collections will provide us with a good understanding about the diversity of annelids.
- Most of the knowledge about the biology, anatomy and behavior of annelid groups is based on a limited number of species for each family. This is more obvious in modern biology that mainly focuses on model organisms, especially in the fields of comparative physiology and morphology. Studies based on a larger number of taxa and including some of the microscopic modern technics (phase contrast, scanning electron microscopy, transmission electron microscopy, confocal laser scanning microscopy, tomography and 3D reconstructions) are needed.
- It is recommended to undertake an integrative approach for delimiting and documenting species. This includes the combination of morphological, molecular and biological data. For several groups of annelids, understanding the reproductive and developmental features has also shown to be helpful for species delimitation.
- Species descriptions or re-description often requires re-examination of type specimens for species comparisons. Ideally, a thorough examination of specimens from different geographical areas and ecological features is needed in order to establish species boundaries and to assess intraspecific variability.
- Obtaining genetic information for at least the type species (preferably from type locality) of each genus is needed, as this would allow generic revisions. Specimens for genetic analysis should be collected from the type localities and vouchers deposited.
- For species with wide geographical or bathymetric distribution, population genetic studies are necessary to reveal potential cryptic species. In this line, the advent of high-throughput sequencing methods has a lot to offer for the generation of species delimitation datasets.
- Molecular taxonomy is revealing hidden diversity at a high speed, but formal taxonomic descriptions are lagging behind the molecular work. Therefore, we should increase the efforts made in describing the species encountered after molecular methods. Only trained taxonomist can undertake the development of a proper account and documentation of these lineages, as these activities often require revision of genera, revision of old literature, synonymies and varying terminology, re-examination of museum types, etc.
- Policy should take care of biodiversity, and governments should invest in systems and in training the next generation of taxonomists. The taxonomic work has been neglected for decades and we are suffering a loss of taxonomic knowledge. This has a direct impact on the speed with which species are described, but also on the quality of the biodiversity assessments and the studies based on those. We need to train and sustain more systematists able to discover, describe, identify and classify species.
- We need to promote, care and engage with museum collections and public databases. It is imperative that the type material (holotype, type series, additional specimens showing intraspecific variability, and DNA extractions) is always deposited in properly curated permanent museum collection(s) where it is maintained in optimal conditions. Museums and researchers need to commit to open-access databases such as World Register of Marine Species—WoRMS, Ocean Biodiversity Information System—OBIS, National Center for Biotechnology Information—Genbank, or the Global Biodiversity Information Facility—GBIF, that offer invaluable information for research projects.
2.4. This Special Issue
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kojima, S. Paraphyletic Status of Polychaeta Suggested by Phylogenetic Analysis Based on the Amino Acid Sequences of Elongation Factor-1 Alpha. Mol. Phylogenetics Evol. 1998, 9, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Martin, P. On the Origin of the Hirudinea and the Demise of the Oligochaeta. Proc. R. Soc. Lond. B 2001, 268, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.; Rouse, G.; Hutchings, P.; Colgan, D. Assessing the Usefulness of Histone H3, U2 SnRNA and 28S RDNA in Analyses of Polychaete Relationships. Aust. J. Zool. 1999, 47, 499. [Google Scholar] [CrossRef]
- Rouse, G.W.; Fauchald, K. Cladistics and Polychaetes. Zool. Scr. 1997, 26, 139–204. [Google Scholar] [CrossRef]
- McHugh, D. Molecular Evidence That Echiurans and Pogonophorans Are Derived Annelids. Proc. Nat. Acad. Sci. USA 1997, 94, 8006–8009. [Google Scholar] [CrossRef] [PubMed]
- McHugh, D. Molecular Phylogeny of the Annelida. Can. J. Zool. 2000, 78, 1873–1884. [Google Scholar] [CrossRef]
- Purschke, G.; Hessling, R.; Westheide, W. The Phylogenetic Position of the Clitellata and the Echiura-on the Problematic Assessment of Absent Characters. J. Zool. Syst. Evol. Res. 2000, 38, 165–173. [Google Scholar] [CrossRef]
- Rouse, G.W.; Pleijel, F. Polychaetes; Oxford University Press: London, UK, 2001; pp. 1–354. [Google Scholar]
- Bleidorn, C.; Vogt, L.; Bartolomaeus, T. New Insights into Polychaete Phylogeny (Annelida) Inferred from 18S RDNA Sequences. Mol. Phylogenetics Evol. 2003, 29, 279–288. [Google Scholar] [CrossRef]
- Bleidorn, C.; Vogt, L.; Bartolomaeus, T. A Contribution to Sedentary Polychaete Phylogeny Using 18S RRNA Sequence Data. J. Zool. Syst. Evol. Res. 2003, 41, 186–195. [Google Scholar] [CrossRef]
- Rousset, V.; Rouse, G.W.; Siddall, M.E.; Tillier, A.; Pleijel, F. The Phylogenetic Position of Siboglinidae (Annelida) Inferred from 18S RRNA, 28S RRNA and Morphological Data. Cladistics 2004, 20, 518–533. [Google Scholar] [CrossRef]
- Struck, T.H.; Paul, C.; Hill, N.; Hartmann, S.; Hösel, C.; Kube, M.; Lieb, B.; Meyer, A.; Tiedemann, R.; Purschke, G.; et al. Phylogenomic Analyses Unravel Annelid Evolution. Nature 2011, 471, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Struck, T.H.; Golombek, A.; Weigert, A.; Franke, F.A.; Westheide, W.; Purschke, G.; Bleidorn, C.; Halanych, K.M. The Evolution of Annelids Reveals Two Adaptive Routes to the Interstitial Realm. Curr. Biol. 2015, 25, 1993–1999. [Google Scholar] [CrossRef] [PubMed]
- Weigert, A.; Helm, C.; Meyer, M.; Nickel, B.; Arendt, D.; Hausdorf, B.; Santos, S.R.; Halanych, K.M.; Purschke, G.; Bleidorn, C.; et al. Illuminating the Base of the Annelid Tree Using Transcriptomics. Mol. Biol. Evol. 2014, 31, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Andrade, S.C.S.; Novo, M.; Kawauchi, G.Y.; Worsaae, K.; Pleijel, F.; Giribet, G.; Rouse, G.W. Articulating “Archiannelids”: Phylogenomics and Annelid Relationships, with Emphasis on Meiofaunal Taxa. Mol. Biol. Evol. 2015, 32, 2860–2875. [Google Scholar] [CrossRef]
- Parry, L.; Tanner, A.; Vinther, J. The Origin of Annelids. Palaeontology 2014, 57, 1091–1103. [Google Scholar] [CrossRef]
- Peterson, K.J.; Cotton, J.A.; Gehling, J.G.; Pisani, D. The Ediacaran Emergence of Bilaterians: Congruence between the Genetic and the Geological Fossil Records. Philos. Trans. R. Soc. B 2008, 363, 1435–1443. [Google Scholar] [CrossRef]
- Martin, P.; Kaygorodova, I.; Sherbakov, D.Y.; Verheyen, E. Rapidly Evolving Lineages Impede the Resolution of Phylogenetic Relationships among Clitellata (Annelida). Mol. Phylogenetics Evol. 2000, 15, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Bleidorn, C.; Eeckhaut, I.; Podsiadlowski, L.; Schult, N.; McHugh, D.; Halanych, K.M.; Milinkovitch, M.C.; Tiedemann, R. Mitochondrial Genome and Nuclear Sequence Data Support Myzostomida as Part of the Annelid Radiation. Mol. Phylogenetics Evol. 2007, 24, 1690–1701. [Google Scholar] [CrossRef]
- Bleidorn, C.; Podsiadlowski, L.; Zhong, M.; Eeckhaut, I.; Hartmann, S.; Halanych, K.M.; Tiedemann, R. On the Phylogenetic Position of Myzostomida: Can 77 Genes Get It Wrong? BMC Evol. Biol. 2009, 9, 150. [Google Scholar] [CrossRef]
- Struck, T.H. The Impact of Paralogy on Phylogenomic Studies—A Case Study on Annelid Relationships. PLoS ONE 2013, 8, e62892. [Google Scholar] [CrossRef]
- Weigert, A.; Golombek, A.; Gerth, M.; Schwarz, F.; Struck, T.H.; Bleidorn, C. Evolution of Mitochondrial Gene Order in Annelida. Mol. Phylogenetics Evol. 2016, 94, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Laumer, C.E.; Bekkouche, N.; Kerbl, A.; Goetz, F.; Neves, R.C.; Sørensen, M.V.; Kristensen, R.M.; Hejnol, A.; Dunn, C.W.; Giribet, G.; et al. Spiralian Phylogeny Informs the Evolution of Microscopic Lineages. Curr. Biol. 2015, 25, 2000–2006. [Google Scholar] [CrossRef]
- Beesley, P.L.; Ross, G.J.B.; Glasby, C.J. Polychaetes & Allies: The Southern Synthesis. Fauna of Australia. Polychaeta, Myzostomida, Pogonophora, Echiura, Sipuncula; CSIRO Publishing: Melbourne, Australia, 2000; Volume 44, pp. 1–465. [Google Scholar]
- Reisinger, E. Zweiter Unterstamm der Vermes Polymera. Allgemeine Einleitung zur Naturgeschichte der Vermes Polymera. In Handbuch der Zoologie; Part 6, Vermes Polymera; DeGruyter & Co.: Leipzig/Berlin, Germany, 1928; Volume 2.2, pp. 1–32. [Google Scholar]
- Hempelmann, F. Erste und Zweite Klasse der Vermes Polymera (Annelida). Archiannelida = UrRingelwürmer und Polychaeta = Borstenwürmer. In Handbuch der Zoologie; Part 7, Vermes Polymera; Kükenthal, W., Krumbach, T., Eds.; DeGruyter & Co.: Leipzig/Berlin, Germany, 1928; Volume 2.2, pp. 1–212. [Google Scholar]
- Michaelsen, W. Dritte Klasse der Vermes Polymera (Annelida). Clitellata = Gürtelwürmer. 1. Ordnung der Clitellata: Oligochaeta = Regenwürmer und Verwandte. In Handbuch der Zoologie; Part 8, Vermes Polymera; Kükenthal, W., Krumbach, T., Eds.; DeGruyter & Co.: Leipzig/Berlin, Germany, 1928; Volume 2.2, pp. 1–118. [Google Scholar]
- Scriban, I.A.; Autrum, H. 2. Ordnung Der Clitellata: Hirudinea = Egel. In Handbuch der Zoologie; Part 8, Vermes Polymera; Kükenthal, W., Krumbach, T., Eds.; DeGruyter & Co.: Leipzig/Berlin, Germany, 1928; Volume 2.2, pp. 119–352. [Google Scholar]
- Purschke, G.; Böggemann, M.; Westheide, W. Basal Groups and Pleistoannelica, Sedentaria. In Handbook of Zoology; Annelida; De Gruyter: Berlin, Germany, 2019; Volume 1, pp. 1–472. [Google Scholar]
- Purschke, G.; Böggemann, B.; Westheide, W. Pleistoannelida, Sedentaria II. In Handbook of Zoology; Annelida; De Gruyter: Berlin, Germany, 2019; Volume 2, pp. 1–465. [Google Scholar]
- Purschke, G.; Böggemann, B.; Westheide, W. Pleistoannelida, Sedentaria III and Errantia I. In Handbook of Zoology; Annelida; De Gruyter: Berlin, Germany, 2021; Volume 3, pp. 1–480. [Google Scholar]
- Belan, T.A. Marine Environmental Quality Assessment Using Polychaete Taxocene Characteristics in Vancouver Harbour. Mar. Environ. Res. 2004, 57, 89–101. [Google Scholar] [CrossRef]
- Surugiu, V. The Use of Polychaetes as Indicators of Eutrophication and Organic Enrichment of Coastal Waters: A Study Case–Romanian Black Sea Coast. An. Ştiinţifice ale Univ. “Al.I. Cuza” Iaşi s. Biol. Anim. 2005, 51, 55–62. [Google Scholar]
- Fründ, H.-C.; Graefe, U.; Tischer, S. Earthworms as Bioindicators of Soil Quality. In Biology of Earthworms; Springer: Berlin/Heidelberg, Germany, 2011; pp. 261–278. [Google Scholar]
- Olsgard, F.; Brattegard, T.; Holthe, T. Polychaetes as Surrogates for Marine Biodiversity: Lower Taxonomic Resolution and Indicator Groups. Biodiv. Conserv. 2003, 12, 1033–1049. [Google Scholar] [CrossRef]
- Gladstone, W.; Murray, B.R.; Hutchings, P. Promising yet Variable Performance of Cross-Taxon Biodiversity Surrogates: A Test in Two Marine Habitats at Multiple Times. Biodiv. Conserv. 2020, 29, 3067–3089. [Google Scholar] [CrossRef]
- Bocchetti, R.; Fattorini, D.; Gambi, M.; Regoli, F. Trace Metal Concentrations and Susceptibility to Oxidative Stress in the Polychaete Sabella spallanzanii (Gmelin)(Sabellidae): Potential Role of Antioxidants in Revealing Stressful Environmental Conditions in the Mediterranean. Arch. Environ. Contam. Toxicol. 2004, 46, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Poirier, L.; Berthet, B.; Amiard, J.-C.; Jeantet, A.-Y.; Amiard-Triquet, C. A Suitable Model for the Biomonitoring of Trace Metal Bioavailabilities in Estuarine Sediments: The Annelid Polychaete Nereis diversicolor. J. Mar. Biol. Assoc. UK 2006, 86, 71–82. [Google Scholar] [CrossRef]
- Dean, H.K. The Use of Polychaetes (Annelida) as Indicator Species of Marine Pollution: A Review. Rev. Biol. Trop. 2008, 56, 11–38. [Google Scholar]
- Unrine, J.M.; Hunyadi, S.E.; Tsyusko, O.V.; Rao, W.; Shoults-Wilson, W.A.; Bertsch, P.M. Evidence for Bioavailability of Au Nanoparticles from Soil and Biodistribution within Earthworms (Eisenia fetida). Environ. Sci. Technol. 2010, 44, 8308–8313. [Google Scholar] [CrossRef]
- Jamieson, B.G.M.; Rouse, G.W.; Pleijel, F. Reproductive Biology and Phylogeny of Annelida; Science Publishers: Enfield, UK, 2006; Volume 4, pp. 1–698. [Google Scholar]
- Cole, V.J.; Chick, R.C.; Hutchings, P.A. A Review of Global Fisheries for Polychaete Worms as a Resource for Recreational Fishers: Diversity, Sustainability and Research Needs. Rev. Fish Biol. Fish. 2018, 28, 543–565. [Google Scholar] [CrossRef]
- Datta, D.; Talapatra, S.N.; Swarnakar, S. Bioactive Compounds from Marine Invertebrates for Potential Medicines-an Overview. Int. Let. Nat. Sci. 2015, 7, 42–61. [Google Scholar] [CrossRef]
- Rodrigo, A.P.; Costa, P.M. The Hidden Biotechnological Potential of Marine Invertebrates: The Polychaeta Case Study. Environ. Res. 2019, 173, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Bax, N.; Williamson, A.; Aguero, M.; Gonzalez, E.; Geeves, W. Marine Invasive Alien Species: A Threat to Global Biodiversity. Mar. Pol. 2003, 27, 313–323. [Google Scholar] [CrossRef]
- Zenetos, A.; Çinar, M.; Pancucci-Papadopoulou, M.; Harmelin, J.; Furnari, G.; Andaloro, F.; Bellou, N.; Streftaris, N.; Zibrowius, H. Annotated List of Marine Alien Species in the Mediterranean with Records of the Worst Invasive Species. Mediterr. Mar. Sci. 2005, 6, 63–118. [Google Scholar] [CrossRef]
- Molnar, J.L.; Gamboa, R.L.; Revenga, C.; Spalding, M.D. Assessing the Global Threat of Invasive Species to Marine Biodiversity. Front. Ecol. Environ. 2008, 6, 485–492. [Google Scholar] [CrossRef]
- Ekman, S. Zoogeography of the Sea; Sidgwick & Jackson, Ltd.: London, UK, 1953; Volume 6, pp. 1–240. [Google Scholar] [CrossRef]
- Knox, G. The Distribution of Polychaetes within the Indo-Pacific. In Proceedings of the 8th Pacific Science Congress, Quezon City, Philippines, 16–28 November 1953; National Research Council of the Philippines: Quezon City, Philippines, 1957; Volume 3, pp. 403–411. [Google Scholar]
- Hartman, O. Catalogue of the Polychaeta Annelids of the World. Allan Hancock Found. Publ. 1959, 23, 1–628. [Google Scholar]
- Day, J.H. A Monograph on the Polychaeta of Southern Africa. Part 1. Errantia; British Museum (Natural History): London, UK, 1967; pp. 1–458. [Google Scholar]
- Day, J.H. A Monograph on the Polychaeta of Southern Africa. Part II. Sedentaria; British Museum (Natural History): London, UK, 1967; pp. 459–878. [Google Scholar]
- Fauvel, P. Polychète Errantes. Faune Fr. 1923, 5, 1–488. [Google Scholar]
- Fauvel, P. Polychètes Sédentaires. Addenda Aux Errantes, Archiannélides, Myzostomaires. Faune Fr. 1927, 16, 1–494. [Google Scholar]
- Williams, S.J. The Status of Terebellides stroemi (Polychaeta; Trichobranchidae) as a Cosmopolitan Species, Based on a Worldwide Morphological Survey, Including Description of New Species. In Proceedings of the First International Polychaete Conference, Sydney, Australia, 4–9 July 1983; pp. 118–142. [Google Scholar]
- Eckelbarger, K.; Grassle, J. Spermatogenesis, Sperm Storage and Comparative Sperm Morphology in Nine Species of Capitella, Capitomastus and Capitellides (Polychaeta: Capitellidae). Mar. Biol. 1987, 95, 415–429. [Google Scholar] [CrossRef]
- Chambers, S.J.; Woodham, A. A New Species of Chaetozone (Polychaeta: Cirratulidae) from Deep Water in the Northeast Atlantic, with Comments on the Diversity of the Genus in Cold Northern Waters. Hydrobiologia 2003, 496, 41–48. [Google Scholar] [CrossRef]
- Hutchings, P.; Karageorgopoulos, P. Designation of a Neotype of Marphysa sanguinea (Montagu, 1813) and a Description of a New Species of Marphysa from Eastern Australia. Hydrobiologia 2003, 496, 87–94. [Google Scholar] [CrossRef]
- Koh, B.S.; Bhaud, M.R.; Jirkov, I.A. Two New Species of Owenia (Annelida: Polychaeta) in the Northern Part of the North Atlantic Ocean and Remarks on Previously Erected Species from the Same Area. Sarsia 2003, 88, 175–188. [Google Scholar] [CrossRef]
- Ford, E.; Hutchings, P. An Analysis of Morphological Characters of Owenia Useful to Distinguish Species: Description of Three New Species of Owenia (Oweniidae: Polychaeta) from Australian Waters. Mar. Ecol. 2005, 26, 181–196. [Google Scholar] [CrossRef]
- Nygren, A.; Parapar, J.; Pons, J.; Meißner, K.; Bakken, T.; Kongsrud, J.A.; Oug, E.; Gaeva, D.; Sikorski, A.; Johansen, R.A.; et al. A Mega-Cryptic Species Complex Hidden among One of the Most Common Annelids in the North East Atlantic. PLoS ONE 2018, 13, e0198356. [Google Scholar] [CrossRef] [PubMed]
- Grosse, M.; Bakken, T.; Nygren, A.; Kongsrud, J.A.; Capa, M. Species Delimitation Analyses of NE Atlantic Chaetozone (Annelida, Cirratulidae) Reveals Hidden Diversity among a Common and Abundant Marine Annelid. Mol. Phylogenetics Evol. 2020, 149, 106852. [Google Scholar] [CrossRef]
- Parapar, J.; Capa, M.; Nygren, A.; Moreira, J. To Name but a Few: Descriptions of Five New Species of Terebellides (Annelida, Trichobranchidae) from the North East Atlantic. ZooKeys 2020, 992, 1–58. [Google Scholar] [CrossRef]
- Nygren, A. Cryptic Polychaete Diversity: A Review. Zool. Scr. 2014, 43, 172–183. [Google Scholar] [CrossRef]
- Hutchings, P.; Kupriyanova, E. Cosmopolitan Polychaetes–Fact or Fiction? Personal and Historical Perspectives. Invertebr. Syst. 2018, 32, 1–9. [Google Scholar] [CrossRef]
- Hutchings, P.; Lavesque, N. I Know Who You Are, but Do Others Know? Why Correct Scientific Names Are so Important for the Biological Sciences. Zoosymposia 2020, 19, 151–163. [Google Scholar] [CrossRef]
- Hendrix, P.F.; Callaham Jr, M.A.; Drake, J.M.; Huang, C.-Y.; James, S.W.; Snyder, B.A.; Zhang, W. Pandora’s Box Contained Bait: The Global Problem of Introduced Earthworms. Ann. Rev. Ecol. Evol. Syst. 2008, 39, 593–613. [Google Scholar] [CrossRef]
- Çinar, M.E. Alien Polychaete Species Worldwide: Current Status and Their Impacts. J. Mar. Biol. Ass. 2013, 93, 1257–1278. [Google Scholar] [CrossRef]
- De Quatrefages, A. Histoire Naturelle Des Annéles Marine et d’eau Douce. Annélides et Géphyriens; Librarie Encyclopédique de Roret: Paris, France, 1866; Volume 2, pp. 1–588. [Google Scholar]
- Uschakov, P.V. Polychaeta of the Far Eastern Seas of the U.S.S.R. Keys to the Fauna of the USSR. No. 56; Zoological Institute of the Academy of Sciences of the USSR: Moscow, Russia, 1955; pp. 1–433. [Google Scholar]
- Hartman, O. Atlas of the Errantiate Polychaetous Annelids from California; Allan Hancock Foundation, University of Southern California: Los Angeles, LA, USA, 1968; pp. 1–828. [Google Scholar]
- Hartman, O. Atlas of the Sedentariate Polychaetous Annelids from California; Allan Hancock Foundation, University of Southern California: Los Angeles LA, USA, 1969; pp. 1–812. [Google Scholar]
- Hartmann-Schröder, G. Annelida, Borstenwürmer, Polychaeta. Tierwelt Dtschl. 1971, 58, 1–594. [Google Scholar]
- Dales, R.P. Annelids; Hutchinson University Library: London, UK, 1963; pp. 1–200. [Google Scholar]
- Dales, R.P. The polychaete stomodeum and phylogeny. In Essays on Polychaetous Annelids in Memory of Dr. Olga Hartman; Reish, D.J., Fauchald, K., Eds.; Allan Hancock Foundation, University of Southern California: Los Angeles, LA, USA, 1977; pp. 525–546. [Google Scholar]
- Fauchald, K. The Polychaete Worms. Definitions and Keys to the Orders, Families and Genera. Nat. Hist. Mus. Los Angeles Cty. Sci. Ser. 1977, 28, 1–188. [Google Scholar]
- Pettibone, M.H. Annelida. In Synopsis and Classification of living Organisms; Parker, S.P., Ed.; McGraw-Hill Book Co: New York, NY, USA, 1982; Volume 2, pp. 1–43. [Google Scholar]
- Hartmann-Schröder, G. Annelida, Borstenwürmer, Polychaeta. In Die Tierwelt Deutschlands und der Angrenzenden Meeresteile; 2nd revised ed.; Gustav Fischer: Jena, Germany, 1996; pp. 1–648. [Google Scholar]
- Fauchald, K.; Rouse, G. Polychaete Systematics: Past and Present. Zool. Scr. 1997, 26, 71–138. [Google Scholar] [CrossRef]
- Rouse, G.W.; Pleijel, F. Current Problems in Polychaete Systematics. Hydrobiologia 2003, 496, 175–189. [Google Scholar] [CrossRef]
- Eibye-Jacobsen, D.; Nielsen, C. The Rearticulation of Annelids. Zool. Scr. 1996, 25, 275–282. [Google Scholar] [CrossRef]
- Bartolomaeus, T.; Purschke, G.; Hausen, H. Polychaete Phylogeny Based on Morphological Data–a Comparison of Current Attempts. Hydrobiologia 2005, 535, 341–356. [Google Scholar]
- Struck, T.H.; Schult, N.; Kusen, T.; Hickman, E.; Bleidorn, C.; McHugh, D.; Halanych, K.M. Annelida phylogeny and the status of Sipuncula and Echiura. BMC Evol. Biol. 2007, 7, 57. [Google Scholar] [CrossRef]
- Struck, T.H.; Nesnidal, M.P.; Purschke, G.; Halanych, K.M. Detecting Possibly Saturated Positions in 18S and 28S Sequences and Their Influence on Phylogenetic Reconstruction of Annelida (Lophotrochozoa). Mol. Phylogenetics Evol. 2008, 48, 628–645. [Google Scholar] [CrossRef] [PubMed]
- Zrzavý, J.; Říha, P.; Piálek, L.; Janouškovec, J. Phylogeny of Annelida (Lophotrochozoa): Total-Evidence Analysis of Morphology and Six Genes. BMC Evol. Biol. 2009, 9, 189. [Google Scholar] [CrossRef]
- Appeltans, W.; Ahyong, S.T.; Anderson, G.; Angel, M.V.; Artois, T.; Bailly, N.; Bamber, R.; Barber, A.; Bartsch, I.; Berta, A. The Magnitude of Global Marine Species Diversity. Curr. Biol. 2012, 22, 2189–2202. [Google Scholar] [CrossRef] [PubMed]
- Aguado, M.; Capa, M.; Oceguera-Figueroa, A.; Rouse, G. Annelida. In The Tree of Life: Evolution and Classification of Living Organisms; Vargas, P., Zardoya, R., Eds.; Sinauer Associates: Sunderland, MA, USA, 2014; pp. 254–269. [Google Scholar]
- Weigert, A.; Bleidorn, C. Current Status of Annelid Phylogeny. Org. Divers. Evol. 2016, 16, 345–362. [Google Scholar] [CrossRef]
- WoRMS Annelida. 2021. Available online: http://marinespecies.org/aphia.php?p=taxdetails&id=882 (accessed on 13 March 2021).
- Parapar, J.; Martínez, A.; Moreira, J. On the Systematics and Biodiversity of the Opheliidae and Scalibregmatidae. Diversity 2021, 13, 87. [Google Scholar] [CrossRef]
- Zanol, J.; Carrera-Parra, L.F.; Steiner, T.M.; Amaral, A.C.Z.; Wiklund, H.; Ravara, A.; Budaeva, N. The Current State of Eunicida (Annelida) Systematics and Biodiversity. Diversity 2021, 13, 74. [Google Scholar] [CrossRef]
- Glasby, C.J.; Erséus, C.; Martin, P. Annelids in Extreme Aquatic Environments: Diversity, Adaptations and Evolution. Diversity 2021, 13, 98. [Google Scholar] [CrossRef]
- Hutchings, P.; Carrerette, O.; Nogueira, J.M.; Hourdez, S.; Lavesque, N. The Terebelliformia-Recent Developments and Future Directions. Diversity 2021, 13, 60. [Google Scholar] [CrossRef]
- Martinsson, S.; Erséus, C. Cryptic Clitellata: Molecular Species Delimitation of Clitellate Worms (Annelida): An Overview. Diversity 2021, 13, 36. [Google Scholar] [CrossRef]
- Meca, M.A.; Zhadan, A.; Struck, T.H. The Early Branching Group of Orbiniida Sensu Struck et al., 2015: Parergodrilidae and Orbiniidae. Diversity 2021, 13, 29. [Google Scholar] [CrossRef]
- Parapar, J.; Mortimer, K.; Capa, M.; Moreira, J. On the Systematics and Biodiversity of the Palaeoannelida. Diversity 2021, 13, 41. [Google Scholar] [CrossRef]
- Schulze, A.; Kawauchi, G.Y. How Many Sipunculan Species Are Hiding in Our Oceans? Diversity 2021, 13, 43. [Google Scholar] [CrossRef]
- Worsaae, K.; Kerbl, A.; Domenico, M.D.; Gonzalez, B.C.; Bekkouche, N.; Martínez, A. Interstitial Annelida. Diversity 2021, 13, 77. [Google Scholar] [CrossRef]
- Capa, M.; Kupriyanova, E.; Nogueira, J.M.M.; Bick, A.; Tovar-Hernández, M.A. Fanworms: Yesterday, Today and Tomorrow. Diversity 2021, in press. [Google Scholar]
- Grosse, M.; Zhadan, A.; Langeneck, J.; Fiege, D.; Martínez, A. Still Digging: Advances and Perspectives in the Study of the Diversity of Several Sedentarian Annelid Families. Diversity 2021, in press. [Google Scholar]
- Martin, D.; Aguado, M.T.; Fernández Alamo, M.A.; Britayev, T.; Böggemann, M.; Capa, M.; Faulwetter, S.; Fukuda, M.V.; Helm, C.; Petti, M.A.V.; et al. An Approach to the Diversity of Phyllodocida (Annelida: Errantia), with a Focus on Glyceridae, Goniadidae, Nephtyidae, Polynoidae, Sphaerodoridae, Syllidae and the Holoplanktonic Families. Diversity 2021, in press. [Google Scholar]
- Struck, T. Phylogeny. Annelida, Basal Groups: Pleistoannelida, Sedentaria I. In Handbook of Zoology; De Gruyter: Berlin, Germany, 2019; Volume 1, pp. 37–68. [Google Scholar]
- Blake, J.A.; Maciolek, N.J.; Meissner, K. Spionidae Grube, 1850. In Handbook of Zoology; Annelida; Volume 2: Pleistoannelida, Sedentaria II; De Gruyter: Berlin, Germany, 2019; pp. 1–102. [Google Scholar]
- Blake, J.A.; Maciolek, N.J. Poecilochaetidae Hannerz, 1956. In Handbook of Zoology; Annelida; Volume 2: Pleistoannelida, Sedentaria II; De Gruyter: Berlin, Germany, 2019; pp. 103–119. [Google Scholar]
- Blake, J.A.; Maciolek, N.J. Uncispionidae Green, 1982. In Handbook of Zoology; Annelida; Volume 2: Pleistoannelida, Sedentaria II; De Gruyter: Berlin, Germany, 2019; pp. 136–144. [Google Scholar]
- Blake, J.A.; Maciolek, N.J. Trochochaetidae Pettibone,1963. In Handbook of Zoology; Annelida; Volume 2: Pleistoannelida, Sedentaria II.; De Gruyter: Berlin, Germany, 2019; pp. 120–135. [Google Scholar]
- Capa, M.; Hutchings, P. Sabellariidae. In Hanbook of Zoology; Volume 2: Pleistoannelida, Sedentaria II; De Gruyter: Berlin, Germany, 2019; pp. 144–163. [Google Scholar]
- Li, Y.; Kocot, K.M.; Whelan, N.V.; Santos, S.R.; Waits, D.S.; Thornhill, D.J.; Halanych, K.M. Phylogenomics of Tubeworms (Siboglinidae, Annelida) and Comparative Performance of Different Reconstruction Methods. Zool. Scr. 2017, 46, 200–213. [Google Scholar] [CrossRef]
- Costello, M.J.; Wilson, S.; Houlding, B. Predicting Total Global Species Richness Using Rates of Species Description and Estimates of Taxonomic Effort. Syst. Biol. 2012, 61, 871. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, B.; Perrard, A.; Bouchet, P. 21 Years of Shelf Life between Discovery and Description of New Species. Curr. Biol. 2012, 22, R943–R944. [Google Scholar] [CrossRef]
- Hutchings, P.A. Major Issues Facing Taxonomy—A Personal Perspective. Megataxa 2020, 1, 46–48. [Google Scholar] [CrossRef]
- Goldstein, P.Z.; DeSalle, R. Integrating DNA Barcode Data and Taxonomic Practice: Determination, Discovery, and Description. Bioessays 2011, 33, 135–147. [Google Scholar] [CrossRef]
- Capa, M.; Pons, J.; Hutchings, P. Cryptic Diversity, Intraspecific Phenetic Plasticity and Recent Geographical Translocations in Branchiomma (Sabellidae, Annelida). Zol. Scr. 2013, 42, 637–655. [Google Scholar] [CrossRef]
- Seixas, V.C.; Zanol, J.; Magalhães, W.F.; Paiva, P.C. Genetic Diversity of Timarete punctata (Annelida: Cirratulidae): Detection of Pseudo-Cryptic Species and a Potential Biological Invader. Estuar. Coast. Shelf. Sci. 2017, 197, 214–220. [Google Scholar] [CrossRef]
- Martinsson, S.; Erséus, C. Cryptic Diversity in Supposedly Species-Poor Genera of Enchytraeidae (Annelida: Clitellata). Zool. J. Linn. Soc. 2018, 183, 749–762. [Google Scholar] [CrossRef]
- Marchán, D.F.; Fernández, R.; Domínguez, J.; Díaz Cosín, D.J.; Novo, M. Genome-Informed Integrative Taxonomic Description of Three Cryptic Species in the Earthworm Genus Carpetania (Oligochaeta, Hormogastridae). Syst. Biodivers. 2020, 18, 203–215. [Google Scholar] [CrossRef]
- Gunton, L.M.; Kupriyanova, E.K.; Alvestad, T.; Avery, L.; Blake, J.A.; Biriukova, O.; Böggemann, M.; Borisova, P.; Budaeva, N.; Burghardt, I.; et al. Annelids of the Eastern Australian Abyss Collected by the 2017 RV ‘Investigator’ Voyage. Zookeys 2021, 1020, 1–198. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capa, M.; Hutchings, P. Annelid Diversity: Historical Overview and Future Perspectives. Diversity 2021, 13, 129. https://doi.org/10.3390/d13030129
Capa M, Hutchings P. Annelid Diversity: Historical Overview and Future Perspectives. Diversity. 2021; 13(3):129. https://doi.org/10.3390/d13030129
Chicago/Turabian StyleCapa, María, and Pat Hutchings. 2021. "Annelid Diversity: Historical Overview and Future Perspectives" Diversity 13, no. 3: 129. https://doi.org/10.3390/d13030129
APA StyleCapa, M., & Hutchings, P. (2021). Annelid Diversity: Historical Overview and Future Perspectives. Diversity, 13(3), 129. https://doi.org/10.3390/d13030129






