Effects of Longer Droughts on Holm Oak Quercus ilex L. Acorn Pests: Consequences for Infestation Rates, Seed Biomass and Embryo Survival
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Areas
2.2. Study Species
2.3. Field Sampling and Acorn Classification
2.4. Acorn Crop and Infestation Rates
2.5. Meteorological Data
2.6. Statistical Analyses
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Logan, J.A.; Régnière, J.; Powell, J.A. Assessing the impacts of global warming on forest pest dynamics. Front. Ecol. Environ. 2003, 1, 130–137. [Google Scholar] [CrossRef]
- Pureswaran, D.S.; Roques, A.; Battisti, A. Forest insects and climate change. Curr. For. Rep. 2018, 4, 35–50. [Google Scholar] [CrossRef]
- McKone, M.J.; Kelly, D.; Lee, W.G. Effect of climate change on mast-seeding species: Frequency of mass flowering and escape from specialist insect seed predators. Glob. Chang. Biol. 1998, 4, 591–596. [Google Scholar] [CrossRef]
- Voigt, W.; Perner, J.; Davis, A.J.; Eggers, T.; Schumacher, J.; Bährmann, R.; Fabian, B.; Heinrich, W.; Köhler, G.; Lichter, D.; et al. Trophic levels are differentially sensitive to climate. Ecology 2003, 84, 2444–2453. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, D.; Wang, L.; Fang, J.; Sun, W.; Ren, B. Effects of altered precipitation on insect community composition and structure in a meadow steppe. Ecol. Entomol. 2014, 39, 453–461. [Google Scholar] [CrossRef]
- Solbreck, C.; Knape, J. Seed production and predation in a changing climate—New roles for resource and seed predator feedback. Ecology 2017, 98, 2301–2311. [Google Scholar] [CrossRef] [PubMed]
- Lewis, O.T.; Gripenberg, S. Insect seed predators and environmental change. J. Appl. Ecol. 2008, 45, 1593–1599. [Google Scholar] [CrossRef]
- Giorgi, F.; Lionello, P. Climate change projections for the Mediterranean region. Glob. Planet. Chang. 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Serrano, J.P.; Díaz, F.J.A.; García, J.A.G. Analysis of extreme temperature events over the Iberian Peninsula during the 21st century using dynamic climate projections chosen using max-stable processes. Atmosphere 2020, 11, 506. [Google Scholar] [CrossRef]
- Poncet, B.N.; Garat, P.; Manel, S.; Bru, N.; Sachet, J.M.; Roques, A.; Despres, L. The effect of climate on masting in the European larch and on its specific seed predators. Oecologia 2009, 159, 527–537. [Google Scholar] [CrossRef]
- Bonal, R.; Hernández, M.; Espelta, J.M.; Muñoz, A.; Aparicio, J.M. Unexpected consequences of a drier world: Evidence that delay in late summer rains biases the population sex ratio of an insect. R. Soc. Open Sci. 2015, 2. [Google Scholar] [CrossRef]
- Bogdziewicz, M.; Canelo, T.; Bonal, R. Rainfall and host reproduction regulate population dynamics of a specialist seed predator. Ecol. Entomol. 2019, 45. [Google Scholar] [CrossRef]
- Renner, S.S.; Zohner, C.M. Climate change and phenological mismatch in trophic interactions among plants, insects, and vertebrates. Annu. Rev. Ecol. Evol. Syst. 2018, 49, 165–182. [Google Scholar] [CrossRef]
- Simberloff, D.; Dayan, T. The guild concept and the structure of ecological communities. Annu. Rev. Ecol. Syst. 1991, 22, 115–143. [Google Scholar] [CrossRef]
- Ruiz-Carbayo, H.; Bonal, R.; Pino, J.; Espelta, J.M. Zero-sum landscape effects on acorn predation associated with shifts in granivore insect community in new holm oak (Quercus ilex) forests. Divers. Distrib. 2018, 1–14. [Google Scholar] [CrossRef]
- Conrad, A.O.; Crocker, E.V.; Li, X.; Thomas, W.R.; Ochuodho, T.O.; Holmes, T.P.; Nelson, C.D. Threats to oaks in the eastern United States: Perceptions and expectations of experts. J. For. 2020, 118, 14–27. [Google Scholar] [CrossRef]
- Galko, J.; Økland, B.; Kimoto, T.; Rell, S.; Zúbrik, M.; Kunca, A.; Vakula, J.; Gubka, A.; Nikolov, C. Testing temperature effects on woodboring beetles associated with oak dieback. Biol. Bratisl. 2018, 73, 361–370. [Google Scholar] [CrossRef]
- Lewis, V.R. Within-tree distribution of acorns infested by Curculio occidentis (Coleoptera: Curculionidae) and Cydia latiferreana (Lepidoptera: Tortricidae) on the coast live oak. Environ. Entomol. 1992, 21, 975–982. [Google Scholar] [CrossRef]
- Maetô, K. Raletionships between size and mortality of Quercus mongolica var. grosseserrata acorns due to pre-dispersal infestation by frugivorous insects. Jpn. For. Soc. 1995, 77, 213–219. [Google Scholar]
- Csóka, G.; Hirka, A.H. Direct effects of carpophagous insects on the germination ability and early abscission of oak acorns. Acta Silv. Lign. Hung. 2006, 2, 57–67. [Google Scholar]
- Bonal, R.; Muñoz, A.; Díaz, M. Satiation of predispersal seed predators: The importance of considering both plant and seed levels. Evol. Ecol. 2007, 21, 367–380. [Google Scholar] [CrossRef]
- Xiao, Z.; Harris, M.K.; Zhang, Z. Acorn defenses to herbivory from insects: Implications for the joint evolution of resistance, tolerance and escape. For. Ecol. Manag. 2007, 238, 302–308. [Google Scholar] [CrossRef]
- Gibson, L.P. Biology and life history of acorn-infesting weevils of the genus Conotrachelus (Coleoptera: Curculionidae). Annu. Entomol. Soc. Am. 1964, 57, 521–526. [Google Scholar] [CrossRef]
- Leiva, M.J.; Fernández-Alés, R. Holm-oak (Quercus ilex subsp. ballota) acorns infestation by insects in Mediterranean dehesas and shrublands: Its effect on acorn germination and seedling emergence. For. Ecol. Manag. 2005, 212, 221–229. [Google Scholar] [CrossRef]
- Moreno, G.; Pulido, F.J. The functioning, management and persistence of Dehesas. In Agroforestry in Europe: Current Status and Future Prospects; Rigueiro-Rodríguez, A., McAdam, J., Mosquera-Losada, M.R., Eds.; Springer Science and Business Media B.V: Berlin, Germany, 2009; pp. 127–160. ISBN 978-1-4020-8271-9. [Google Scholar]
- Bonal, R.; Muñoz, A.; María Espelta, J. Mismatch between the timing of oviposition and the seasonal optimum. The stochastic phenology of Mediterranean acorn weevils. Ecol. Entomol. 2010, 35, 270–278. [Google Scholar] [CrossRef]
- Espelta, J.M.; Arias-Leclaire, H.; Fernandez-Martinez, M.; Doblas-Miranda, E.; Muñoz, A.; Bonal, R. Beyond predator satiation: Masting but also the effects of rainfall stochasticity on weevils drive acorn predation. Ecosphere 2017, 8. [Google Scholar] [CrossRef]
- Branco, M.; Branco, C.; Merouani, H.; Almeida, M.H. Germination success, survival and seedling vigour of Quercus suber acorns in relation to insect damage. For. Ecol. Manag. 2002, 166, 159–164. [Google Scholar] [CrossRef]
- Tejerina, D.; García-Torres, S.; Cabeza de Vaca, M.; Vázquez, F.M.; Cava, R. Acorns (Quercus rotundifolia Lam.) and grass as natural sources of antioxidants and fatty acids in the “montanera” feeding of Iberian pig: Intra- and inter-annual variations. Food Chem. 2011, 124, 997–1004. [Google Scholar] [CrossRef]
- Bonal, R.; Hernández, M.; Ortego, J.; Muñoz, A.; Espelta, J.M. Positive cascade effects of forest fragmentation on acorn weevils mediated by seed size enlargement. Insect Conserv. Divers. 2012, 5, 381–388. [Google Scholar] [CrossRef]
- Bonal, R.; Muñoz, A. Seed growth suppression constrains the growth of seed parasites: Premature acorn abscission reduces Curculio elephas larval size. Ecol. Entomol. 2008, 33, 31–36. [Google Scholar] [CrossRef]
- Bartlow, A.W.; Agosta, S.J.; Curtis, R.; Yi, X.; Steele, M.A. Acorn size and tolerance to seed predators: The multiple roles of acorns as food for seed predators, fruit for dispersal and fuel for growth. Integr. Zool. 2018, 13, 251–266. [Google Scholar] [CrossRef]
- Torres-Vila, L.M.; Cruces Caldera, E.; Rodríguez-Molina, M.; Sánchez Gónzalez, A.; Delgado Valiente, E.; Ferrero García, J.J.; Ponce Escudero, F.; Palo Nuñez, E.; Barrena Galnán, M.; Aza Barrero, M.; et al. Daños, distribución espacial de los principales carpófagos de encina y alcornoque en Extremadura: Curculio elephas Gyllenhal, Cydia fagiglandana Zeller y Cydia triangulella Goeze. Bol. San. Veg. Plagas 2006, 32, 45–56. [Google Scholar]
- Desouhant, E.; Debouzie, D.; Ploye, H.; Menu, F. Clutch size manipulations in the chestnut weevil, Curculio elephas: Fitness of oviposition strategies. Oecologia 2000, 122, 493–499. [Google Scholar] [CrossRef]
- Bonal, R.; Muñoz, A. Seed weevils living on the edge: Pressures and conflicts over body size in the endoparasitic Curculio larvae. Ecol. Entomol. 2009, 34, 304–309. [Google Scholar] [CrossRef]
- Espelta, J.M.; Bonal, R.; Sánchez-Humanes, B. Pre-dispersal acorn predation in mixed oak forests: Interspecific differences are driven by the interplay among seed phenology, seed size and predator size. J. Ecol. 2009, 97, 1416–1423. [Google Scholar] [CrossRef]
- Bonal, R.; Espelta, J.M.; Vogler, A.P. Complex selection on life-history traits and the maintenance of variation in exaggerated rostrum length in acorn weevils. Oecologia 2011, 167, 1053–1061. [Google Scholar] [CrossRef]
- Menu, F. Strategies of emergence in the chestnut weevil Curculio elephas (Coleoptera: Curculionidae). Oecologia 1993, 96, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Torres-Vila, L.M.; Cruces Caldera, E.; Sánchez Gónzalez, A.; Ferrero García, J.J.; Ponce Escudero, F.; Martín Vertedor, D.; Aza Barrero, C.; Rodríguez Corbacho, F.; Barrena Galán, F. Dinámica poblacional y daños de Curculio elephas Gyllenhal (Col.: Curculionidae), Cydia fagiglandana Zeller, Cydia triangulella Goeze y Pattunene fasciana L. (Lep.: Tortricidae) sobre Quercus y Castanea en Extremadura. Boletín Sanid. Veg. Plagas 2008, 34, 329–341. [Google Scholar]
- Ito, F.; Higashi, S. An indirect mutualism between oaks and wood ants via aphids. J. Anim. Ecol. 1991, 60, 463–470. [Google Scholar] [CrossRef]
- ESRI ArcGIS Desktop; Versión 10; Sistemas Ambientales: Redlands, CA, USA, 2011.
- Pulido, F.J. Herbivorismo y Regeneración de la Encina (Quercus ilex L.) en Bosques y Dehesas. Ph.D. Thesis, University of Extremadura, Badajoz, Spain, 1999. [Google Scholar]
- Yu, X.; Zhou, H.; Luo, T. Spatial and temporal variations in insect-infested acorn fall in a Quercus liaotungensis forest in North China. Ecol. Res. 2003, 18, 155–164. [Google Scholar] [CrossRef]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Wickham, H. The Split-apply-combine strategy for data analysis. J. Stat. Software 2011, 40, 1–29. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mixed Effects Models. R Package Version 3.1-137. 2018. Available online: https://CRAN.R-project.org/package=nlme (accessed on 20 January 2021).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous inference in general parametric models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Pulido, F.J.; Díaz, M. Recruitment of a mediterranean oak: A whole-cycle approach. Ecoscience 2005, 12, 92–102. [Google Scholar] [CrossRef]
- Bonal, R.; Muñoz, A. Multi-trophic effects of ungulate intraguild predation on acorn weevils. Oecologia 2007, 152, 533–540. [Google Scholar] [CrossRef]
- Fukumoto, H.; Kajimura, H. Effects of asynchronous acorn production by co-occurring Quercus trees on resource utilization by acorn-feeding insects. J. For. Res. 2011, 16, 62–67. [Google Scholar] [CrossRef]
- Debouzie, D.; Heizmann, A.; Desouhant, E.; Menu, F. Interference at several temporal and spatial scales between two chestnut insects. Oecologia 1996, 108, 151–158. [Google Scholar] [CrossRef]
- Guidone, L.; Valentini, N.; Rolle, L.; Me, G.; Tavella, L. Early nut development as a resistance factor to the attacks of Curculio nucum (Coleoptera: Curculionidae). Annu. Appl. Biol. 2007, 150, 323–329. [Google Scholar] [CrossRef]
- Dickerson, A.K.; Shankles, P.G.; Hu, D.L. Raindrops push and splash flying insects. Phys. Fluids 2014, 26. [Google Scholar] [CrossRef]
- Chen, C.; Harvey, J.A.; Biere, A.; Gols, R. Rain downpours affect survival and development of insect herbivores: The specter of climate change? Ecology 2019, 100, 1–10. [Google Scholar] [CrossRef]
- Peguero, G.; Bonal, R.; Sol, D.; Muñoz, A.; Sork, V.L.; Espelta, J.M. Tropical insect diversity: Evidence of greater host specialization in seed-feeding weevils. Ecology 2017, 98, 2180–2190. [Google Scholar] [CrossRef] [PubMed]
- Espelta, J.M.; Cortés, P.; Molowny-Horas, R.; Sánchez-Humanes, B.; Retana, J. Masting mediated by summer drought reduces acorn predation in mediterranean oak forests. Ecology 2008, 89, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Caignard, T.; Kremer, A.; Firmat, C.; Nicolas, M.; Venner, S.; Delzon, S. Increasing spring temperatures favor oak seed production in temperate areas. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]



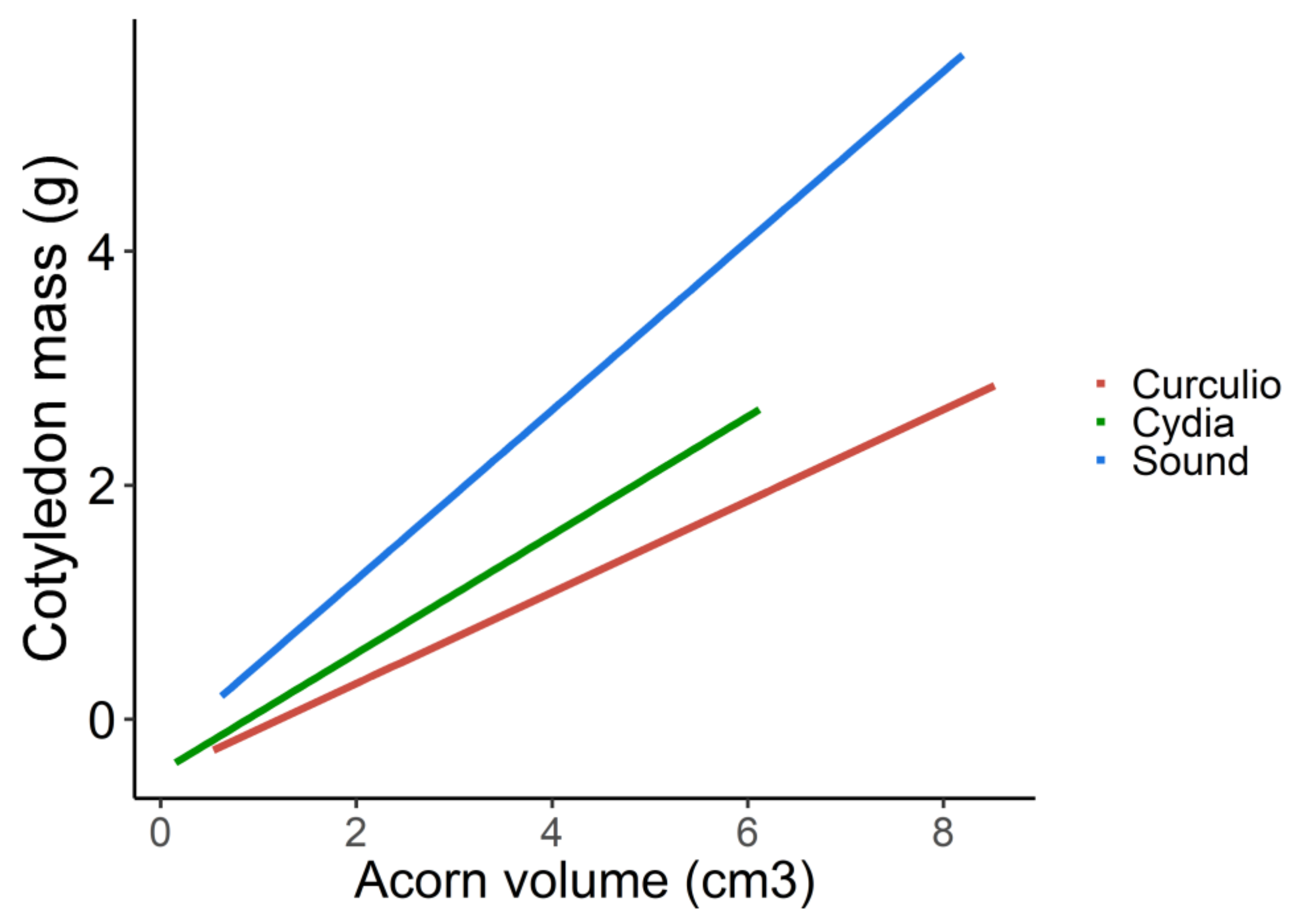
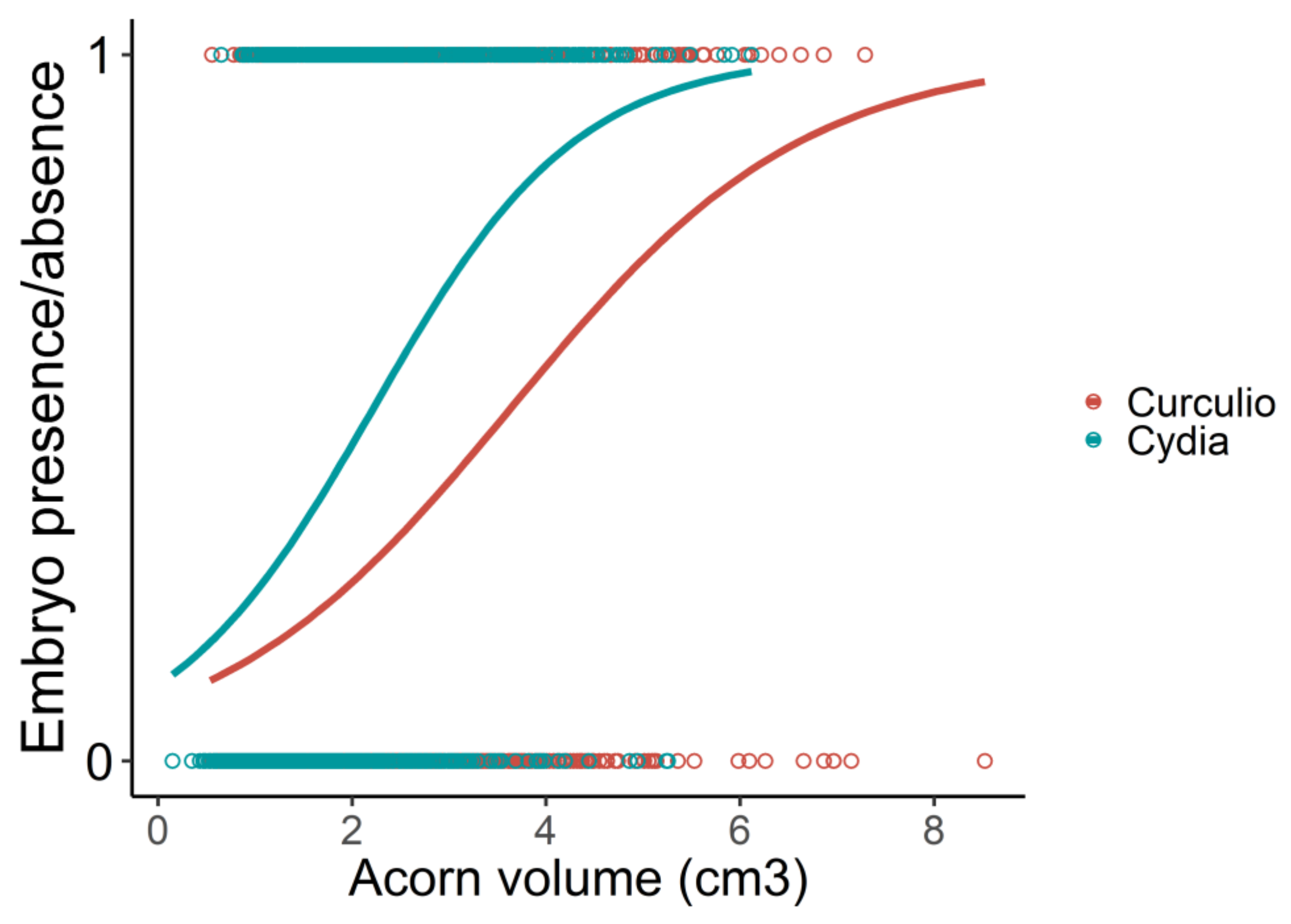
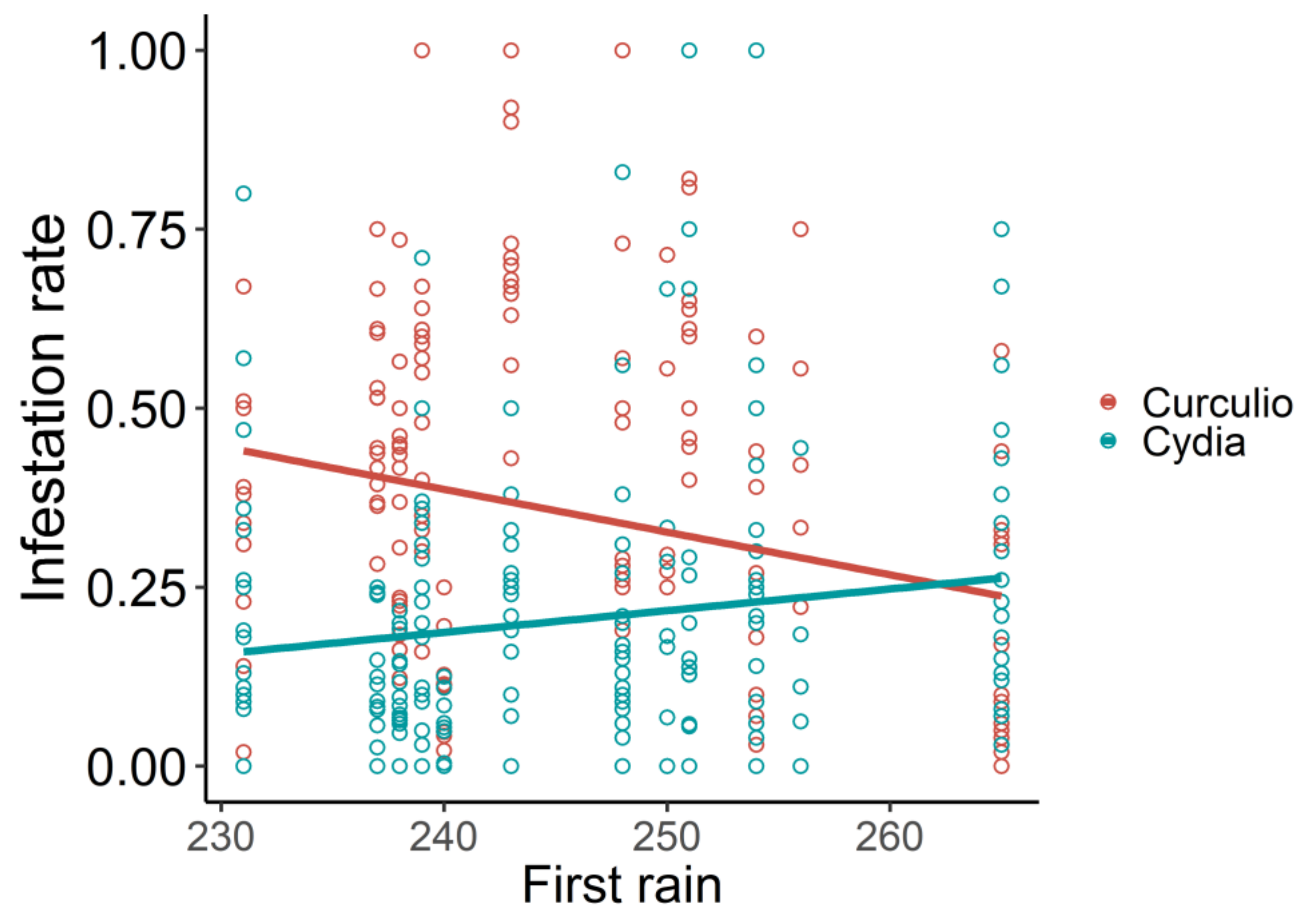
| Type of Acorn | Estimate | Z | p |
|---|---|---|---|
| Cydia–Curculio | −0.318 | −8.266 | <0.001 |
| Sound–Curculio | 0.440 | 14.932 | <0.001 |
| Sound–Cydia | 0.758 | 20.899 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canelo, T.; Gaytán, Á.; Pérez-Izquierdo, C.; Bonal, R. Effects of Longer Droughts on Holm Oak Quercus ilex L. Acorn Pests: Consequences for Infestation Rates, Seed Biomass and Embryo Survival. Diversity 2021, 13, 110. https://doi.org/10.3390/d13030110
Canelo T, Gaytán Á, Pérez-Izquierdo C, Bonal R. Effects of Longer Droughts on Holm Oak Quercus ilex L. Acorn Pests: Consequences for Infestation Rates, Seed Biomass and Embryo Survival. Diversity. 2021; 13(3):110. https://doi.org/10.3390/d13030110
Chicago/Turabian StyleCanelo, Tara, Álvaro Gaytán, Carlos Pérez-Izquierdo, and Raúl Bonal. 2021. "Effects of Longer Droughts on Holm Oak Quercus ilex L. Acorn Pests: Consequences for Infestation Rates, Seed Biomass and Embryo Survival" Diversity 13, no. 3: 110. https://doi.org/10.3390/d13030110
APA StyleCanelo, T., Gaytán, Á., Pérez-Izquierdo, C., & Bonal, R. (2021). Effects of Longer Droughts on Holm Oak Quercus ilex L. Acorn Pests: Consequences for Infestation Rates, Seed Biomass and Embryo Survival. Diversity, 13(3), 110. https://doi.org/10.3390/d13030110






