Abstract
During long periods with no precipitation, a sulfidic spring (Smrdljivec) appears in the dry bed of the Reka River before sinking into the karst underground. The study characterizes the area’s geological setting, development of microbial communities and an ecotone, and impact on the vulnerable karst ecosystem. Geological mapping of the area, stable isotopic analyses, field measurements, and physico-chemical and toxicity analyses were applied to elucidate the environmental conditions. The spring’s microbial diversity was assessed using cultivation methods, microscopy, and metagenomics. Sulfur compounds in the spring probably originate from coal layers in the vicinity. Metagenomic analyses revealed 175 distinct operational taxonomic units in spring water and biofilms. Proteobacteria predominated in developed biofilms, and a “core” microbiome was represented by methylotrophs, including Methylobacter, Methylomonas, and Methylotenera. Diatoms represented an important component of biofilm biomass. A combination of environmental factors and climatic conditions allows the formation and accessibility of emerging biodiversity hotspots and ecotones. Details of their dynamic nature, global impact, and distribution should be highlighted further and given more protection.
1. Introduction
Currently, climatological and hydrological extremes seem to occur more frequently than during the recent past [1]. Under such conditions, biota must adapt to environmental changes in order to ensure survival and successful colonization [2]. Biota are affected by climate change [3], and clear indications of accelerated warming have been observed during the last 120 years [4]. Organisms need to adapt in order to survive environmental changes that affect their habitat, displaying various responses ranging from phenological displacements [5] to changes in species distribution and community composition [2], or even to extinctions [6]. It is also known that ecosystem type, biomass, productivity, or community composition react differently to environmental changes. In the case of abrupt changes, distinct parts of an ecosystem will respond in different ways [7]. For instance, despite the common view that sensitive environments such as the Arctic are particularly susceptible to environmental changes, it was shown that Arctic benthic species were surprisingly robust in terms of habitat losses compared to taxonomic groups that are under pressure from ocean warming and acidification [8].
Karst caves, particularly semi-closed systems with a stable climate, e.g., constant temperature, provide another relevant example of an environment and accompanying biota that are sensitive to changes. However, the vulnerability of subterranean biota to changes most probably depend upon the extent of its adaptation, its evolutionary history, and its phylogeny [9]. Karst, comprising a landscape that lacks a surface-water drainage network and displays pronounced groundwater vulnerability, frequently faces extreme hydrological conditions ranging between complete dryness and total flooding [10]. Extreme environmental conditions represent a threat for species diversity in karst [11].
Changing environmental conditions can reveal hidden habitats that, in combination with the formation of environmental gradients, can lead to the emergence of ecotones, which commonly represent hotspots of species richness and abundance. Springs are one such example, being considered ecotones between a soil ecosystem, surface water, and groundwater [12,13]. During extremely dry periods, a sulfidic spring (first observed in 2017) appears in the bed of the Reka River upstream of where the river sinks into the karst caves of Škocjanske jame (Slovenia). Terrestrial sulfidic springs host diverse microbial communities by serving as stable conduits for geochemically diverse and nutrient-rich subsurface waters. The microorganisms that colonize terrestrial springs likely originate from groundwater, but may also originate from the surface. Therefore, the biogeographic distribution of microbial communities colonizing sulfidic springs is likely controlled by a combination of spring geochemistry and surface and subsurface transport mechanisms, and not necessarily by geographic proximity to other springs. The composition of a core microbial community could provide a way to monitor changes in diversity if a system is threatened by anthropogenic processes or climate change [14].
The objective of this study was to analyze the recharge area of the sulfidic spring, the chemical signature of which is overwhelmed at times of high flow in the Reka River. As such, it offers only a temporary and short-lived habitat and ecotone for biota, and presents only brief opportunities during which it is possible to estimate its microbial diversity and its impact upon the karst river ecosystem as a whole.
2. Materials and Methods
2.1. Discovery and Site Description
On 12 July 2017 an unusual spring exhibiting colorful (white, greenish yellow, and brown) biofilms, accompanied by the distinctive odor of hydrogen sulfide, was observed during routine park nature protection supervision on the right bank of the dry bed of the Reka River at a site known locally as Vir (“spring” in English). The sulfidic spring became visible when the water level of the Reka was extremely low, and the river had shrunk to occupy only the deepest part of its bed. Several orifices emitting gases were observed along about 7 m of the river bank (Figure 1). Spring (WGS84: 45°39′47.8506″ N, 13°59′56.5326″ E, 321.9 m a.s.l.), which was given the name Smrdljivec (“stinker” in English), appeared around 500 m upstream of where the Reka River sinks at Škocjanske jame (Škocjan Caves). With its main source below the Snežnik Mountain in SW Slovenia, the Reka River transmits water from a surface catchment of more than 350 km2 towards the ponors in Škocjanske jame. The mean annual discharge upstream of the ponors is 8.95 m3/s, but during times of high flow, it can reach 387 m3/s [15,16]. In the final part of its surface course, the Reka River flows in a 5 km-long blind valley, which ends in the Škocjanske jame ponor [17]. The caves were entered on UNESCO’s list of natural and cultural World Heritage Sites in 1986 and added to the Ramsar List of Wetlands of International Importance in 1999. Subsequently, the area was incorporated into UNESCO’s Man and the Biosphere Programme as part of the World Network of Biosphere Reserves in 2004.

Figure 1.
Location of the Smrdljivec spring (1—the Reka River, 2—main orifice of the Smrdljivec spring, 3—direction of the water flow before it sinks into the gravel).
The Smrdljivec spring lies in the core area of Regijski park Škocjanske jame (Škocjan Caves Regional Park). This area covers 403 ha and the buffer zone, which overlaps with the Reka River catchment area, amounts to 45,000 ha. The core area includes 52 valuable natural features of national importance, comprising 14 “areas”, 13 “sites”, and 25 “caves”. Most are related to the hydrology, geomorphology, geology, and biodiversity of the karst, especially in and around the caves. Škocjanske jame (the Škocjan Caves) represent the most outstanding site of natural value within the protected area [18].
2.2. Geology of the Smrdljivec Spring Area
The surface and underground karst of the Škocjanske jame area developed on and within 400–500 m of bedded Cretaceous limestones containing rare rudist bivalve fossils, known as the Sežana Formation (Figure 2), K22−4, [19,20,21]. Above the Sežana Formation, the 250–400 m-thick Lipica Formation (K24−5) consists of bedded and massive limestones with rudists (K24−5). Above this, the 50–300 m-thick Liburnian Formation (K-Pc) comprises bedded and platy limestones with thin coal beds occurring locally. Below the Sežana Formation (Figure 2 cross-section A–B), the massive and partly recrystallized limestones [19] of the Repen Formation (K21,2) are less than 200 m thick, with their base about 450 m below the horizon associated with the Smrdljivec spring.
Fossils of characean algae are noticeably abundant in certain beds of the Liburnian Formation, sufficiently that the rock can be described as characean limestone [19]. This rock type was deposited in restricted shelf areas of lagoonal type, whereas coal seams with high sulfur content were formed during phases with brackish- and fresh-water conditions. The source material for the coal layers, most of which are developed in the lower and middle parts of the Liburnian Formation [19], probably accumulated aquatic plant debris, especially characeans [22].
Black coals in the Liburnian Formation contain extremely high levels (up to 12%) of sulfur [22]. The coal is determined as being meta-bituminous (R: 1.0—vitrinite reflectance in %) [23], and is located within dark, slightly bituminous, limestones [19]. Coal formerly extracted at the now-disused coal mine at Vremski Britof, about 2.5 km SE of the Smrdljivec spring, was classified as a high-quality black coal with a high heat content, averaging 35,590 kJ/kg [22]. Other information indicates that coal mining took place even closer—2 km SE—to Škocjanske jame, where five coal layers (d = 0.2–1.9 m) were found within the Liburnian Formation, across a 3.0 × 0.8 km area [24]. Coal layers were accessed by deep shafts penetrating to about 60–65 m below the surface. After the First World War, coal exploration drilling revealed coal layers 1–1.6 m thick at 500 m depth [25]. The mouth of the Smrdljivec spring is in limestones of the Lipica Formation (K24−5) (cross-section A–B in Figure 2), but the source of the sulfur must be within another lithological unit, most probably the coal beds in the Liburnian Formation.
Tectonically the Škocjanske jame area belongs to the northern part of the External Dinarides, where folding and overthrusting are typically towards the S and SW [26]. The most important fault in the area is the Divača Fault (Figure 2), which shows predominantly dextral strike-slip movement [19,21,27] and which is part of the Dinaric (NW–SE) Fault System [27]. As shown on the cross-section A–B (Figure 2), the Divača Fault also exhibits vertical normal fault movement. Additionally the Krgunca and Risnik faults were originally mapped and named in the area NW of Škocjanske jame [28], and detailed structural geological mapping on the surface above the cave, performed by one of the authors, confirmed their continuation into the study area (Figure 2). Vertical displacement on the Krgunca Fault in the area close to the spring shows relative lowering of the SW block, which is the block that contains the Smrdljivec spring. Details of any possible continuation of the Risnik Fault into the locality of the spring are inconclusive.
It has been confirmed that bedding-plane slips are important tectonic structures in guiding the development of cave passages in the Škocjanske jame area [15,21,29,30]. In addition, not only the faults with Dinaric (NW–SE) orientation but also zones of almost N–S-oriented fissures can remain tectonically active [20,30]. Considering the strongly tectonized nature of the Liburnian Formation limestones [31], sulfur compounds could be transported in solution from coal layers within the formation, along Dinaric-orientation fault planes and N–S-oriented fissure zones, following bedding plane slips through the older Cretaceous limestones from north to south (cross-section A–B, Figure 2). Conversely, if sulfur compounds originate from the coal layers within Liburnian Formation beds south of the spring site, transport paths are possibly related to faults following the Dinaric trend and to N–S-oriented fissure zones.
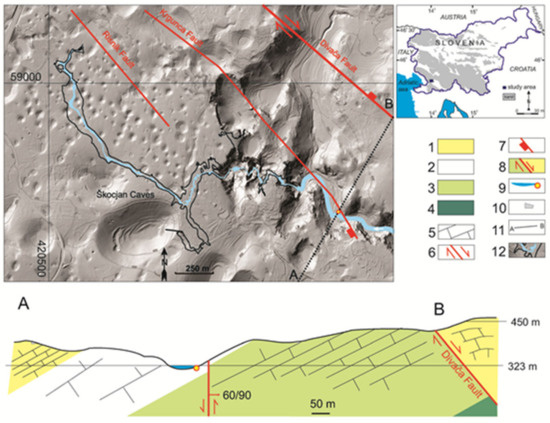
Figure 2.
Morphological situation of the Smrdljivec spring and Škocjanske jame with principal faults and geological cross-section A–B. 1—Liburnian Formation (K-Pc) (d = 50–300 m), 2—Lipica Formation (K24−5) (d = 250–400 m), 3—Sežana Formation (K22−4) (d = 400–500 m), 4—Repen Formation (K21,2) (d < 200 m), 5—Limestone bedding planes in cross-section A–B, 6—Fault with horizontal movement (on ground-plan), 7—Fault with sense of vertical movement (on ground-plan), 8—Fault downthrow and uplift direction in the A–B cross-section, 9—Reka River, with the position of the Smrdljivec spring, 10—road, 11—line of geological cross-section A–B, 12—Škocjanske jame ground plan with the extent of the underground River Reka. LiDAR topography with 50 m contour lines [32].
Figure 2.
Morphological situation of the Smrdljivec spring and Škocjanske jame with principal faults and geological cross-section A–B. 1—Liburnian Formation (K-Pc) (d = 50–300 m), 2—Lipica Formation (K24−5) (d = 250–400 m), 3—Sežana Formation (K22−4) (d = 400–500 m), 4—Repen Formation (K21,2) (d < 200 m), 5—Limestone bedding planes in cross-section A–B, 6—Fault with horizontal movement (on ground-plan), 7—Fault with sense of vertical movement (on ground-plan), 8—Fault downthrow and uplift direction in the A–B cross-section, 9—Reka River, with the position of the Smrdljivec spring, 10—road, 11—line of geological cross-section A–B, 12—Škocjanske jame ground plan with the extent of the underground River Reka. LiDAR topography with 50 m contour lines [32].
2.3. Physicochemical and Toxicity Analyses
On-site measurements of water temperature (T), electrical conductivity (EC), pH, and dissolved oxygen (DO) were performed (WTW Multiline Multi 3620 IDS, WTW Oxi 3310 IDS).
The discharge of the Smrdljivec spring was estimated at 10 L/min (18 July 2017).
Chemical analyses for hardness, alkalinity, Cl, K, NH4, NO2, NO3, SO4, H2S, PO4, Fe, and SiO2 were performed using a YSI 9300 spectrophotometer according to the manufacturer’s instructions (YSI), and for Ca and Mg according to Standard methods [33] (sampled 18 July 2017, 24 July 2017, 28 July 2021). When toxicity of a water sample (sampled 24 August 2017) was assessed using Daphnia magna (SIST EN ISO 6341: 2013), chemical analyses were carried out at the National Laboratory of Health, Environment and Food, Koper (Slovenia): Al (SIST EN ISO 17294-2: 2005), Ca (SIST EN ISO 17294-2: 2005), Cl (ISO 10304-1:2007), Cr (SIST EN ISO 17294-2: 2005), Cu (SIST EN ISO 17294-2: 2005), Fe (SIST EN ISO 17294-2: 2005), H2S (SIST ISO 13358: 2000), K (SIST EN ISO 17294-2: 2005), Mg (SIST EN ISO 17294-2: 2005), Mn (SIST EN ISO 17294-2: 2005), NH4 (ISO 7150-1:1984), Ni (SIST EN ISO 17294-2: 2005), NO2 (SIST EN 26777: 1996), NO3 (HM075-HPLC: 2012), Pb (SIST EN ISO 17294-2: 2005), SiO2 (SM 4500-SiO2-C: 2005), SO4 (ISO 10304-1: 2007), TOC, total organic carbon (ISO 8245: 1999), and Zn (SIST EN ISO 17294-2: 2005).
Due to the Smrdljivec spring’s vicinity to the Reka River, a sampling campaign for stable isotopes of both water bodies was carried out to elucidate whether the spring and the Reka River share contributions from a common water source. Water samples from the river and spring were collected on 18 July 2017, 30 July 2017, 1 August 2017, 9 August 2017, 24 August 2017, and 31 August 2017. For comparison, values of stable isotopes were aligned along the local meteoric water line for Postojna. Monthly composite precipitation samples were collected in Postojna (45°46′31.55″ N, 14°12′47.64″ E, 553 m a.s.l., about 21 km from the Smrdljivec spring) between August 2015 and October 2017 [34].
Stable isotopes of oxygen and hydrogen were measured by Picarro 2130i (Picarro Inc., Santa Clara, CA, USA) water isotope analyzer according to previously described protocols [35,36]. The analyses were performed at the Institute of Hydrology of the Slovak Academy of Sciences (Slovak Republic). The samples were analyzed in triplicate and seven injections were used per sample. The values were expressed in the conventional δ notation (‰) with respect to Vienna Standard Mean Ocean Water. The analytical accuracy was better than 0.2‰ for δ18O and better than 1.0‰ for δ2H.
2.4. Sampling and Microbiological Analyses
Two types of samples were collected at the spring: water and biofilm. Waters from the main orifice of the Smrdljivec spring and from the Reka River were sampled aseptically for comparison (18 July 2017, 24 August 2017, and 28 July 2021). Ready-to-use microbiological media Compact Dry (Nissui Pharmaceutical, Tokyo, Japan) were used to estimate the concentration of heterotrophic aerobic bacteria (Compact Dry TC), coliforms and E. coli (Compact Dry EC), and enterococci (Compact Dry ETC) directly from one milliliter of the water sample. A total of 100 mL of water collected from the Smrdljivec spring on 28 July 2021 was passed through a 0.45 µm pore filter, and the filter (with captured microbial cells on its surface) was placed onto the above-mentioned selective media for E. coli and enterococci. EC, ETC, and one set of TC plates was incubated for 48 h at 37 °C, and another set of TC for 7 days at 20 °C. Concentrations of bacteria were expressed as Colony-Forming Units (CFU) per milliliter. Colonies indicative for E. coli (Enterobacteriaceae) on EC plates (28 July 2021) from the Smrdljivec spring were subjected to identification by Api20E (Biomérieux, Marcy-l’Étoile, France). The ATP content in water samples was estimated using AquaSnap Total testing instruments (Hygiena, Camarillo, CA, USA) and expressed in RLU, Relative Light Units, where 1 RLU equates to 1 fmol of ATP per milliliter.
In addition, water from the main orifice of the Smrdljivec spring was sampled for metagenomics analyses on 18 July 2017 and 24 August 2017. Water (0.5 L) was filtered through 0.22 μm pore-size filters (47 mm in diameter, Millipore, Burlington, MA, USA). Filters were used to isolate total community DNA with a MoBio PowerWater DNA Isolation kit (MoBio, Vancouver, BC, Canada).
Biofilms, white at the vicinity of the main orifice and brown downstream from the main orifice, were sampled on 18 July 2017 for metagenomics and algological analyses, and on 28 July 2021, brown biofilm was sampled downstream from the main orifice, but only for algological analyses. Biofilm DNA was isolated using a MoBio Power Biofilm®DNA Isolation kit (MoBio, Vancouver, BC, Canada)
Field-sampled material for algological analyses was fixed in 4% (v/v) aqueous formalin. Algae and cyanobacteria were identified by examination under a Nikon Eclipse TE 300 phase-contrast microscope (Nikon, Tokyo, Japan) equipped with a digital camera and Nikon software NIS Elements (Nikon, Tokyo, Japan). Cells were identified according to conventional keys [37,38,39,40,41,42,43].
2.5. Amplicon Sequencing, and Bioinformatics Analysis
Amplification of 16S rDNA, library preparation and IonTorrent sequencing were performed at Omega, Ljubljana (Slovenia). The Ion Torrent platform implements simultaneous sequencing of multiple 16S rRNA regions and provides consensus sequences as a result. It was used to identify primarily the most important and abundant members of the community, rather than organisms that are present in extremely low percentages. Each sample was sequenced once. Ion 16S Metagenomics Kit (ThermoFisher Scientific, Waltham, MA, USA) was used for amplification of regions V2-4-8 and V3-6, 7-9 of 16S rRNA in two separate PCR reactions consisting of an initial denaturation at 95 °C for 10 min, 25 cycles of denaturation at 95 °C for 30 s, annealing at 58 °C for 20 s, and polymerization at 72 °C for 20 s, followed by a final extension step at 72 °C for 7 min.
Amplified DNA was purified using Agencourt AMPure XP (Beckman Coulter, Brea, CA, USA). The purified PCR products were quantified on LabChip GX Touch using HT DNA High Sensitivity Reagent Kit (both PerkinElmer, Waltham, MA, USA). Fifty nanogram amplicons were processed to make the DNA library using Ion Plus Fragment Library Kit and barcode adapters Ion Xpress Barcode Adapters Kit (both ThermoFisher Scientific, Waltham, MA, USA). Size distribution and number of processed libraries were evaluated using the LabChip GX instrument (PerkinElmer, Waltham, MA, USA). Each sample was adjusted to 20 picomolar concentration. Equal volumes of all samples were combined and processed in emulsion PCR and enrichment steps with One-Touch 2 and One-Touch ES systems using the Ion PGM Hi- QT View OT2 kit (all ThermoFisher Scientific, Waltham, MA, USA). Finally, the libraries were sequenced in a 316 v2 chip on the Ion Personal Genome Machine (PGM) using the Ion PGM Hi- QT View Sequencing Kit according to the manufacturer’s protocol (all ThermoFisher Scientific Waltham, MA, USA).
Raw reads were analyzed using the cloud service Ion Reporter Software v.5.10 (ThermoFisher Scientific, Waltham, MA, USA), which implements QIIME’s open-source bioinformatics pipeline [44]. Primer sequences were removed from the reads and reads shorter than 150 bp were removed from the dataset. Sequences were aligned against the Curated Greengenes 16S reference library v13.5. The minimum alignment coverage was set to 90% and the read abundance filter set to 10 reads. Genus-level operational taxonomic units (OTUs) were determined at 97% sequence identity and species-level OTUs at 99% sequence identity. Rarefaction curves were determined based on Chao1 metrics. Vegan Community Ecology [45] and Bioconductor [46] packages were used within an R free-software environment [47] to calculate the alpha-diversity measures Chao1, ACE, and Shannon index, to produce heatmaps, and to generate a Venn diagram.
3. Results and Discussion
3.1. Environment of the Spring
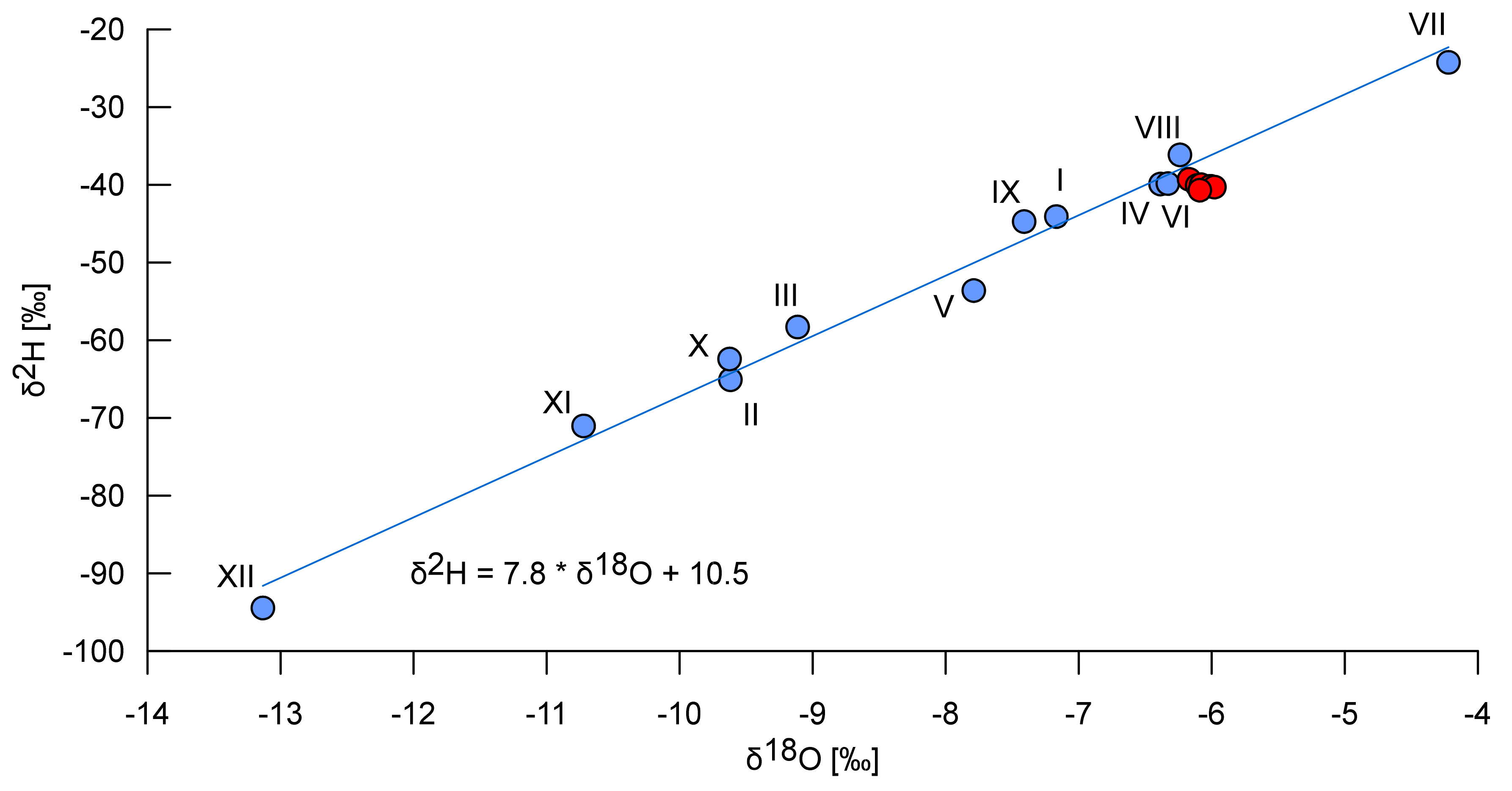
The isotopic composition of water samples from the Smrdljivec spring was the same as that of the Reka River (Table 1). It corresponded to spring-summer precipitation, and exhibited a slight evaporation signal (red symbols in Figure 3). Temperature and EC between the two water bodies were also comparable. These results indicate that, while still underground, the sulfide-rich water reaching the Smrdljivec spring from the coal-bearing beds must mix with a larger quantity of water similar to that in the main Reka River. This results in a reduction of temperature differentials, buffering of the spring water’s expected chemical signature, and a higher pH for the spring water than would be expected. Because of the dilution caused by mixing, the most noticeable differences that survive between the two water bodies are the presence of hydrogen sulfide and the elevated iron content of the Smrdljivec spring water (Table 2).

Table 1.
Results of stable isotope analysis.
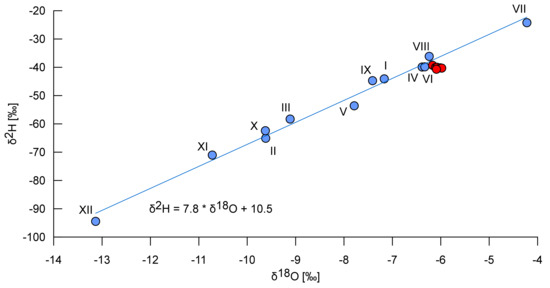
Figure 3.
Isotopic composition of the mean monthly precipitation (the blue symbols) at Postojna calculated as weighted averages from monthly samples collected between September 2015 and November 2017 (the Roman numerals indicate months) with the equation of the local meteoric water line, and isotopic composition of the Reka River’s water in July and August 2017 (red dots).

Table 2.
Physicochemical and microbiological analyses of the Smrdljivec spring and the Reka River.
Less evident differences relate to the presence of silica (3.4–5.1 mg/L for the Smrdljivec spring, 1.8–2.8 mg/L for the Reka River) and phosphate (<0.05–0.16 mg/L for the Smrdljivec spring, <0.05 mg/L for the Reka River). This indicates that at least part of the recharge area of the Smrdljivec spring differs from that of the Reka River. This can be attributed to sulfur compounds, largely in the form of hydrogen sulfide, and heavy metals, which likely originate, and are leached and transported, from stratigraphically higher coal layers. Generally, the concentration of major ions was comparably low in both water bodies (Table 2).
Bioassays using D. magna as a test animal to assess acute toxicity are commonly used in aquatic toxicology [48] and in a wide range of environmental applications such as assessing the toxicity of (un)treated gold-mine effluents [49] or estimating levels of arsenic contamination in underground water [50]. In total, 10% immobilization of D. magna occurred at 100% of the Smrdljivec spring water sample concentration. This value (10% immobilization) coincides with the maximum of the permitted control range, so the spring water was not considered toxic. DO measured during the laboratory test was 8.33 mg/L (20.2 °C). Heavy metals are also mentioned in the legislation relating to drinking water quality assessment and, interestingly, of those tested, the concentrations of Al, Fe, Mn, and Ni exceeded the recommended limits (Table 2). Even if H2S was successfully degassed from the water, the potential use of the spring to provide drinking water would be unlikely (Table 2).
3.2. Microbial Diversity
ATP values indicated abundant microbial biomass in both water bodies and values were higher in the Reka River samples (up to 107 CFU/mL) than in those from the Smrdljivec spring (from 105 to 106 CFU/mL, based on the Hygiena biomass estimation chart). Similar relationships were also observed when using cultivable bacterial indicators, common heterotrophic aerobic bacteria, and coliforms. The presence of cultivable coliforms was detected during the first round of sampling in 2017, and application of membrane filtration to the 2021 water samples revealed the presence of E. coli (4 CFU/100 mL) in the Smrdljivec spring (with % ID = 99.8 for two isolates and % ID = 98.9 for two isolates). Isolates indicative for enterococci were observed in both water bodies (Table 1). However, the members of Escherichia and Enterococcus were already identified from isolated DNA in sulfidic water and white biofilm in samples from July 2017 as a minor fraction with a relative low frequency, 0.01% and 0.02% for water, and 0.08% and 0.08% for white biofilm, respectively (Table S1 in Supplementary Materials).
Whereas the presence of E. coli in sulfidic springs had not been documented before, the resilience of E. coli in open environments has been discussed previously [51,52]. It is known that pathogenic E. coli strains were found to grow and survive in natural environments for long periods, and can become naturalized in some non-host environments [53], growing even at 8–10 °C in robust biofilms [54]. It has previously been shown that naturalized wastewater strains of E. coli are distributed globally, sharing a common genetic background with a genome enriched in stress-response elements, and also sharing virulence and antibiotic resistance genes with enteric pathotypes [53]. In open environments, the growth and survival of E. coli can be restricted by availability of nutrients and energy sources, but studies have also shown that E. coli O157:H7 can survive at low carbon concentrations in sterile freshwaters [55]. The presence of microbial indicators for water quality in the Smrdljivec spring, though at lower levels than in the Reka River (Table 1), supports the likelihood of a connection between the two water bodies.
Biofilms begin to develop close to the Smrdljivec spring orifice and gradually become denser and more abundant downstream. White biofilm predominates closer to the spring, whereas downstream from the orifices they are brownish as well as denser. 16S metagenomics analyses were conducted to describe the taxonomic composition of bacteria found in spatially different biofilms and within the water emerging from the orifice. Unfortunately, DNA from the water sampled from the spring orifice on 18 July 2017 was not of sufficient quality or quantity to provide reliable data for the metagenome at that site. However, the next sampling (24 August 2017) of the water from the same site provided enough sequences for analysis. Further characterization of the Smrdljivec spring in terms of diversity, dynamics, and species succession was limited in this study due to the short time period in which it was available for research (a few weeks per year).
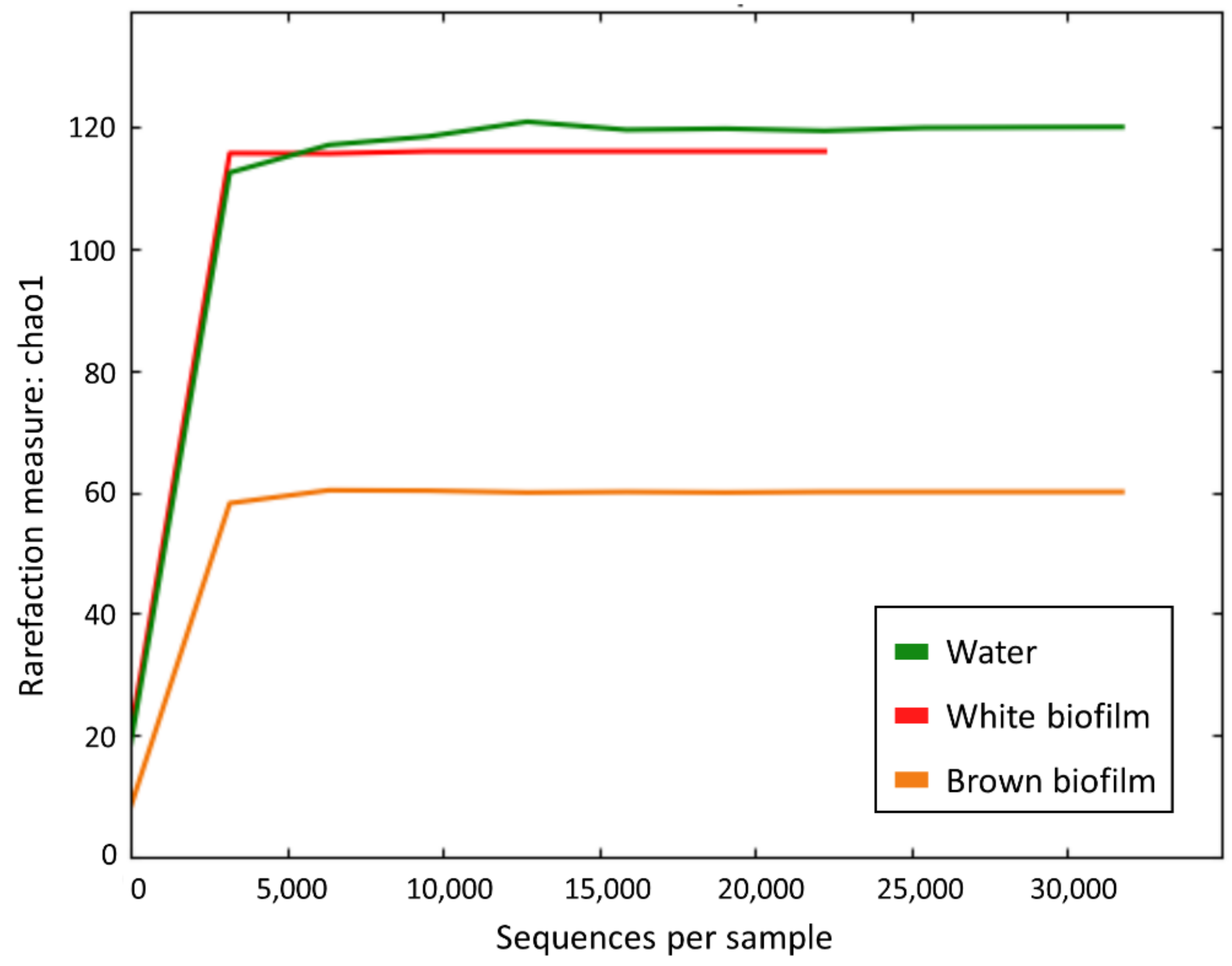
Quality filtration of sequence readings gave a total of 325,005 sequences in the studied samples (Table 3). The number of OTUs was lowest in the sampled brown biofilm (60), higher in the white biofilm (116), and still higher in the water (120), and was in accordance with values of alpha-diversity parameters based upon application of an abundance-based coverage estimator (ACE) and Shannon index (Table 3).

Table 3.
Number of reads, OTUs, richness, and diversity indexes for water, white biofilm and brown biofilm samples from the Smrdljivec spring.
The rarefaction curves (Figure 4) indicated that each species accumulation curve reaches an asymptote, and thus, that it is unlikely that further sampling would yield any additional species.
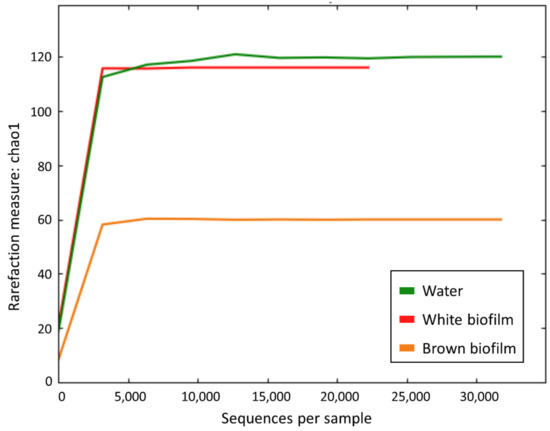
Figure 4.
Rarefaction curves based on Chao1 metrics for water, white biofilm, and brown biofilm samples.
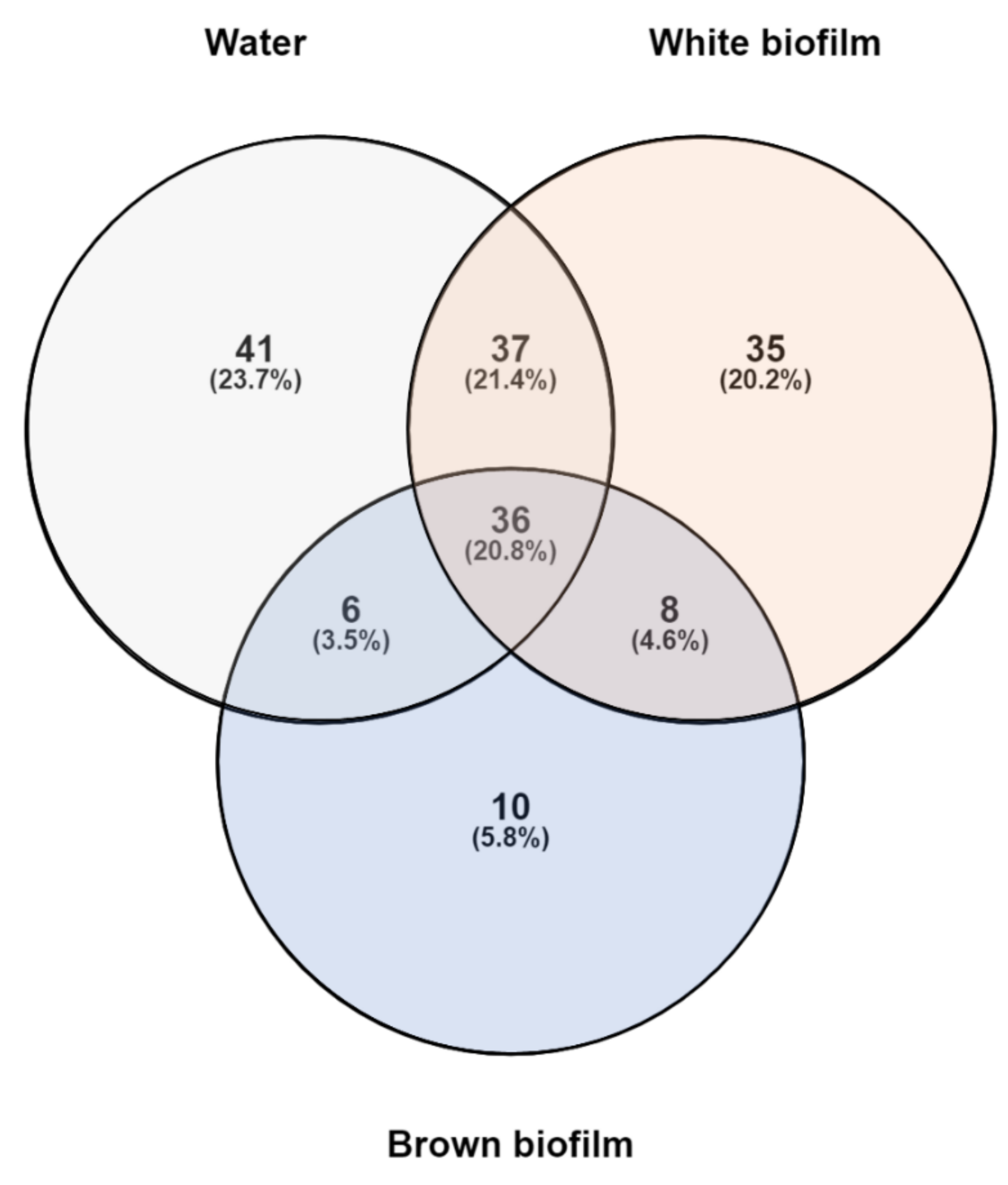
Altogether, the samples included 175 distinct OTUs. Their distribution in individual samples is presented in Figure 5. Thirty-six OTUs (20.8%) were common to all samples. Among them, six had a relative abundance > 0.5% in each of the three samples and can be interpreted as the “core” microbiome. Three of these OTUs were included in the top five taxa in all three field samples and corresponded to Methylomonas (abundance range 4.7–31.7%), Methylotenera (3.9–27.0%), and Methylobacter (3.2–10.1%). The remaining OTUs were Methylophilus (0.5–5.9%), Sulfuricurvum (0.6–14.3%), and Haemophilus (1.7–3.8%). All of these have also been found previously in coal mines: Haemophilus [56]; Methylobacter [57]; Methylomonas [58]; Methylophilus [59]; Methylotenera [60]; Sulfuricurvum [61].
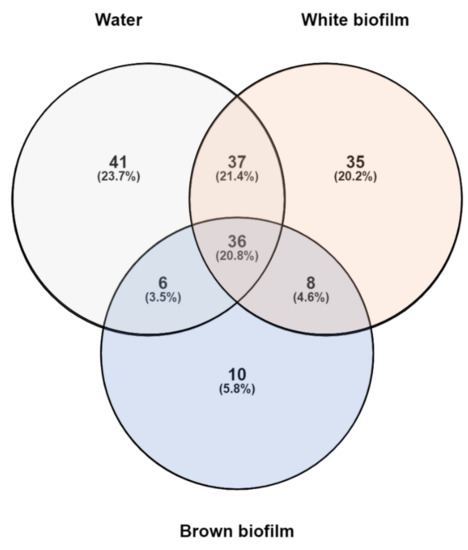
Figure 5.
Prokaryotic OTU distribution in water, white biofilm, and brown biofilm samples from the Smrdljivec spring. The Venn diagram indicates the number of distinct and shared OTUs.
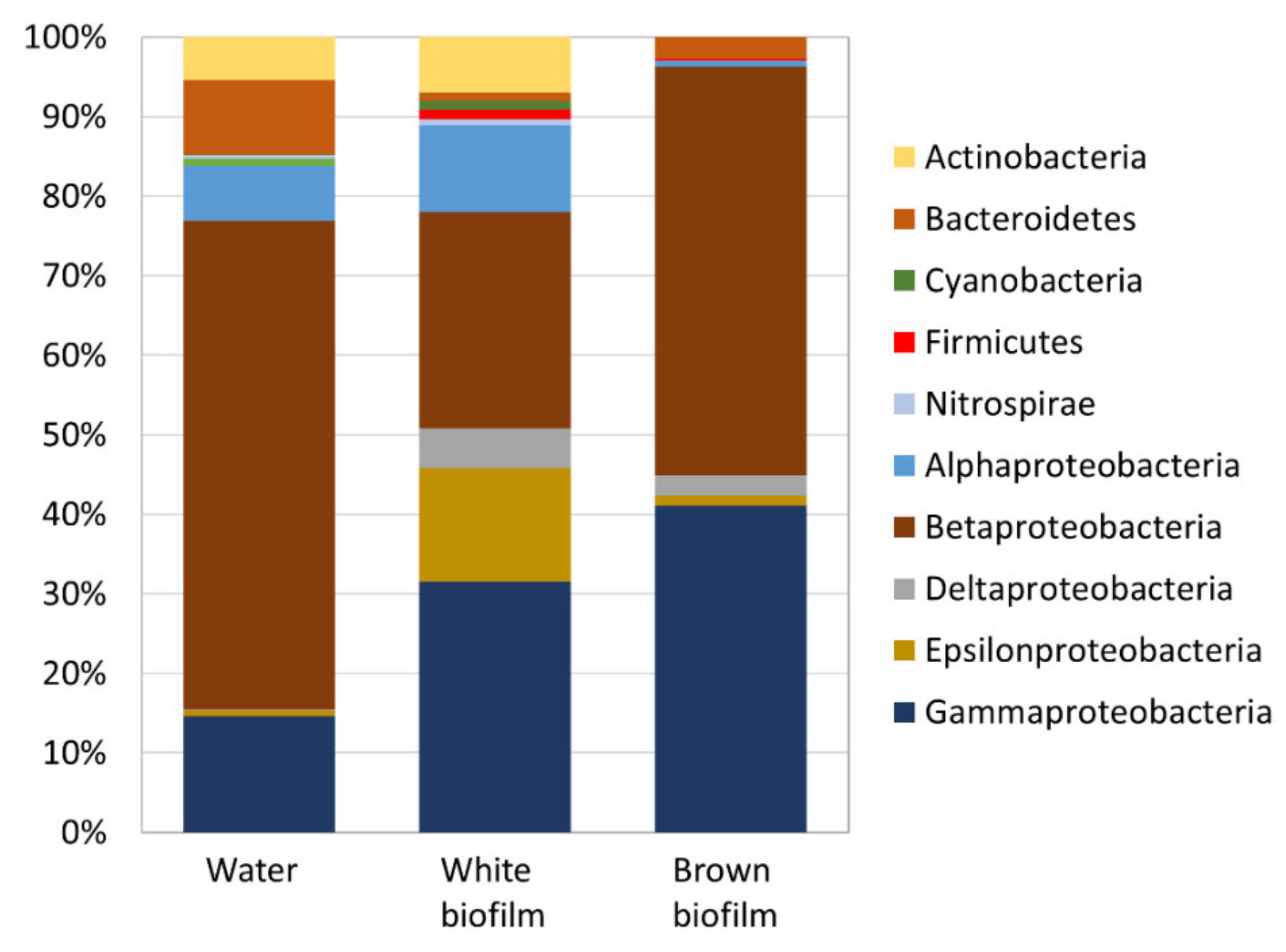
The above OTUs corresponded to seven bacterial phyla out of which Proteobacteria dominated all samples (Figure 6). In the water sample, Bacteroidetes (9.5%) and Actinobacteria (5.4%) were abundant alongside Proteobacteria (84.6%). In the white biofilm sample, Actinobacteria (7.0%) were abundant in addition to Proteobacteria (88.9%). The brown biofilm was the least diverse at phylum level and Proteobacteria (97.0%) were overwhelmingly dominant, followed by Bacteroidetes (2.7%).
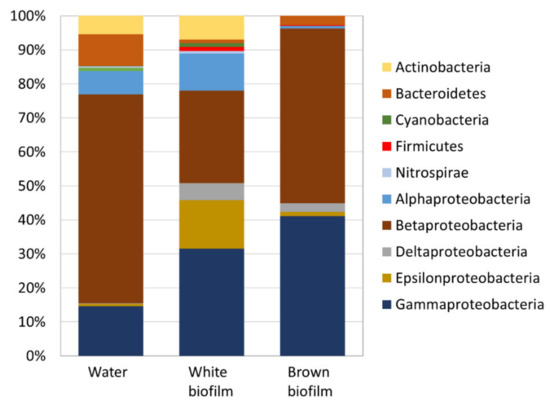
Figure 6.
Relative abundance of bacterial phyla in water and biofilm samples from the Smrdljivec spring. The phylum of Proteobacteria is subdivided into class levels (Alpha-, Beta-, Delta-, Epsilon-, Gamma-).
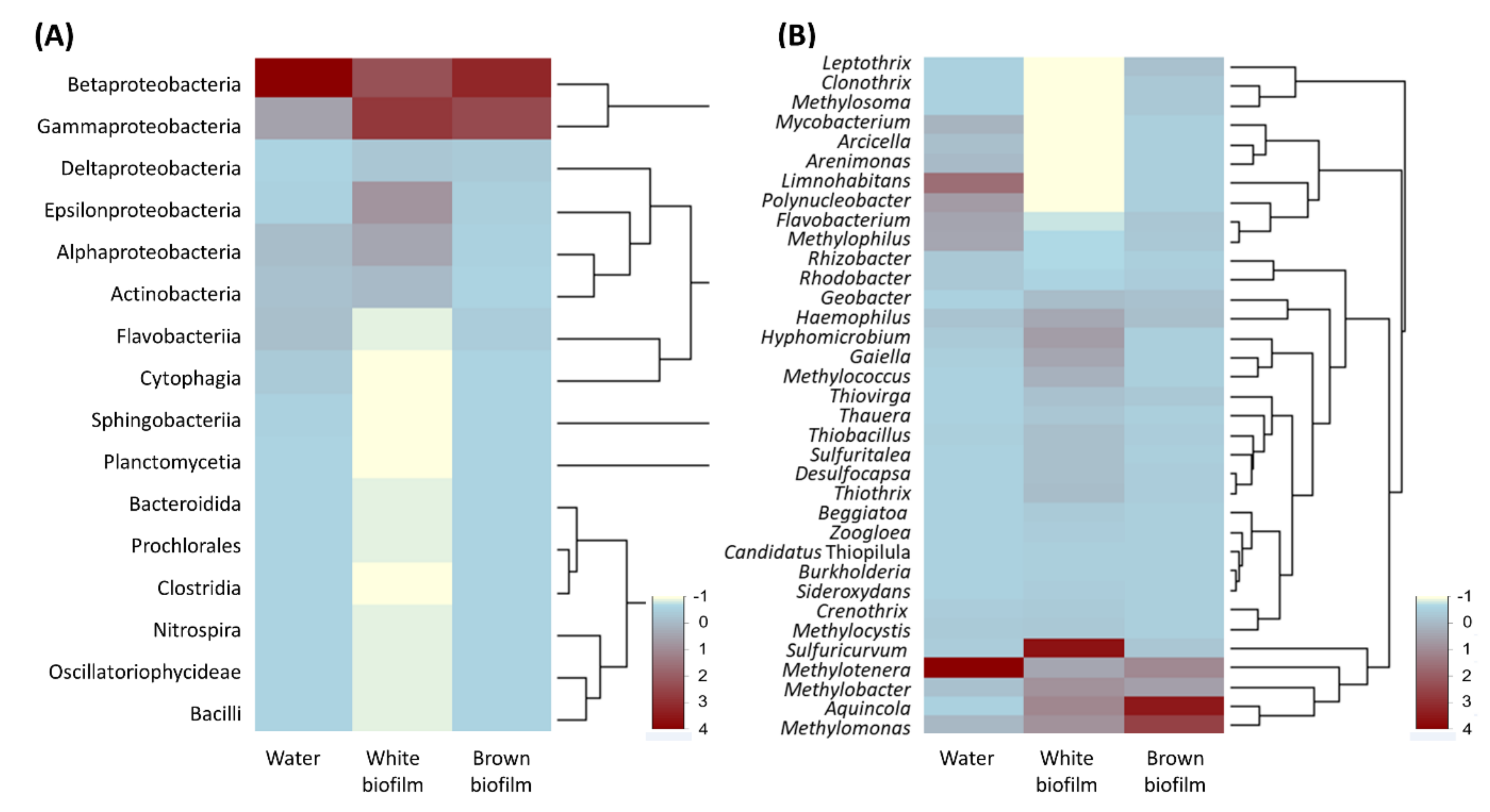
Relative abundances of bacterial classes detected in samples are represented in Figure 7A. Betaproteobacteria (62.1%) dominated the water sample, followed by Gammaproteobacteria (14.8%). Other abundant groups were Alphaproteobacteria (6.9%), Flavobacteriia (6.2%), Actinobacteria (5.5%), and Cytophagia (2.7%). Apart from the above six classes, seven other classes were present in the water sample, but their relative abundance ranged between 0.01 and 0.6%. The white biofilm had comparatively less Betaproteobacteria (27.2%) and a higher abundance of Gammaproteobacteria (27.2%). These were followed in abundance by Epsilonproteobacteria (14.3%), Alphaproteobacteria (10.9%), Actinobacteria (7.0%), and Deltaproteobacteria (5.0%). Apart from those listed above, nine other classes were present in the biofilm, and their relative abundances were between 0.05 and 0.8%. The brown biofilm sample was the least diverse at class level, being composed of members of nine bacterial classes. It was dominated by Betaproteobacteria (51.4%) and Gammaproteobacteria (41.1%), followed in abundance by Deltaproteobacteria (2.5%), Flavobacteriia (2.4%), and Epsilonproteobacteria (1.3%). Four other classes had relative abundances between 0.03 and 0.7%.
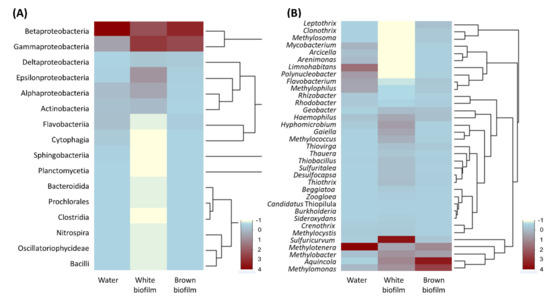
Figure 7.
Heat map analysis of the relative abundance of members of prokaryotic communities in water, white biofilm, and brown biofilm in the Smrdljivec spring, at class (A) and genus (B) level. Listed are all phylotypes detected at class level and phylotypes whose relative abundance at genus level was >0.5% in at least one of the samples.
At genus level (Figure 7B), the water sample included 120 genera, the white biofilm sample contained 116 genera, and the brown biofilm sample showed 60 genera. As mentioned above, Methylomonas, Methylobacter, Methylotenera, Methylococcus, Haemophilus, and Sulfuricurvum were common to all samples.
Members of Methylotenera (29.5%) dominated the water sample, which also contained other methylotrophs, including (as mentioned above) Methylomonas (3.3%), Methylobacter (2.3%), and Methylococcus (0.04%), as well as Methylophilus (5.9%) and Methylocystis (0.1%). The abundance of methylotrophs suggests that cycling of one-carbon compounds is an important process in this habitat. Associations between the above genera have been described previously, for example in eutrophic lake waters, where Methylobacter transfers methane-derived carbon to Methylotenera in the presence of nitrate [62]. The remaining common OTUs, Haemophilus and Sulfuricurvum, were less abundant in the sample (1.8% and 0.6%, respectively). Limnohabitans (13.9%) and Polynucleobacter (7.5%) were the second and the third most abundant genera in the water sample, respectively. Betaproteobacterial Limnohabitans [63] are planktonic bacteria that inhabit the water column of freshwater lakes, reservoirs, and streams, and have also been found in a coal mine environment [64]. At a species level, Limnohabitans phylotypes were affiliated with all four validly described species of the genus L. australis, L. curvus, L. parvus, and L. planctonicus. Polynucleobacter phylotypes (7.5%) are among the most frequently detected phylotypes in freshwater ecosystems [65]. Two major phylotypes of this genus have been detected in stream waters elsewhere—P. cosmopolitanus and P. neccesarius—and these correspond to ultramicrobacteria that are subjected to grazing by flagellate predators [66]. Another abundant freshwater group, Flavobacterium (6.2%), has also been found in a coalmine environment [67].
The most abundant genus-level OTU in the white biofilm sample belonged to Sulfuricurvum, a sulfur oxidizer from the class Epsilonproteobacteria. This genus was represented almost exclusively by a phylotype affiliated with S. kujiense, a sulfur-oxidizing chemolithotroph that was originally isolated from an underground crude-oil storage cavity partially filled with groundwater [68]. Sulfuricurvum phylotypes were previously reported from cave biofilms in the Frasassi sulfidic cave system [69] and the Aquasanta Therme cave system [70] in Italy, in the Lower Kane Cave and Glenwood Hot Springs in the USA [71], and in filaments found in high-Arctic thermal springs in Svalbard [72]. However, Sulfuricurvum did not represent the major phylotype encountered in any of the systems studied. The white biofilm sample contained other sulfur oxidizers, albeit at low abundance, namely betaproteobacterial Sulfuritalea (2.2%) and Thiobacillus (2.3%), and gammaproteobacterial Thiothrix (2.5%), Thiovirga (2.1%), and Beggiatoa (1.5%). In terms of relative abundance, Sulfuricurvum phylotypes were followed by Aquincola (6.2%), Methylobacter (5.4%), Methylomonas (5.4%), and Hyphomicrobium (4.7%).
The composition of major OTUs in the brown biofilm shared some similarities with that of a water sample. Methylomonas (24.8%), Methylotenera (11.9%), and Methylobacter (8.1%) were among the top five taxa. However, the dominant OTU in the brown biofilm was Aquincola (31.5%). At species level, this phylotype corresponded to the only species of this genus—A. tetracarbonis. Not much is known about the physiology of this microbe, apart from its extraordinary ability to utilize tertiary butyl moieties present in gasoline [73]. However, analysis of its genome showed that it includes a large cluster of genes for extracellular polysaccharide biosynthesis, and it has been shown to have the ability to flocculate in wastewater treatment systems [74]. In these terms, it is likely that this microbe contributes to the formation of brown biofilm. Other phylotypes that could be associated with brown biofilm formation were Leptothrix (2.2%) and Clonothrix (1.2%), two filamentous bacteria that oxidize and accumulate either manganese or iron, both of which were found in high concentrations in the stream from the Smrdljivec spring. In addition, the iron reducer Geobacter (2.2%) and iron reducer Ferriphaselus (0.8%) were present in the brown biofilm.
The Smrdljivec spring (sulfidic water and associated biofilms) contains a diverse microbial community. However, more successive sampling campaigns should be conducted at the Smrdljivec spring and the results compared with those of other similar sites whose results were not available or could not be properly classified for comparison at the time of writing (i.e., ecotone, sulfidic spring, coal, and karst) in order to consider this spring as a global diversity hotspot.
Some algae, specifically diatoms, can thrive well and compose biofilms in an environment with sulfide. In a previous study of phototrophs at the outflow of cold sulfidic water from an artesian borehole at Sovra (Slovenia), some algal taxa (Caloneis tenuis, Frustulla vulgaris, Gomphonema sp., Navicula radiosa, Oscillatoria sp., and Tribonema vulgare) that can tolerate and live along an environmental gradient formed by hydrogen sulfide were identified [75]. Microscopic analysis revealed 26 algal and cyanobacterial taxa in both biofilm types in the Smrdljivec spring. In biofilms related to sub-to anoxic habitats, as is the case at the Smrdljivec spring, the associated phototrophs can have a notable impact on the environment (e.g., by oxygenic photosynthesis) and the overall community structure, as well as contributing to eutrophication. In white biofilm sampled at the vicinity of the main spring outlet, diatoms (Bacillariophyceae) prevailed in the community (77.8%), whereas their abundance was lower (45.5%) in brown biofilm downstream from the outlets. As well as diatoms, cyanobacteria were identified in the white biofilm, whereas other phototrophic taxa (Chlorophyceae, Chrysophyceae, Euglenoidea, and Zygnematophyceae) were also present in the brown type. Craticula cuspidate, Navicula sp., Nitzschia sp., Oscillatoria limosa, and Phormidium sp. were identified in both biofilm types (Table 4). The identified phototrophs are commonly found in freshwater ecosystems [76], but some of them have also been reported previously in similar environments, e.g., Achnanthidium in springs with hydrogen sulfide [77] and elevated heavy metals [78], Euglena in cold sulfidic springs [79], Oscillatoria limosa in sulfidic mineral springs [80], and Phormidium in various sulfidic environments [81].

Table 4.
Algae and cyanobacteria associated with biofilms at the Smrdljivec spring during sampling campaigns in the summers of 2017 and 2021.
An important aspect of biofilms is the carbon cycling between different groups of microorganisms. Microalgae can produce and excrete various extracellular polymeric substances (EPS) into their immediate environment during their life cycle [82]. EPS initiate cell adhesion to a substrate, promote the formation of microbial aggregates, immobilize cells in close proximity in a matrix to protect cells from desiccation and toxic substances, play a role in extracellular energy flow and carbon sinks [82], and are, therefore, an important source of nutrients for microorganisms in biofilms [83].
The sulfidic karst spring Žveplenica (NW Slovenia), where the spring output is a mixture of local precipitation and sulfide-rich deep water, is considered a biodiversity-rich ecotone between anoxic sulfidic water and oxygenated water [84,85]. Ecotones may also represent speciation centers [86]. Interestingly, a diverse fauna particularly rich in copepods was found in the Žveplenica spring. This indicated that some invertebrates can successfully tolerate hypoxic to anoxic conditions and certain H2S concentrations [84]. Due to the resilience of these organisms and the partial resemblance of both springs, their presence can also be anticipated at the Smrdljivec spring. Future activities to check the invertebrate community and expand knowledge of the spring’s total diversity should include a wider survey of the area. The Smrdljivec spring represents a practically ideal site to study succession patterns of biota related to formation of a geochemical gradient.
4. Conclusions
Sulfidic springs are interesting natural phenomena as well as biodiversity hotspots. The iron-sulfidic Smrdljivec spring, which becomes visible only during dry periods, is an example of an emerging habitat that drives ecotone formation. It is a dynamic environment with a biomass that can contribute to additional eutrophication both of the Reka River ecosystem and of the karst underground downstream. A microbial community has developed from the dominating phylum of Proteobacteria. At genus level, the water sample was dominated by the methylotroph Methylotenera, the white biofilm was dominated by the sulfur oxidizer Sulphuricurvum, and the brown biofilm was dominated by Aquincola, previously reported as a freshwater organism. The “core” microbiome of the Smrdljivec spring is represented by methylotrophs, specifically Methylobacter, Methylomonas, and Methylotenera. In the developed biofilm, phototrophic microorganisms contributed notably to the overall biomass, and diatoms represented the most important group. E. coli was isolated from the sulfidic water. As an ecotone, the Smrdljivec spring represents only a small area, and its protection could be cost effective, but due to its location, it is likely to be covered by flood sediments in the near future. The spring lies in the core protected area of Regijski park Škocjanske jame, where sulfidic springs were previously unknown. As such, the site will be recommended to the Institute of the Republic of Slovenia for Nature Conservation, to be listed as a valuable natural feature as a part of Slovenia’s natural heritage.
Supplementary Materials
The following is available online at https://www.mdpi.com/article/10.3390/d13120655/s1, Table S1: OUT list.
Author Contributions
J.M. and A.O.-M. conceived and designed the experiments; J.M., A.O.-M., S.S., S.Š., R.C., and T.Z. performed fieldwork and collected samples; J.M., S.S., L.H., and T.E. analyzed the samples in the laboratory and in the field; J.M., A.O.-M., S.Š., T.E., and L.P. analyzed the data, J.M. contributed reagents and materials; all authors wrote the paper. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are openly available in in the NCBI Sequence Read Archive under the BioProject accession number PRJNA769834 corresponding to BioSample accession numbers SAMN22170884 (water sample), SAMN22170885 (white biofilm sample), and SAMN22170886 (brown biofilm sample).
Acknowledgments
The authors acknowledge financial support from the Slovenian Research Agency (research core funding No. P6-0119 and No. P1-0245). The projects “Development of research infrastructure for the international competitiveness of the Slovenian RRI space–RI-SI-LifeWatch and RI-SI-EPOS”, co-financed by the Republic of Slovenia, Ministry of Education, Science and Sport and the European Union from the European Regional Development Fund, enabled some extra environmental analyses. Our thanks also to Samo Šturm, Franjo Drole, and Mitja Prelovšek for providing fieldwork assistance, to Simon Koren, Nataša Toplak and Lilijana Rušnjak for laboratory support, and to David Lowe for assistance with English language editing.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Qing, Y.; Wang, S.; Zhang, B.; Wang, Y. Ultra-high resolution regional climate projections for assessing changes in hydrological extremes and underlying uncertainties. Clim. Dynam. 2020, 55, 2031–2051. [Google Scholar] [CrossRef]
- Schippers, P.; Abarca, E.L.; Verboom, J.; Wamelink, G.W.W.; Vos, C.C.; de Boer, W.F.; Harvey, J.A.; Essens, T.; Grashof-Bokdam, C.J.; WallisDeVries, M.F.; et al. Biodiversity conservation in climate change driven transient communities. Biodivers. Conserv. 2021, 30, 2885–2906. [Google Scholar] [CrossRef]
- Hoffmann, S.; Irl, S.D.H.; Beierkuhnlein, C. Predicted climate shifts within terrestrial protected areas worldwide. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IPCC. Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; Shukla, P., Skea, J., Calvo Buendia, E., Masson-Delmotte, V., Pörtner, H., Roberts, D., Zhai, P., Slade, R., Connors, S., van Diemen, R., et al., Eds.; IPCC: Geneva, Switzerland, 2019; p. 864, In press. [Google Scholar]
- Root, T.L.; Price, J.; Hall, K.R.; Schneider, S.H.; Rosenzweig, C.; Pounds, J. Fingerprints of global warming on wild animals and plants. Nature 2003, 421, 57–60. [Google Scholar] [CrossRef]
- Roman-Palacios, C.; Wiens, J.J. Recent responses to climate change reveal the drivers of species extinction and survival. Proc. Natl. Acad. Sci. USA 2020, 117, 4211–4217. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.G.; Calder, W.J.; Cumming, G.S.; Hughes, T.P.; Jentsch, A.; LaDeau, S.L.; Lenton, T.M.; Shuman, B.N.; Turetsky, M.R.; Ratajczak, Z.; et al. Climate change, ecosystems and abrupt change: Science priorities. Philos. Trans. R. Soc. B. 2020, 375, 20190105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Renaud, P.E.; Wallhead, P.; Kotta, J.; Wtodarska-Kowalczuk, M.; Bellerby, R.G.J.; Ratsep, M.; Slagstad, D.; Kuklinski, P. Arctic sensitivity? Suitable habitat for benthic taxa is surprisingly robust to climate change. Front. Mar. Sci. 2019, 6, 538. [Google Scholar] [CrossRef]
- Mammola, S.; Piano, E.; Cardoso, P.; Vernon, P.; Dominguez-Villar, D.; Culver, D.C.; Pipan, T.; Isaia, M. Climate change going deep: The effects of global climatic alterations on cave ecosystems. Anthr. Rev. 2019, 6, 98–116. [Google Scholar] [CrossRef]
- Kovačič, G.; Petrič, M.; Ravbar, N. Evaluation and quantification of the effects of climate and vegetation cover change on karst water sources: Case studies of two springs in South-Western Slovenia. Water 2020, 12, 3087. [Google Scholar] [CrossRef]
- Retter, A.; Karwautz, C.; Griebler, C. Groundwater microbial communities in times of climate change. Curr. Issues. Mol. Biol. 2021, 41, 509–538. [Google Scholar] [CrossRef]
- Lai, G.G.; Padedda, B.M.; Ector, L.; Wetzel, C.E.; Luglie, A.; Cantonati, M. Mediterranean karst springs: Diatom biodiversity hotspots under the pressure of hydrological fluctuation and nutrient enrichment. Plant. Biosyst. 2020, 154, 673–684. [Google Scholar] [CrossRef]
- Cantonati, M.; Fureder, L.; Gerecke, R.; Juttner, I.; Cox, E.J. Crenic habitats, hotspots for freshwater biodiversity conservation: Toward an understanding of their ecology. Freshw. Sci. 2012, 31, 463–480. [Google Scholar] [CrossRef]
- Headd, B.; Engel, A.S. Biogeographic congruency among bacterial communities from terrestrial sulfidic springs. Front. Microbiol. 2014, 5, 473. [Google Scholar] [CrossRef] [Green Version]
- Mihevc, A. Speleogeneza Divaškega Krasa; Založba ZRC: Ljubljana, Slovenia, 2001; p. 180. [Google Scholar]
- Kranjc, A. Škocjanske jame. In Kras, Pokrajina, Življenje, Ljudje; Kranjc, A., Ed.; Založba ZRC: Ljubljana, Slovenia, 1999; pp. 77–82. [Google Scholar]
- Gams, I. Kras v Sloveniji v Prostoru in Času; Založba ZRC: Ljubljana, Slovenia, 2004; p. 515. [Google Scholar]
- Programme for Protection and Development of the Škocjan Caves Park for the 2019–2023 Period; The Škocjan Caves Park Public Agency: Škocjan, Slovenia, 2018.
- Jurkovšek, B.; Toman, M.; Ogorelec, B.; Šribar, L.; Drobne, K.; Poljak, M.; Šribar, L. Geological Map of the Southern Part of the Trieste-Komen Plateau, Cretaceous and Paleogene Carbonate Rocks 1:50,000; Inštitut za Geologijo, Geotehniko in Geofiziko: Ljubljana, Slovenia, 1996. [Google Scholar]
- Jurkovšek, B. Geološka Karta Krasa 1:100,000; Geološki Zavod Slovenije: Ljubljana, Slovenia, 2013.
- Šebela, S. Structural geology of the Škocjan Caves. Acta Carsol. 2009, 38, 165–177. [Google Scholar] [CrossRef] [Green Version]
- Hamrla, M. O pogojih nastanka premogišč na Krasu. Geologija 1959, 5, 180–264. [Google Scholar]
- Hamrla, M. Light reflectance of some Slovenian coals. Geologija 1987, 28–29, 293–317. [Google Scholar]
- Geocaching.com. Opuščen Rudnik Črnega Premoga Vremski Britof. Available online: https://www.geocaching.com/geocache/GC8B52A_opuscen-rudnik-crnega-premoga-vremski-britof?guid=1d18a9da-4d74-4f70-b803-5adef69c815c (accessed on 10 September 2021).
- Malec, T. Rudnik v Vremski Dolini. Available online: http://www.tatjana-malec.si/default.asp?Id=420 (accessed on 10 September 2021).
- Poljak, M. Structural-Tectonic Map of Slovenia, Based on Basic Geological Map of SFR Yugoslavia 1:100000; Mladinska Knjiga: Ljubljana, Slovenia, 2000. [Google Scholar]
- Placer, L. Poenostavljena strukturno-geološka karta Krasa. Geologija 2015, 58, 89–93. [Google Scholar] [CrossRef]
- Žvab Rožič, P.; Čar, J.; Rožič, B. Geological structure of the Divača area and its influence on speleogenesis and hydrogeology of Kačna jama. Acta Carsol. 2015, 44, 153–168. [Google Scholar] [CrossRef] [Green Version]
- Knez, M. Vpliv Lezik na Razvoj Kraških Jam: Primer Velike Doline, Škocjanske Jame; Založba ZRC: Ljubljana, Slovenia, 1996; p. 186. [Google Scholar]
- Knez, M. The influence of bedding-planes on the development of Karst caves (a study of Velika Dolina at Škocjanske Jame caves, Slovenia). Carbonate. Evaporite. 1998, 13, 121–131. [Google Scholar] [CrossRef]
- Gospodarič, R. Cave sediments and Škocjanske jame speleogenesis. Acta Carsol. 1984, 12, 27–48. [Google Scholar]
- Slovenian Environment Agency (ARSO). Lidar Data. Available online: http://gis.arso.gov.si/evode/profile.aspx?id=atlas_voda_Lidar@Arso&culture=en-US (accessed on 10 September 2021).
- Clesceri, L.S.; Greenberg, A.E.; Eaton, A.D. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Mulec, J.; Oarga-Mulec, A.; Holko, L.; Pašić, L.; Kopitar, A.N.; Eleršek, T.; Mihevc, A. Microbiota entrapped in recently-formed ice: Paradana Ice Cave, Slovenia. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Holko, L. Syringe life and memory effects in isotopic analyses performed by liquid water isotopic analysers—A case study for natural waters from central Europe. Isot. Environ. Health Stud. 2016, 52, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Coplen, T.B.; Wassenaar, L.I. LIMS for Lasers 2015 for achieving long-term accuracy and precision of δ2 H, δ17 O, and δ18 O of waters using laser absorption spectrometry. Rapid Commun. Mass Spectom. 2015, 29, 2122–2130. [Google Scholar] [CrossRef] [PubMed]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota 1. Teil: Chroococcales; Spectrum Akademischer: Heidelberg/Berlin, Germany, 2000; p. 548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota 2. Teil: Oscillatoriales; Elsevier, Spektrum Akademischer: München, Germany, 2005; p. 759. [Google Scholar]
- Komárek, J. Cyanoprokaryota 3. Teil: Heterocytous genera; Springer Spektrum: Berlin, Heidelberg, 2013; p. 1130. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 1. Teil: Naviculaceae; Gustav Fischer: Stuttgart, Germany; New York, NY, USA, 1986; p. 876. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae; Gustav Fischer: Stuttgart, Germany; New York, NY, USA, 1988; p. 596. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 3. Teil: Centrales, Fragilariaceae, Eunotiaceae; Gustav Fischer: Stuttgart, Germany; New York, NY, USA, 1991; p. 576. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 4. Teil: Achnanthaceae, Kritische Ergänzungen zu Achnanthes s.l., Navicula s. str., Gomphonema; Gustav Fischer: Stuttgart, Germany; New York, NY, USA, 1991; p. 468. [Google Scholar]
- Caporaso, J.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.; Costello, E.; Fierer, N.; Pena, A.; Goodrich, J.; Gordon, J.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oksanen, J.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’hara, R.; Simpson, G.; Solymos, P.; Stevens, M.; Wagner, H. Vegan: Community ecology package. R Package Version 2019, 2, 5–6. [Google Scholar]
- Huber, W.; Carey, V.; Gentleman, R.; Anders, S.; Carlson, M.; Carvalho, B.; Bravo, H.; Davis, S.; Gatto, L.; Girke, T.; et al. Orchestrating high-throughput genomic analysis with Bioconductor. Nat. Methods 2015, 12, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Team RC. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- OECD. Test No. 202: Daphnia sp. Acute Immobilisation Test., OECD Guidelines for the Testing of Chemicals, Section 2; OECD Publishing: Paris, France, 2004. [Google Scholar]
- Foudhaili, T.; Jaidi, R.; Neculita, C.M.; Rosa, E.; Triffault-Bouchet, G.; Veilleux, E.; Coudert, L.; Lefebvre, O. Effect of the electrocoagulation process on the toxicity of gold mine effluents: A comparative assessment of Daphnia magna and Daphnia pulex. Sci. Total. Environ. 2020, 708, 134739. [Google Scholar] [CrossRef]
- Medunić, G.; Fiket, Ž.; Ivanić, M. Arsenic contamination status in Europe, Australia, and other parts of the world. In Arsenic in Drinking Water and Food; Srivastava, S., Ed.; Springer: Singapore, 2020; pp. 183–233. [Google Scholar]
- Van Elsas, J.D.; Semenov, A.V.; Costa, R.; Trevors, J.T. Survival of Escherichia coli in the environment: Fundamental and public health aspects. ISME J. 2011, 5, 173–183. [Google Scholar] [CrossRef] [Green Version]
- Jang, J.; Hur, H.G.; Sadowsky, M.J.; Byappanahalli, M.N.; Yan, T.; Ishii, S. Environmental Escherichia coli: Ecology and public health implications—A review. J. Appl. Microbiol. 2017, 123, 570–581. [Google Scholar] [CrossRef] [Green Version]
- Zhi, S.; Banting, G.; Stothard, P.; Ashbolt, N.J.; Checkley, S.; Meyer, K.; Otto, S.; Neumann, N.F. Evidence for the evolution, clonal expansion and global dissemination of water treatment-resistant naturalized strains of Escherichia coli in wastewater. Water Res. 2019, 156, 208–222. [Google Scholar] [CrossRef]
- Ingle, D.J.; Clermont, O.; Skurnik, D.; Denamur, E.; Walk, S.T.; Gordon, D.M. Biofilm formation by and thermal niche and virulence characteristics of Escherichia spp. Appl. Environ. Microb. 2011, 77, 2695–2700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vital, M.; Hammes, F.; Egli, T. Escherichia coli O157 can grow in natural freshwater at low carbon concentrations. Environ. Microbiol. 2008, 10, 2387–2396. [Google Scholar] [CrossRef]
- Guo, H.; Liu, R.; Yu, Z.; Zhang, H.; Yun, J.; Li, Y.; Liu, X.; Pan, J. Pyrosequencing reveals the dominance of methylotrophic methanogenesis in a coal bed methane reservoir associated with Eastern Ordos Basin in China. Int. J. Coal Geol. 2012, 93, 56–61. [Google Scholar] [CrossRef]
- Wolinska, A.; Pytlak, A.; Stepniewska, Z.; Kuzniar, A.; Piasecki, C. Identification of methanotrophic bacteria community in the Jastrzebie-Moszczenica coal mine by fluorescence in situ hybridization and PCR techniques. Pol. J. Environ. Stud. 2013, 22, 275–282. [Google Scholar]
- Bowman, J. Methylomonas. In Bergey’s Manual of Systematics of Archaea and Bacteria; Whitman, W., Rainey, F., Kampfer, P., Trujillo, M., Chun, J., DeVos, P., Hedlund, B., Dedysh, S., Eds.; Wiley Online Library: Hoboken, NJ, USA, 2015; pp. 1–10. [Google Scholar]
- Giri, D.D.; Kumar, A.; Shukla, P.N.; Singh, R.; Singh, P.K.; Pandey, K.D. Salt stress tolerance of methylotrophic bacteria Methylophilus sp. and Methylobacterium sp. isolated from coal mine spoils. Pol. J. Environ. Stud. 2013, 62, 273–280. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Cui, X.; Zhang, Y.; Yu, Z. Bioconversion of coal to methane by microbial communities from soil and from an opencast mine in the Xilingol grassland of northeast China. Biotechnol. Biofuels 2019, 12, 236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadnikov, V.V.; Mardanov, A.V.; Beletsky, A.V.; Antsiferov, D.V.; Kovalyova, A.A.; Karnachuk, O.V.; Ravin, N.V. Sulfur-oxidizing bacteria dominate in the water from a flooded coal mine shaft in Kuzbass. Microbiology 2019, 88, 120–123. [Google Scholar] [CrossRef]
- Van Grinsven, S.; Damste, J.S.S.; Harrison, J.; Polerecky, L.; Villanueva, L. Nitrate promotes the transfer of methane-derived carbon from the methanotroph Methylobacter sp. to the methylotroph Methylotenera sp. in eutrophic lake water. Limnol. Oceanogr. 2021, 66, 878–891. [Google Scholar] [CrossRef]
- Hahn, M.W.; Kasalicky, V.; Jezbera, J.; Brandt, U.; Jezberova, J.; Simek, K. Limnohabitans curvus gen. nov., sp nov., a planktonic bacterium isolated from a freshwater lake. Int. J. Syst. Evol. Micr. 2010, 60, 1358–1365. [Google Scholar] [CrossRef]
- Han, B.; Chen, Y.; Abell, G.; Jiang, H.; Bodrossy, L.; Zhao, J.; Murrell, J.C.; Xing, X.H. Diversity and activity of methanotrophs in alkaline soil from a Chinese coal mine. Fems. Microbiol. Ecol. 2009, 70, 196–207. [Google Scholar] [CrossRef] [Green Version]
- Nuy, J.K.; Hoetzinger, M.; Hahn, M.W.; Beisser, D.; Boenigk, J. Ecological differentiation in two major freshwater bacterial taxa along environmental gradients. Front. Microbiol. 2020, 11, 154. [Google Scholar] [CrossRef]
- Boenigk, J.; Stadler, P.; Wiedlroither, A.; Hahn, M.W. Strain-specific differences in the grazing sensitivities of closely related ultramicrobacteria affiliated with the Polynucleobacter cluster. Appl. Environ. Microb. 2004, 70, 5787–5793. [Google Scholar] [CrossRef] [Green Version]
- Millar, W. Heterotrophic bacterial population in acid coal mine water: Flavobacterium acidurans sp. n. Int. J. Syst. Bacteriol. 1973, 23, 142–150. [Google Scholar] [CrossRef] [Green Version]
- Kodama, Y.; Watanabe, K. Sulfuricurvum kujiense gen. nov., sp nov., a facultatively anaerobic, chemolithoautotrophic, sulfur-oxidizing bacterium isolated from an underground crude-oil storage cavity. Int. J. Syst. Evol. Micr. 2004, 54, 2297–2300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macalady, J.L.; Dattagupta, S.; Schaperdoth, I.; Jones, D.S.; Druschel, G.K.; Eastman, D. Niche differentiation among sulfur-oxidizing bacterial populations in cave waters. ISME J. 2008, 2, 590–601. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, T.L.; Jones, D.S.; Schaperdoth, I.; Macalady, J.L. Metagenomic insights into S(0) precipitation in a terrestrial subsurface lithoautotrophic ecosystem. Front. Microbiol. 2015, 5, 756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossmassler, K.; Hanson, T.E.; Campbell, B.J. Diverse sulfur metabolisms from two subterranean sulfidic spring systems. FEMS Microbiol. Lett. 2016, 363, fnw162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reigstad, L.J.; Jorgensen, S.L.; Lauritzen, S.E.; Schleper, C.; Urich, T. Sulfur-oxidizing chemolithotrophic proteobacteria dominate the microbiota in high Arctic thermal springs on Svalbard. Astrobiology 2011, 11, 665–678. [Google Scholar] [CrossRef]
- Lechner, U.; Brodkorb, D.; Geyer, R.; Hause, G.; Hartig, C.; Auling, G.; Fayolle-Guichard, F.; Piveteau, P.; Muller, R.H.; Rohwerder, T. Aquincola tertiaricarbonis gen. nov., sp. nov., a tertiary butyl moiety-degrading bacterium. Int. J. Syst. Evol. Micr. 2007, 57, 1295–1303. [Google Scholar] [CrossRef] [Green Version]
- Yu, D.; Xia, M.; Zhang, L.; Song, Y.; Duan, Y.; Yuan, T.; Yao, M.; Wu, L.; Tian, C.; Wu, Z.; et al. RpoN (σ54) is required for floc formation but not for extracellular polysaccharide biosynthesis in a floc-forming Aquincola tertiaricarbonis strain. Appl. Environ. Microb. 2017, 83, e00709-17. [Google Scholar] [CrossRef] [Green Version]
- Eleršek, T.; Mulec, J. The algal community at an ecocline of a cold sulphidic spring (Sovra artesian borehole, Slovenia). Environ. Earth. Sci. 2014, 71, 5255–5261. [Google Scholar] [CrossRef]
- AlgaeBase. Available online: https://www.algaebase.org/ (accessed on 9 October 2021).
- Leira, M.; Meijide-Failde, R.; Torres, E. Diatom communities in thermo-mineral springs of Galicia (NW Spain). Diatom. Res. 2017, 32, 29–42. [Google Scholar] [CrossRef]
- Luis, A.T.; Teixeira, P.; Almeida, S.F.P.; Ector, L.; Matos, J.X.; Ferreira da Silva, E.A. Impact of acid mine drainage (AMD) on water quality, stream sediments and periphytic diatom communities in the surrounding streams of Aljustrel mining area (Portugal). Water Air Soil Poll. 2009, 200, 147–167. [Google Scholar] [CrossRef]
- Yanagawa, K.; Haraguchi, A.; Yoshitake, K.; Asamatsu, K.; Harano, M.; Yamashita, K.; Ishibashi, J. Ubiquity of Euglena mutabilis population in three ecologically distinct acidic habitats in Southwestern Japan. Water 2021, 13, 1570. [Google Scholar] [CrossRef]
- Danilova, E.; Barkhutova, D.; Bryanskaya, A.; Namsaraev, Z.; Namsaraev, B. Effect of environmental conditions on the distribution of functional groups of microorganisms in the Khoito-Gol mineral springs (East Sayan). Contemp. Probl. Ecol. 2009, 2, 33–39. [Google Scholar] [CrossRef]
- Lumian, J.E.; Jungblut, A.D.; Dillion, M.L.; Hawes, I.; Doran, P.T.; Mackey, T.J.; Dick, G.J.; Grettenberger, C.L.; Sumner, D.Y. Metabolic capacity of the Antarctic cyanobacterium Phormidium pseudopriestleyi that sustains oxygenic photosynthesis in the presence of hydrogen sulfide. Genes 2021, 12, 426. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Zheng, Y. Overview of microalgal extracellular polymeric substances (EPS) and their applications. Biotechnol. Adv. 2016, 34, 1225–1244. [Google Scholar] [CrossRef] [PubMed]
- Barranguet, C.; Veuger, B.; Van Beusekom, S.A.M.; Marvan, P.; Sinke, J.J.; Admiraal, W. Divergent composition of algal-bacterial biofilms developing under various external factors. Eur. J. Phycol. 2005, 40, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Mulec, J.; Oarga, A.; Schiller, E.; Persoiu, A.; Holko, L.; Sebela, S. Assessment of the physical environment of epigean invertebrates in a unique habitat: The case of a karst sulfidic spring, Slovenia. Ecohydrology 2015, 8, 1326–1334. [Google Scholar] [CrossRef]
- Mulec, J.; Summers Engel, A. Karst spring microbial diversity differs across an oxygen-sulphide ecocline and reveals potential for novel taxa discovery. Acta Carsol. 2019, 48, 129–143. [Google Scholar] [CrossRef] [Green Version]
- Kark, S. Ecotones and ecological gradients. In Ecological Systems: Selected Entries from the Encyclopedia of Sustainability Science and Technology; Leemans, R., Ed.; Springer: New York, NY, USA, 2013; pp. 147–160. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).