Acclimatization Drives Differences in Reef-Building Coral Calcification Rates
Abstract
1. Introduction
2. Materials and Methods
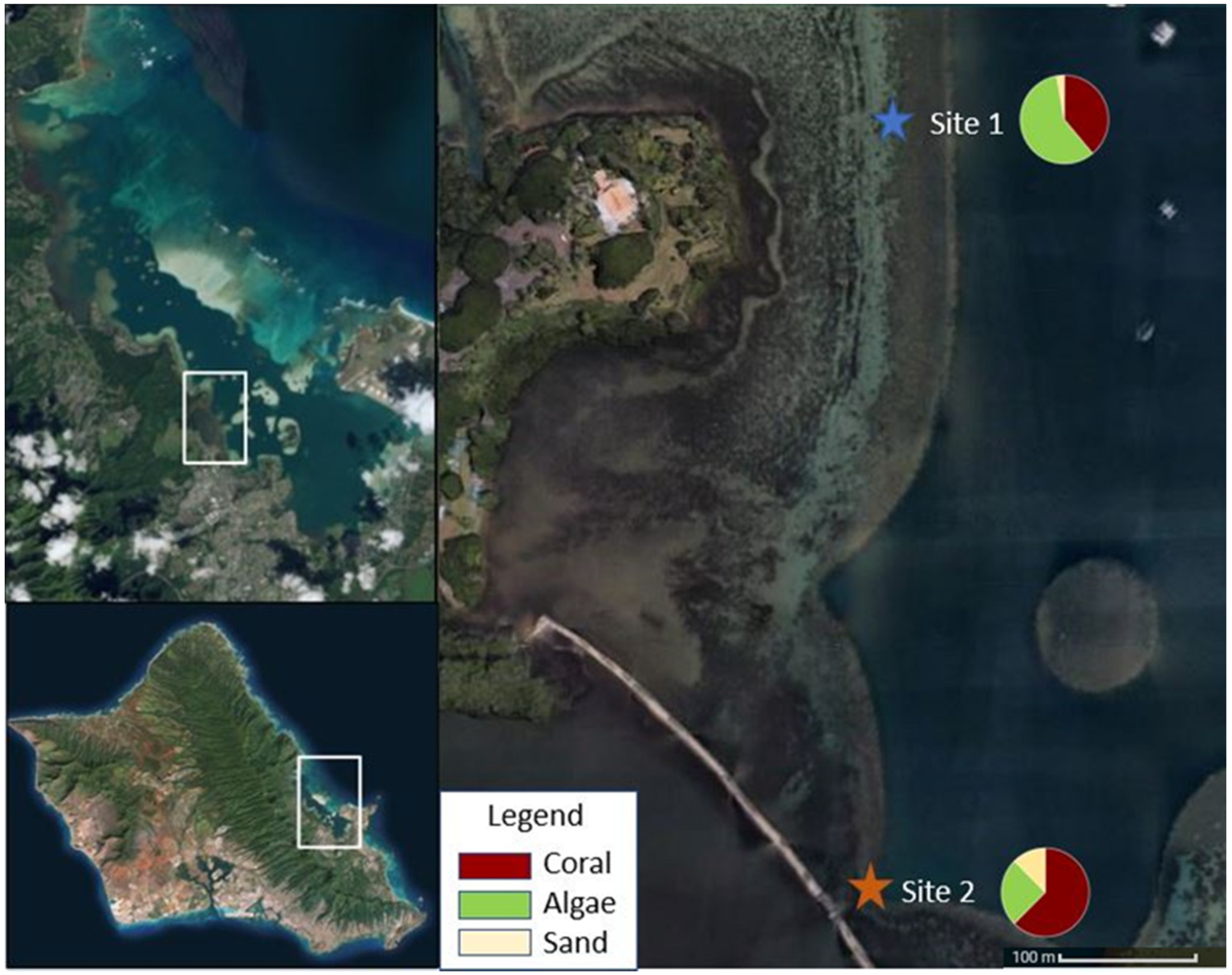
2.1. Study Site
2.2. Environmental Parameters
2.3. Experimental Setup
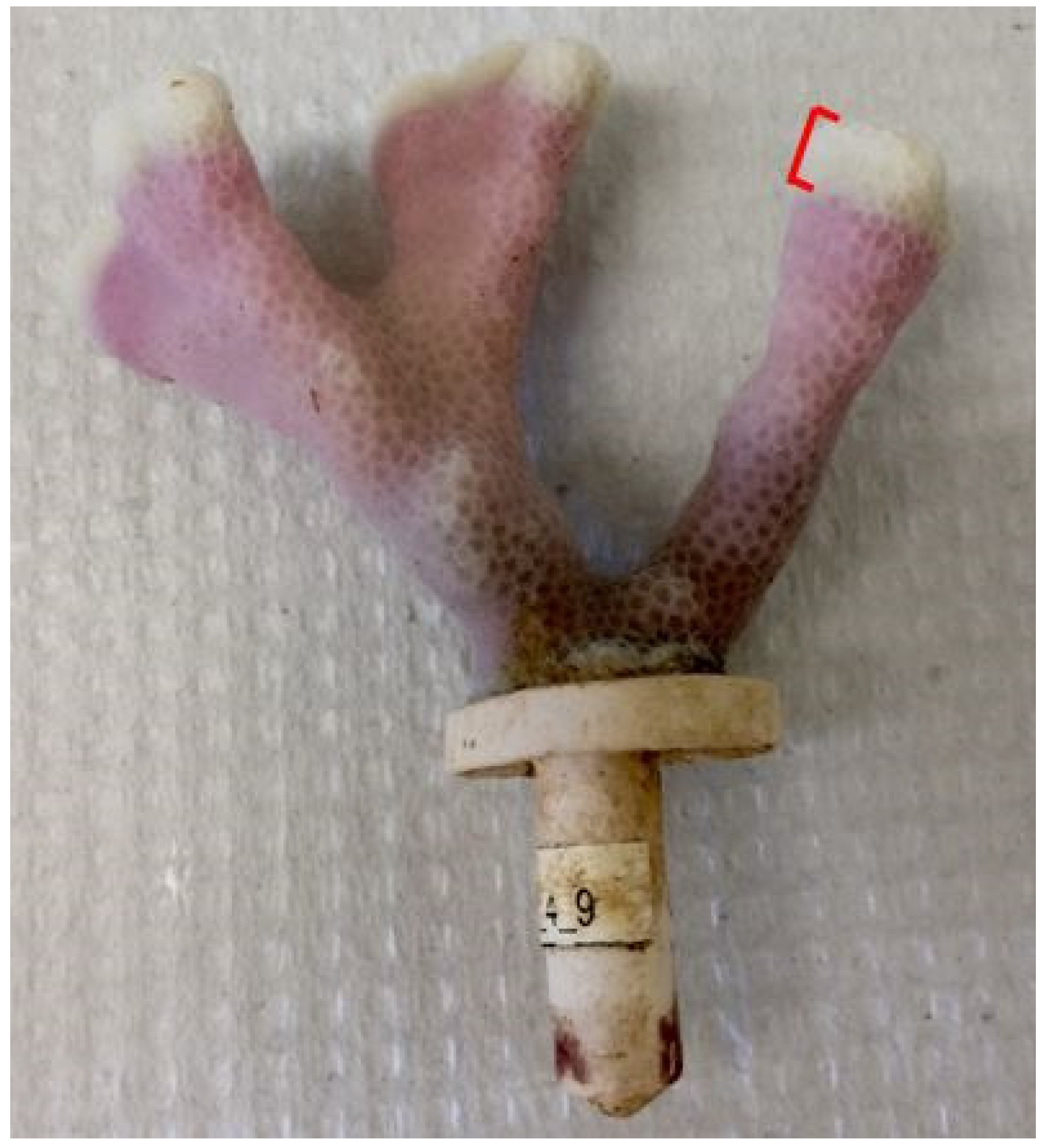
2.3.1. Coral Growth Measurements: Linear Extension
2.3.2. Coral Growth Measurements: Dry Skeletal Weight
2.4. Statistical Analysis
3. Results
3.1. Environmental Parameters
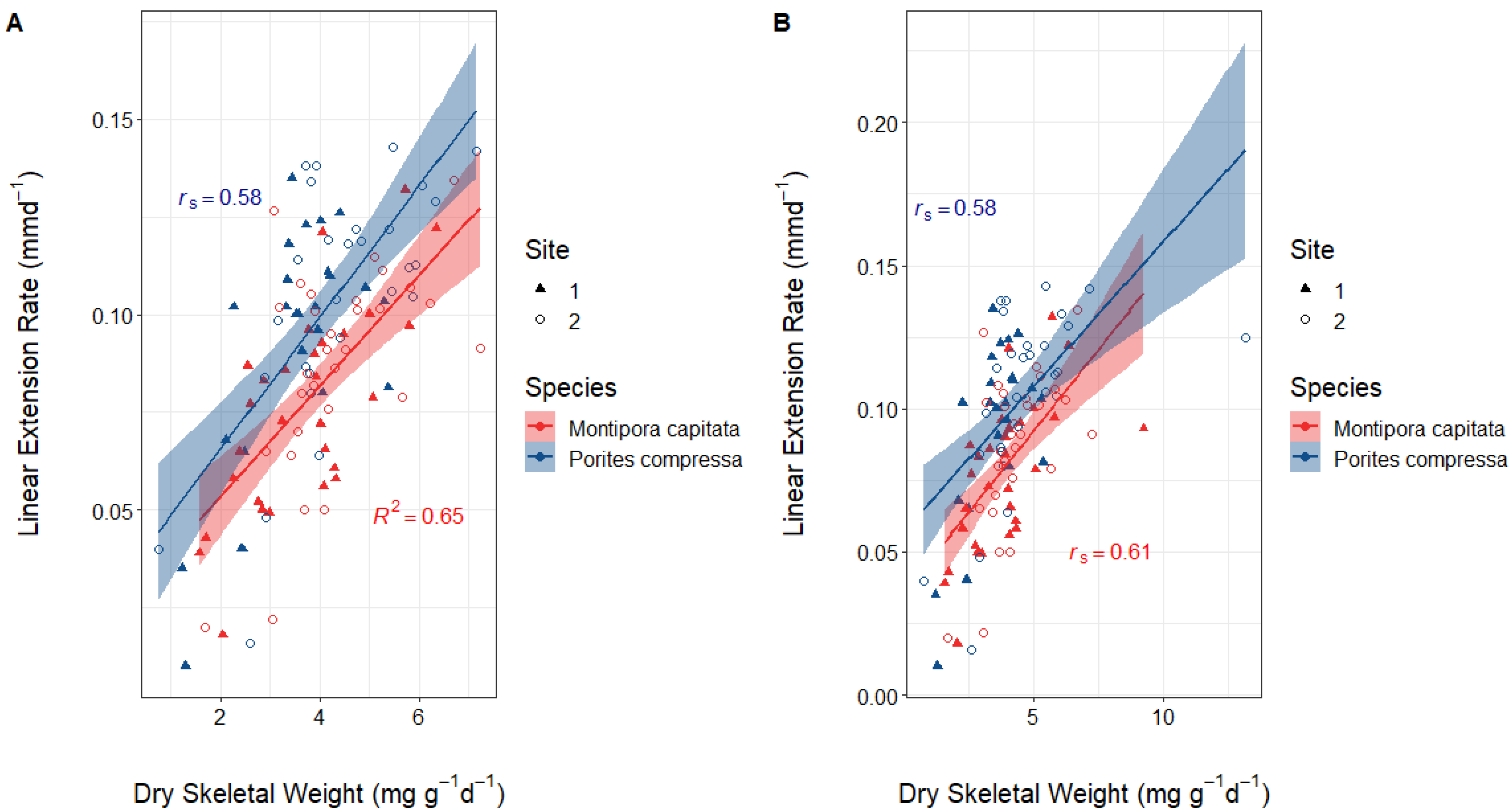
3.2. Correlation between Dry Skeletal Weight and Linear Extension Rates
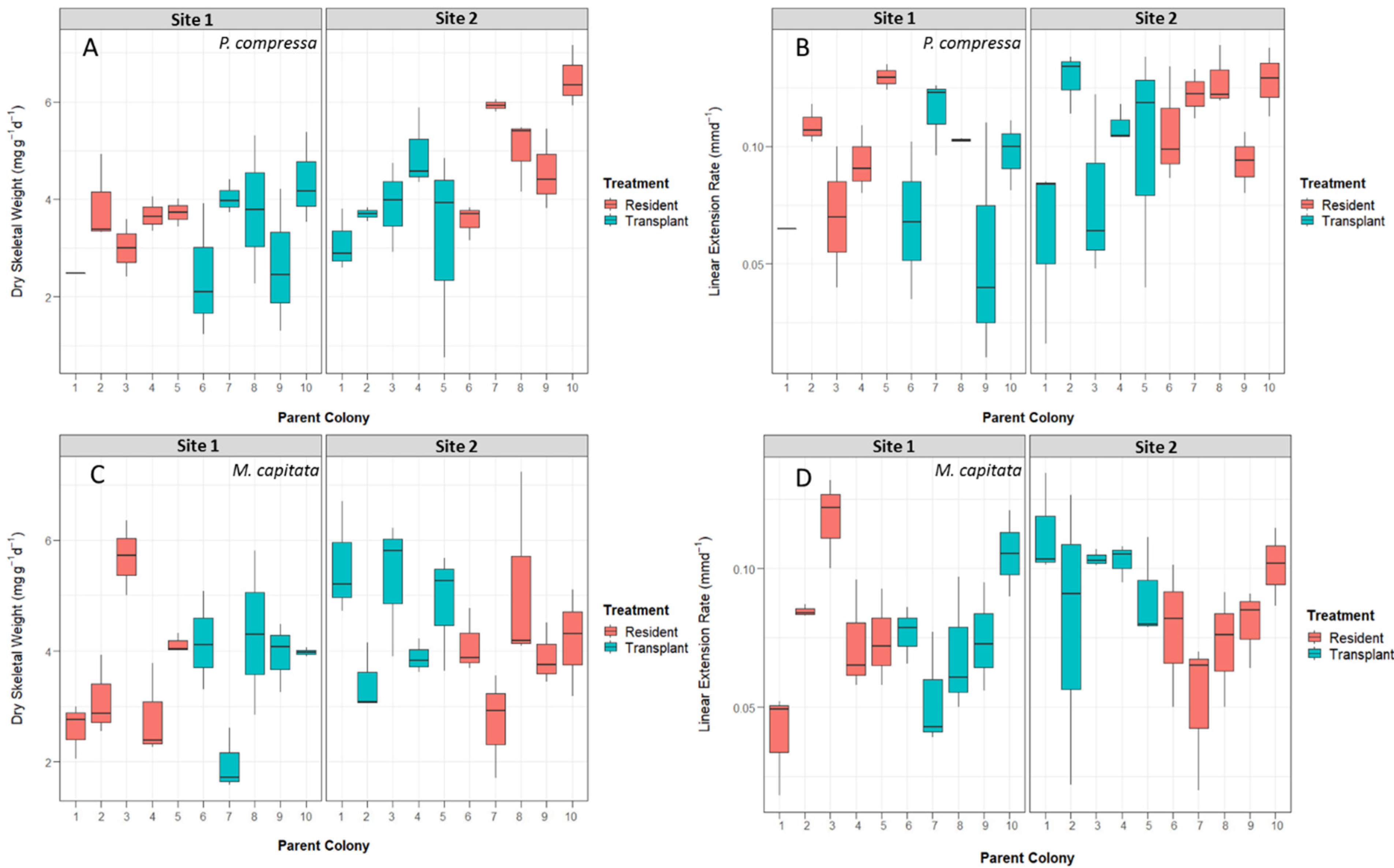
3.3. Coral Growth Measurements
3.4. Reciprocal Transplant Results
3.4.1. Montipora capitata Results
3.4.2. Porites compressa Results
4. Discussion
4.1. Environmental Parameters
4.2. Reciprocal Transplant
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoegh-Guldberg, O.; Mumby, P.J.; Hooten, A.J.; Steneck, R.S.; Greenfield, P.; Gomez, E.; Harvell, C.D.; Sale, P.F.; Edwards, A.J.; Caldeira, K.; et al. Coral reefs under rapid climate change and ocean acidification. Science 2007, 318, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Kerry, J.T.; Álvarez-Noriega, M.; Álvarez-Romero, J.G.; Anderson, K.D.; Baird, A.H.; Babcock, R.C.; Beger, M.; Bellwood, D.R.; Berkelmans, R.; et al. Global warming and recurrent mass bleaching of corals. Nature 2017. [Google Scholar] [CrossRef]
- Bonaldo, R.M.; Hay, M.E. Seaweed-coral interactions: Variance in seaweed allelopathy, coral susceptibility, and potential effects on coral resilience. PLoS ONE 2014. [Google Scholar] [CrossRef]
- Mumby, P.J.; Wolff, N.H.; Bozec, Y.M.; Chollett, I.; Halloran, P. Operationalizing the resilience of coral reefs in an era of climate change. Conserv. Lett. 2014. [Google Scholar] [CrossRef]
- West, J.M.; Salm, R.V. Resistance and Resilience to Coral Bleaching: Implications for Coral Reef Conservation and Management. Conserv. Biol. 2003, 17, 956–967. [Google Scholar] [CrossRef]
- Coles, S.L.; Brown, B.E. Coral bleaching–Capacity for acclimatization and adaptation. Adv. Mar. Biol. 2003, 46, 183–223. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.C.; Starger, C.J.; McClanahan, T.R.; Glynn, P.W. Corals’ adaptive response to climate change. Nature 2004. [Google Scholar] [CrossRef]
- Pandolfi, J.M.; Connolly, S.R.; Marshall, D.J.; Cohen, A.L. Projecting coral reef futures under global warming and ocean acidification. Science 2011, 333, 418–422. [Google Scholar] [CrossRef]
- Putnam, H.M.; Ritson-Williams, R.; Cruz, J.A.; Davidson, J.M.; Gates, R.D. Nurtured by nature: Considering the role of environmental and parental legacies in coral ecological performance. BioRxiv 2018. [Google Scholar] [CrossRef]
- Drury, C.; Manzello, D.; Lirman, D. Genotype and local environment dynamically influence growth, disturbance response and survivorship in the threatened coral, Acropora cervicornis. PLoS ONE 2017. [Google Scholar] [CrossRef]
- Bahr, K.D.; Jokiel, P.L.; Toonen, R.J. The unnatural history of Kāne’ohe bay: Coral reef resilience in the face of centuries of anthropogenic impacts. PeerJ 2015. [Google Scholar] [CrossRef] [PubMed]
- Jury, C.P.; Toonen, R.J. Adaptive responses and local stressor mitigation drive coral resilience in warmer, more acidic oceans. Proc. R. Soc. B Biol. Sci. 2019, 286. [Google Scholar] [CrossRef] [PubMed]
- Pastorok, R.; Bilyard, G. Effects of sewage pollution on coral-reef communities. Mar. Ecol. Prog. Ser. 1985. [Google Scholar] [CrossRef]
- Jokiel, P.L.; Hunter, C.L.; Taguchi, S.; Watarai, L. Ecological impact of a fresh-water “reef kill” in Kaneohe Bay, Oahu, Hawaii. Coral Reefs 1993. [Google Scholar] [CrossRef]
- Bahr, K.D.; Rodgers, K.S.; Jokiel, P.L. Impact of three bleaching events on the reef resiliency of Kāne’ohe Bay, Hawai’i. Front. Mar. Sci. 2017. [Google Scholar] [CrossRef]
- Jokiel, P.L. Illustrated Scientific Guide To Kane’Ohe Bay, Oahu; Hawaii Institute of Marine Biology: Honolulu, Hawaii, 1991. [Google Scholar]
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Barnhill, K.A.; Bahr, K.D. Coral resilience at Malauka’a fringing reef, Kāne’ohe Bay, O’ahu after 18 years. J. Mar. Sci. Eng. 2019, 7, 311. [Google Scholar] [CrossRef]
- Ritson-Williams, R.; Gates, R.D. Coral community resilience to successive years of bleaching in Kāne‘ohe Bay, Hawai‘i. Coral Reefs 2020, 39, 757–769. [Google Scholar] [CrossRef]
- De Villemereuil, P.; Gaggiotti, O.E.; Mouterde, M.; Till-Bottraud, I. Common garden experiments in the genomic era: New perspectives and opportunities. Heredity 2016, 116, 249–254. [Google Scholar] [CrossRef]
- Smith, J.E.; Brainard, R.; Carter, A.; Grillo, S.; Edwards, C.; Harris, J.; Lewis, L.; Obura, D.; Rohwer, F.; Sala, E.; et al. Re-evaluating the health of coral reef communities: Baselines and evidence for human impacts across the central pacific. Proc. R. Soc. B Biol. Sci. 2016. [Google Scholar] [CrossRef]
- Gattuso, J.P.; Payri, C.E.; Pichon, M.; Delesalle, B.; Frankignoulle, M. Primary production, calcification, and air-sea CO2 fluxes of a macroalgal-dominated coral reef community (Moorea, French Polynesia). J. Phycol. 1997, 33, 729–738. [Google Scholar] [CrossRef]
- Carilli, J.E.; Norris, R.D.; Black, B.A.; Walsh, S.M.; McField, M. Local stressors reduce coral resilience to bleaching. PLoS ONE 2009. [Google Scholar] [CrossRef]
- Gattuso, J.P.; Frankignoulle, M.; Wollast, R. Carbon and carbonate metabolism in coastal aquatic ecosystems. Annu. Rev. Ecol. Syst. 1998. [Google Scholar] [CrossRef]
- Jury, C.P.; Delano, M.N.; Toonen, R.J. High heritability of coral calcification rates and evolutionary potential under ocean acidification. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Rodgers, K.S.; Jokiel, P.L.; Brown, E.K.; Hau, S.; Sparks, R. Over a Decade of Change in Spatial and Temporal Dynamics of Hawaiian Coral Reef Communities. Pac. Sci. 2015. [Google Scholar] [CrossRef]
- Gardner, W.D. Sediment trap dynamics and calibration: A laboratory evaluation. J. Mar. Res. 1980, 38, 17–39. [Google Scholar]
- Storlazzi, C.D.; Field, M.E.; Bothner, M.H. The use (and misuse) of sediment traps in coral reef environments: Theory, observations, and suggested protocols. Coral Reefs 2011, 30, 23–38. [Google Scholar] [CrossRef]
- Dickson, A.G.; Sabine, C.L.; Christian, J.R. Guide to Best Practices for Ocean CO2 Measurements; North Pacific Marine Science Organization: Sidney, British Columbia, 2007. [Google Scholar]
- Pierrot, D.; Lewis, E.; Wallace, D.W.R. MS Excel program developed for CO2 system calculations. In ORNL/CDIAC-105a. Carbon Dioxide Inf. Anal. Cent; Oak Ridge National Laboratory US Department Energy: Oak Ridge, TN, USA, 2006. [Google Scholar]
- Barnhill, K.A.; Brown, C.; McGowan, A.E.; Bahr, K.D. Reciprocal transplant coral growth rates for Porites compressa and Montipora capitata in Kāne’ohe Bay, O’ahu, Hawai’i. Pangaea 2020. [Google Scholar] [CrossRef]
- Barnes, D.J. Coral skeletons: An explanation of their growth and structure. Science 1970, 170, 1305–1308. [Google Scholar] [CrossRef]
- Rodgers, K.S.; Cox, E.F. The effects of trampling on Hawaiian corals along a gradient of human use. Biol. Conserv. 2003. [Google Scholar] [CrossRef]
- Lamberts, A.E. Alizarin Deposition by Corals; University of Hawai’i: Honolulu, HI, USA, 1973. [Google Scholar]
- Jokiel, P.L.; Maragos, J.; Franzisket, L. Coral growth: Buoyant weight technique. Coral Reefs Res. Methods 1978, 1, 529–541. [Google Scholar]
- Rstudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2019. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Hebbali, A. olsrr: Tools for Building OLS Regression Models. R Package Version 0.5.1. Available online: https://olsrr.rsquaredacademy.com/ (accessed on 1 September 2020).
- Luke, S.G. Evaluating significance in linear mixed-effects models in R. Behav. Res. Methods 2017, 49, 1494–1502. [Google Scholar] [CrossRef]
- Kassambara, A. ggpubr: “ggplot2” Based Publication Ready Plots. R Package Version 0.2.4. 2019. Available online: https://rpkgs.datanovia.com/ggpubr/ (accessed on 10 July 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 9780387981406. [Google Scholar]
- Barshis, D.J.; Ladner, J.T.; Oliver, T.A.; Seneca, F.O.; Traylor-Knowles, N.; Palumbi, S.R. Genomic basis for coral resilience to climate change. Proc. Natl. Acad. Sci. USA 2013. [Google Scholar] [CrossRef]
- Jokiel, P.L.; Coles, S.L. Effects of Heated Effluent on Hermatypic Corals at Kahe Point, Oahu. Pacific Sci. 1974, 28, 1–18. [Google Scholar]
- Jokiel, P.L.; Coles, S.L. Effects of temperature on the mortality and growth of Hawaiian reef corals. Mar. Biol. 1977. [Google Scholar] [CrossRef]
- Safaie, A.; Silbiger, N.J.; McClanahan, T.R.; Pawlak, G.; Barshis, D.J.; Hench, J.L.; Rogers, J.S.; Williams, G.J.; Davis, K.A. High frequency temperature variability reduces the risk of coral bleaching. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Coles, S.L.; Jokiel, P.L. Effects of Salinity on Coral Reefs. In Pollution in Tropical Aquatic Systems; CRC Press: Boca Raton, FL, USA, 1992; pp. 147–166. [Google Scholar]
- Möhlenkamp, P.; Beebe, C.K.; McManus, M.A.; Kawelo, A.H.; Kotubetey, K.; Lopez-Guzman, M.; Nelson, C.E.; Alegado, R.A. Ku Hou Kuapa: Cultural restoration improves water budget and water quality dynamics in He’eia Fishpond. Sustainable 2018, 11, 161. [Google Scholar] [CrossRef]
- Kleypas, J.A.; McManu, J.W.; Mene, L.A.B. Environmental limits to coral reef development: Where do we draw the line? Am. Zool. 1999. [Google Scholar] [CrossRef]
- Guinotte, J.M.; Buddemeier, R.W.; Kleypas, J.A. Future coral reef habitat marginality: Temporal and spatial effects of climate change in the Pacific basin. Coral Reefs 2003, 22, 551–558. [Google Scholar] [CrossRef]
- Uthicke, S.; Furnas, M.; Lønborg, C. Coral reefs on the edge? Carbon chemistry on inshore reefs of the great barrier reef. PLoS ONE 2014. [Google Scholar] [CrossRef]
- Enochs, I.C.; Formel, N.; Manzello, D.; Morris, J.; Mayfield, A.B.; Boyd, A.; Kolodziej, G.; Adams, G.; Hendee, J. Coral persistence despite extreme periodic pH fluctuations at a volcanically acidified Caribbean reef. Coral Reefs 2020, 39, 523–528. [Google Scholar] [CrossRef]
- Gagliano, M.; McCormick, M.I.; Moore, J.A.; Depczynski, M. The basics of acidification: Baseline variability of pH on Australian coral reefs. Mar. Biol. 2010. [Google Scholar] [CrossRef]
- Gray, S.E.C.; DeGrandpre, M.D.; Langdon, C.; Corredor, J.E. Short-term and seasonal pH, p CO 2 and saturation state variability in a coral-reef ecosystem. Global Biogeochem. Cycles 2012. [Google Scholar] [CrossRef]
- Rogers, C. Responses of coral reefs and reef organisms to sedimentation. Mar. Ecol. Prog. Ser. 1990. [Google Scholar] [CrossRef]
- Fabricius, K.E.; De’ath, G.; Humphrey, C.; Zagorskis, I.; Schaffelke, B. Intra-annual variation in turbidity in response to terrestrial runoff on near-shore coral reefs of the Great Barrier Reef. Estuar. Coast. Shelf Sci. 2013. [Google Scholar] [CrossRef]
- Kinsey, D.W.; Davies, P.J. Effects of elevated nitrogen and phosphorus on coral reef growth. Limnol. Oceanogr. 1979, 24, 935–940. [Google Scholar] [CrossRef]
- Fabricius, K.E. Effects of terrestrial runoff on the ecology of corals and coral reefs: Review and synthesis. Mar. Pollut. Bull. 2005. [Google Scholar] [CrossRef]
- Allemand, D.; Tambutté, É.; Zoccola, D.; Tambutté, S. Coral calcification, cells to reefs. In Coral Reefs: An Ecosystem in Transition; Springer: Berlin/Heidelberg, Germany, 2011; ISBN 9789400701137. [Google Scholar]
- Gladfeiter, E.H. Skeletal development in Acropora cervicornis: I. Patterns of calcium carbonate accretion in the axial corallite. Coral Reefs 1982. [Google Scholar] [CrossRef]
- Jokiel, P.L.; Jury, C.P.; Kuffner, I.B. Coral calcification and ocean acidification. In Coral Reefs at the Crossroads; Springer: Berlin/Heidelberg, Germany, 2016; pp. 7–45. [Google Scholar]
- Dodge, R.E.; Brass, G.W. Skeletal extension, density and calcification of the reef coral, Montastrea annularis: St. Croix, US Virgin Islands. Bull. Mar. Sci. 1984, 34, 288–307. [Google Scholar]
- Cox, E.F. The effects of a selective corallivore on growth rates and competition for space between two species of Hawaiian corals. J. Exp. Mar. Bio. Ecol. 1986. [Google Scholar] [CrossRef]
- Grottoli, A.G. Variability of stable isotopes and maximum linear extension in reef-coral skeletons at Kaneohe Bay, Hawaii. Mar. Biol. 1999. [Google Scholar] [CrossRef]
- Bahr, K.D.; Jokiel, P.L.; Rodgers, K.S. Relative sensitivity of five Hawaiian coral species to high temperature under high-pCO2 conditions. Coral Reefs 2016. [Google Scholar] [CrossRef]
- Carricart-Ganivet, J.P.; Cabanillas-Terán, N.; Cruz-Ortega, I.; Blanchon, P. Sensitivity of calcification to thermal stress varies among genera of massive reef-building corals. PLoS ONE 2012. [Google Scholar] [CrossRef] [PubMed]
- Coles, S.L.; Bahr, K.D.; Rodgers, K.S.; May, S.L.; McGowan, A.E.; Tsang, A.; Bumgarner, J.; Han, J.H. Evidence of acclimatization or adaptation in Hawaiian corals to higher ocean temperatures. PeerJ 2018. [Google Scholar] [CrossRef]
| Variable | Site 1 | Site 2 |
|---|---|---|
| * Temperature (°C) | 27.72 ± 0.94 | 27.48 ± 0.96 |
| * Salinity (ppt) | 32.91 ± 2.20 | 29.68 ± 2.86 |
| Turbidity (NTU) | 0.38 ± 0.17 | 0.97 ± 0.39 |
| Sediment Accumulation (mg cm−2 hr−1) | 0.34 ± 0.09 | 0.66 ± 0.29 |
| pH (pHT) | 8.28 ± 0.21 | 8.26 ± 0.10 |
| * ΩAragonite | 2.13 ± 0.50 | 1.73 ± 0.56 |
| Dry Skeletal Weight (mg g−1 d−1) | Linear Extension (mm d−1) | |||
|---|---|---|---|---|
| P. compressa | M. capitata | P. compressa | M. capitata | |
| Site 1 Resident | 3.51 ± 0.67 | 3.67 ± 1.25 | 0.097 ± 0.026 | 0.078 ± 0.028 |
| Site 1 Transplant | 3.43 ± 1.29 | 4.03 ± 1.78 | 0.086 ± 0.034 | 0.074 ± 0.022 |
| Site 2 Resident | 5.59 ± 2.31 | 4.02 ± 1.17 | 0.115 ± 0.019 | 0.077 ± 0.024 |
| Site 2 Transplant | 3.76 ± 1.15 | 4.61 ± 1.11 | 0.095 ± 0.037 | 0.098 ± 0.025 |
| Fixed Effect | Estimate | Standard Error | t Value |
| Intercept | 1.83 | 0.92 | 1.99 |
| * Destination Site | 1.62 | 0.57 | 2.85 |
| Treatment Transplant | 1.29 | 1.26 | 1.02 |
| Destination Site: Treatment Transplant | −1.30 | 0.79 | −1.66 |
| Random Effects | Variance | Standard Deviation | |
| Original Site: Parent Colony (Intercept) | 0.392 | 0.63 | |
| Residual | 0.989 | 0.99 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barnhill, K.A.; Jogee, N.; Brown, C.; McGowan, A.; Rodgers, K.; Bryceson, I.; Bahr, K. Acclimatization Drives Differences in Reef-Building Coral Calcification Rates. Diversity 2020, 12, 347. https://doi.org/10.3390/d12090347
Barnhill KA, Jogee N, Brown C, McGowan A, Rodgers K, Bryceson I, Bahr K. Acclimatization Drives Differences in Reef-Building Coral Calcification Rates. Diversity. 2020; 12(9):347. https://doi.org/10.3390/d12090347
Chicago/Turabian StyleBarnhill, Kelsey Archer, Nadia Jogee, Colleen Brown, Ashley McGowan, Ku’ulei Rodgers, Ian Bryceson, and Keisha Bahr. 2020. "Acclimatization Drives Differences in Reef-Building Coral Calcification Rates" Diversity 12, no. 9: 347. https://doi.org/10.3390/d12090347
APA StyleBarnhill, K. A., Jogee, N., Brown, C., McGowan, A., Rodgers, K., Bryceson, I., & Bahr, K. (2020). Acclimatization Drives Differences in Reef-Building Coral Calcification Rates. Diversity, 12(9), 347. https://doi.org/10.3390/d12090347







