Revisiting the Evolution of Arboreal Life in Oribatid Mites
Abstract
1. Introduction
2. Material and Methods
2.1. Data Acquisition
2.2. Analysis
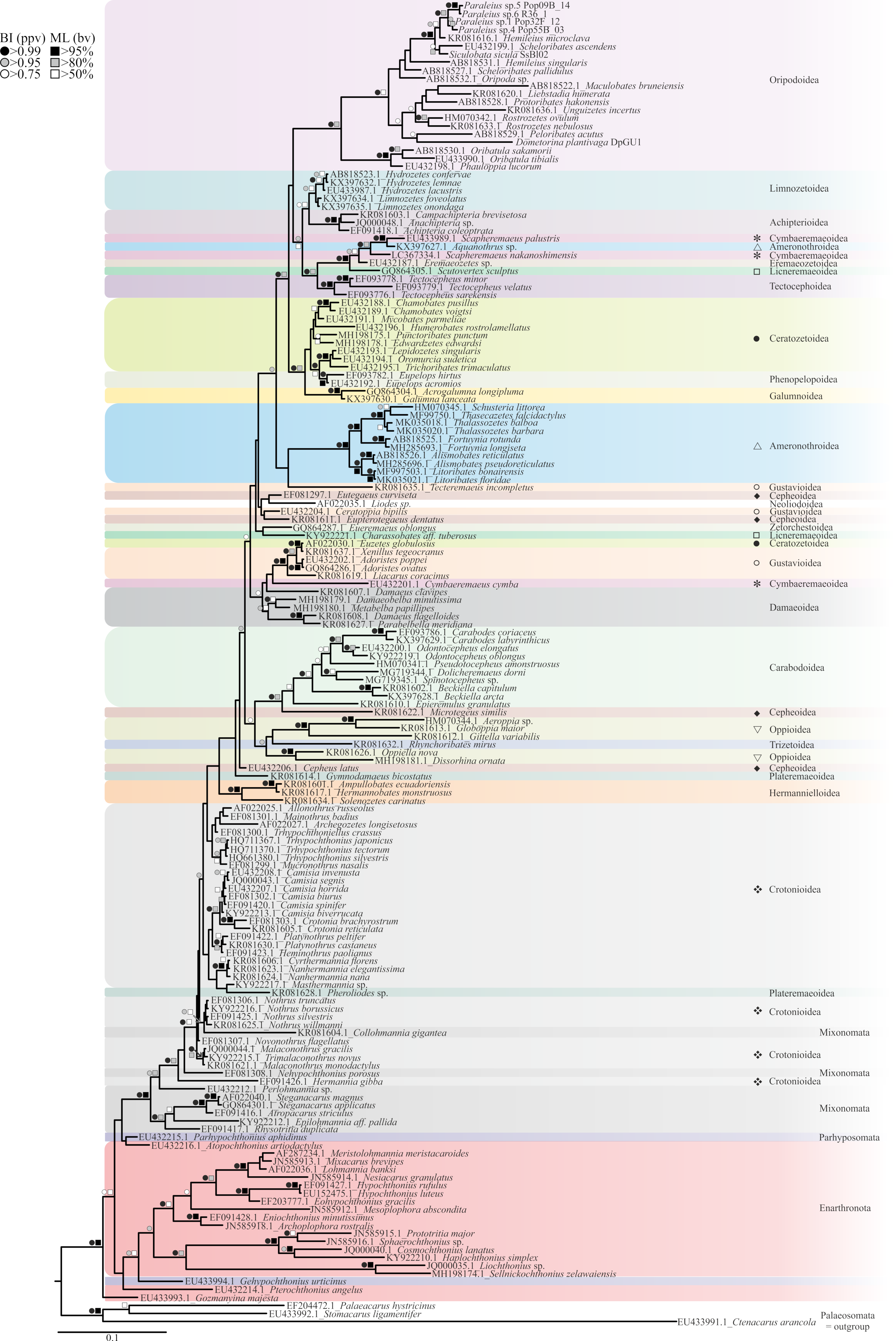
3. Results
4. Discussion
4.1. Revisiting the Tree-Living Lifestyle in Oribatids
4.2. Capitate Sensillus—A Special Morphological Feature of Tree-Dwelling Oribatid Mites?
4.3. Discussing the Term “Arboreal” in Oribatid Mites
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ruf, A. A maturity index for predatory soil mites (Mesostigmata: Gamasina) as an indicator of environmental impacts of pollution on forest soils. Appl. Soil Ecol. 1998, 9, 447–452. [Google Scholar] [CrossRef]
- Van Straalen, N.M. Evaluation of bioindicator systems derived from soil arthropod communities. Appl. Soil Ecol. 1998, 9, 429–437. [Google Scholar] [CrossRef]
- Behan-Pelletier, V.M. Oribatid mite biodiversity in agroecosystems: Role for bioindication. Agric. Ecosyst. Environ. 1999, 74, 411–423. [Google Scholar] [CrossRef]
- Gergócs, V.; Hufnagel, L. Application of oribatid mites as indicators. Appl. Ecol. Environ. Res. 2009, 7, 79–98. [Google Scholar] [CrossRef]
- Kraus, D.; Bütler, R.; Krumm, F.; Lachat, T.; Larrieu, L.; Mergner, U.; Paillet, Y.; Schuck, A.; Winter, S. Catalogue of Tree Microhabitats: Reference Field List; European Forest Institute: Joensuu, Finland, 2016. [Google Scholar]
- Paillet, Y.; Archaux, F.; Boulanger, V.; Debaive, N.; Fuhr, M.; Gilg, O.; Gosselin, F.; Guilbert, E. Snags and large trees drive higher tree microhabitat densities in strict forest reserves. For. Ecol. Manag. 2017, 389, 176–186. [Google Scholar] [CrossRef]
- Larrieu, L.; Paillet, Y.; Winter, S.; Bütler, R.; Kraus, D.; Krumm, F.; Lachat, T.; Micheli, A.K.; Regnery, B.; Vandekerkhove, K. Tree related microhabitats in temperate and Mediterranean European forests: A hierarchical typology for inventory standardization. Ecol. Indic. 2018, 84, 194–207. [Google Scholar] [CrossRef]
- Wunderle, I. Die Oribatiden-Gemeinschaften (Acari) der verschiedenen Habitate eines Buchenwaldes. Carolinea 1992, 50, 79–144. [Google Scholar]
- Travé, J. Écologie et biologie des Oribates (Acariens) saxicoles et arboricoles. Vie et Milieu Suppl. 1963, 14, 267. [Google Scholar]
- Niedbala, W. Tree moss-mite fauna in the outskirts of Poznan. In Oribatei and Their Role in the Process of the Soil Formation; Lithuanian Academy of Sciences: Vilnius, Lithuania, 1970; pp. 103–112. [Google Scholar]
- Aoki, J.I. Soil mites (oribatids) climbing trees. In Proceedings of the 3rd International Congress of Acarology, Prague 1971; Daniel, M., Rosický, B., Eds.; Springer: Dordrecht, The Netherlands, 1973; pp. 59–65. [Google Scholar]
- Behan-Pelletier, V.M.; Winchester, N.N. Arboreal oribatid mite diversity: Colonizing the canopy. Appl. Soil Ecol. 1998, 9, 45–54. [Google Scholar] [CrossRef]
- Winchester, N.N.; Behan-Pelletier, V.; Ring, R.A. Arboreal specificiy, diversity and abundance of canopy-dwelling oribatid mites (Acari: Oribatida). Pedobiologia 1999, 43, 391–400. [Google Scholar]
- Behan-Pelletier, V.M.; Walter, D.E. Biodiversity of oribatid mites (Acari: Oribatida) in tree canopies and litter. In Invertebrates as Webmasters in Ecosystems; Coleman, D.C., Hendrix, P.F., Eds.; CABI Publishing: Wallingford, UK, 2000; pp. 187–202. [Google Scholar]
- Lindo, Z.; Winchester, N.N. Oribatid mite communities and foliar litter decomposition in canopy suspended soils and forest floor habitats of western redcedar forests, Vancouver Island, Canada. Soil Biol. Biochem. 2007, 39, 2957–2966. [Google Scholar] [CrossRef]
- Arroyo, J.; Kenny, J.; Bolger, T. Variation between mite communities in Irish forest types—Importance of bark and moss cover in canopy. Pedobiologia 2013, 56, 241–250. [Google Scholar] [CrossRef]
- Michel, A.K.; Winter, S.; Linde, A. The effect of tree dimension on the diversity of bark microhabitat structures and bark use in Douglas-fir (Pseudotsuga menziesii var. menziesii). Can. J. For. Res. 2011, 41, 300–308. [Google Scholar] [CrossRef]
- Heethoff, M.; Domes, K.; Laumann, M.; Maraun, M.; Norton, R.A.; Scheu, S. High genetic divergences indicate ancient separation of parthenogenetic lineages of the oribatid mite Platynothrus peltifer (Acari, Oribatida). J. Evol. Biol. 2007, 20, 392–402. [Google Scholar] [CrossRef]
- Pfingstl, T.; Baumann, J.; Lienhard, A. The Caribbean enigma: The presence of unusual cryptic diversity in intertidal mites (Arachnida, Acari, Oribatida). Org. Divers. Evol. 2019, 19, 609–623. [Google Scholar] [CrossRef]
- Schäffer, S.; Kerschbaumer, M.; Koblmüller, S. Multiple new species: Cryptic diversity in the widespread mite species Cymbaeremaeus cymba (Oribatida, Cymbaeremaeidae). Mol. Phylogenet. Evol. 2019, 135, 185–192. [Google Scholar] [CrossRef]
- Maraun, M.; Erdmann, G.; Schulz, G.; Norton, R.A.; Scheu, S.; Domes, K. Multiple convergent evolution of arboreal life in oribatid mites indicates the primacy of ecology. Proc. R. Soc. B 2009, 276, 3219–3227. [Google Scholar] [CrossRef] [PubMed]
- Seyd, E.L.; Seaward, M.R.D. The association of oribatid mites and lichens. Zool. J. Linn. Soc. 1984, 80, 369–420. [Google Scholar] [CrossRef]
- André, H.M. Associations between corticolous microarthropod communities and epiphytic cover on bark. Holarct. Ecol. 1985, 8, 113–119. [Google Scholar] [CrossRef]
- Nicolai, V. The bark of trees: Thermal properties, microclimate and fauna. Oecologia 1986, 69, 148–160. [Google Scholar] [CrossRef]
- Root, H.T.; McGee, G.G.; Norton, R.A. Arboreal mite communities on epiphytic lichens of the Adirondack Mountains of New York. Northeast. Nat. 2007, 14, 425–438. [Google Scholar] [CrossRef]
- Taylor, A.R.; Ranius, T. Tree hollows harbour a specialised oribatid mite fauna. J. Insect Conserv. 2014, 18, 39–55. [Google Scholar] [CrossRef]
- Salavatulin, V. Microhabitat distribution of arboreal oribatid mites (Oribatida), associated with the Siberian pine (Pinus sibirica) of Western Siberia. Exp. Appl. Acarol. 2019, 78, 469–483. [Google Scholar] [CrossRef] [PubMed]
- Vanek, J. Arboreal oribatid mites (Acari, Oribatei) in young stands of Abies alba. Sbornik Vysoke Skoly Zemedelske a Lesnicke Fakulty v Praha 1972, 5, 291–300. [Google Scholar]
- Krivolutsky, D.A. Arboricular (Tree-dwelling) oribatid mites as bioindicators of environment quality. Dokl. Biol. Sci. 2004, 399, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Behan-Pelletier, V.M.; John, M.G.S.; Winchester, N. Canopy Oribatida: Tree specific or microhabitat specific? Eur. J. Soil Biol. 2008, 44, 220–224. [Google Scholar] [CrossRef]
- Klimov, P.B.; O’Connor, B.M.; Chetverikov, P.E.; Bolton, S.J.; Pepato, A.R.; Mortazavi, A.; Tolstikov, A.V.; Bauchan, G.R.; Ochoa, R. Comprehensive phylogeny of acariform mites (Acariformes) provides insights on the origin of the four-legged mites (Eriophyoidea), a long branch. Mol. Phylogenet. Evol. 2018, 119, 105–117. [Google Scholar] [CrossRef]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2019, 20, 1160–1166. [Google Scholar] [CrossRef]
- Capella-Gutiérrez, S.; Silla-Martínez, J.M.; Gabaldón, T. TrimAl: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef]
- Weigmann, G. Hornmilben (Oribatida); Goecke and Evers: Keltern, Germany, 2006. [Google Scholar]
- Lefort, V.; Longueville, J.-E.; Gascuel, O. SMS: Smart Model Selection in PhyML. MBE 2017, 34, 2422–2424. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate Maximum-Likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarization in Bayesian phylogenetics using Tracer1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Maddison, W.P.; Maddison, D.R. Mesquite: A Modular System for Evolutionary Analysis. Version 3.51. Available online: http://www.mesquiteproject.org (accessed on 28 June 2018).
- Halliday, R.B.; Majka, C.G. Clarification of the status of the genus names Leiodes Latreille 1796 (Coleoptera), Liodes von Heyden 1826 (Acari) and Neoliodes Berlese 1888 (Acari). Zootaxa 2010, 2600, 61–65. [Google Scholar] [CrossRef]
- Schatz, H.; Behan-Pelletier, V.M.; O’Connor, B.M.; Norton, R.A. Suborder Oribatida van der Hammen, 1968. In Animal Biodiversity: An Outline of Higher-Level Classification and Survey of Taxonomic Richness; Zhang, Z.Q., Ed.; Zootaxa 3148; Magnolia Press: Auckland, New Zealand, 2011; pp. 141–147. [Google Scholar]
- Pagel, M. Inferring the historical patterns of biological evolution. Nature 1999, 401, 877–884. [Google Scholar] [CrossRef]
- Krause, A.; Pachl, P.; Schulz, G.; Lehmitz, R.; Seniczak, A.; Schaefer, I.; Scheu, S.; Maraun, M. Convergent evolution of aquatic life by sexual and parthenogenetic oribatid mites. Exp. Appl. Acarol. 2016, 70, 439–453. [Google Scholar] [CrossRef]
- Pachl, P.; Lindl, A.C.; Krause, A.; Scheu, S.; Schaefer, I.; Maraun, M. The tropics as ancient cradle of oribatid mite diversity. Acarologia 2017, 57, 309–322. [Google Scholar] [CrossRef]
- Schäffer, S.; Koblmüller, S.; Pfingstl, T.; Sturmbauer, C.; Krisper, G. Ancestral state reconstruction reveals multiple independent evolution of diagnostic morphological characters in the “Higher Oribatida” (Acari), conflicting with current classification schemes. BMC Evol. Biol. 2010, 10, 246. [Google Scholar] [CrossRef]
- Lindo, Z.; Winchester, N.N. A comparison of microarthropod assemblages with emphasis on oribatid mites in canopy suspended soils and forest floors associated with ancient western redcedar trees. Pedobiologia 2006, 50, 31–41. [Google Scholar] [CrossRef]
- Subias, L.S. Taxonomía y Ecología de los Oribátidos Saxícolas y Arborícolas de la Sierra de Guadarrama (Acarida, Oribatida). Ph.D. Thesis, Universidad Complutense de Madrid, Madrid, Spain, 1977; p. 375. [Google Scholar]
- Wallwork, J.A. The Distribution and Diversity of Soil Fauna; Academic Press: London, UK; New York, NY, USA; San Francisco, CA, USA, 1976. [Google Scholar]
- Gjelstrup, P. Epiphytic cryptostigmatid mites on some beech- and birch-trees in Denmark. Pedobiologia 1979, 19, 1–8. [Google Scholar]
- Krivolutsky, D.A.; Lebedeva, N.V. Oribatid mites (Oribatei, Acariformes) in bird feathers: Non-passerines. Acta Zool. Litu. 2004, 14, 26–47. [Google Scholar] [CrossRef]
- Krivolutsky, D.A.; Lebedeva, N.V. Oribatid mites (Oribatei, Acariformes) in bird feathers: Passeriformes. Acta Zool. Litu. 2004, 14, 19–38. [Google Scholar] [CrossRef]
- Lehmitz, R.; Russell, D.; Hohberg, K.; Christian, A.; Xylander, W.E. Wind dispersal of oribatid mites as a mode of migration. Pedobiologia 2011, 54, 201–207. [Google Scholar] [CrossRef]
- Walter, D.E.; Behan-Pelletier, V.M. Systematics and ecology of Adhaesozetes polyphyllos sp.nov. (Acari: Oribatida: Licneremaeoidea), a leaf-inhabiting mite from Australian rainforests. Can. J. Zool. 1993, 71, 1024–1040. [Google Scholar] [CrossRef]
- Lindo, Z.; Clayton, M.; Behan-Pelletier, V.M. Systematics and ecology of the genus Dendrozetes (Acari: Oribatida: Peloppiidae) from arboreal habitats in Western North America. Zootaxa 2010, 2403, 10–22. [Google Scholar] [CrossRef]
- Bayartogtokh, B.; Itioka, T.; Kitora, H.; Meleng, P.; Shimano, S. New findings of poronotic oribatid mites (Acari: Oribatida) from the high canopy of a Bornean tropical rain forest. Int. J. Acarol. 2020, 46, 73–82. [Google Scholar] [CrossRef]
- Van der Hammen, L. Glossary of Acarological Terminology. Vol. I. General Terminology; Dr. W. Junk Publishers: The Hague, The Netherlands, 1980; p. 244. [Google Scholar]
- Alberti, G.; Moreno, A.I.; Kratzmann, M. The fine structure of trichobothria in moss mites with special emphasis on Acrogalumna lonigpluma (Berlese, 1904) (Oribatida, Acari, Arachnida). Acta Zool. 1994, 75, 57–74. [Google Scholar] [CrossRef]
- Norton, R.A.; Palacios-Vargas, J.G. Nueva Belba (Oribatei: Damaeidae) de musgos epifitos de Mexico. Folia Entomol. Mex. 1982, 52, 61–73. [Google Scholar]
- Olszanowski, Z.; Clayton, M.R.; Humble, L.M. New species of the genus Camisia (Acari: Oribatida): An arboreal mite with enclosed sensilli. Can. Entomol. 2002, 134, 707–721. [Google Scholar] [CrossRef]
- Schatz, H.; Behan-Pelletier, V. Global diversity of oribatids (Oribatida: Acari: Arachnida). Hydrobiologia 2008, 595, 323–328. [Google Scholar] [CrossRef]
- Behan-Pelletier, V.M.; Walter, D.E. Phylleremus n. gen., from leaves of deciduous trees in eastern Australia (Oribatida: Licneremaeoidea). Zootaxa 2007, 1386, 1–17. [Google Scholar] [CrossRef]
- Walter, D.E.; O´Dowd, D.J. Beneath biodiversity: Factors influencing the diversity and abundance of canopy mites. Selbyana 1995, 16, 12–20. [Google Scholar]
- Niedbala, W. Oribatei (Acari) of Spitzbergen. Bull. Acad. Polon. Sci. Série Sci. Biologique 1971, 19, 737–742. [Google Scholar]
- Erdmann, G.; Otte, V.; Langel, R.; Scheu, S.; Maraun, M. The trophic structure of bark-living oribatid mite communities analysed with stable isotopes (15N, 13C) indicates strong niche differentiation. Exp. Appl. Acarol. 2007, 41, 1–10. [Google Scholar] [CrossRef]
- Bauer, D.M.; Bell, K.P.; Nelson, E.J.; Calhoun, A.J. Managing small natural features: A synthesis of economic issues and emergent opportunities. Biol. Conserv. 2017, 211, 80–87. [Google Scholar] [CrossRef]
- Lindenmayer, D.B. Conserving large old trees as small natural features. Biol. Conserv. 2017, 211, 51–59. [Google Scholar] [CrossRef]
- Hunter, M.L. Conserving small natural features with large ecological roles: An introduction and definition. Biol. Conserv. 2017, 211, 1–2. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schäffer, S.; Koblmüller, S.; Krisper, G. Revisiting the Evolution of Arboreal Life in Oribatid Mites. Diversity 2020, 12, 255. https://doi.org/10.3390/d12060255
Schäffer S, Koblmüller S, Krisper G. Revisiting the Evolution of Arboreal Life in Oribatid Mites. Diversity. 2020; 12(6):255. https://doi.org/10.3390/d12060255
Chicago/Turabian StyleSchäffer, Sylvia, Stephan Koblmüller, and Günther Krisper. 2020. "Revisiting the Evolution of Arboreal Life in Oribatid Mites" Diversity 12, no. 6: 255. https://doi.org/10.3390/d12060255
APA StyleSchäffer, S., Koblmüller, S., & Krisper, G. (2020). Revisiting the Evolution of Arboreal Life in Oribatid Mites. Diversity, 12(6), 255. https://doi.org/10.3390/d12060255





