Diversification of African Rainforest Restricted Clades: Piptostigmateae and Annickieae (Annonaceae)
Abstract
1. Introduction
2. Material and Methods
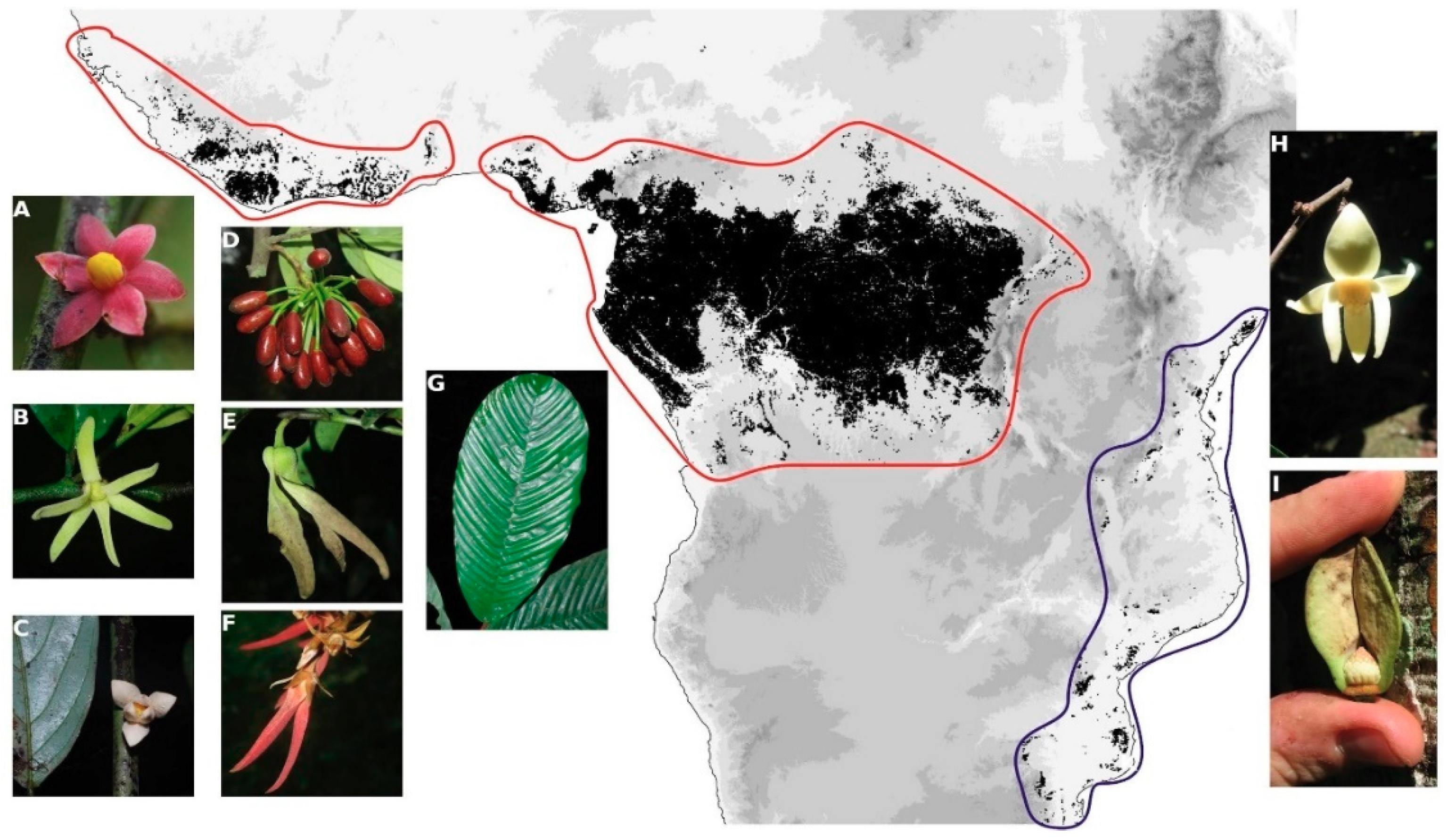
2.1. Taxon and Nuclear Marker Sampling
2.2. Contig Assembly, Alignment and Paralog Identification
2.3. Exons and Introns Selection for Molecular Dating
2.4. Molecular Dating
2.5. Diversification Analyses
3. Results
3.1. Phylogenomics
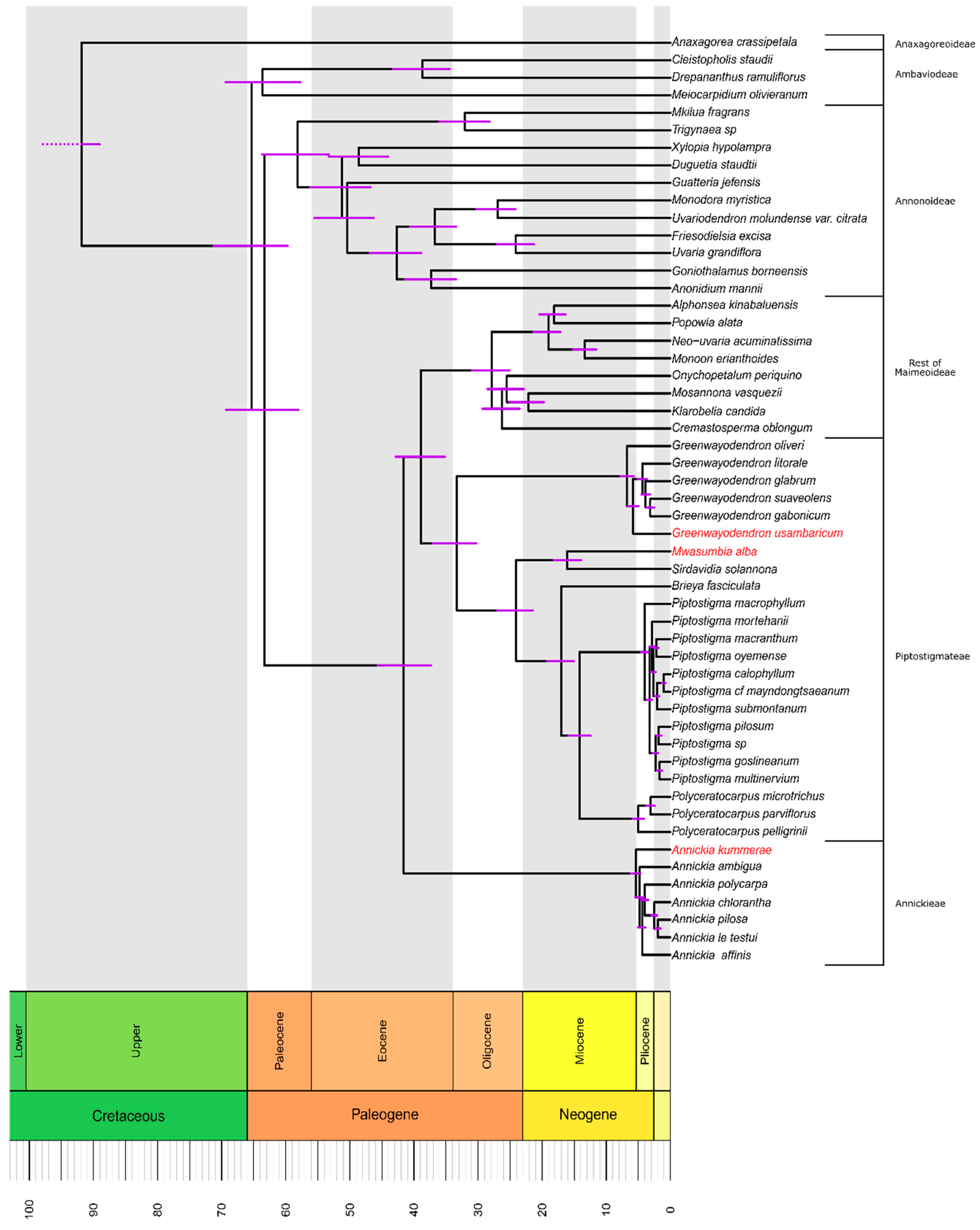
3.2. Molecular Dating and Divergence Times
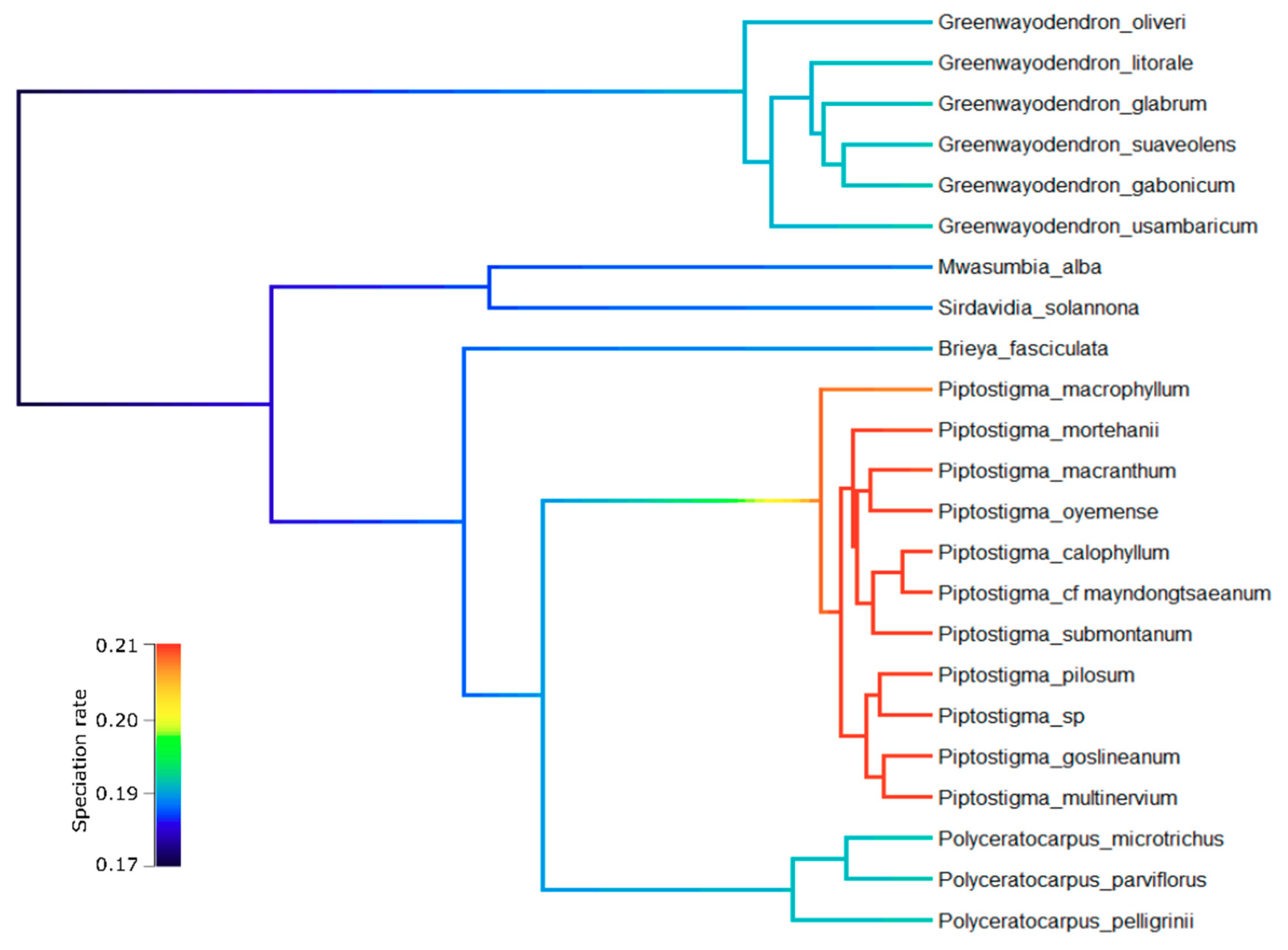
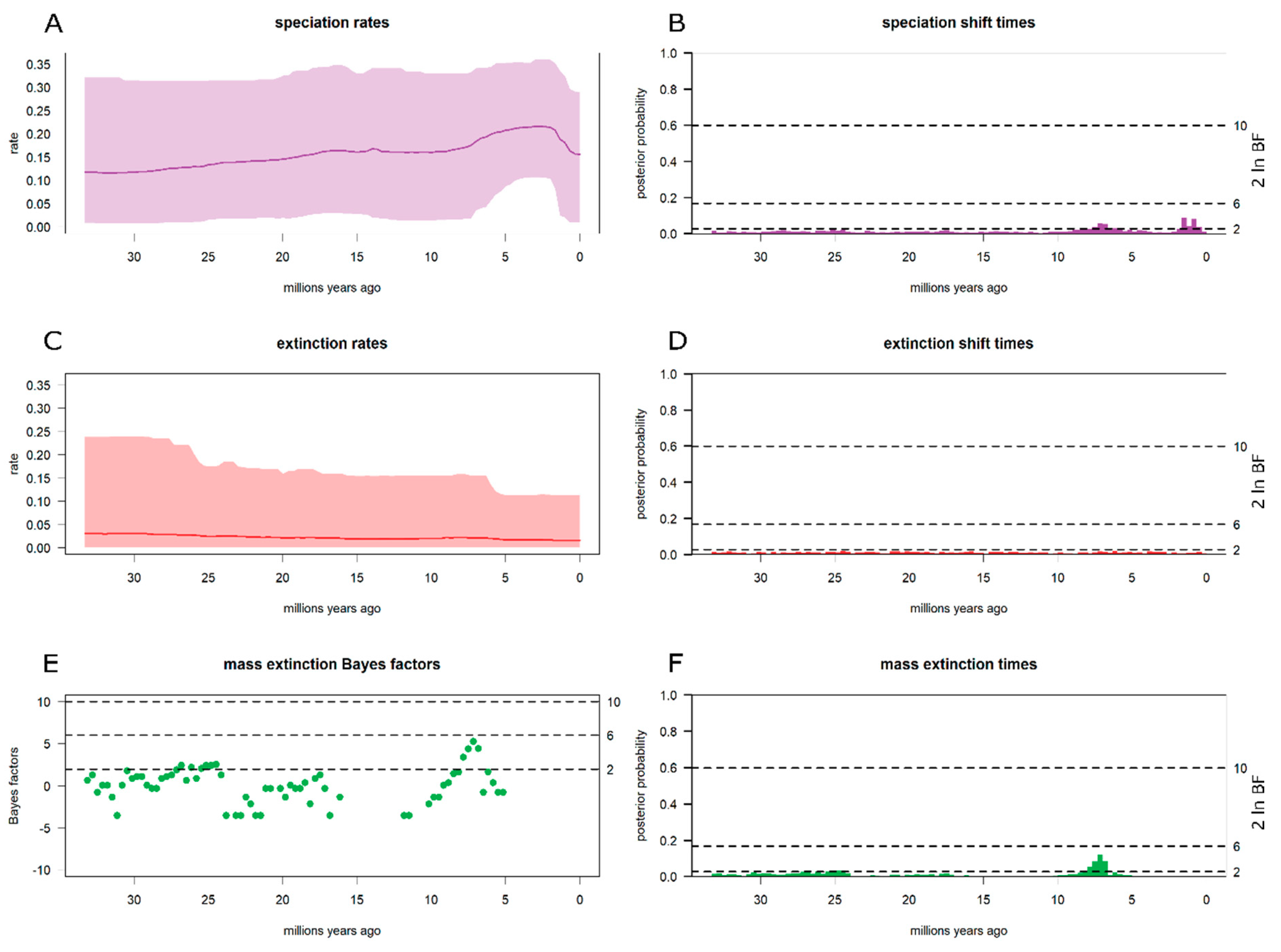
3.3. Diversification Analyses
4. Discussion
4.1. Temporal Estimates of African Rainforest Vicariance
4.2. Piptostigmateae Diversification
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Whitmore, T.C. An Introduction to Tropical Rain Forests; Clarendon Press: Oxford, UK, 1998; ISBN 978-0-19-850147-3. [Google Scholar]
- Morley, R.J. Origin and Evolution of Tropical Rain Forests; John Wiley & Sons: New York, NY, USA, 2000; ISBN 978-0-471-98326-2. [Google Scholar]
- Linder, H.P. Plant diversity and endemism in sub-Saharan tropical Africa. J. Biogeogr. 2001, 28, 169–182. [Google Scholar] [CrossRef]
- Klopper, R.R.; Gautier, L.; Chatelain, C.; Smith, G.F.; Spichiger, R. Floristics of the Angiosperm flora of Sub-Saharan Africa: An analysis of the African plant checklist and database. Taxon 2007, 56, 201–208. [Google Scholar] [CrossRef]
- Sosef, M.S.M.; Dauby, G.; Blach-Overgaard, A.; van der Burgt, X.; Catarino, L.; Damen, T.; Deblauwe, V.; Dessein, S.; Dransfield, J.; Droissart, V.; et al. Exploring the floristic diversity of tropical Africa. BMC Biol. 2017, 15, 15. [Google Scholar] [CrossRef] [PubMed]
- Linder, H.P. The evolution of African plant diversity. Front. Ecol. Evol. 2014, 2, 38. [Google Scholar] [CrossRef]
- Plana, V.; Gascoigne, A.; Forrest, L.L.; Harris, D.; Pennington, R.T. Pleistocene and pre-Pleistocene Begonia speciation in Africa. Mol. Phylogenet. Evol. 2004, 31, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Auvrey, G.; Harris, D.J.; Richardson, J.E.; Newman, M.F.; Särkinen, T.E. Phylogeny and dating of Aframomum (Zingiberaceae). In Diversity, Phylogeny, and Evolution in the Monocotyledons; Seberg, O., Peterson, G., Barfod, A., Davis, J.I., Eds.; Aarhus University Press: Aarhus, Denmark, 2010; pp. 287–305. ISBN 978-87-7934-398-6. [Google Scholar]
- Couvreur, T.L.; Chatrou, L.W.; Sosef, M.S.; Richardson, J.E. Molecular phylogenetics reveal multiple tertiary vicariance origins of the African rain forest trees. BMC Biol. 2008, 6, 54. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, T.L.P.; Porter-Morgan, H.; Wieringa, J.J.; Chatrou, L.W. Little ecological divergence associated with speciation in two African rain forest tree genera. BMC Evol. Biol. 2011, 11, 296. [Google Scholar] [CrossRef] [PubMed]
- Sosef, M.S.M. Refuge Begonias: Taxonomy, phylogeny and historical biogeography of Begonia sect. Loasibegonia and sect. Scutobegonia in relation to glacial rain forest refuges in Africa. Studies in Begonia V. Wagening. Agric. Univ. Pap. 1994, 94, 1–306. [Google Scholar]
- Maley, J.; Brenac, P. Vegetation dynamics, palaeoenvironments and climatic changes in the forests of western Cameroon during the last 28,000 years B.P. Rev. Palaeobot. Palynol. 1998, 99, 157–187. [Google Scholar] [CrossRef]
- Plana, V. Mechanisms and tempo of evolution in the African Guineo–Congolian rainforest. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2004, 359, 1585–1594. [Google Scholar] [CrossRef]
- Hardy, O.J.; Born, C.; Budde, K.; Daïnou, K.; Dauby, G.; Duminil, J.; Ewédjé, E.-E.B.; Gomez, C.; Heuertz, M.; Koffi, G.K.; et al. Comparative phylogeography of African rain forest trees: A review of genetic signatures of vegetation history in the Guineo-Congolian region. Comptes Rendus Geosci. 2013, 345, 284–296. [Google Scholar] [CrossRef]
- Richards, P.W. Africa, the “Odd man out”. In Tropical Forest Ecosystems of Africa and South America: A Comparative Review; Meggers, B.J., Ayensu, E.S., Duckworth, W.D., Eds.; Smithsonian Institution Press: Washington, DC, USA, 1973; ISBN 978-0-87474-125-4. [Google Scholar]
- Couvreur, T.L.P. Odd man out: Why are there fewer plant species in African rain forests? Plant Syst. Evol. 2015, 301, 1299–1313. [Google Scholar] [CrossRef]
- Axelrod, D.I.; Raven, P.H. Late Cretaceous and Tertiary vegetation history of Africa. In Biogeography and Ecology of Southern Africa; Werger, M.J.A., Ed.; W. Junk bv Publishers: The Hague, The Netherlands, 1978; pp. 77–130. ISBN 978-94-009-9951-0. [Google Scholar]
- Richards, P.W. The Tropical Rain Forest: An Ecological Study, 2nd ed.; Cambridge University Press: Cambridge, UK, 1996; ISBN 978-0-521-42194-2. [Google Scholar]
- Bobe, R. The evolution of arid ecosystems in eastern Africa. J. Arid Environ. 2006, 66, 564–584. [Google Scholar] [CrossRef]
- White, F. The Guineo-Congolian region and its relationships to other phytochoria. Bull. Jard. Bot. Natl. Belg. 1979, 49, 11–55. [Google Scholar] [CrossRef]
- Wasser, S.K.; Lovett, J.C. Biogeography and Ecology of the Rainforests of Eastern Africa; Lovett, J.C., Wasser, S.K., Eds.; Cambridge University Press: Cambridge, UK, 1993; ISBN 978-0-521-06898-7. [Google Scholar]
- Burgess, N.D.; Clarke, G.P.; Rodgers, W.A. Coastal forests of eastern Africa: Status, endemism patterns and their potential causes. Biol. J. Linn. Soc. 1998, 64, 337–367. [Google Scholar] [CrossRef]
- Burgess, N.D.; Clarke, G.P. Coastal Forests of Eastern Africa; IUCN - The World Conservation Union, Publications Services Unit: Cambridge, UK, 2000; ISBN 978-2-8317-0436-4. [Google Scholar]
- Dimitrov, D.; Nogués-Bravo, D.; Scharff, N. Why Do Tropical Mountains Support Exceptionally High Biodiversity? The Eastern Arc Mountains and the Drivers of Saintpaulia Diversity. PLoS ONE 2012, 7, e48908. [Google Scholar] [CrossRef] [PubMed]
- Pokorny, L.; Riina, R.; Mairal, M.; Meseguer, A.S.; Culshaw, V.; Cendoya, J.; Serrano, M.; Carbajal, R.; Ortiz, S.; Heuertz, M.; et al. Living on the edge: Timing of Rand Flora disjunctions congruent with ongoing aridification in Africa. Front. Genet. 2015, 6, 154. [Google Scholar] [CrossRef]
- Tosso, F.; Hardy, O.J.; Doucet, J.-L.; Daïnou, K.; Kaymak, E.; Migliore, J. Evolution in the Amphi-Atlantic tropical genus Guibourtia (Fabaceae, Detarioideae), combining NGS phylogeny and morphology. Mol. Phylogenet. Evol. 2018, 120, 83–93. [Google Scholar] [CrossRef]
- Lovett, J.C. Climatic history and forest distribution in eastern Africa. In Biogeography and Ecology of the Rainforests of Eastern Africa; Lovett, J.C., Wasser, S.K., Eds.; Cambridge University Press: Cambridge, UK, 1993; pp. 23–29. ISBN 0-521-43083-6. [Google Scholar]
- Morley, R.J. Cretaceous and Tertiary climate change and the past distribution of megathermal rainforests. In Tropical Rainforest Responses to Climatic Change; Bush, M.B., Flenley, J., Eds.; Praxis Publishing: Chichester, UK, 2007; pp. 1–31. ISBN 978-3-642-05382-5. [Google Scholar]
- Willis, K.; McElwain, J. The Evolution of Plants, 2nd ed.; Oxford University Press: Oxford, UK, 2014; ISBN 978-0-19-929223-3. [Google Scholar]
- Linder, H.P. East African Cenozoic vegetation history. Evol. Anthropol. Issues News Rev. 2017, 26, 300–312. [Google Scholar] [CrossRef]
- Davis, C.C.; Bell, C.D.; Fritsch, P.W.; Mathews, S. Phylogeny of Acridocarpus-Brachylophon (Malpighiaceae): Implications for Tertiary tropical floras and Afroasian biogeography. Evolution 2002, 56, 2395–2405. [Google Scholar] [CrossRef]
- Coetzee, J.A. African flora since the terminal Jurassic. In Biological Relationships between Africa and South America; Goldblatt, P., Ed.; Yale University Press: New Haven, CT, USA, 1993; pp. 37–61. ISBN 978-0-300-05375-3. [Google Scholar]
- Jacobs, B.F.; Kingston, J.D.; Jacobs, L.L. The Origin of Grass-Dominated Ecosystems. Ann. Mo. Bot. Gard. 1999, 86, 590–643. [Google Scholar] [CrossRef]
- Voelker, G.; Outlaw, R.K.; Bowie, R.C.K. Pliocene forest dynamics as a primary driver of African bird speciation: African forest refugia. Glob. Ecol. Biogeogr. 2010, 19, 111–121. [Google Scholar] [CrossRef]
- Tolley, K.A.; Townsend, T.M.; Vences, M. Large-scale phylogeny of chameleons suggests African origins and Eocene diversification. Proc. R. Soc. B Biol. Sci. 2013, 280, 20130184. [Google Scholar] [CrossRef]
- Loader, S.P.; Pisani, D.; Cotton, J.A.; Gower, D.J.; Day, J.J.; Wilkinson, M. Relative time scales reveal multiple origins of parallel disjunct distributions of African caecilian amphibians. Biol. Lett. 2007, 3, 505–508. [Google Scholar] [CrossRef]
- Chatrou, L.W.; Pirie, M.D.; Erkens, R.H.J.; Couvreur, T.L.P.; Neubig, K.M.; Abbott, J.R.; Mols, J.B.; Maas, J.W.; Saunders, R.M.K.; Chase, M.W. A new subfamilial and tribal classification of the pantropical flowering plant family Annonaceae informed by molecular phylogenetics. Bot. J. Linn. Soc. 2012, 169, 5–40. [Google Scholar] [CrossRef]
- Guo, X.; Tang, C.C.; Thomas, D.C.; Couvreur, T.L.P.; Saunders, R.M.K. A mega-phylogeny of the Annonaceae: Taxonomic placement of five enigmatic genera and support for a new tribe, Phoenicantheae. Sci. Rep. 2017, 7, 7323. [Google Scholar] [CrossRef] [PubMed]
- Sonké, B.; Couvreur, T.L. Tree diversity of the Dja Faunal Reserve, southeastern Cameroon. Biodivers. Data J. 2014, e1049. [Google Scholar] [CrossRef]
- Doyle, J.A.; Le Thomas, A. Cladistic analysis and pollen evolution in Annonaceae. Acta Bot. Gallica 1994, 141, 149–170. [Google Scholar] [CrossRef][Green Version]
- Couvreur, T.L.P.; Forest, F.; Baker, W.J. Origin and global diversification patterns of tropical rain forests: Inferences from a complete genus-level phylogeny of palms. BMC Biol. 2011, 9, 44. [Google Scholar] [CrossRef]
- Thomas, D.C.; Tang, C.C.; Saunders, R.M.K. Historical biogeography of Goniothalamus and Annonaceae tribe Annoneae: Dispersal-vicariance patterns in tropical Asia and intercontinental tropical disjunctions revisited. J. Biogeogr. 2017, 44, 2862–2876. [Google Scholar] [CrossRef]
- Couvreur, T.L.P.; Helmstetter, A.J.; Koenen, E.J.M.; Brandão, R.D.; Little, S.; Sauquet, H.; Erkens, R.H.J. Phylogenomics of the major tropical plant family Annonaceae using targeted enrichment of nuclear genes. Front. Plant Sci. 2019, 1941. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, T.L.P.; van der Ham, R.W.J.M.; Mbele, Y.M.; Mbago, F.M.; Johnson, D.M. Molecular and Morphological Characterization of a New Monotypic Genus of Annonaceae, Mwasumbia from Tanzania. Syst. Bot. 2009, 34, 266–276. [Google Scholar] [CrossRef]
- Couvreur, T.L.P.; Niangadouma, R.; Sonké, B.; Sauquet, H. Sirdavidia, an extraordinary new genus of Annonaceae from Gabon. PhytoKeys 2015, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Ghogue, J.-P.; Sonké, B.; Couvreur, T.L.P. Taxonomic revision of the African genera Brieya and Piptostigma (Annonaceae). Plant Ecol. Evol. 2017, 150, 173–216. [Google Scholar] [CrossRef]
- Lissambou, B.-J.; Hardy, O.J.; Atteke, C.; Stevart, T.; Dauby, G.; Mbatchi, B.; Sonke, B.; Couvreur, T.L.P. Taxonomic revision of the African genus Greenwayodendron (Annonaceae). PhytoKeys 2018, 55–93. [Google Scholar] [CrossRef]
- Marshall, A.R.; Couvreur, T.L.P.; Summers, A.L.; Deere, N.J.; Luke, W.R.Q.; Ndangalasi, H.J.; Sparrow, S.; Johnson, D.M. A new species in the tree genus Polyceratocarpus (Annonaceae) from the Udzungwa Mountains of Tanzania. PhytoKeys 2016, 63–76. [Google Scholar] [CrossRef]
- Versteegh, C.P.C.; Sosef, M.S.M. Revision of the African genus Annickia (Annonaceae). Syst. Geograph. Plants 2007, 91–118. [Google Scholar] [CrossRef]
- Johnson, M.G.; Gardner, E.M.; Liu, Y.; Medina, R.; Goffinet, B.; Shaw, A.J.; Zerega, N.J.C.; Wickett, N.J. HybPiper: Extracting Coding Sequence and Introns for Phylogenetics from High-Throughput Sequencing Reads Using Target Enrichment. Appl. Plant Sci. 2016, 4, 1600016. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Castresana, J. Selection of Conserved Blocks from Multiple Alignments for Their Use in Phylogenetic Analysis. Mol. Biol. Evol. 2000, 17, 540–552. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Mai, U.; Mirarab, S. TreeShrink: Fast and accurate detection of outlier long branches in collections of phylogenetic trees. BMC Genom. 2018, 19, 272. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.A.; Brown, J.W.; Walker, J.F. So many genes, so little time: A practical approach to divergence-time estimation in the genomic era. PLoS ONE 2018, 13, e0197433. [Google Scholar] [CrossRef] [PubMed]
- Darriba, D.; Posada, D.; Kozlov, A.M.; Stamatakis, A.; Morel, B.; Flouri, T. ModelTest-NG: A new and scalable tool for the selection of DNA and protein evolutionary models. bioRxiv 2009, 612903. [Google Scholar] [CrossRef]
- Drummond, A.J.; Suchard, M.A.; Xie, D.; Rambaut, A. Bayesian Phylogenetics with BEAUti and the BEAST 1.7. Mol. Biol. Evol. 2012, 29, 1969–1973. [Google Scholar] [CrossRef]
- Pirie, M.D.; Doyle, J.A. Dating clades with fossils and molecules: The case of Annonaceae: Dating clades in Annonaceae. Bot. J. Linn. Soc. 2012, 169, 84–116. [Google Scholar] [CrossRef]
- Russel, P.M.; Brewer, B.J.; Klaere, S.; Bouckaert, R.R. Model Selection and Parameter Inference in Phylogenetics Using Nested Sampling. Syst. Biol. 2018, 68, 219–233. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior Summarization in Bayesian Phylogenetics Using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef]
- Bouckaert, R.; Heled, J.; Kühnert, D.; Vaughan, T.; Wu, C.-H.; Xie, D.; Suchard, M.A.; Rambaut, A.; Drummond, A.J. BEAST 2: A Software Platform for Bayesian Evolutionary Analysis. PLoS Comput. Biol. 2014, 10, e1003537. [Google Scholar] [CrossRef]
- Paradis, E.; Claude, J.; Strimmer, K. APE: Analyses of Phylogenetics and Evolution in R language. Bioinformatics 2004, 20, 289–290. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: http://www.R-project.org (accessed on 15 May 2019).
- Morlon, H.; Lewitus, E.; Condamine, F.L.; Manceau, M.; Clavel, J.; Drury, J. RPANDA: An R package for macroevolutionary analyses on phylogenetic trees. Methods Ecol. Evol. 2016, 7, 589–597. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002; ISBN 978-0-387-22456-5. [Google Scholar]
- Höhna, S.; May, M.R.; Moore, B.R. TESS: An R package for efficiently simulating phylogenetic trees and performing Bayesian inference of lineage diversification rates. Bioinformatics 2016, 32, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Rabosky, D.L. Automatic Detection of Key Innovations, Rate Shifts, and Diversity-Dependence on Phylogenetic Trees. PLoS ONE 2014, 9, e89543. [Google Scholar] [CrossRef] [PubMed]
- Rabosky, D.L.; Mitchell, J.S.; Chang, J. Is BAMM Flawed? Theoretical and Practical Concerns in the Analysis of Multi-Rate Diversification Models. Syst. Biol. 2017, 66, 477–498. [Google Scholar] [CrossRef]
- Plummer, M.; Best, N.; Cowles, K.; Vines, K. CODA: Convergence diagnosis and output analysis for MCMC. R News 2006, 6, 7–11. [Google Scholar]
- Culshaw, V.; Stadler, T.; Sanmartín, I. Exploring the power of bayesian birth-death skyline models to detect mass extinction events from phylogenies with only extant taxa. Evolution 2019, 73, 1133–1150. [Google Scholar] [CrossRef]
- Kass, R.E.; Raftery, A.E. Bayes Factors. J. Am. Stat. Assoc. 1995, 90, 773–795. [Google Scholar] [CrossRef]
- Richardson, J.E.; Chatrou, L.W.; Mols, J.B.; Erkens, R.H.J.; Pirie, M.D. Historical biogeography of two cosmopolitan families of flowering plants: Annonaceae and Rhamnaceae. Philos. Trans. R. Soc. Lond. B Ser. 2004, 359, 1495–1508. [Google Scholar] [CrossRef]
- Xue, B.; Guo, X.; Landis, J.B.; Sun, M.; Tang, C.C.; Soltis, P.S.; Soltis, D.E.; Saunders, R.M.K. Accelerated diversification correlated with functional traits shapes extant diversity of the early divergent angiosperm family Annonaceae. Mol. Phylogenet. Evol. 2020, 142, 106659. [Google Scholar] [CrossRef]
- Migliore, J.; Kaymak, E.; Mariac, C.; Couvreur, T.L.P.; Lissambou, B.; Piñeiro, R.; Hardy, O.J. Pre-Pleistocene origin of phylogeographical breaks in African rain forest trees: New insights from Greenwayodendron (Annonaceae) phylogenomics. J. Biogeogr. 2018, 46, 212–223. [Google Scholar] [CrossRef]
- Lopes, J.C.; Chatrou, L.W.; Mello-Silva, R.; Rudall, P.J.; Sajo, M.G. Phylogenomics and evolution of floral traits in the Neotropical tribe Malmeeae (Annonaceae). Mol. Phylogenet. Evol. 2018, 118, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.C.; Chatrou, L.W.; Stull, G.W.; Johnson, D.M.; Harris, D.J.; Thongpairoj, U.; Saunders, R.M.K. The historical origins of palaeotropical intercontinental disjunctions in the pantropical flowering plant family Annonaceae. Perspect. Plant Ecol. Evol. Syst. 2015, 17, 1–16. [Google Scholar] [CrossRef]
- Shevenell, A.E.; Kennett, J.P.; Lea, D.W. Middle Miocene ice sheet dynamics, deep-sea temperatures, and carbon cycling: A Southern Ocean perspective. Geochem. Geophys. Geosyst. 2008, 9. [Google Scholar] [CrossRef]
- Flower, B.P.; Kennett, J.P. The middle Miocene climatic transition: East Antarctic ice sheet development, deep ocean circulation and global carbon cycling. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1994, 108, 537–555. [Google Scholar] [CrossRef]
- Jacobs, B.F. Palaeobotanical studies from tropical Africa: Relevance to the evolution of forest, woodland and savannah biomes. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2004, 359, 1573–1583. [Google Scholar] [CrossRef]
- Sepulchre, P.; Ramstein, G.; Fluteau, F.; Schuster, M.; Tiercelin, J.-J.; Brunet, M. Tectonic Uplift and Eastern Africa Aridification. Science 2006, 313, 1419–1423. [Google Scholar] [CrossRef]
- Senut, B.; Pickford, M.; Ségalen, L. Neogene desertification of Africa. Comptes Rendus Geosci. 2009, 341, 591–602. [Google Scholar] [CrossRef]
- Sepulchre, P.; Ramstein, G.; Schuster, M. Modelling the impact of tectonics, surface conditions and sea surface temperatures on Saharan and sub-Saharan climate evolution. Comptes Rendus Geosci. 2009, 341, 612–620. [Google Scholar] [CrossRef]
- Prömmel, K.; Cubasch, U.; Kaspar, F. A regional climate model study of the impact of tectonic and orbital forcing on African precipitation and vegetation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2013, 369, 154–162. [Google Scholar] [CrossRef]
- Sommerfeld, A.; Prömmel, K.; Cubasch, U. The East African Rift System and the impact of orographic changes on regional climate and the resulting aridification. Int. J. Earth Sci. 2016, 105, 1779–1794. [Google Scholar] [CrossRef]
- Moucha, R.; Forte, A.M. Changes in African topography driven by mantle convection. Nat. Geosci. 2011, 4, 707–712. [Google Scholar] [CrossRef]
- Hamon, N.; Sepulchre, P.; Lefebvre, V.; Ramstein, G. The role of eastern Tethys seaway closure in the Middle Miocene Climatic Transition (ca. 14 Ma). Clim. Past 2013, 9, 2687–2702. [Google Scholar] [CrossRef]
- Retallack, G.J.; Dugas, D.P.; Bestland, E.A. Fossil Soils and Grasses of a Middle Miocene East-African Grassland. Science 1990, 247, 1325–1328. [Google Scholar] [CrossRef]
- Jacobs, B.F.; Pan, A.D.; Scotese, C.R. A review of the Cenozoic vegetation history of Africa. In Cenozoic Mammals of Africa; Werdelin, L., Sanders, J., Eds.; University of California Press: Berkeley, CA, USA, 2010; pp. 57–72. ISBN 978-0-520-25721-4. [Google Scholar]
- Mankga, L.T.; Yessoufou, K.; Chitakira, M. On the origin and diversification history of the African genus Encephalartos. S. Afr. J. Bot. 2020, 130, 231–239. [Google Scholar] [CrossRef]
- Bonnefille, R. Cenozoic vegetation, climate changes and hominid evolution in tropical Africa. Glob. Planet. Chang. 2010, 72, 390–411. [Google Scholar] [CrossRef]
- Fer, I.; Tietjen, B.; Jeltsch, F.; Trauth, M.H. Modelling vegetation change during Late Cenozoic uplift of the East African plateaus. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2017, 467, 120–130. [Google Scholar] [CrossRef]
- Hassanin, A.; Delsuc, F.; Ropiquet, A.; Hammer, C.; Jansen van Vuuren, B.; Matthee, C.; Ruiz-Garcia, M.; Catzeflis, F.; Areskoug, V.; Nguyen, T.T.; et al. Pattern and timing of diversification of Cetartiodactyla (Mammalia, Laurasiatheria), as revealed by a comprehensive analysis of mitochondrial genomes. C. R. Biol. 2012, 335, 32–50. [Google Scholar] [CrossRef]
- Portillo, F.; Branch, W.R.; Conradie, W.; Rödel, M.-O.; Penner, J.; Barej, M.F.; Kusamba, C.; Muninga, W.M.; Aristote, M.M.; Bauer, A.M.; et al. Phylogeny and biogeography of the African burrowing snake subfamily Aparallactinae (Squamata: Lamprophiidae). Mol. Phylogenet. Evol. 2018, 127, 288–303. [Google Scholar] [CrossRef]
- Pickford, M.; Senut, B.; Mourer-Chauviré, C. Early Pliocene Tragulidae and peafowls in the Rift Valley, Kenya: Evidence for rainforest in East Africa. Comptes Rendus Palevol 2004, 3, 179–189. [Google Scholar] [CrossRef]
- Jaramillo, C.; Rueda, M.J.; Mora, G. Cenozoic Plant Diversity in the Neotropics. Science 2006, 311, 1893–1896. [Google Scholar] [CrossRef]
- Pan, A.D.; Jacobs, B.F.; Dransfield, J.; Baker, W.J. The fossil history of palms (Arecaceae) in Africa and new records from the Late Oligocene (28-27 Mya) of north-western Ethiopia. Bot. J. Linn. Soc. 2006, 151, 69–81. [Google Scholar] [CrossRef]
- Faye, A.; Pintaud, J.-C.; Baker, W.J.; Vigouroux, Y.; Sonke, B.; Couvreur, T.L.P. Phylogenetics and diversification history of African rattans (Calamoideae, Ancistrophyllinae). Bot. J. Linn. Soc. 2016, 182, 256–271. [Google Scholar] [CrossRef]
- Erkens, R.H.J.; Mennega, E.A.; Westra, L.Y.T. A concise bibliographic overview of Annonaceae. Bot. J. Linn. Soc. 2012, 169, 41–73. [Google Scholar] [CrossRef]
- Massoni, J.; Couvreur, T.L.; Sauquet, H. Five major shifts of diversification through the long evolutionary history of Magnoliidae (angiosperms). BMC Evol. Biol. 2015, 15, 49. [Google Scholar] [CrossRef]
- Boyce, C.K.; Brodribb, T.J.; Feild, T.S.; Zwieniecki, M.A. Angiosperm leaf vein evolution was physiologically and environmentally transformative. Proc. Biol. Sci. 2009, 276, 1771–1776. [Google Scholar] [CrossRef] [PubMed]
- Brodribb, T.J.; Feild, T.S. Leaf hydraulic evolution led a surge in leaf photosynthetic capacity during early angiosperm diversification. Ecol. Lett. 2010, 13, 175–183. [Google Scholar] [CrossRef]
- Koenen, E.J.M.; Clarkson, J.J.; Pennington, T.D.; Chatrou, L.W. Recently evolved diversity and convergent radiations of rainforest mahoganies (Meliaceae) shed new light on the origins of rainforest hyperdiversity. New Phytol. 2015, 207, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Kainulainen, K.; Razafimandimbison, S.G.; Wikström, N.; Bremer, B. Island hopping, long-distance dispersal and species radiation in the Western Indian Ocean: Historical biogeography of the Coffeeae alliance (Rubiaceae). J. Biogeogr. 2017, 44, 1966–1979. [Google Scholar] [CrossRef]
- Janssens, S.B.; Knox, E.B.; Huysmans, S.; Smets, E.F.; Merckx, V.S.F.T. Rapid radiation of Impatiens (Balsaminaceae) during Pliocene and Pleistocene: Result of a global climate change. Mol. Phylogenet. Evol. 2009, 52, 806–824. [Google Scholar] [CrossRef]
- Faye, A.; Deblauwe, V.; Mariac, C.; Richard, D.; Sonké, B.; Vigouroux, Y.; Couvreur, T.L.P. Phylogeography of the genus Podococcus (Palmae/Arecaceae) in Central African rain forests: Climate stability predicts unique genetic diversity. Mol. Phylogenet. Evol. 2016, 105, 126–138. [Google Scholar] [CrossRef]
- Monthe, F.K.; Migliore, J.; Duminil, J.; Bouka, G.; Demenou, B.B.; Doumenge, C.; Blanc-Jolivet, C.; Ekué, M.R.M.; Hardy, O.J. Phylogenetic relationships in two African Cedreloideae tree genera (Meliaceae) reveal multiple rain/dry forest transitions. Perspect. Plant Ecol. Evol. Syst. 2019, 37, 1–10. [Google Scholar] [CrossRef]
- Baker, W.J.; Couvreur, T.L.P. Global biogeography and diversification of palms sheds light on the evolution of tropical lineages. I. Historical biogeography. J. Biogeogr. 2013, 40, 274–285. [Google Scholar] [CrossRef]
- Armstrong, K.E.; Stone, G.N.; Nicholls, J.A.; Valderrama, E.; Anderberg, A.A.; Smedmark, J.; Gautier, L.; Naciri, Y.; Milne, R.; Richardson, J.E. Patterns of diversification amongst tropical regions compared: A case study in Sapotaceae. Front. Genet. 2014, 5, 362. [Google Scholar] [CrossRef] [PubMed]
- Bardon, L.; Chamagne, J.; Dexter, K.G.; Sothers, C.A.; Prance, G.T.; Chave, J. Origin and evolution of Chrysobalanaceae: Insights into the evolution of plants in the Neotropics. Bot. J. Linn. Soc. 2013, 171, 19–37. [Google Scholar] [CrossRef]
- Antonelli, A.; Zizka, A.; Silvestro, D.; Scharn, R.; Cascales-Miñana, B.; Bacon, C.D. An engine for global plant diversity: Highest evolutionary turnover and emigration in the American tropics. Front. Genet. 2015, 6, 130. [Google Scholar] [CrossRef]
- de la Estrella, M.; Forest, F.; Wieringa, J.J.; Fougère-Danezan, M.; Bruneau, A. Insights on the evolutionary origin of Detarioideae, a clade of ecologically dominant tropical African trees. New Phytol. 2017, 214, 1722–1735. [Google Scholar] [CrossRef] [PubMed]
- Rabosky, D.L. Extinction Rates Should Not Be Estimated from Molecular Phylogenies. Evolution 2010, 64, 1816–1824. [Google Scholar] [CrossRef]
- Beaulieu, J.M.; O’Meara, B.C. Extinction can be estimated from moderately sized molecular phylogenies. Evolution 2015, 69, 1036–1043. [Google Scholar] [CrossRef]
- Droissart, V.; Dauby, G.; Hardy, O.J.; Deblauwe, V.; Harris, D.J.; Janssens, S.; Mackinder, B.A.; Blach-Overgaard, A.; Sonké, B.; Sosef, M.S.M.; et al. Beyond trees: Biogeographical regionalization of tropical Africa. J. Biogeogr. 2018, 45, 1153–1167. [Google Scholar] [CrossRef]
| Models | Log Likelihood | AICc | ΔAICc | λ0 | α | µ0 | β |
|---|---|---|---|---|---|---|---|
| Exponential λ (no μ) | −64.9652 | 134.531 | 0 | 0.224 | −0.074 | - | - |
| Constant λ/constant μ | −66.0258 | 136.651 | 2.120 | 0.241 | - | 0.200 | - |
| Constant λ/exponential μ | −64.9436 | 137.150 | 2.619 | 0.198 | - | 0.057 | 0.008 |
| Exponential λ/constant μ | −64.9652 | 137.194 | 2.663 | 0.224 | −0.074 | 1.98 × 10−7 | - |
| Constant λ (no μ) | −68.1224 | 138.435 | 3.904 | 0.127 | - | - | - |
| Exponential λ/exponential μ | −64.9652 | 140.153 | 5.622 | 0.224 | −0.074 | 2.20 × 10−6 | 0.023 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brée, B.; Helmstetter, A.J.; Bethune, K.; Ghogue, J.-P.; Sonké, B.; Couvreur, T.L.P. Diversification of African Rainforest Restricted Clades: Piptostigmateae and Annickieae (Annonaceae). Diversity 2020, 12, 227. https://doi.org/10.3390/d12060227
Brée B, Helmstetter AJ, Bethune K, Ghogue J-P, Sonké B, Couvreur TLP. Diversification of African Rainforest Restricted Clades: Piptostigmateae and Annickieae (Annonaceae). Diversity. 2020; 12(6):227. https://doi.org/10.3390/d12060227
Chicago/Turabian StyleBrée, Baptiste, Andrew J. Helmstetter, Kévin Bethune, Jean-Paul Ghogue, Bonaventure Sonké, and Thomas L. P. Couvreur. 2020. "Diversification of African Rainforest Restricted Clades: Piptostigmateae and Annickieae (Annonaceae)" Diversity 12, no. 6: 227. https://doi.org/10.3390/d12060227
APA StyleBrée, B., Helmstetter, A. J., Bethune, K., Ghogue, J.-P., Sonké, B., & Couvreur, T. L. P. (2020). Diversification of African Rainforest Restricted Clades: Piptostigmateae and Annickieae (Annonaceae). Diversity, 12(6), 227. https://doi.org/10.3390/d12060227





