Diversity and Distribution Patterns of Geometrid Moths (Geometridae, Lepidoptera) in Mongolia
Abstract
1. Introduction
Study Review
2. Materials and Methods
- -
- With all of the words: Mongol (in English Mongolia, in German Mongolei, thus it was better to use only Mongol);
- -
- With at least one of the words: Geometrid OR Larentiinae OR Desmobathrinae OR Ennominae OR Archiearinae OR Geometrinae OR Oenochrominae OR Orthostixinae OR Sterrhinae;
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Subfamily | Species | Author | Year | Reference |
|---|---|---|---|---|
| Archiearinae | Archiearis notha | Hübner | 1802 | [34] |
| Archiearinae | Archiearis parthenias | Linnaeus | 1761 | [34] |
| Archiearinae | Archiearis parthenias sajana | Prout | 1912 | [46] |
| Archiearinae | Leucobrephos middendorfii | Ménétriés | 1858 | [41] |
| Desmobathrinae | Gypsochroa renitidata | Hübner | 1817 | [57] |
| Ennominae | Abraxas grossulariata | Linnaeus | 1758 | [21,34,46,63,65] |
| Ennominae | Abraxas grossulariata dsungarica | Wehrli | 1939 | [38] |
| Ennominae | Alcis deversata | Staudinger | 1892 | [34,39,46,63,65] |
| Ennominae | Alcis extinctaria | Eversmann | 1851 | [23,34,36,39,65,91] |
| Ennominae | Alcis jubata | Thunberg | 1788 | [37] |
| Ennominae | Alcis repandata | Linnaeus | 1758 | [65] |
| Ennominae | Alloharpina conjungens | Alphéraky | 1892 | [33] |
| Ennominae | Amraica superans | Butler | 1878 | [33] |
| Ennominae | Angerona prunaria | Linnaeus | 1758 | [24,34,46,63,65] |
| Ennominae | Angerona prunaria kentearia | Staudinger | 1892 | [39] |
| Ennominae | Angerona prunaria mongoligena | Bryk | 1949 | [62] |
| Ennominae | Apeira syringaria | Linnaeus | 1758 | [63] |
| Ennominae | Apocheima hispidaria | Denis & Schiffermüller | 1775 | [34] |
| Ennominae | Apocolotois almatensis | Djakonov | 1952 | [39] |
| Ennominae | Apocolotois smirnovi | Romanoff | 1885 | [39] |
| Ennominae | Arichanna barteli | Prout | 1915 | [32,45] |
| Ennominae | Arichanna melanaria | Linnaeus | 1758 | [34,46,65,91] |
| Ennominae | Arichanna melanaria decolorata | Staudinger | 1892 | [45] |
| Ennominae | Arichanna melanaria praeolivina | Wehrli | 1933 | [39] |
| Ennominae | Aspitates conspersaria | Staudinger | 1901 | [23,45] |
| Ennominae | Aspitates curvaria | Eversmann | 1852 | [1,8,14] |
| Ennominae | Aspitates forbesi | Munroe | 1963 | [65] |
| Ennominae | Aspitates gilvaria | Denis & Schiffermüller | 1775 | [23,24,34,36,63,91] |
| Ennominae | Aspitates gilvaria minimus | Vojnits | 1975 | [39] |
| Ennominae | Aspitates insignis | Alphéraky | 1883 | [36,39] |
| Ennominae | Aspitates kozhantchikovi | Munroe | 1963 | [36,65] |
| Ennominae | Aspitates mongolicus | Vojnits | 1975 | [39,65] |
| Ennominae | Aspitates mundataria | Stoll | 1782 | [34,46,63,65] |
| Ennominae | Aspitates mundataria uncinataria | Vojnits | 1975 | [39] |
| Ennominae | Aspitates obscurata | Wehrli | 1953 | [33,34,39] |
| Ennominae | Aspitates staudingeri | Vojnits | 1975 | [39] |
| Ennominae | Aspitates taylorae sibirica | Djakonov | 1955 | [36,65] |
| Ennominae | Aspitates tristrigaria | Bremer & Grey | 1853 | [34,37] |
| Ennominae | Astegania honesta | Prout | 1908 | [34] |
| Ennominae | Biston betularia | Linnaeus | 1758 | [34,46,63,91] |
| Ennominae | Biston betularia sibiricus | Fuchs | 1899 | [37] |
| Ennominae | Cabera exanthemata | Scopoli | 1763 | [23,34,46,65] |
| Ennominae | Cabera exanthemata hamica | Wehrli | 1939 | [39] |
| Ennominae | Cabera pusaria | Linnaeus | 1758 | [34,39,63] |
| Ennominae | Calcaritis pallida | Hedemann | 1881 | [47] |
| Ennominae | Chariaspilates formosaria | Eversmann | 1837 | [37] |
| Ennominae | Charissa agnitaria | Staudinger | 1897 | [55] |
| Ennominae | Charissa ambiguata | Duponchel | 1830 | [34,36,46,65] |
| Ennominae | Charissa ambiguata ophthalmicata | Lederer | 1853 | [39] |
| Ennominae | Charissa beljaevi | Erlacher et al., 2017 | 2017 | [55] |
| Ennominae | Charissa bidentatus | Shchetkin & Viidalepp | 1980 | [46] |
| Ennominae | Charissa creperaria | Erschoff | 1877 | [34,55,65] |
| Ennominae | Charissa difficilis | Alphéraky | 1883 | [21,24,34,39,65] |
| Ennominae | Charissa gozmanyi | Vojnits | 1975 | [14] |
| Ennominae | Charissa macguffini | Smiles | 1979 | [65] |
| Ennominae | Charissa ochrofasciata | Staudinger | 1895 | [21,30,34,36,39,55,65] |
| Ennominae | Charissa remmi | Viidalepp | 1988 | [56,63] |
| Ennominae | Charissa sibiriata | Guenée | 1858 | [21,24,30,34,36] |
| Ennominae | Charissa subsplendidaria | Wehrli | 1922 | [63,92] |
| Ennominae | Charissa turfosaria | Wehrli | 1922 | [30,34,39,45,62] |
| Ennominae | Charissa vastaria | Staudinger | 1892 | [30,34] |
| Ennominae | Chiasmia aestimaria | Hübner | 1809 | [65] |
| Ennominae | Chiasmia aestimaria kuldschana | Wehrli | 1940 | [39] |
| Ennominae | Chiasmia clathrata | Linnaeus | 1758 | [23,24,26,34,36,46,63,65,91] |
| Ennominae | Chiasmia clathrata djakonovi | Kardakoff | 1928 | [38,39] |
| Ennominae | Chiasmia saburraria | Eversmann | 1851 | [21,34,65] |
| Ennominae | Chiasmia saburraria kenteata | Staudinger | 1892 | [38] |
| Ennominae | Cleora cinctaria | Denis & Schiffermüller | 1775 | [34,46,63] |
| Ennominae | Colotois pennaria | Linnaeus | 1760 | [46] |
| Ennominae | Deileptenia ribeata | Clerck | 1759 | [63] |
| Ennominae | Digrammia rippertaria | Duponchel | 1830 | [34] |
| Ennominae | Ectropis crepuscularia | Denis & Schiffermüller | 1775 | [34,46] |
| Ennominae | Eilicrinia orias | Wehrli | 1933 | [45] |
| Ennominae | Elophos banghaasi | Wehrli | 1922 | [30,34,45] |
| Ennominae | Ematurga atomaria | Linnaeus | 1758 | [23,24,34,36,46,65] |
| Ennominae | Ematurga atomaria krassnojarscensis | Fuchs | 1899 | [39] |
| Ennominae | Ennomos autumnaria | Werneburg | 1859 | [46] |
| Ennominae | Epione repandaria | Hufnagel | 1767 | [34] |
| Ennominae | Epione vespertaria | Linnaeus | 1767 | [34,39] |
| Ennominae | Epirranthis diversata | Denis & Schiffermüller | 1775 | [63] |
| Ennominae | Erannis jacobsoni | Djakonov | 1926 | [34,46,65] |
| Ennominae | Gnophopsodos ravistriolaria | Wehrli | 1922 | [36] |
| Ennominae | Gnophopsodos ravistriolaria ravistriolaria | Wehrli | 1922 | [55] |
| Ennominae | Gnophopsodos stemmataria | Eversmann | 1848 | [39] |
| Ennominae | Gnophopsodos tholeraria | Püngeler | 1901 | [50] |
| Ennominae | Gnophos bipartitus | Vojnits | 1975 | [39] |
| Ennominae | Gnophos rubefactaria | Püngeler | 1902 | [37] |
| Ennominae | Heliomata glarearia | Denis & Schiffermüller | 1775 | [46] |
| Ennominae | Hypomecis punctinalis | Scopoli | 1763 | [46] |
| Ennominae | Hypomecis roboraria | Denis & Schiffermüller | 1775 | [23,34,39,63] |
| Ennominae | Hypoxystis pluviaria | Fabricius | 1787 | [34,46,63] |
| Ennominae | Isturgia altaica | Vojnits | 1978 | [43] |
| Ennominae | Isturgia arenacearia | Denis & Schiffermüller | 1775 | [63,91] |
| Ennominae | Isturgia arenacearia mongolica | Vojnits | 1974 | [38] |
| Ennominae | Isturgia falsaria | Alphéraky | 1892 | [34] |
| Ennominae | Isturgia halituaria | Guenée | 1858 | [48] |
| Ennominae | Isturgia kaszabi | Vojnits | 1974 | [38] |
| Ennominae | Isturgia murinaria | Denis & Schiffermüller | 1775 | [34,36] |
| Ennominae | Isturgia murinaria uralica | Wehrli | 1937 | [63] |
| Ennominae | Jankowskia bituminaria | Lederer | 1853 | [65] |
| Ennominae | Jankowskia bituminaria raddensis | Wehrli | 1941 | [93] |
| Ennominae | Lomaspilis marginata | Linnaeus | 1758 | [23,34,46,65] |
| Ennominae | Lomaspilis opis amurensis | Hedemann | 1881 | [38] |
| Ennominae | Lomographa buraetica | Staudinger | 1892 | [34] |
| Ennominae | Lomographa temerata | Denis & Schiffermüller | 1775 | [46] |
| Ennominae | Lycia hirtaria | Clerck | 1759 | [63] |
| Ennominae | Lycia lapponaria | Boisduval | 1840 | [37] |
| Ennominae | Macaria alternata | Denis & Schiffermüller | 1775 | [34,46,91] |
| Ennominae | Macaria artesiaria | Denis & Schiffermüller | 1775 | [34,38] |
| Ennominae | Macaria brunneata | Thunberg | 1784 | [36,38,91] |
| Ennominae | Macaria circumflexaria | Eversmann | 1848 | [38,46,63,91] |
| Ennominae | Macaria costimaculata | Graeser | 1888 | [34] |
| Ennominae | Macaria latefasciata | Staudinger | 1896 | [21,34] |
| Ennominae | Macaria liturata | Clerck | 1759 | [65] |
| Ennominae | Macaria liturata pressaria | Christoph | 1893 | [37] |
| Ennominae | Macaria loricaria | Eversmann | 1837 | [36] |
| Ennominae | Macaria notata | Linnaeus | 1758 | [34,63] |
| Ennominae | Macaria notata kirina | Wehrli | 1940 | [38] |
| Ennominae | Macaria serenaria | Staudinger | 1896 | [21,34] |
| Ennominae | Macaria signaria | Hübner | 1809 | [38,46] |
| Ennominae | Macaria wauaria | Linnaeus | 1758 | [34,36] |
| Ennominae | Megalycinia strictaria | Lederer | 1853 | [21,34,39,46,63] |
| Ennominae | Megametopon piperatum | Alphéraky | 1892 | [34,39,65] |
| Ennominae | Narraga fasciolaria | Hufnagel | 1767 | [34,63] |
| Ennominae | Odontopera bidentata | Clerck | 1759 | [21,35,40,47,66] |
| Ennominae | Odontopera bidentata exsul | Tchetrerikov | 1905 | [36,39] |
| Ennominae | Odontopera bidentata rava | Vojnits | 1975 | [39,65] |
| Ennominae | Ourapteryx persica | Ménétriés | 1832 | [34] |
| Ennominae | Ourapteryx sambucaria | Linnaeus | 1758 | [63,65] |
| Ennominae | Perconia strigillaria | Hübner | 1787 | [46,63] |
| Ennominae | Petrophora kaszabi | Vojnits | 1978 | [43] |
| Ennominae | Phaselia narynaria | Oberthür | 1913 | [49] |
| Ennominae | Phaselia serrularia | Eversmann | 1847 | [65] |
| Ennominae | Phthonandria emaria | Bremer | 1864 | [39] |
| Ennominae | Plagodis dolabraria | Linnaeus | 1767 | [34] |
| Ennominae | Plagodis pulveraria | Linnaeus | 1758 | [21,34,65] |
| Ennominae | Plagodis pulveraria singularis | Vojnits | 1975 | [39] |
| Ennominae | Pleogynopteryx bituminaria | Lederer | 1853 | [21,34,39] |
| Ennominae | Pseudopanthera macularia | Linnaeus | 1758 | [34] |
| Ennominae | Pseudopanthera macularia cryptica | Beljaev | 1997 | [94] |
| Ennominae | Selenia dentaria | Fabricius | 1775 | [39] |
| Ennominae | Selenia dentaria alpestris | Wehrli | 1940 | [37] |
| Ennominae | Selenia ononica | Kostjuk | 1991 | [37] |
| Ennominae | Selenia sordidaria | Leech | 1897 | [39] |
| Ennominae | Selenia tetralunaria | Hufnagel | 1767 | [34,36,46,63] |
| Ennominae | Siona lineata | Scopoli | 1763 | [23,26,34,36,39,46,63,65] |
| Ennominae | Spartopteryx kindermannaria | Staudinger | 1871 | [36,39,46] |
| Ennominae | Xandrames dholaria | Moore | 1868 | [33] |
| Ennominae | Yezognophos vittaria | Thunberg | 1792 | [65] |
| Geometrinae | Chlorissa viridata | Linnaeus | 1758 | [34] |
| Geometrinae | Dyschloropsis impararia | Guenée | 1858 | [21,24,34,40,41,65] |
| Geometrinae | Geometra papilionaria | Linnaeus | 1758 | [40,46,63] |
| Geometrinae | Geometra papilionaria herbacearia | Ménétriés | 1859 | [41,65] |
| Geometrinae | Hemistola chrysoprasaria | Esper | 1794 | [46,63] |
| Geometrinae | Hemistola chrysoprasaria lissas | Prout | 1912 | [40] |
| Geometrinae | Hemistola zimmermanni | Hedemann | 1879 | [34,40,41] |
| Geometrinae | Hemithea aestivaria | Hübner | 1799 | [46] |
| Geometrinae | Jodis lactearia | Linnaeus | 1758 | [37] |
| Geometrinae | Microloxia herbaria | Hübner | 1813 | [34,65] |
| Geometrinae | Microloxia herbaria advolata | Eversmann | 1837 | [41] |
| Geometrinae | Thalera chlorosaria | Graeser | 1890 | [34,40,41,91] |
| Geometrinae | Thalera fimbrialis | Scopoli | 1763 | [63] |
| Geometrinae | Thetidia atyche | Prout | 1935 | [40,41] |
| Geometrinae | Thetidia chlorophyllaria | Hedemann | 1879 | [37] |
| Geometrinae | Thetidia correspondens | Alpheraky | 1883 | [49] |
| Geometrinae | Thetidia volgaria | Guenée | 1858 | [21,34,40,46,65] |
| Geometrinae | Thetidia volgaria mongolica | Staudinger | 1897 | [41] |
| Larentiinae | Acasis appensata | Eversmann | 1842 | [46,65] |
| Larentiinae | Anticlea badiata | Denis & Schiffermüller | 1775 | [34,63] |
| Larentiinae | Anticlea derivata | Denis & Schiffermüller | 1775 | [24,34,46,63] |
| Larentiinae | Aplocera plagiata roddi | Vasilenko | 1995 | [59] |
| Larentiinae | Baptria tibiale | Esper | 1804 | [34,42] |
| Larentiinae | Camptogramma bilineata | Linnaeus | 1758 | [46] |
| Larentiinae | Carsia sororiata | Hübner | 1813 | [23,34,36] |
| Larentiinae | Catarhoe cuculata | Hufnagel | 1767 | [37,46,59,63] |
| Larentiinae | Catarhoe rubidata | Denis & Schiffermüller | 1775 | [46] |
| Larentiinae | Chloroclysta miata | Linnaeus | 1758 | [36] |
| Larentiinae | Cidaria distinctata | Staudinger | 1892 | [37] |
| Larentiinae | Cidaria fulvata | Forster | 1771 | [34,44,63,65] |
| Larentiinae | Coenocalpe lapidata | Hübner | 1809 | [21,23,34,36,46,65] |
| Larentiinae | Coenotephria korschunovi | Viidalepp | 1976 | [34] |
| Larentiinae | Colostygia aptata | Hübner | 1813 | [34,65] |
| Larentiinae | Cosmorhoe ocellata | Linnaeus | 1758 | [37] |
| Larentiinae | Dysstroma citrata | Linnaeus | 1761 | [34,46,63,65] |
| Larentiinae | Dysstroma citrata septentrionalis | Heydemann | 1929 | [36] |
| Larentiinae | Dysstroma citratum kamtshadalarium | Beljaev & Vasilenko | 2002 | [48] |
| Larentiinae | Dysstroma infuscata | Tengström | 1869 | [65] |
| Larentiinae | Dysstroma latefasciata | Blöcker | 1908 | [34,44,65] |
| Larentiinae | Dysstroma pseudimmanata | Heydemann | 1929 | [31,34,44] |
| Larentiinae | Dysstroma truncata | Hufnagel | 1767 | [23,31,34,44,65,91] |
| Larentiinae | Dysstroma truncata transbaicalensis | Heydemann | 1929 | [36] |
| Larentiinae | Ecliptopera capitata | Herrich-Schäffer | 1839 | [63] |
| Larentiinae | Ecliptopera dimita | Prout | 1938 | [37] |
| Larentiinae | Ecliptopera umbrosaria | Motschulsky | 1861 | [34] |
| Larentiinae | Ecliptoptera oblongata | Guenée | 1858 | [44] |
| Larentiinae | Electrophaes chimakaleparia | Oberthür | 1893 | [44] |
| Larentiinae | Electrophaes corylata | Thunberg | 1792 | [46,65] |
| Larentiinae | Entephria caesiata | Denis & Schiffermüller | 1775 | [34,36,44] |
| Larentiinae | Entephria kuznetzovi | Viidalepp | 1976 | [34,45] |
| Larentiinae | Entephria tzygankovi | Wehrli | 1929 | [36] |
| Larentiinae | Epirrhoe alternata | Müller | 1764 | [23,34,36] |
| Larentiinae | Epirrhoe hastulata | Hübner | 1790 | [34,36,44,46] |
| Larentiinae | Epirrhoe hastulata reducta | Djakonov | 1929 | [48] |
| Larentiinae | Epirrhoe pupillata | Thunberg | 1788 | [23,34,36,44,46,63,65,91] |
| Larentiinae | Epirrhoe tristata | Linnaeus | 1758 | [23,34,46] |
| Larentiinae | Epirrita autumnata | Borkhausen | 1794 | [21,34,34] |
| Larentiinae | Epirrita autumnata smetanini | Beljaev & Vasilenko | 2002 | [48] |
| Larentiinae | Epirrita autumnata tunkunata | Bang-Haas | 1910 | [36] |
| Larentiinae | Esakiopteryx volitans | Butler | 1878 | [44] |
| Larentiinae | Eulithis mellinata | Fabricius | 1787 | [34] |
| Larentiinae | Eulithis populata | Linnaeus | 1758 | [36,44,63,91] |
| Larentiinae | Eulithis prunata | Linnaeus | 1758 | [34,44,46] |
| Larentiinae | Eulithis pyraliata | Denis & Schiffermüller | 1775 | [23,34,44,46,63,65] |
| Larentiinae | Eulithis pyropata | Hübner | 1809 | [91] |
| Larentiinae | Eulithis testata | Linnaeus | 1761 | [23,34,44,46,63] |
| Larentiinae | Euphyia coangulata | Prout | 1914 | [21,23,24,34,36,44,65] |
| Larentiinae | Euphyia intersecta | Staudinger | 1882 | [21,23,34] |
| Larentiinae | Euphyia unangulata | Haworth | 1809 | [34,46,63,65] |
| Larentiinae | Eupithecia selinata | Herrich-Schäffer | 1861 | [34] |
| Larentiinae | Eupithecia absinthiata | Clerck | 1759 | [95] |
| Larentiinae | Eupithecia actaeata | Walderdorff | 1869 | [52] |
| Larentiinae | Eupithecia addictata | Dietze | 1908 | [37] |
| Larentiinae | Eupithecia aggregata | Guenée | 1858 | [37] |
| Larentiinae | Eupithecia amplexata | Christoph | 1881 | [34,65] |
| Larentiinae | Eupithecia anikini | Mironov & Galsworthy | 2014 | [52] |
| Larentiinae | Eupithecia aporia | Vojnits | 1975 | [41,45] |
| Larentiinae | Eupithecia assimilata | Doubleday | 1856 | [52] |
| Larentiinae | Eupithecia bastelbergeri | Dietze | 1910 | [52] |
| Larentiinae | Eupithecia biornata | Christoph | 1867 | [34,65] |
| Larentiinae | Eupithecia bohatschi | Staudinger | 1897 | [25,34,65] |
| Larentiinae | Eupithecia carpophilata | Staudinger | 1897 | [34,65] |
| Larentiinae | Eupithecia catharinae | Vojnits | 1969 | [65] |
| Larentiinae | Eupithecia centaureata | Denis & Schiffermüller | 1775 | [34,63,65] |
| Larentiinae | Eupithecia chingana | Wehrli | 1926 | [45] |
| Larentiinae | Eupithecia corroborata | Dietze | 1908 | [36] |
| Larentiinae | Eupithecia denotata | Hübner | 1813 | [34] |
| Larentiinae | Eupithecia despectaria | Lederer | 1853 | [34,37] |
| Larentiinae | Eupithecia dissertata | Püngeler | 1905 | [34,36,65] |
| Larentiinae | Eupithecia djakonovi | Shchetkin | 1956 | [37] |
| Larentiinae | Eupithecia dolosa | Vojnits | 1977 | [45] |
| Larentiinae | Eupithecia ericeata | Rambur | 1833 | [52,65] |
| Larentiinae | Eupithecia extensaria | Freyer | 1844 | [36,65] |
| Larentiinae | Eupithecia fennoscandica | Knaben | 1949 | [36,96] |
| Larentiinae | Eupithecia fuscicostata | Christoph | 1887 | [65] |
| Larentiinae | Eupithecia graciliata | Dietze | 1906 | [34] |
| Larentiinae | Eupithecia hannemanni | Vojnits & De Laever | 1973 | [65] |
| Larentiinae | Eupithecia holti | Viidalepp | 1973 | [34,65,97] |
| Larentiinae | Eupithecia illaborata | Dietze | 1904 | [52] |
| Larentiinae | Eupithecia impolita | Vojnits | 1980 | [52] |
| Larentiinae | Eupithecia inculta | Vojnits | 1975 | [65] |
| Larentiinae | Eupithecia indigata | Hübner | 1813 | [63] |
| Larentiinae | Eupithecia innotata | Hufnagel | 1767 | [21,34,65] |
| Larentiinae | Eupithecia intricata | Zetterstedt | 1839 | [34] |
| Larentiinae | Eupithecia inveterata | Vojnits | 1987 | [65] |
| Larentiinae | Eupithecia irriguata | Hübner | 1813 | [65] |
| Larentiinae | Eupithecia kozlovi | Viidalepp | 1973 | [34,97] |
| Larentiinae | Eupithecia kuldschaensis | Staudinger | 1892 | [34,65] |
| Larentiinae | Eupithecia laboriosa | Vojnits | 1977 | [65] |
| Larentiinae | Eupithecia lariciata | Freyer | 1841 | [34,36,65] |
| Larentiinae | Eupithecia leptogrammata | Staudinger | 1882 | [65] |
| Larentiinae | Eupithecia linariata | Denis & Schiffermüller | 1775 | [65] |
| Larentiinae | Eupithecia mima | Mironov | 1989 | [65] |
| Larentiinae | Eupithecia minusculata | Alphéraky | 1883 | [34,65] |
| Larentiinae | Eupithecia mongolica | Vojnits | 1974 | [65] |
| Larentiinae | Eupithecia morosa | Vojnits | 1976 | [65] |
| Larentiinae | Eupithecia munguata | Mironov & Galsworthy | 2014 | [52] |
| Larentiinae | Eupithecia necessaria | Vojnits | 1977 | [41,45] |
| Larentiinae | Eupithecia nephelata | Staudinger | 1897 | [21,23,34,65] |
| Larentiinae | Eupithecia nobilitata | Staudinger | 1882 | [36,65] |
| Larentiinae | Eupithecia olgae | Mironov | 1986 | [52] |
| Larentiinae | Eupithecia opisthographata | Dietze | 1906 | [34] |
| Larentiinae | Eupithecia perfuscata | Vojnits | 1975 | [65] |
| Larentiinae | Eupithecia pernotata | Guenée | 1858 | [48] |
| Larentiinae | Eupithecia pimpinellata | Hübner | 1813 | [34,65] |
| Larentiinae | Eupithecia propria | Vojnits | 1977 | [65] |
| Larentiinae | Eupithecia pusillata | Denis & Schiffermüller | 1775 | [52] |
| Larentiinae | Eupithecia pygmaeata | Hübner | 1799 | [65] |
| Larentiinae | Eupithecia recens | Dietze | 1904 | [34,36] |
| Larentiinae | Eupithecia relaxata | Dietze | 1904 | [65] |
| Larentiinae | Eupithecia repentina | Vojnits & De Laever | 1978 | [52] |
| Larentiinae | Eupithecia rubellata | Dietze | 1904 | [41,45] |
| Larentiinae | Eupithecia saisanaria | Staudinger | 1882 | [52] |
| Larentiinae | Eupithecia satyrata | Hübner | 1813 | [36] |
| Larentiinae | Eupithecia selinata | Herrich-Schäffer | 1861 | [95] |
| Larentiinae | Eupithecia simpliciata | Haworth | 1809 | [52] |
| Larentiinae | Eupithecia sinuosaria | Eversmann | 1848 | [23,34,36] |
| Larentiinae | Eupithecia subbrunneata | Dietze | 1904 | [52] |
| Larentiinae | Eupithecia subexiguata | Vojnits | 1974 | [65] |
| Larentiinae | Eupithecia subfuscata | Haworth | 1809 | [34] |
| Larentiinae | Eupithecia suboxydata | Staudinger | 1897 | [65,98] |
| Larentiinae | Eupithecia subtacincta | Hampson | 1895 | [37] |
| Larentiinae | Eupithecia subumbrata | Denis & Schiffermüller | 1775 | [23,34,65] |
| Larentiinae | Eupithecia succenturiata | Linnaeus | 1758 | [95] |
| Larentiinae | Eupithecia sutiliata | Christoph | 1877 | [65] |
| Larentiinae | Eupithecia thalictrata | Püngeler | 1902 | [52] |
| Larentiinae | Eupithecia undata | Freyer | 1840 | [65] |
| Larentiinae | Eupithecia veratraria | Herrich-Schäffer | 1848 | [95] |
| Larentiinae | Eupithecia vicina | Mironov | 1989 | [65] |
| Larentiinae | Eupithecia virgaureata | Doubleday | 1861 | [21,23,34,65] |
| Larentiinae | Eupithecia vulgata | Haworth | 1809 | [21,23,34] |
| Larentiinae | Eupithecia vulgata lepsaria | Staudinger | 1882 | [37] |
| Larentiinae | Eupithecis unedonata | Mabille | 1868 | [33] |
| Larentiinae | Eustroma reticulatum obsoleta | Djakonov | 1929 | [48] |
| Larentiinae | Gagitodes sagittata | Fabricius | 1787 | [44,46,63] |
| Larentiinae | Gagitodes sagittata albiflua | Prout | 1939 | [48] |
| Larentiinae | Horisme aemulata | Hübner | 1813 | [23,34,46,65] |
| Larentiinae | Horisme aquata | Hübner | 1813 | [23,34,36,46,65,91] |
| Larentiinae | Horisme falcata | Bang-Haas | 1907 | [25,27,34,36,63,65] |
| Larentiinae | Horisme incurvaria | Erschoff | 1877 | [34,36,65] |
| Larentiinae | Horisme lucillata | Guenée | 1858 | [23,34] |
| Larentiinae | Horisme parcata | Püngeler | 1909 | [65] |
| Larentiinae | Horisme scotosiata | Guenée | 1858 | [21,23,34,63,65] |
| Larentiinae | Horisme tersata | Denis & Schiffermüller | 1775 | [34,65] |
| Larentiinae | Horisme tersata tetricata | Guenée | 1858 | [37] |
| Larentiinae | Horisme vitalbata | Denis & Schiffermüller | 1775 | [21,23,34,36,46,65] |
| Larentiinae | Hydrelia flammeolaria | Hufnagel | 1767 | [44,46] |
| Larentiinae | Hydria cervinalis | Scopoli | 1763 | [34] |
| Larentiinae | Hydria undulata | Linnaeus | 1758 | [34,65] |
| Larentiinae | Hydriomena furcata | Thunberg | 1784 | [21,23,34,36,44] |
| Larentiinae | Hydriomena impluviata | Denis & Schiffermüller | 1775 | [21,34,36] |
| Larentiinae | Hydriomena impluviata djakonovi | Beljaev & Vasilenko | 2002 | [48] |
| Larentiinae | Hydriomena ruberata | Freyer | 1831 | [65] |
| Larentiinae | Juxtephria consentaria | Freyer | 1846 | [36,44,65] |
| Larentiinae | Kyrtolitha obstinata | Staudinger | 1892 | [34] |
| Larentiinae | Laciniodes denigrata abiens | Prout | 1938 | [33] |
| Larentiinae | Lampropteryx albigirata | Kollar | 1848 | [65] |
| Larentiinae | Lampropteryx jameza | Butler | 1898 | [37] |
| Larentiinae | Lampropteryx minna | Butler | 1881 | [44,45,65] |
| Larentiinae | Lampropteryx suffumata | Denis & Schiffermüller | 1775 | [63] |
| Larentiinae | Leptostegna tenerata | Christoph | 1881 | [99] |
| Larentiinae | Lithostege coassata mongolica | Vojnits | 1978 | [42] |
| Larentiinae | Lithostege coassata ochraceata | Staudinger | 1897 | [42,65] |
| Larentiinae | Lithostege mesoleucata | Püngeler | 1899 | [34,42] |
| Larentiinae | Lithostege pallescens | Staudinger | 1897 | [21,34] |
| Larentiinae | Lobophora halterata | Hufnagel | 1767 | [44,46] |
| Larentiinae | Martania taeniata | Stephens | 1831 | [44] |
| Larentiinae | Mesoleuca albicillata | Linnaeus | 1758 | [34,37,44,46] |
| Larentiinae | Mesotype verberata | Scopoli | 1763 | [44] |
| Larentiinae | Nebula lamata | Staudinger | 1897 | [21,34] |
| Larentiinae | Nebula mongoliata | Staudinger | 1897 | [21,34,44,65] |
| Larentiinae | Odezia atrata | Linnaeus | 1758 | [23,34] |
| Larentiinae | Orthonama obstipata | Fabricius | 1794 | [34] |
| Larentiinae | Pelurga comitata | Linnaeus | 1758 | [34,44,63,65] |
| Larentiinae | Pelurga taczanowskiaria | Oberthür | 1880 | [63,91] |
| Larentiinae | Perizoma alchemillata | Linnaeus | 1758 | [34,36,44] |
| Larentiinae | Perizoma bifaciata | Haworth | 1809 | [65] |
| Larentiinae | Perizoma blandiata | Denis & Schiffermüller | 1775 | [23,34] |
| Larentiinae | Perizoma hydrata | Treitschke | 1829 | [36,44,65] |
| Larentiinae | Perizoma minorata | Treitschke | 1828 | [46] |
| Larentiinae | Phibalapteryx virgata | Hufnagel | 1767 | [34,36,42,91] |
| Larentiinae | Photoscotosia palaearctica | Staudinger | 1882 | [23,34] |
| Larentiinae | Plemyria rubiginata | Denis & Schiffermüller | 1775 | [34,44,65] |
| Larentiinae | Plesioscotosia pulchrata | Alphéraky | 1883 | [23,34] |
| Larentiinae | Povilasia kashghara | Moore | 1878 | [51] |
| Larentiinae | Pseudentephria remmi | Viidalepp | 1976 | [35] |
| Larentiinae | Pseudobaptria corydalaria | Graeser | 1889 | [34] |
| Larentiinae | Rheumaptera hastata | Linnaeus | 1758 | [34,36,44,46,65] |
| Larentiinae | Rheumaptera subhastata | Nolcken | 1870 | [36] |
| Larentiinae | Rheumaptera subhastata commixta | Matsumura | 1925 | [48] |
| Larentiinae | Schistostege nubilaria | Hübner | 1799 | [23,34,36,42,65] |
| Larentiinae | Scotopteryx chenopodiata | Linnaeus | 1758 | [23,34,46,63,65] |
| Larentiinae | Scotopteryx chenopodiata sibirica | Bang-Haas | 1907 | [42] |
| Larentiinae | Scotopteryx golovushkini | Kostjuk | 1991 | [65] |
| Larentiinae | Scotopteryx sinensis | Alphéraky | 1883 | [23,34] |
| Larentiinae | Scotopteryx transbaicalica | Djakonov | 1955 | [28,34,36] |
| Larentiinae | Spargania luctuata | Denis & Schiffermüller | 1775 | [23,34,44,63,65] |
| Larentiinae | Stamnodes danilovi | Erschoff | 1877 | [21,23,34,36,42,65] |
| Larentiinae | Stamnodes danilovi djakonovi | Alphéraky | 1916 | [33] |
| Larentiinae | Stamnodes pauperaria | Eversmann | 1848 | [65] |
| Larentiinae | Thera obeliscata | Hübner | 1787 | [34,91] |
| Larentiinae | Thera variata | Denis & Schiffermüller | 1775 | [23,34] |
| Larentiinae | Trichopterigia consobrinaria | Leech | 1891 | [44] |
| Larentiinae | Trichopteryx carpinata | Borkhausen | 1794 | [65] |
| Larentiinae | Xanthorhoe abrasaria | Herrich-Schäffer | 1855 | [36,44,65] |
| Larentiinae | Xanthorhoe deflorata | Erschoff | 1877 | [23,34,44,65] |
| Larentiinae | Xanthorhoe montanata | Denis & Schiffermüller | 1775 | [34,36,46] |
| Larentiinae | Xanthorhoe quadrifasiata tannuensis | Prout | 1924 | [45,63] |
| Larentiinae | Xanthorhoe sajanaria | Prout | 1914 | [36,44] |
| Larentiinae | Xanthorhoe sajanaria djakonovi | Vasilenko | 1995 | [100] |
| Larentiinae | Xanthorhoe spadicearia | Denis & Schiffermüller | 1775 | [44,46] |
| Larentiinae | Xanthorhoe stupida aridela | Prout | 1937 | [37] |
| Larentiinae | Zola terranea | Butler | 1879 | [34] |
| Sterrhinae | Cleta jacutica (Axel Hausmann: probably only one Cleta species occurring in Mongolia) | Viidalepp | 1976 | [36] |
| Sterrhinae | Cleta perpusillaria | Eversmann | 1847 | [65] |
| Sterrhinae | Cyclophora albipunctata | Hufnagel | 1767 | [46] |
| Sterrhinae | Cyclophora pendularia | Clerck | 1759 | [46] |
| Sterrhinae | Glossotrophia rufotinctata | Prout | 1913 | [49] |
| Sterrhinae | Holarctias rufinaria | Staudinger | 1861 | [58] |
| Sterrhinae | Idaea aureolaria | Denis & Schiffermüller | 1775 | [23,34,46] |
| Sterrhinae | Idaea biselata extincta | Staudinger | 1897 | [101] |
| Sterrhinae | Idaea muricata | Hufnagel | 1967 | [34] |
| Sterrhinae | Idaea muricata minor | Sterneck | 1727 | [40] |
| Sterrhinae | Idaea nitidata | Herrich-Schäffer | 1861 | [37] |
| Sterrhinae | Idaea nudaria | Christoph | 1881 | [37] |
| Sterrhinae | Idaea pallidata | Denis & Schiffermüller | 1775 | [34,40] |
| Sterrhinae | Idaea rufaria | Hübner | 1799 | [65] |
| Sterrhinae | Idaea rusticata | Denis & Schiffermüller | 1775 | [40,63] |
| Sterrhinae | Idaea serpentata | Hufnagel | 1767 | [23,34,36,41,63] |
| Sterrhinae | Idaea straminata | Borkhausen | 1794 | [34,91] |
| Sterrhinae | Idaea straminata sibirica | Djakonov | 1926 | [40] |
| Sterrhinae | Ochodontia adustaria | Fischer de Waldheim | 1840 | [23,34,65] |
| Sterrhinae | Rhodometra sacraria | Linnaeus | 1767 | [34] |
| Sterrhinae | Rhodostrophia jacularia | Hübner | 1813 | [21,23,34,36,40,41,63,65] |
| Sterrhinae | Rhodostrophia tyugui | Vasilenko | 1998 | [64] |
| Sterrhinae | Rhodostrophia ustyuzhanini | Vasilenko | 2006 | [49] |
| Sterrhinae | Rhodostrophia vibicaria | Clerck | 1759 | [34,46,63] |
| Sterrhinae | Scopula aequifasciata | Christoph | 1881 | [47] |
| Sterrhinae | Scopula albiceraria | Herrich-Schäffer | 1847 | [21,25,34,65] |
| Sterrhinae | Scopula albiceraria vitellinaria | Eversmann | 1851 | [40,41] |
| Sterrhinae | Scopula beckeraria | Lederer | 1853 | [34,40,41,63,65] |
| Sterrhinae | Scopula beckeraria amataria | Wehrli | 1927 | [36,40,65] |
| Sterrhinae | Scopula cajanderi | Herz | 1903 | [41,46] |
| Sterrhinae | Scopula caricaria | Reutti | 1853 | [46] |
| Sterrhinae | Scopula contramutata | Prout | 1920 | [34] |
| Sterrhinae | Scopula cumulata | Alpheraky | 1883 | [65] |
| Sterrhinae | Scopula decorata | Denis & Schiffermüller | 1775 | [21,23,34,41,63,65] |
| Sterrhinae | Scopula decorata przewalskii | Viidalepp | 1975 | [36,40,65] |
| Sterrhinae | Scopula dignata | Guenée | 1858 | [34] |
| Sterrhinae | Scopula floslactata | Haworth | 1809 | [37] |
| Sterrhinae | Scopula frigidaria | Möschler | 1860 | [47] |
| Sterrhinae | Scopula immorata | Linnaeus | 1758 | [23,34,36,40,46,63,65] |
| Sterrhinae | Scopula immutata contramutata | Prout | 1913 | [58] |
| Sterrhinae | Scopula impersonata | Walker | 1861 | [34] |
| Sterrhinae | Scopula impersonata macescens | Butler | 1879 | [40,41] |
| Sterrhinae | Scopula incanata | Linnaeus | 1758 | [34,41,65] |
| Sterrhinae | Scopula latelineata | Graeser | 1892 | [49] |
| Sterrhinae | Scopula marginepunctata | Goeze | 1781 | [23,34,63] |
| Sterrhinae | Scopula nigropunctata | Hufnagel | 1767 | [34] |
| Sterrhinae | Scopula nigropunctata subcandidata | Walker | 1863 | [37] |
| Sterrhinae | Scopula ornata | Scopoli | 1763 | [34,41,46] |
| Sterrhinae | Scopula permutata | Staudinger | 1897 | [34,39,65] |
| Sterrhinae | Scopula rubiginata | Hufnagel | 1767 | [34,40,41,63,65,91] |
| Sterrhinae | Scopula ternata | Schrank | 1802 | [25,34,36,46] |
| Sterrhinae | Scopula tessellaria | Boisduval | 1840 | [65] |
| Sterrhinae | Scopula umbelaria | Hübner | 1813 | [34,46,63] |
| Sterrhinae | Scopula umbelaria graeseri | Prout | 1935 | [41,65] |
| Sterrhinae | Scopula virgulata | Denis & Schiffermüller | 1775 | [23,34,40,41,46,63,65,91] |
| Sterrhinae | Scopula virgulata substrigaria | Staudinger | 1900 | [36] |
| Sterrhinae | Timandra griseata | Petersen | 1902 | [46] |
| Sterrhinae | Timandra paralias | Prout | 1935 | [34,40] |
| Sterrhinae | Timandra recompta | Prout | 1930 | [40,63] |
References
- Summerville, K.S.; Ritter, L.M.; Crist, T.O. Forest moth taxa as indicators of lepidopteran richness and habitat disturbance: A preliminary assessment. Biol. Conserv. 2004, 116, 9–18. [Google Scholar] [CrossRef]
- Pöyry, J.; Lindgren, S.; Salminen, J.; Kuussaari, M. Responses of butterfly and moth species to restored cattle grazing in semi-natural grasslands. Biol. Conserv. 2005, 122, 465–478. [Google Scholar] [CrossRef]
- Bachand, M.; Pellerin, S.; Côté, S.D.; Moretti, M.; De Cáceres, M.; Brousseau, P.-M.; Cloutier, C.; Hébert, C.; Cardinal, É.; Martin, J.-L. Species indicators of ecosystem recovery after reducing large herbivore density: Comparing taxa and testing species combinations. Ecol. Indic. 2014, 38, 12–19. [Google Scholar] [CrossRef]
- Enkhtur, K.; Pfeiffer, M.; Lkhagva, A.; Boldgiv, B. Response of moths (Lepidoptera: Heterocera) to livestock grazing in Mongolian rangelands. Ecol. Indic. 2017, 72, 667–674. [Google Scholar] [CrossRef]
- Frequently Asked Questions|The Lepidopterists’ Society. Available online: https://www.lepsoc.org/content/frequently-asked-questions#9 (accessed on 17 January 2020).
- Hahn, M.; Brühl, C.A. The secret pollinators: An overview of moth pollination with a focus on Europe and North America. Arthropod Plant Interact. 2016, 10, 21–28. [Google Scholar] [CrossRef]
- Scoble, M.J. The Lepidoptera. Form, Function and Diversity; Oxford University Press: Oxford, UK, 1992. [Google Scholar]
- Ashton, L.A.; Kitching, R.L.; Maunsell, S.; Bito, D.; Putland, D. Macrolepidopteran assemblages along an altitudinal gradient in subtropical rainforest-exploring indicators of climate change. Mem. Qld. Mus. 2011, 55, 375–389. [Google Scholar]
- Choi, S.-W. Patterns of species description and species richness of Geometrid moths (Lepidoptera: Geometridae) on the Korean peninsula. Zool. Sci. 2006, 23, 155–160. [Google Scholar] [CrossRef]
- Alonso-Rodríguez, A.M.; Finegan, B.; Fiedler, K. Neotropical moth assemblages degrade due to oil palm expansion. Biodivers. Conserv. 2017, 26, 2295–2326. [Google Scholar] [CrossRef]
- Hilt, N.; Fiedler, K. Diversity and composition of Arctiidae moth ensembles along a successional gradient in the Ecuadorian Andes. Divers. Distrib. 2005, 11, 387–398. [Google Scholar] [CrossRef]
- Brehm, G.; Murillo-Ramos, L.; Sihvonen, P.; Hausmann, A.; Schmidt, B.C.; Õunap, E.; Moser, A.; Mörtter, R.; Bolt10, D.; Bodner11, F. New World geometrid moths (Lepidoptera: Geometridae): Molecular phylogeny, biogeography, taxonomic updates and description of 11 new tribes. Arthropod Syst. Philogeny 2019, 77, 457–486. [Google Scholar]
- Sihvonen, P.; Siljander, M. Species diversity and geographical distribution of Scopulini moths (Lepidoptera: Geometridae, Sterrhinae) on a world-wide scale. Biodivers. Conserv. 2005, 14, 703–721. [Google Scholar] [CrossRef]
- Pfeiffer, M.; Dulamsuren, C.; Wesche, K. Grasslands and Shrublands of Mongolia. In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Enkhnasan, D.; Boldgiv, B. Biogeography of predaceous diving beetles (Coleoptera, Dytiscidae) of Mongolia. ZooKeys 2019, 853, 87. [Google Scholar] [CrossRef] [PubMed]
- WWF About Mongolia|WWF. Available online: http://mongolia.panda.org/en/about_mongolia/ (accessed on 27 December 2019).
- Paknia, O.; Grundler, M.; Pfeiffer, M. Species richness and niche differentiation of darkling beetles (Coleoptera: Tenebrionidae) in Mongolian steppe ecosystems. In Steppe Ecosystems: Biological Diversity, Management and Restoration, 1st ed.; Chapter: Species Richness and Niche Differentiation of Darkling beetles (Coleoptera: Tenebrionidae) in Mongolian Steppe Ecosystems; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 47–72. [Google Scholar]
- Pfeiffer, M.; Dulamsuren, C.; Jäschke, Y.; Wesche, K. Grasslands of China and Mongolia: Spatial extent, land use and conservation. In Grasslands of the World: Diversity, Management and Conservation; CRC Press: Boca Raton, FL, USA, 2018; pp. 170–198. [Google Scholar]
- Suzuki, Y. Conflict between mining development and nomadism in Mongolia. In The Mongolian Ecosystem Network; Springer: Tokyo, Japan, 2013; pp. 269–294. [Google Scholar]
- Staudinger, O. Lepidopteren des Kentei-Gebirges. Dtsch. Entomol. Z. Iris 1892, 5, 300–393. [Google Scholar]
- Staudinger, O. Ueber Lepidopteren von Uliassutai. Dtsch. Entomol. Z. Iris 1896, 9, 240–283. [Google Scholar]
- Staudinger, O. Neue Lepidopteren des palaearktischen Faunengebiets. Dtsch. Entomol. Z. Iris 1899, 12, 354–356. [Google Scholar]
- Staudinger, O.; Rebel, H. Catalog der Lepidopteren des Palaearctischen Faunengebietes, 3. aufl. des Cataloges der Lepidopteren des Europaischen Faunengebietes, Berlin, 1901. Distrib. Recurvaric Nanella 1901, 2, 155. [Google Scholar]
- Alberti, B. Lepidopteren aus der Mongolischen Volksrepublik. ergebnisse der Mongolisch-Deutschen Biologischen Expeditionen seit 1962, nr. 58. Dtsch. Entomol. Z. 1971, 18, 361. [Google Scholar]
- Moucha, J. Ergebnisse der 1. mongolische-tschechoslowakischen entomologisch-botanischen Expedition. 2. Lepidoptera. Acta Faun. Entomol. Musei Natl. Pragae 1967, 12, 35–42. [Google Scholar]
- Grum-Grshimailo, G. Butterflies collected in the vicinity of Troitskosavsk in 1896–1897 and in northern Mongolia in 1907. Tr. Troitskosavsky Dep. Russ. Geographer. Ob-VA 1911, 13, 65–67. [Google Scholar]
- Djakonov, A.M. To the knowledge of the fauna of Geometridae of the Minusinsk Territory. Yearb. State Mus. Named NM Martyanov 1926, 4, 1–78. [Google Scholar]
- Djakonov, A.M. New and little known Lepidoptera, Geometridae of the fauna of the USSR. Tr. Zool. Inst. Akad. Nauk SSSR 1955, 18, 314–319. [Google Scholar]
- Dietze, K. Biologie der Eupithecien (Tafeln &Text); R. Friedländer & Sohn: Berlin, Germany, 1910; Volume 1, pp. 83–86. [Google Scholar]
- Wehrli, E. Ueber neue schweizerische und zentral asiatische Gnophos-Arten und mikroskopische Bearbeitung einzelner Gruppen der Gattung. Dtsch. Entomol. Z. Iris 1922, 36, 1–30. [Google Scholar]
- Heydemann, F. Monographie der palaarktischen Arten des Subgenus Dysstroma Hub.(truncata-citrata-Gruppe) der Gattung Cidaria. Mitt. Münchner Entomol. Ges. 1929, 19, 207–292. [Google Scholar]
- Prout, L.B. Die Spanner des Palaearktischen Faunengebietes. In Die Gross-Schmetterlinge der Erde; Seitz, A., Ed.; Verlag A. Kernen: Stuttgart, Germany, 1912; Volume 4, pp. 1–479. [Google Scholar]
- Wehrli, E. Die Spanner des Palaearktischen Faunengebietes. In Die Gross-Schmetterlinge der Erde; Seitz, A., Ed.; Verlag A. Kernen: Stuttgart, Germany, 1939–1954; Volume 4, pp. 254–766. [Google Scholar]
- Viidalepp, J. On the fauna of Geometrid moths (Lepidoptera, Geometridae) of the Mongolian People’s Republic. Insects Mong. 1975, 3, 438–490. [Google Scholar]
- Viidalepp, J.R. New genera and species of geometrid moths (Lepidoptera, Geometridae) from Southern Siberia and Mongolia. Insects Mong. 1976, 381–402. [Google Scholar]
- Viidalepp, J.; Soljanikov, V.P. On the geometrid moths (Lepidoptera, Geometridae) of the northern part of the Mongolian People’s Republic. Insects Mong. 1977, 5, 620–641. [Google Scholar]
- Viidalepp, J. Checklist of the Geometridae (Lepidoptera) of the Former U.S.S.R.; Apollo Books: Stenstrup, Denmark, 1996; ISBN 978-87-88757-05-7. [Google Scholar]
- Vojnits, A. Abraxini and Semiothisini species from Mongolia (Lepidoptera, Geometridae: Ennominae). Természettud. Múz. Evk 1974, 66, 281–287. [Google Scholar]
- Vojnits, A.M. Ennominae species from Mongolia, II.(Lepidoptera, Geometridae). In Annales historico-naturales Musei nationalis hungarici; Népm\Huvelési Propaganda Iroda: Budapest, Hungary; Volume 67, pp. 183–206.
- Vojnits, A. Geometrinae and Sterrhinae from Mongolia (Lepidoptera, Geometridae). In Annales historico-naturales Musei nationalis hungarici; Népm\Huvelési Propaganda Iroda: Budapest, Hungary, 1976; Volume 68, pp. 169–174. [Google Scholar]
- Vojnits, A. Archieariinae, Rhodometrinae, Geometrinae II, Sterrhinae II and Ennominae III (Lepidoptera, Geometridae) from Mongolia. Ann. Hist. Nat. Musei Nat. Hung. 1977, 69, 165–175. [Google Scholar]
- Vojnits, A.M. Larentiinae (Lepidoptera, Geometridae) from Mongolia. I. Ann. Hist. Nat. Musei Natl. Hung. A Termtud. Muz. Evkv. 1978, 70, 191–195. [Google Scholar]
- Vojnits, A. Tephrina altaica sp. n. from Mongolia (Ennominae IV: Lepidoptera, Geometridae). Folia Entomol. Hung. Rovart. Kozl. Ser. Nova 1978, 31, 205–207. [Google Scholar]
- Vojnits, A.M. Larentiinae from Mongolia II (Lepidoptera, Geometridae). Folia Entomol. Hung. 1979, 32, 207–212. [Google Scholar]
- Scoble, M.J. Natural History Museum (London, E. Geometrid Moths of the World: A Catalogue: (Lepidoptera, Geometridae); CSIRO: Stenstrup, Denmark; Apollo Books: Stenstrup, Denmark; Collingwood, VIC.: Stenstrup, Denmark, 1999; ISBN 978-0-643-06302-0. [Google Scholar]
- Mühlenberg, M.; Enkhmaa, A.; Mühlenberg-Horn, E. Biodiversity Survey at Khonin Nuga Research Station West-Khentey; Ulaanbaatar, Mongolia, 2011. [Google Scholar]
- Dubatolov, V.V.; Dudko, R.Z.; Mordkovich, V.G.; Korsun, O.V.; Chernyshev, S.E.; Logunov, D.V.; Marusik, Y.M.; Legalov, A.A.; Vasilenko, S.V.; Grishina, L.G. Biodiversity of Sokhondinsky Reserve. Arthropods Novosib. Chita 2004, 416, 4. (In Russian) [Google Scholar]
- Beljaev, E.A.; Vasilenko, S.V. An annotated checklist of geometrid moths (Lepidoptera: Geometridae) from the Kamchatka Peninsula and adjacent islands. Entomol. Fenn. 2002, 13, 195–235. [Google Scholar] [CrossRef]
- Vasilenko, S.V. New records of geometer-moths (Lepidoptera, Geometridae) from West Mongolia. Evraziatskii Entomol. Zhurnal Euroasian Entomol. J. 2006, 5, 344–346. (In Russian) [Google Scholar]
- Vasilenko, S.V. Interesting findings of rare geometrid species (Lepidoptera, Geometridae) in the Altai. Entomol. Rev. 2011, 91, 405–409. [Google Scholar] [CrossRef]
- Vasilenko, S.V.; Ivonin, V.V.; Knyazev, S.A. New records of geometrid moths (Lepidoptera, Geometridae) from the Russian Altai. Euroasian Entomol. J. 2017, 16, 99–103. [Google Scholar]
- Mironov, V.; Galsworthy, A. A survey of Eupithecia Curtis, 1825 (Lepidoptera, Geometridae, Larentiinae) in Mongolia with descriptions of two new species. Zootaxa 2014, 3774, 101–130. [Google Scholar] [CrossRef]
- Erlacher, S.; Erlacher, J. A systematic revision of the genus Gnophopsodos Wehrli, 1945, with description of two new species (Lepidoptera: Geometridae). Zootaxa 2016, 4169, 435–456. [Google Scholar] [CrossRef]
- Erlacher, J.; Erlacher, S. Description of the female of Gnophopsodos ravistriolaria (Wehrli, 1922)(Lepidoptera: Geometridae). Zootaxa 2019, 4586, 391–394. [Google Scholar] [CrossRef]
- Erlacher, S.; Palma, L.M.; Erlacher, J. A systematic revision of Charissa, subgenus Pterygnophos Wehrli, 1951, with description of a new species (Lepidoptera: Geometridae). Zootaxa 2017, 4341, 400. [Google Scholar] [CrossRef]
- Makhov, I.A.; Beljaev, E.A. New data on geometrid moths (Lepidoptera: Geometridae) of the Baikal region, Russia. Far East. Entomol. 2019, 391, 1–23. [Google Scholar] [CrossRef]
- Hausmann, A. The Geometrid Moths of Europe; Apollo Books: Stenstrup, Denmark, 2001; Volume 1, ISBN 978-87-88757-35-4. [Google Scholar]
- Hausmann, A. The Geometrid Moths of Europe; Apollo Books: Stenstrup, Denmark, 2004; Volume 2, ISBN 978-87-88757-37-8. [Google Scholar]
- Hausmann, A.; Mironov, V.; Viidalepp, J. The Geometrid Moths of Europe; Apollo Books: Stenstrup, Denmark, 2012; Volume 3, ISBN 978-87-88757-39-2. [Google Scholar]
- Mironov, V. The Geometrid Moths of Europe; Apollo Books: Stenstrup, Denmark, 2003; Volume 4, ISBN 978-87-88757-40-8. [Google Scholar]
- Skou, P.; Sihvonen, P. The Geometrid Moths of Europe; Brill: Leiden, The Netherlands, 2015; Volume 5, ISBN 978-90-04-25220-2. [Google Scholar]
- Müller, B.; Erlacher, S.; Hausmann, A.; Rajaei, H.; Sihvonen, P.; Skou, P. The Geometrid Moths of Europe; Brill: Leiden, The Netherlands, 2019; Volume 6 (Part 1–2), ISBN 978-90-04-25222-6. [Google Scholar]
- Enkhtur, K.; Pfeiffer, M.; Munkhbat, U.; Boldgiv, B. Diversity of moths (Lepidoptera: Heterocera) in north-central Mongolia. Erforsch. Biol. Ressour. Mong. 2020, 14. in press. [Google Scholar]
- Geometridae Collection of SZMN. Available online: http://szmn.eco.nsc.ru/Lepidop/Geometr/Geometr.htm (accessed on 10 March 2020).
- GBIF.org GBIF Occurrence Download. Available online: https://doi.org/10.15468/dl.a9dxof (accessed on 11 November 2019).
- Ratnasingham, S.; Hebert, P.D.N. BOLD: The Barcode of Life Data System (www.barcodinglife.org). Mol. Ecol. Notes 2007, 7, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Google Earth. Available online: https://earth.google.com/web/@0,0,0a,22251752.77375655d,35y,0h,0t,0r/data=KAE (accessed on 8 November 2019).
- Schaffer, M.; Beccaloni, G.; Scoble, M.; Kitching, I.; Simonsen, T.; Robinson, G.; Pitkin, B.; Hine, A.; Lyal, C. The Global Lepidoptera Names Index (LepIndex). World Wide Web Electronic Publication. Available online: https://www.nhm.ac.uk/our-science/data/lepindex/lepindex/ (accessed on 6 November 2019).
- Chao, A.; Colwell, R.K.; Chiu, C.-H.; Townsend, D. Seen once or more than once: Applying Good–Turing theory to estimate species richness using only unique observations and a species list. Methods Ecol. Evol. 2017, 8, 1221–1232. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.C.; Sander, E.L.; Ma, K.H.; Colwell, R.K.; Ellison, A.M. Rarefaction and extrapolation with Hill numbers: A framework for sampling and estimation in species diversity studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef]
- Hsieh, T.C.; Ma, K.H.; Chao, A. iNEXT: An R package for interpolation and extrapolation of species diversity (Hill numbers). Methods Ecol. Evol. 2016. [Google Scholar] [CrossRef]
- Wickham, H.; Hester, J.; Chang, W.; RStudio; R Core team (Some namespace and vignette code extracted from base R). Devtools: Tools to Make Developing R Packages Easier; R package version 2.2.2. 2020. Available online: https://CRAN.R-project.org/package=devtools (accessed on 22 March 2020).
- Wickham, H.; Chang, W.; Henry, L.; Pedersen, T.L.; Takahashi, K.; Wilke, C.; Woo, K.; Yutani, H.; Dunnington, D.; RStudio. ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics; Springer-Verlag: Berlin, Germany, 2016. [Google Scholar]
- Smith, R. ecole: ecole: School of Ecology Package. R package version 0.7-2019. Available online: https://github.com/phytomosaic/ecole (accessed on 22 March 2020).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package; R package version 2.5-6 2019. Available online: https://CRAN.R-project.org/package=vegan (accessed on 22 March 2020).
- Clarke, K.R.; Somerfield, P.J.; Chapman, M.G. On resemblance measures for ecological studies, including taxonomic dissimilarities and a zero-adjusted Bray–Curtis coefficient for denuded assemblages. J. Exp. Mar. Biol. Ecol. 2006, 330, 55–80. [Google Scholar] [CrossRef]
- Chao, A.; Ma, K.H.; Hsieh, T.C.; Chiu, C.H. Online Program SpadeR (Species-Richness Prediction and Diversity Estimation in R). Program and User’s Guide. Available online: http://chao.stat.nthu.edu.tw/wordpress/software_download (accessed on 22 March 2020).
- Chao, A.; Chazdon, R.L.; Colwell, R.K.; Shen, T.-J. Abundance-based similarity indices and their estimation when there are unseen species in samples. Biometrics 2006, 62, 361–371. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2019. Available online: https://worldclim.org/version2 (accessed on 11 November 2019).
- Conservation GIS Data—The Nature Conservancy. Available online: http://maps.tnc.org/gis_data.html (accessed on 15 November 2019).
- Borcard, D.; Gillet, F.; Legendre, P. Numerical Ecology with R.; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-7975-9. [Google Scholar]
- R Core Team R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020.
- Olson, D.M.; Dinerstein, E. The Global 200: Priority ecoregions for global conservation. Ann. Mo. Bot. Gard. 2002, 199–224. [Google Scholar] [CrossRef]
- Aarvik, L.; Bengt Ake, B.; Elven, H.; Ivinskis, P.; Jürivete, U.; Karshol, O.; Mutanen, M.; Savenkov, N. Nordic-Baltic Checklist of Lepidoptera. Nor. J. Entomol. 2017, 3, 1–236. [Google Scholar]
- Redondo, V.M.; Gaston, J.; Gimeno, R. Geometridae Ibericae; BRILL: Leiden, The Netherlands, 2009; ISBN 978-90-04-26101-3. [Google Scholar]
- Polev, V. Landscape distribution of geometer moths of the subfamily Sterrhinae (Lepidoptera, Geometridae) in Kuznetskaya Holl. Euroasian Entomol. J. 2008, 7, 389–392. [Google Scholar]
- Sihvonen, P.; Nupponen, K. Taxonomy of Rhodostrophia jacularia (Hübner, 1813)–a Sterrhinae moth with variable female wing shape (Lepidoptera: Geometridae). Nota Lepidopterol. 2005, 28, 113–122. [Google Scholar]
- Beck, J.; Schwanghart, W.; Khen, C.V.; Holloway, J.D. Predicting geometrid moth diversity in the Heart of Borneo. Insect Conserv. Divers. 2011, 4, 173–183. [Google Scholar] [CrossRef]
- Brehm, G.; Homeier, J.; Fiedler, K. Beta diversity of geometrid moths (Lepidoptera: Geometridae) in an Andean montane rainforest. Divers. Distrib. 2003, 9, 351–366. [Google Scholar] [CrossRef]
- Beck, J.; Khen, C.V. Beta-diversity of geometrid moths from northern Borneo: Effects of habitat, time and space. J. Anim. Ecol. 2007, 76, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Korsun, O.V.; Akulova, G.A.; Gordeev, S.Y.; Gordeeva, T.V.; Budaeva, A.A. Insects of the Onon-Balj National Park (Mongolia). Amurian Zool. J. 2012, 4, 18–25. [Google Scholar]
- Smiles, K.H. A revision of the subgenus Dysgnophos (Lepidoptera: Geometridae). J. Nat. Hist. 1979, 13, 99–119. [Google Scholar] [CrossRef]
- Jiang, N.; Xue, D.; Han, H. A review of Jankowskia Oberthür, 1884, with descriptions of four new species (Lepidoptera: Geometridae, Ennominae). Zootaxa 2010, 2559, 1–16. [Google Scholar] [CrossRef]
- Beljaev, E.A. A new subspecies of the Pseudopanthera macularia L.(Lepidoptera: Geometridae, Ennominae) from South Siberia. Far East. Entomol. 1997, 51, 1–7. [Google Scholar]
- Makhov, I.A. Eupithecia Curtis, 1825 (Geometridae, Larentiinae) of Southern Baikal Siberia. Euroasian Entomol. J. 2015, 14, 149–156. [Google Scholar]
- Vojnits, A.M. Data to the Eupithecia fauna of Asia (Lepidoptera, Geometridae). [Studies on Palaearctic Eupithecia species XV]. Ann. Hist. -Nat. Musei Natl. Hung 1981, 73, 221–237. [Google Scholar]
- Viidalepp, J. Two species of the genus Eupithecia Curt.(Lepidoptera,-Geometridae) from the Mongolian People’s Republic. Entomol. Rev. 1973, 52, 397–399. [Google Scholar]
- Choi, S.-W.; Kim, S.-S. New Records of Seven Eupithecia (Lepidoptera: Geometridae) from Korea. Anim. Syst. Evol. Divers. 2015, 31. [Google Scholar] [CrossRef]
- Vasilenko, S.V. To the knowledge of the fauna of geometrid moths (Lepidoptera: Geometridae) of Tajikistan. Cauc. Entomol. Bull. 2019, 15, 347–354. [Google Scholar] [CrossRef]
- Vasilenko, S.V.; Beljaev, E.A. Review of geometrid moths of the Xanthorhoe incursata (Hübner, 1813 [“1796”]) group (Lepidoptera, Geometridae, Larentiinae) from the Asian part of Russia, with description of a new species. Entomol. Rev. 2017, 97, 1149–1165. [Google Scholar] [CrossRef]
- Sihvonen, P. The Sterrhinae moth fauna of Fenglin Nature Reserve, North-East China. Spixiana 2006, 29, 247–257. [Google Scholar]
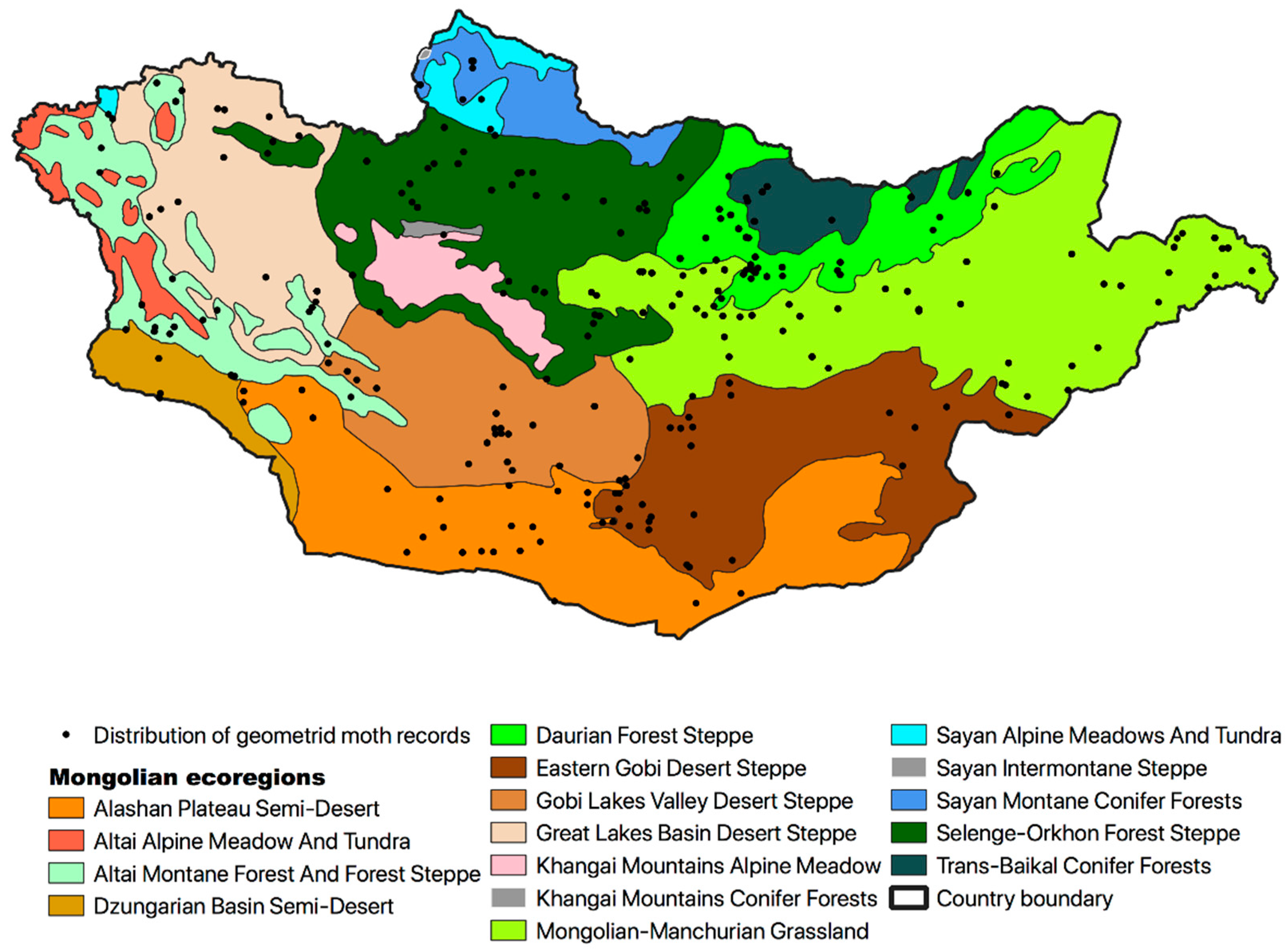
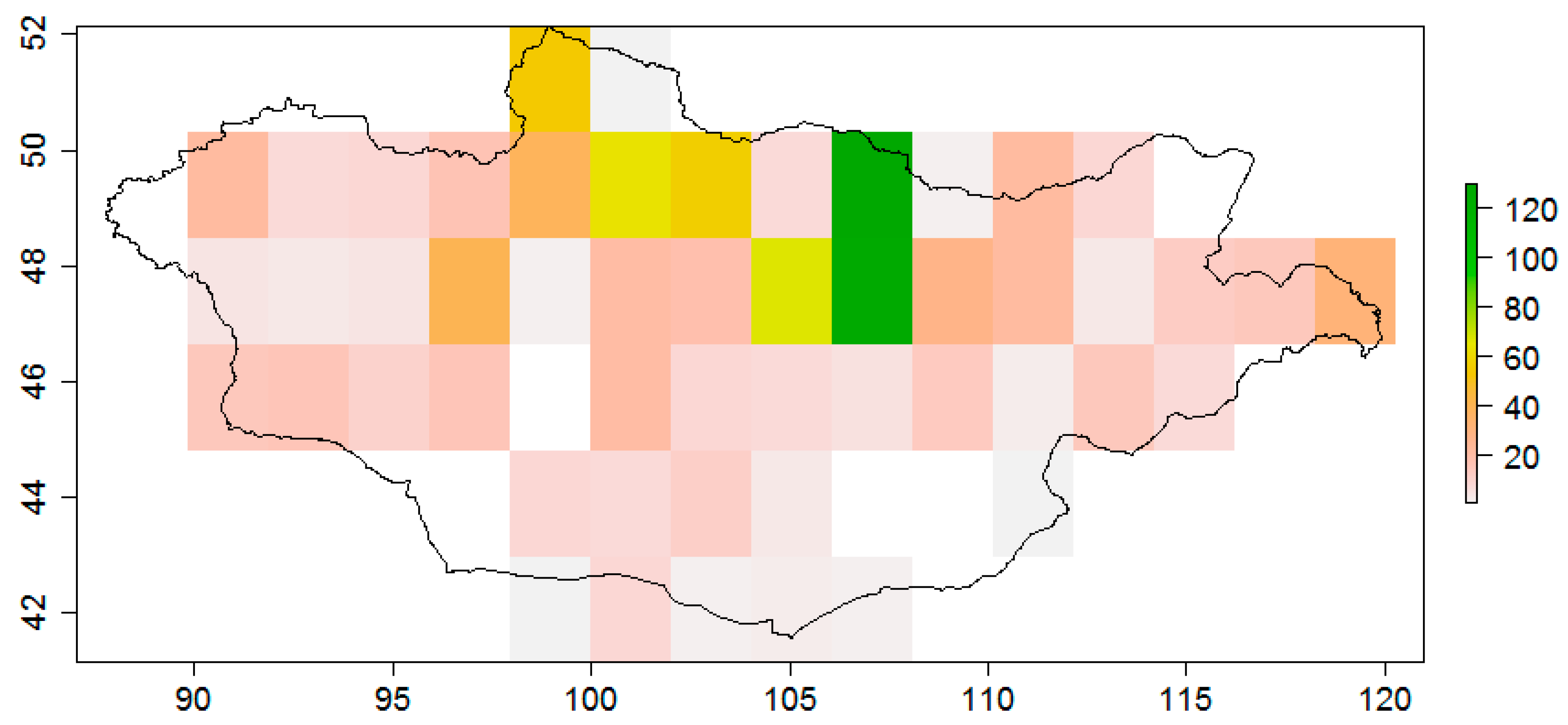
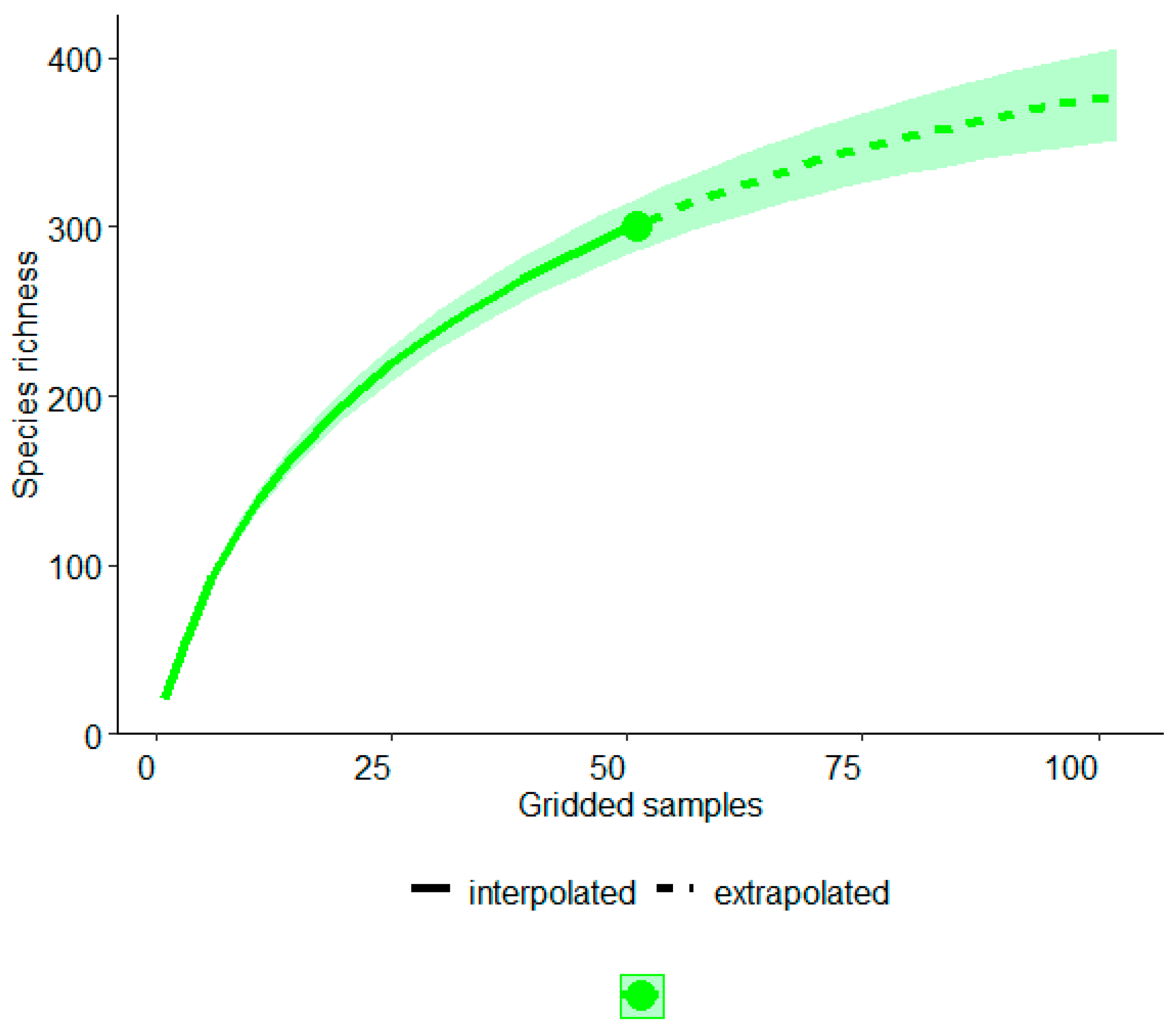
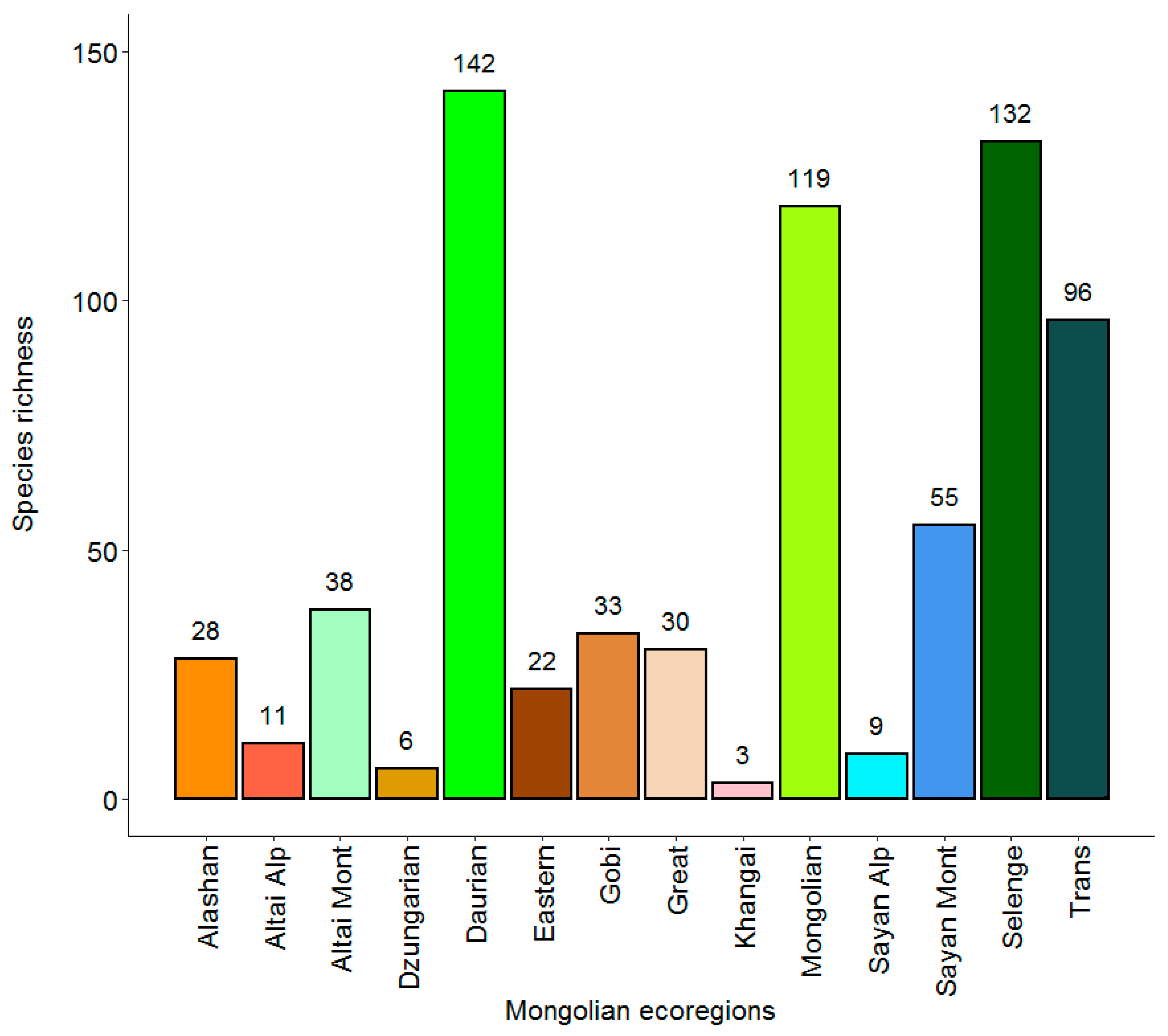
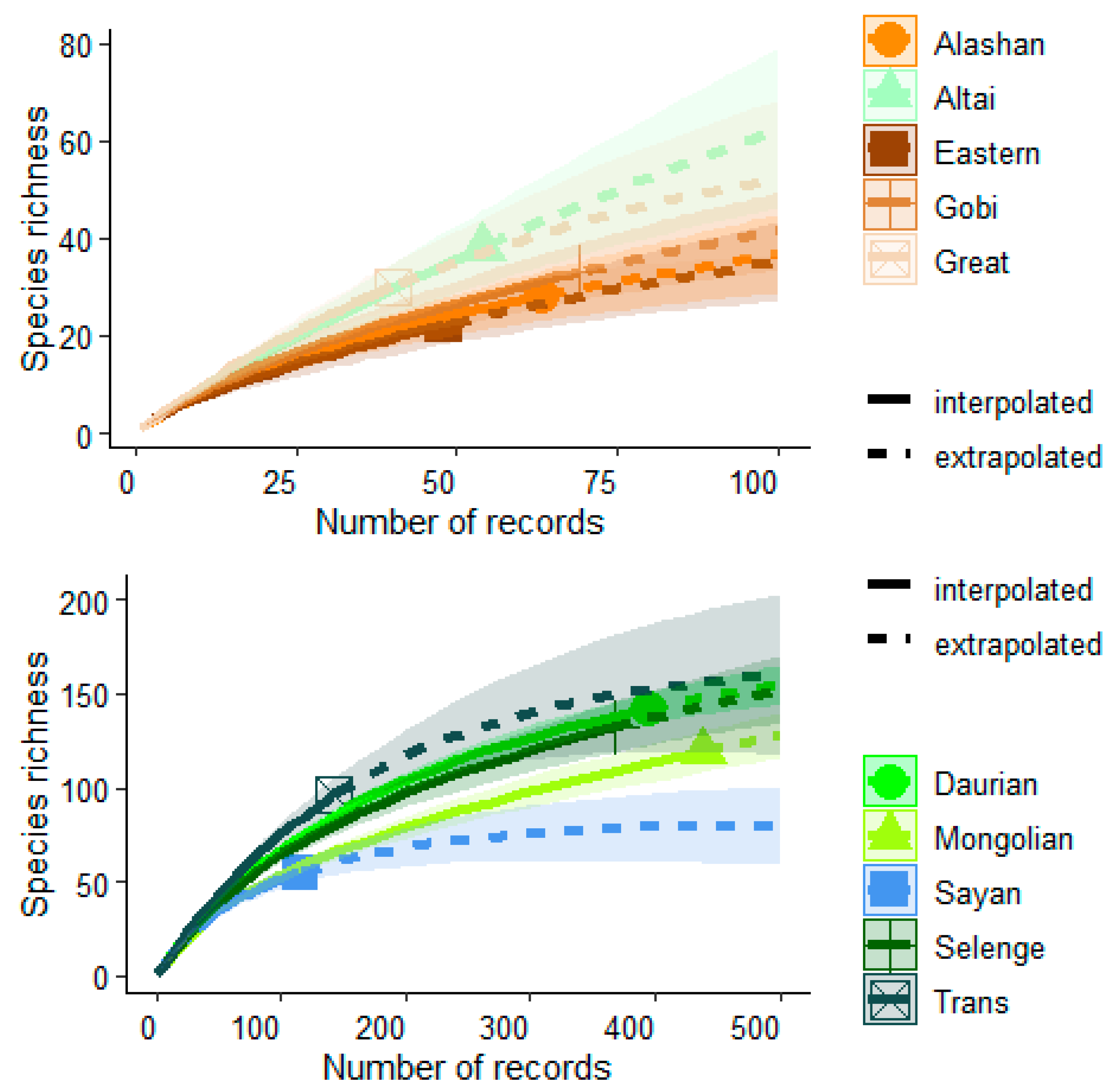

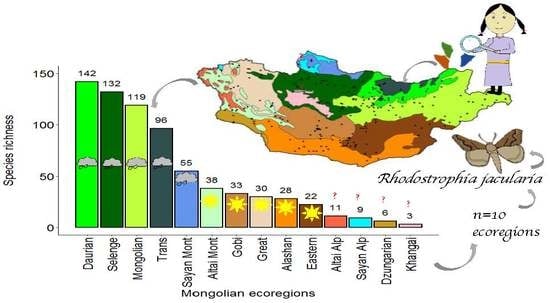
| Ecoregions | Bio1 | Bio2 | Bio5 | Bio6 | Bio7 | Bio10 | Bio11 | Bio12 | Biome [83] |
|---|---|---|---|---|---|---|---|---|---|
| Alashan Plateau Semi-Desert | 5.1 | 14.1 | 28.6 | −20.3 | 49 | 20.6 | −11.7 | 85 | Deserts and Xeric Shrublands |
| Altai Alpine Meadow and Tundra | −4.5 | 12.3 | 17.1 | −28.1 | 45.2 | 10.3 | −20.3 | 199 | Montane Grasslands and Shrublands |
| Altai Montane Forest and Forest Steppe | −1.8 | 13.1 | 20.5 | −26.8 | 47.3 | 13.4 | −18.5 | 148 | Temperate Conifer Forests |
| Dzungarian Basin Semi-Desert | 3.9 | 14 | 27.4 | −23 | 50.4 | 19.6 | −13.9 | 91 | Deserts and Xeric Shrublands |
| Daurian Forest Steppe | −1.5 | 13.9 | 23.7 | −29.1 | 52.9 | 16 | −21 | 306 | Temperate Grasslands, Savannas and Shrublands |
| Eastern Gobi Desert Steppe | 3.3 | 13.4 | 27.6 | −22.5 | 50.1 | 19.8 | −14.7 | 130 | Deserts and Xeric Shrublands |
| Gobi Lakes Valley Desert Steppe | 0.7 | 14.6 | 23.8 | −24.3 | 48.1 | 15.9 | −15.5 | 141 | Deserts and Xeric Shrublands |
| Great Lakes Basin Desert Steppe | −1.6 | 13.5 | 24.2 | −31.7 | 55.9 | 16.6 | −23.1 | 147 | Deserts and Xeric Shrublands |
| Khangai Mountains Alpine Meadow | −5.6 | 14.3 | 17.3 | −30.5 | 47.8 | 9.7 | −22.1 | 261 | Montane Grasslands and Shrublands |
| Mongolian-Manchurian Grassland | 0.3 | 13.6 | 25.4 | −26.4 | 51.8 | 17.6 | −18.7 | 224 | Temperate Grasslands, Savannas and Shrublands |
| Sayan Alpine Meadows and Tundra | −8.4 | 13.6 | 16.3 | −34.9 | 51.2 | 8.5 | −27.3 | 355 | Montane Grasslands and Shrublands |
| Sayan Montane Coniferous Forests | −5.1 | 13.7 | 19.2 | −31.3 | 50.4 | 11.4 | −23.5 | 381 | Temperate Conifer Forests |
| Selenge-Orkhon Forest Steppe | −3.2 | 14.3 | 20.6 | −29.7 | 50.3 | 12.9 | −21.4 | 277 | Temperate Grasslands, Savannas and Shrublands |
| Trans-Baikal Coniferous Forests | −3.3 | 13.4 | 22.1 | −31.1 | 53.2 | 14.6 | −23.3 | 366 | Boreal Forests/Taiga |
| C12(i,j) | Alashan | Altai | Daurian | Eastern | Gobi | Great | Mongolian | Sayan | Selenge | Trans |
|---|---|---|---|---|---|---|---|---|---|---|
| Alashan | 1 | 0.504 | 0.184 | 0.595 | 0.716 | 0.446 | 0.433 | 0.097 | 0.206 | 0.244 |
| Altai | 1 | 0.451 | 0.64 | 0.742 | 0.702 | 0.523 | 0.311 | 0.594 | 0.445 | |
| Daurian | 1 | 0.188 | 0.324 | 0.267 | 0.669 | 0.499 | 0.769 | 0.685 | ||
| Eastern | 1 | 0.942 | 0.644 | 0.533 | 0.127 | 0.424 | 0.141 | |||
| Gobi | 1 | 0.8 | 0.679 | 0.14 | 0.371 | 0.076 | ||||
| Great | 1 | 0.497 | 0.301 | 0.544 | 0.139 | |||||
| Mongolian | 1 | 0.417 | 0.719 | 0.522 | ||||||
| Sayan | 1 | 0.631 | 0.447 | |||||||
| Selenge | 1 | 0.606 | ||||||||
| Trans | 1 |
| Variable | NMDS1 | NMDS2 | r2 | Pr (> 0) |
|---|---|---|---|---|
| Temperature | −0.32277 | 0.94648 | 0.7473 | 0.009 |
| Precipitation | 0.97252 | −0.23281 | 0.9183 | 0.001 |
| Records | 0.73924 | 0.67344 | 0.5096 | 0.095 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Enkhtur, K.; Boldgiv, B.; Pfeiffer, M. Diversity and Distribution Patterns of Geometrid Moths (Geometridae, Lepidoptera) in Mongolia. Diversity 2020, 12, 186. https://doi.org/10.3390/d12050186
Enkhtur K, Boldgiv B, Pfeiffer M. Diversity and Distribution Patterns of Geometrid Moths (Geometridae, Lepidoptera) in Mongolia. Diversity. 2020; 12(5):186. https://doi.org/10.3390/d12050186
Chicago/Turabian StyleEnkhtur, Khishigdelger, Bazartseren Boldgiv, and Martin Pfeiffer. 2020. "Diversity and Distribution Patterns of Geometrid Moths (Geometridae, Lepidoptera) in Mongolia" Diversity 12, no. 5: 186. https://doi.org/10.3390/d12050186
APA StyleEnkhtur, K., Boldgiv, B., & Pfeiffer, M. (2020). Diversity and Distribution Patterns of Geometrid Moths (Geometridae, Lepidoptera) in Mongolia. Diversity, 12(5), 186. https://doi.org/10.3390/d12050186