Feeding Strategies of Co-occurring Newt Species across Different Conditions of Syntopy: A Test of the “Within-Population Niche Variation” Hypothesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Species
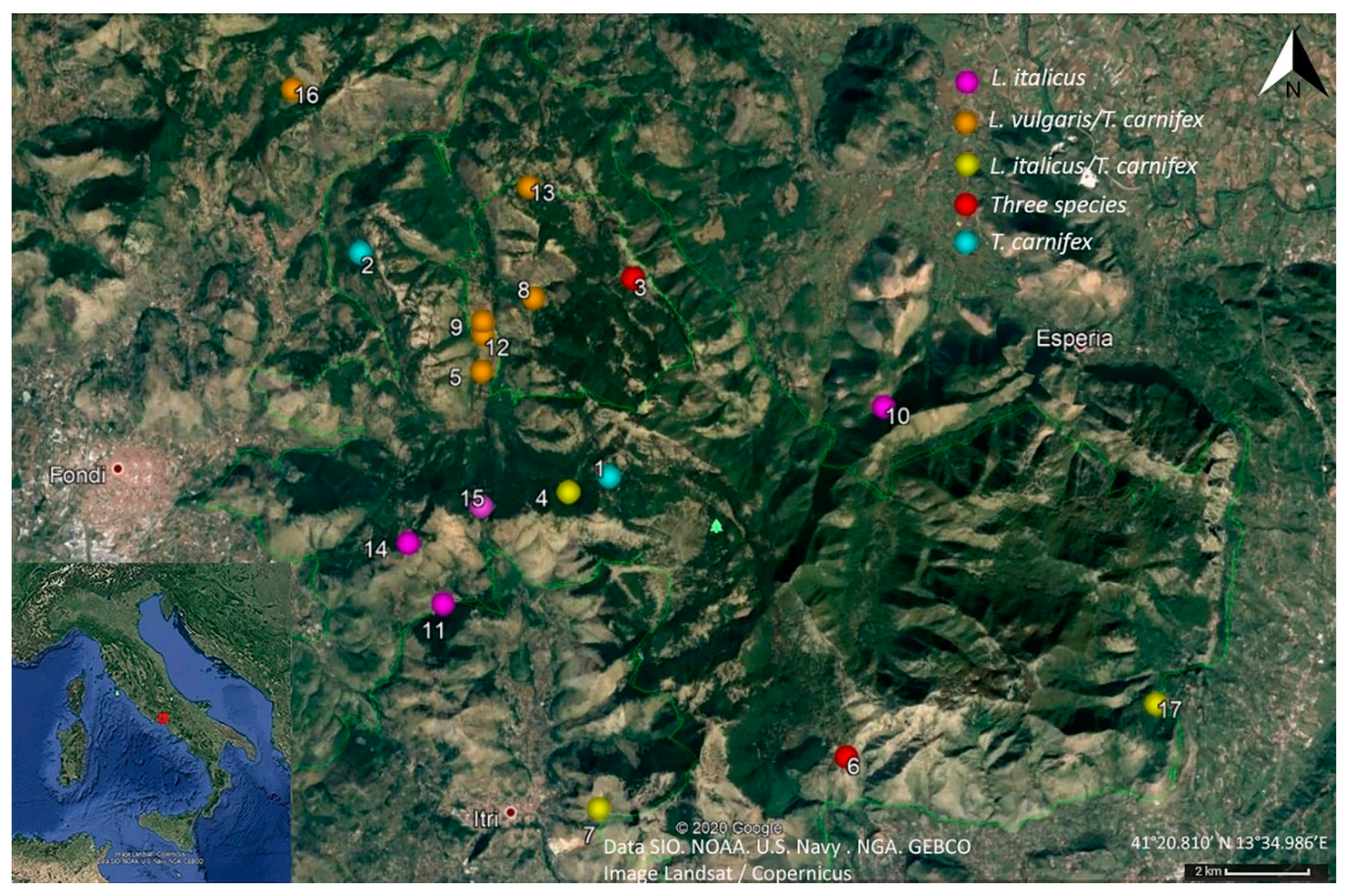
2.2. Study Area
2.3. Sampling
2.4. Data Analyses
3. Results
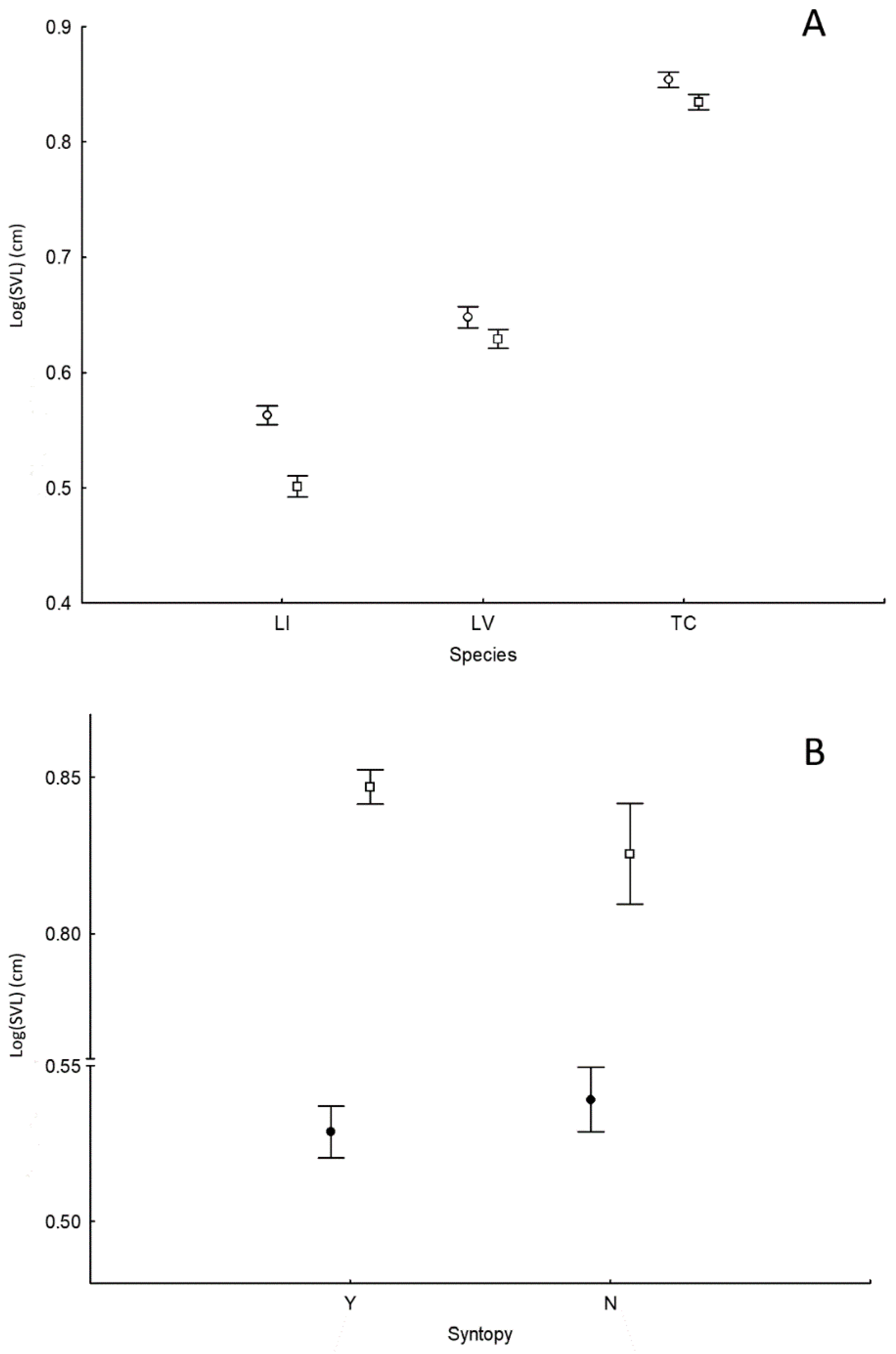
3.1. Body Size
3.2. Diet Analysis
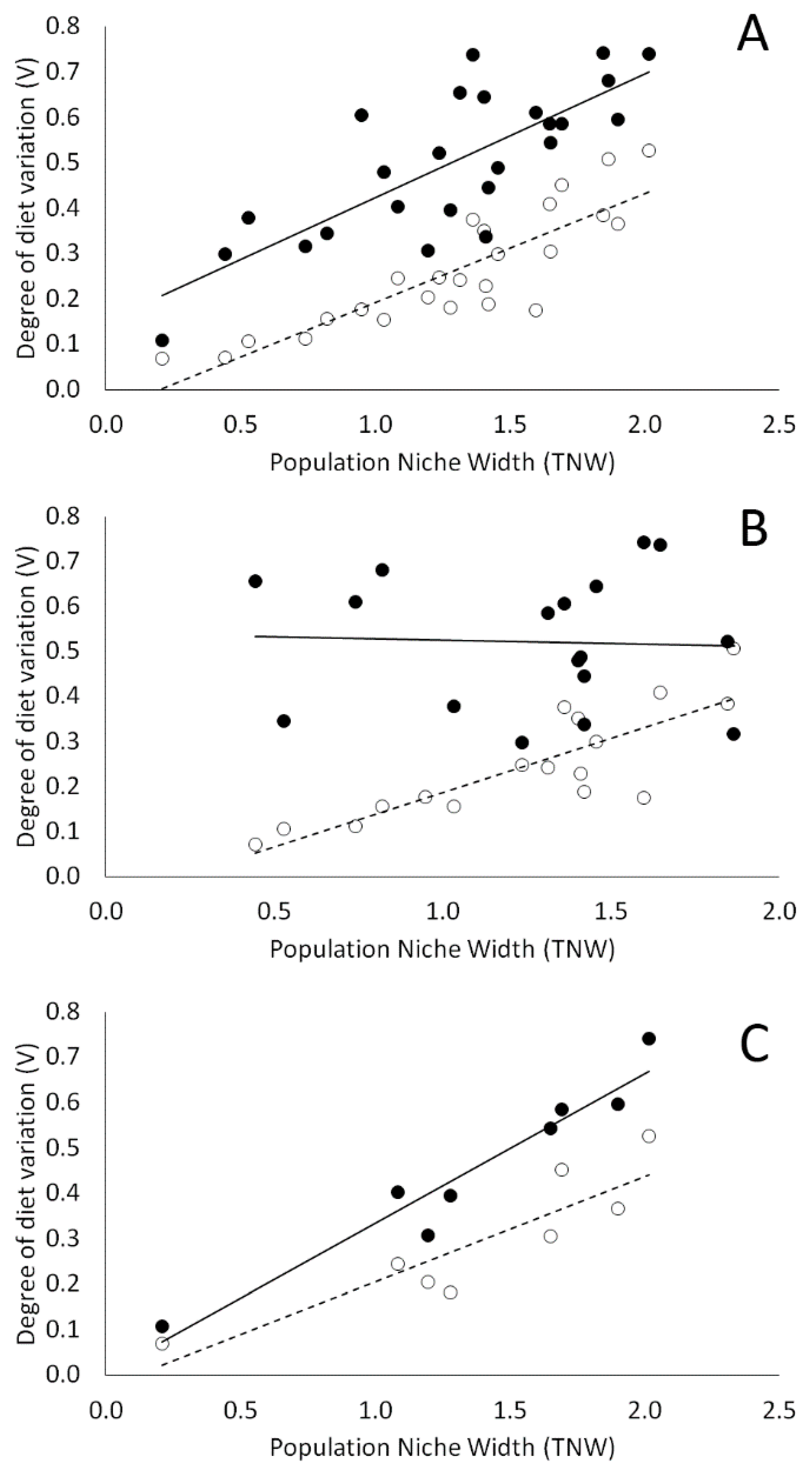
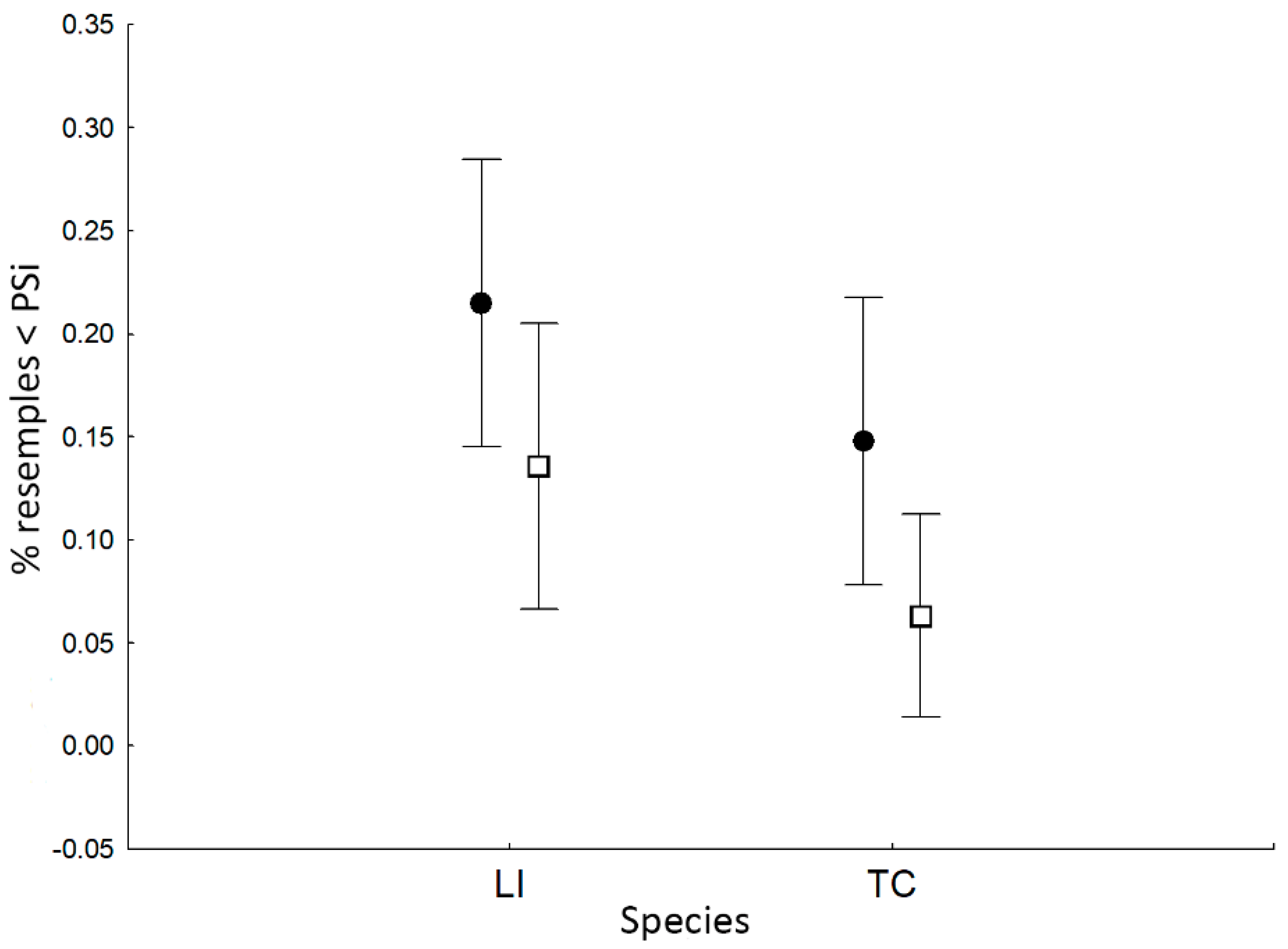
3.3. Individual Specialization and the Effect of Coexisting Species on Within-Population Diet Variation
4. Discussion
4.1. Body Size and Diet
4.2. Individual Specialization and the Effect of Coexisting Species on Within-Population Diet Variation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petranka, J.W. Fish predation: A factor affecting the spatial distribution of a stream-breeding salamander. Copeia 1983, 1983, 624–628. [Google Scholar] [CrossRef]
- Resetarits, W.J. Differences in an ensemble of streamside salamanders (Plethodontidae) above and below a barrier to brook trout. Amphibia-Reptilia 1997, 18, 15–25. [Google Scholar] [CrossRef]
- Wilkins, R.N.; Peterson, N.P. Factors related to amphibian occurrence and abundance in headwater streams draining second-growth Douglas-fir forests in southwestern Washington. For. Ecol. Manag. 2000, 139, 79–91. [Google Scholar] [CrossRef]
- Sánchez-Hernández, J. Reciprocal role of salamanders in aquatic energy flow pathways. Diversity 2020, 12, 32. [Google Scholar] [CrossRef]
- Murphy, M.L.; Hall, J.D. Vaired effects of clear-cut logging on predators and their habitat in small streams of the Cascade Mountains, Oregon. Can. J. Fish. Aq. Sci. 1981, 38, 137–145. [Google Scholar] [CrossRef]
- Marcot, B.G.; Vander Heyden, M. Key ecological functions of wildlife species. In Wildlife-Habitat Relationships in Oregon and Washington; Johnson, D.H., O’Neil, T.A., Eds.; Technical Coordinators Oregon State University Press: Corvallis, OR, USA, 2001. [Google Scholar]
- Schabetsberger, R.; Jersabek, C.D. Alpine newts (Triturus alpestris) as top predators in a high-altitude karst lake: Daily food consumption and impact on the copepod Arctodiaptomus Alpinus. Freshw. Biol. 1995, 33, 47–61. [Google Scholar] [CrossRef]
- Cogălniceanu, D.; Palmer, M.; Ciubuc, C. Feeding in anuran communities on islands in the Danube floodplain. Amphibia-Reptilia 2001, 22, 1–19. [Google Scholar]
- Buono, V.; Bissattini, A.M.; Vignoli, L. Can a cow save a newt? The role of cattle drinking troughs in amphibian conservation. Aquat. Conserv. 2019, 29, 964–975. [Google Scholar] [CrossRef]
- Semlitsch, R.D.; O’Donnell, K.M.; Thompson, F.R., III. Abundance, biomass production, nutrient content, and the possible role of terrestrial salamanders in Missouri Ozark forest ecosystems. Can. J. Zool. 2014, 92, 997–1004. [Google Scholar] [CrossRef]
- Wells, K.D. The Ecology and Behavior of Amphibians; University of Chicago Press: Chicago, IL, USA, 2007. [Google Scholar]
- Davic, R.D.; Welsh JR, H.H. On the ecological roles of salamanders. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 405–434. [Google Scholar] [CrossRef]
- Polis, G.A.; Strong, D.R. Food web complexity and community dynamics. Am. Nat. 1996, 147, 813–846. [Google Scholar] [CrossRef]
- Avery, R.A. Food and feeding relations of three species of Triturus (Amphibia Urodela) during the aquatic phases. Oikos 1968, 19, 408–412. [Google Scholar] [CrossRef]
- Griffiths, R.A. Feeding niche overlap and food selection in smooth and palmate newts, Triturus vulgaris and T. helveticus, at a pond in mid-Wales. J. Anim. Ecol. 1986, 55, 201–214. [Google Scholar] [CrossRef]
- Joly, P.; Giacoma, C. Limitation of similarity and feeding habits in three syntopic species of newts (Triturus, Amphibia). Ecography 1992, 15, 401–411. [Google Scholar] [CrossRef]
- Petranka, J.W. Salamanders of the United States and Canada; Smithsonian Institution Press: Washington, DC, USA, 1998. [Google Scholar]
- Vignoli, L.; Bologna, M.A.; Luiselli, L. Seasonal patterns of activity and community structure in an amphibian assemblage at a pond network with variable hydrology. Acta Oecol. 2007, 31, 185–192. [Google Scholar] [CrossRef]
- Vignoli, L.; Luiselli, L.; Bologna, M.A. Dietary patterns and overlap in an amphibian assemblage at a pond in Mediterranean Central Italy. Vie Milieu 2009, 59, 47–57. [Google Scholar]
- Kopecký, O.; Novak, K.; Vojar, J.; Šusta, F. Food composition of alpine newt (Ichthyosaura alpestris) in the post-hibernation terrestrial life stage. North-West. J. Zool. 2016, 12, 299–303. [Google Scholar]
- Fasola, M.; Canova, L. Feeding habits of Triturus vulgaris, T. cristatus and T. alpestris (Amphibia, Urodela) in the northern Apennines (Italy). Ital. J. Zool. 1992, 59, 273–280. [Google Scholar]
- Lanza, B.; Andreone, F.; Bologna, M.A.; Corti, C.; Razzetti, E. Amphibia; Edizioni Calderini: Bologna, Italy, 2007. [Google Scholar]
- Bologna, M.A.; Capula, M.; Carpaneto, G.M. Anfibi e Rettili Del Lazio; Palombi Editori: Rome, Italy, 2000. [Google Scholar]
- Araújo, M.S.; Bolnick, D.I.; Layman, C.A. The ecological causes of individual specialisation. Ecol. Lett. 2011, 14, 948–958. [Google Scholar] [CrossRef]
- Bolnick, D.I.; Ballare, K.M. Resource diversity promotes among-individual diet variation, but not genomic diversity, in lake stickleback. Ecol. Lett. 2020, 23, 495–505. [Google Scholar] [CrossRef]
- Bolnick, D.I.; Svanbäck, R.; Araújo, M.S.; Persson, L. Comparative support for the niche variation hypothesis that more generalized populations also are more heterogeneous. Proc. Natl. Acad. Sci. USA 2007, 104, 10075–10079. [Google Scholar] [CrossRef] [PubMed]
- Bolnick, D.I.; Ingram, T.; Stutz, W.E.; Snowberg, L.K.; Lau, O.L.; Paull, J.S. Ecological release from interspecific competition leads to decoupled changes in population and individual niche width. Proc. R. Soc. B 2010, 277, 1789–1797. [Google Scholar] [CrossRef] [PubMed]
- Barabás, G.; D’Andrea, R. The effect of intraspecific variation and heritability on community pattern and robustness. Ecol. Lett. 2016, 19, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.P.; Schreiber, S.J.; Levine, J.M. How variation between individuals affects species coexistence. Ecol. Lett. 2016, 19, 825–838. [Google Scholar] [CrossRef] [PubMed]
- Costa-Pereira, R.; Rudolf, V.H.; Souza, F.L.; Araújo, M.S. Drivers of individual niche variation in coexisting species. J. Anim. Ecol. 2018, 87, 1452–1464. [Google Scholar] [CrossRef] [PubMed]
- Cloyed, C.S.; Eason, P.K. Different ecological conditions support individual specialization in closely related, ecologically similar species. Evol. Ecol. 2016, 30, 379–400. [Google Scholar] [CrossRef]
- Bolnick, D.I.; Svanbäck, R.; Fordyce, J.A.; Yang, L.H.; Davis, J.M.; Hulsey, C.D.; Forister, M.L. The ecology of individuals: Incidence and implications of individual specialization. Am. Nat. 2003, 161, 1–28. [Google Scholar] [CrossRef]
- Vignoli, L.; Bissattini, A.M.; Luiselli, L. Food partitioning and the evolution of non-randomly structured communities in tailed amphibians: A worldwide systematic review. Biol. J. Lin. Soc. 2017, 120, 489–502. [Google Scholar] [CrossRef]
- Cloyed, C.S.; Eason, P.K. Feeding limitations in temperate anurans and the niche variation hypothesis. Amphibia-Reptilia 2017, 38, 473–482. [Google Scholar] [CrossRef]
- Toscano, B.J.; Gownaris, N.J.; Heerhartz, S.M.; Monaco, C.J. Personality, foraging behavior and specialization: Integrating behavioral and food web ecology at the individual level. Oecologia 2016, 182, 55–69. [Google Scholar] [CrossRef]
- Cerini, F.; Bologna, M.A.; Vignoli, L. Dragonflies community assembly in artificial habitats: Glimpses from field and manipulative experiments. PLoS ONE 2019, 14. [Google Scholar] [CrossRef] [PubMed]
- Scillitani, G.; Scalera, R.; Carafa, M.; Tripepi, S. Conservation and biology of Triturus italicus in Italy (Amphibia, Salamandridae). It. J. Zool. 2004, 71, 45–54. [Google Scholar] [CrossRef]
- Razzetti, E.; Bernini, F. Triturus vulgaris. In Atlas of Italian Amphibians and Reptiles Societas Herpetologica Italica; Sindaco, R., Doria, G., Razzetti, E., Bernini, F., Eds.; Edizioni Polistampa: Firenze, Italy, 2006. [Google Scholar]
- Novellino, D. An account of basket weaving and the use of fibre plants in the Mount Aurunci Regional Park (Central Italy). In Proceedings of the Fourth International Conference of Ethnobotany (ICEB 2005), Istanbul, Turkey, 21–26 August 2005. [Google Scholar]
- Anzalone, B. Prodromo Della Flora Romana: Elenco Preliminare Delle Piante Vascolari Spontanee Del Lazio; Regione Lazio: Rome, Italy, 1984. [Google Scholar]
- Minutillo, F.; Moraldo, B.; Ross, W. Segnalazioni floristiche italiane. Inform. Bot. Ital. 1985, 17, 124–127. [Google Scholar]
- Romano, A.; Montinaro, G.; Mattoccia, M.; Sbordoni, V. Amphibians of the Aurunci Mountains (Latium, Central Italy). Checklist and conservation guidelines. Acta Herpetol. 2007, 2, 17–25. [Google Scholar]
- Salvidio, S.; Sindaco, R.; Emanueli, L. Feeding habits of sympatric Discoglossus montalentii, Discoglossus sardus and Euproctus montanus during the breeding season. Herpetol. J. 1999, 9, 163–167. [Google Scholar]
- Lizana, M.; Perez-Mellado, V.; Ciudad, M.J. Analysis of the structure of an amphibian community in the central system of Spain. Herpetol. J. 1990, 1, 435–446. [Google Scholar]
- Reques, R.; Tejedo, M. Fenología y hábitats reproductivos de una comunidad de anfibios en la Sierra de Cabra (Córdoba). Rev. Esp. Herp. 1991, 6, 49–54. [Google Scholar]
- Solé, M.; Beckmann, O.; Pelz, B.; Kwet, A.; Engels, W. Stomach-flushing for diet analysis in anurans: An improved protocol evaluated in a case study in Araucaria forests, southern Brazil. Stud. Neotrop. Fauna Environ. 2005, 40, 23–28. [Google Scholar] [CrossRef]
- Newsome, S.D.; Tinker, M.T.; Gill, V.A.; Hoyt, Z.N.; Doroff, A.; Nichol, L.; Bodkin, J.L. The interaction of intraspecific competition and habitat on individual diet specialization: A near range-wide examination of sea otters. Oecologia 2015, 178, 45–59. [Google Scholar] [CrossRef]
- Bolnick, D.I.; Yang, L.H.; Fordyce, J.A.; Davis, J.M.; Svanbäck, R. Measuring individual-level resource specialization. Ecology 2002, 83, 2936–2941. [Google Scholar] [CrossRef]
- Schoener, T.W. The Anolis lizards of Bimini: Resource partitioning in a complex fauna. Ecology 1968, 49, 704–726. [Google Scholar] [CrossRef]
- Svanbäck, R.; Persson, L. Individual diet specialization, niche width and population dynamics: Implications for trophic polymorphisms. J. Anim. Ecol. 2004, 73, 973–982. [Google Scholar] [CrossRef]
- Roughgarden, J. Evolution of niche width. Am. Nat. 1972, 106, 683–718. [Google Scholar] [CrossRef]
- Feinsinger, P.; Spears, E.E.; Poole, R.W. A simple measure of niche breadth. Ecology 1981, 62, 27–32. [Google Scholar] [CrossRef]
- Griffiths, R.A. Newts and Salamanders of Europe; T & AD Poyser: London, UK, 1996. [Google Scholar]
- Dolmen, D.; Koksvik, J.I. Food and feeding habits of Triturus vulgaris (L.) and T. cristatus (Laurenti) (Amphibia) in two bog tarns in central Norway. Amphibia-Reptilia 1983, 4, 17–24. [Google Scholar] [CrossRef]
- Griffiths, R.A.; Mylotte, V.J. Microhabitat selection and feeding relations of smooth and warty newts, Triturus vulgaris and T. cristatus, at an upland pond in mid-Wales. Ecography 1987, 10, 1–7. [Google Scholar] [CrossRef]
- Vignoli, L.; Bombi, P.M.; D’Amen, M.; Bologna, M.A. Seasonal variation in the trophic niche of a heterochronic population of Triturus alpestris apuanus from the south-western Alps. Herpetol. J. 2007, 17, 183–191. [Google Scholar]
- Fasola, M. Resource partitioning by three species of newts during their aquatic phase. Ecography 1993, 16, 73–81. [Google Scholar] [CrossRef]
- Van Valen, L. Morphological variation and width of ecological niche. Am. Nat. 1965, 99, 377–390. [Google Scholar] [CrossRef]
- Salvidio, S.; Crovetto, F.; Costa, A. Individual trophic specialisation in the Alpine newt increases with increasing resource diversity. Ann. Zool. Fenn. 2019, 56, 17–24. [Google Scholar] [CrossRef]

| Effect | SS | DoF | MS | F | p |
|---|---|---|---|---|---|
| Intercept | 130.300 | 1 | 130.300 | 15,218.71 | <0.0001 |
| Site | 0.227 | 16 | 0.014 | 14.18 | <0.0001 |
| Species | 6.605 | 2 | 3.302 | 3301.11 | <0.0001 |
| Sex | 0.145 | 1 | 0.145 | 144.79 | <0.0001 |
| Species × Sex | 0.048 | 2 | 0.024 | 23.99 | <0.0001 |
| Syntopy° | 0.002 | 1 | 0.002 | 1.01 | 0.315 |
| Species × Syntopy° | 0.014 | 1 | 0.014 | 8.48 | 0.004 |
| Error | 0.540 | 540 | 0.001 |
| Effect | SS | DoF | MS | F | p |
|---|---|---|---|---|---|
| Site | 10.177 | 17 | 0.599 | 13.181 | <0.001 |
| Species | 0.600 | 2 | 0.300 | 6.603 | 0.001 |
| Sex | 0.038 | 1 | 0.038 | 0.835 | 0.361 |
| Syntopy° | 0.677 | 1 | 0.677 | 10.305 | 0.001 |
| SVL | 0.007 | 1 | 0.007 | 0.155 | 0.694 |
| Species × Syntopy° | 0.108 | 1 | 0.108 | 1.638 | 0.201 |
| Species × Sex | 0.049 | 2 | 0.024 | 0.534 | 0.586 |
| Sex × Syntopy° | 0.043 | 1 | 0.043 | 0.654 | 0.419 |
| Error | 22.165 | 488 | 0.045 |
| Effect | SS | DoF | F | p |
|---|---|---|---|---|
| Intercept | 0.360 | 1 | 83.692 | 0 |
| Species | 0.022 | 1 | 5.176 | 0.037 |
| Syntopy | 0.0308 | 1 | 7.148 | 0.017 |
| Species × Syntopy | 0.00003 | 1 | 0.008 | 0.930 |
| Error | 0.069 | 16 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirabasso, J.; Bissattini, A.M.; Bologna, M.A.; Luiselli, L.; Stellati, L.; Vignoli, L. Feeding Strategies of Co-occurring Newt Species across Different Conditions of Syntopy: A Test of the “Within-Population Niche Variation” Hypothesis. Diversity 2020, 12, 181. https://doi.org/10.3390/d12050181
Mirabasso J, Bissattini AM, Bologna MA, Luiselli L, Stellati L, Vignoli L. Feeding Strategies of Co-occurring Newt Species across Different Conditions of Syntopy: A Test of the “Within-Population Niche Variation” Hypothesis. Diversity. 2020; 12(5):181. https://doi.org/10.3390/d12050181
Chicago/Turabian StyleMirabasso, Jennifer, Alessandra M. Bissattini, Marco A. Bologna, Luca Luiselli, Luca Stellati, and Leonardo Vignoli. 2020. "Feeding Strategies of Co-occurring Newt Species across Different Conditions of Syntopy: A Test of the “Within-Population Niche Variation” Hypothesis" Diversity 12, no. 5: 181. https://doi.org/10.3390/d12050181
APA StyleMirabasso, J., Bissattini, A. M., Bologna, M. A., Luiselli, L., Stellati, L., & Vignoli, L. (2020). Feeding Strategies of Co-occurring Newt Species across Different Conditions of Syntopy: A Test of the “Within-Population Niche Variation” Hypothesis. Diversity, 12(5), 181. https://doi.org/10.3390/d12050181





