Taxonomic, Phylogenetic, and Functional Diversity of Ferns at Three Differently Disturbed Sites in Longnan County, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Sampling Techniques
2.3. Data Analyses
3. Results and Discussion
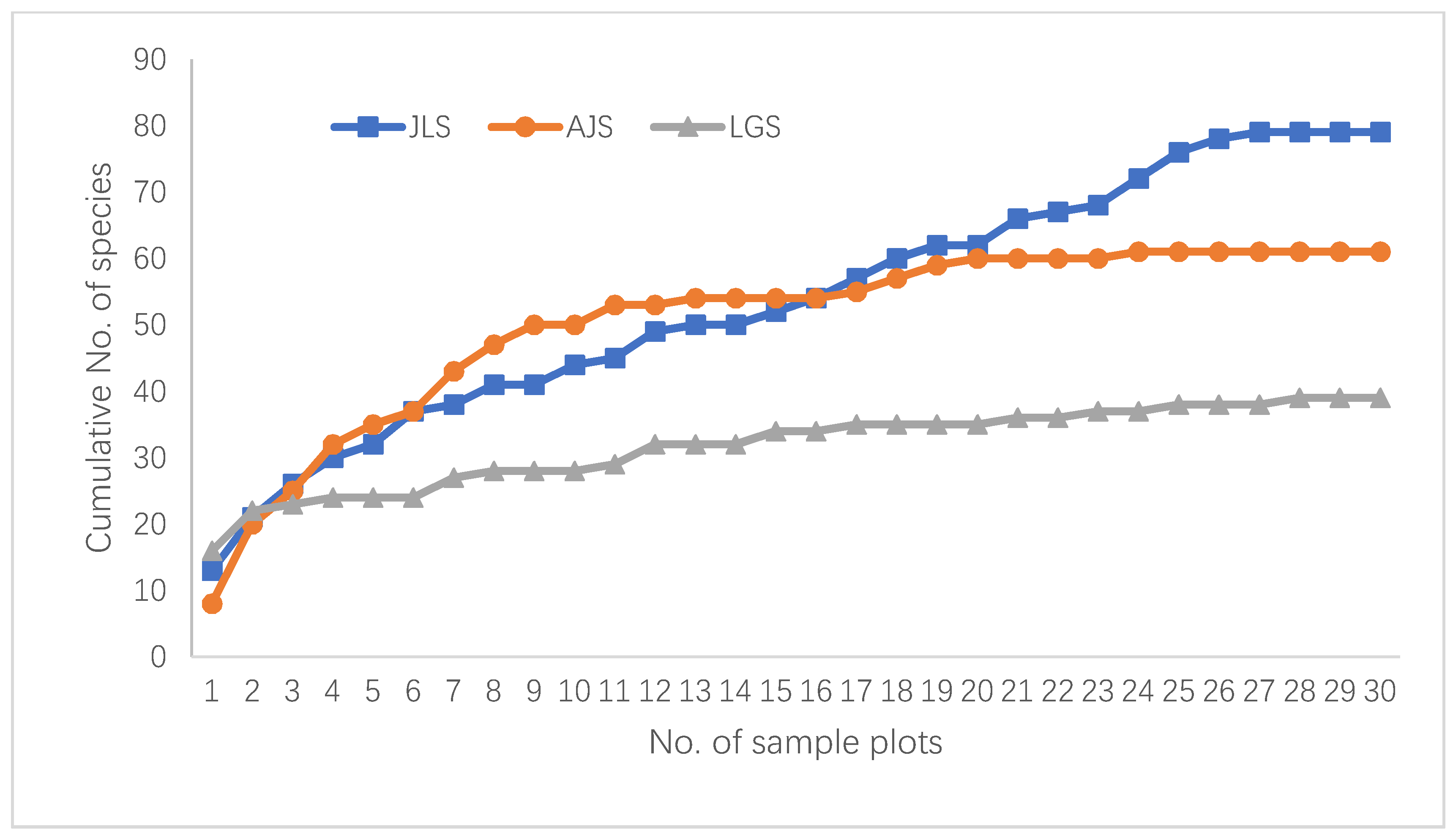
3.1. Sample Coverage
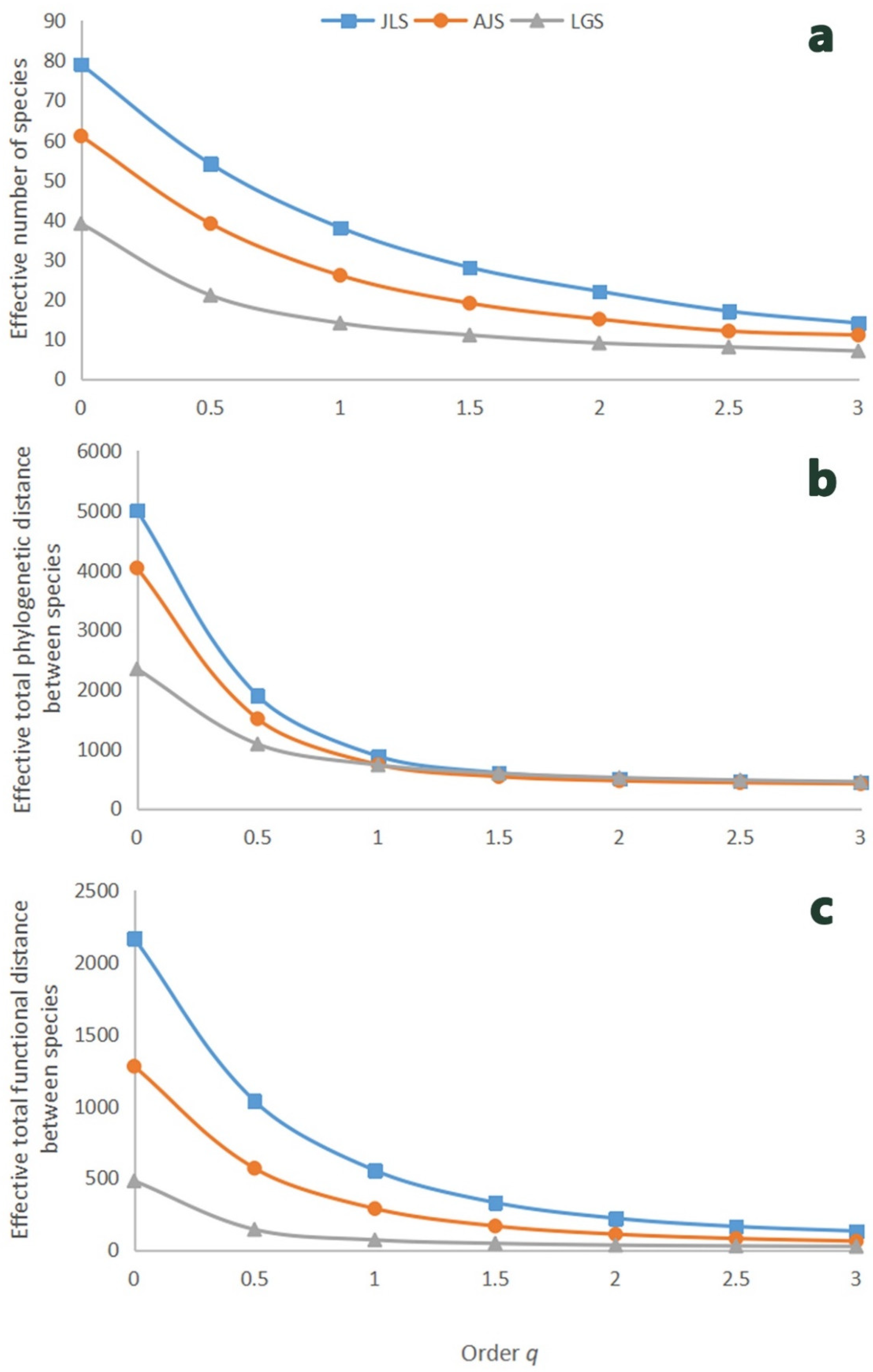
3.2. Taxonomic, Phylogenetic, and Functional Diversity
3.3. Indicator Fern Species for Different Disturbance Levels
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The Pteridophyte Phylogeny Group I. A community-derived classification for extant lycophytes and ferns. J. Syst. Evol. 2016, 54, 563–603. [Google Scholar] [CrossRef]
- Schneider, H.; Schuettpelz, E.; Pryer, K.M.; Cranfill, R.; Magallon, S.; Lupia, R. Ferns diversified in the shadow of angiosperms. Nature 2004, 428, 553–557. [Google Scholar] [CrossRef] [PubMed]
- Testo, W.; Sundue, M. A 4000-species dataset provides new insight into the evolution of ferns. Mol. Phylogenet. Evol. 2016, 105, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Salazar, L.; Homeier, J.; Kessler, M.; Abrahamczyk, S.; Lehnert, M.; Krömer, T.; Kluge, J. Diversity patterns of ferns along elevational gradients in Andean tropical forests. Plant Ecol. Divers. 2013, 8, 13–24. [Google Scholar] [CrossRef]
- Chang, J.; Yoon, I.; Kim, K. Heavy metal and arsenic accumulating fern species as potential ecological indicators in As-contaminated abandoned mines. Ecol. Indic. 2009, 9, 1275–1279. [Google Scholar] [CrossRef]
- Silva, V.L.; Mehltreter, K.; Schmitt, J.L. Ferns as potential ecological indicators of edge effects in two types of Mexican forests. Ecol. Indic. 2018, 93, 669–676. [Google Scholar] [CrossRef]
- Bergeron, A.; Pellerin, S. Pteridophytes as indicators of urban forest integrity. Ecol. Indic. 2014, 38, 40–49. [Google Scholar] [CrossRef]
- Abotsi, K.E.; Bose, R.; Adjossou, K.; Deblauwe, V.; Rouhan, G.; Segla, K.N.; Atsri, K.H.; Kokou, K. Ecological drivers of pteridophyte diversity and distribution in Togo (West Africa). Ecol. Indic. 2020, 108. [Google Scholar] [CrossRef]
- Pouteau, R.; Meyer, J.Y.; Blanchard, P.; Nitta, J.H.; Terorotua, M.; Taputuarai, R. Fern species richness and abundance are indicators of climate change on high-elevation islands: Evidence from an elevational gradient on Tahiti (French Polynesia). Clim. Chang. 2016, 138, 143–156. [Google Scholar] [CrossRef]
- Khine, P.K.; Kluge, J.; Kessler, M.; Miehe, G.; Karger, D.N. Latitude-independent, continent-wide consistency in climate-richness relationships in Asian ferns and lycophytes. J. Biogeogr. 2019, 46, 981–991. [Google Scholar] [CrossRef]
- Kessler, M.; Siorak, Y.; Wunderlich, M.; Wegner, C. Patterns of morphological leaf traits among pteridophytes along humidity and temperature gradients in the Bolivian Andes. Funct. Plant Biol. 2007, 34. [Google Scholar] [CrossRef]
- Carvajal-Hernández, C.I.; Gómez-Díaz, J.A.; Kessler, M.; Krömer, T. Influence of elevation and habitat disturbance on the functional diversity of ferns and lycophytes. Plant Ecol. Divers. 2018, 11, 335–347. [Google Scholar] [CrossRef]
- Weigand, A.; Abrahamczyk, S.; Aubin, I.; Bita-Nicolae, C.; Bruelheide, H.; Carvajal-Hernandez, C.I.; Cicuzza, D.; da Costa, L.E.N.; Csiky, J.; Dengler, J.; et al. Global fern and lycophyte richness explained: How regional and local factors shape plot richness. J. Biogeogr. 2019. [Google Scholar] [CrossRef]
- Lwanga, J.S.; Balmford, A.; Badaza, R. Assessing fern diversity: Relative species richness and its environmental correlates in Uganda. Biodivers. Conserv. 1998, 7, 1387–1398. [Google Scholar] [CrossRef]
- Mehltreter, K.; Walker, L.R.; Sharpe, J.M. Fern Ecology; Cambridge University Press: New York, NY, USA, 2010. [Google Scholar]
- Hernández-Rojas, A.; Kessler, M.; Krömer, T.; Carvajal-Hernández, C.; Weigand, A.; Kluge, J. Richness patterns of ferns along an elevational gradient in the Sierra de Juárez, Oaxaca, Mexico: A comparison with Central and South America. Am. Fern J. 2018, 108, 76–94. [Google Scholar] [CrossRef]
- Grytnes, J.A.; Beaman, J.H. Elevational species richness patterns for vascular plants on Mount Kinabalu, Borneo. J. Biogeogr. 2006, 33, 1838–1849. [Google Scholar] [CrossRef]
- Luna-Vega, I.; Tejero-Diez, J.D.; Contreras-Medina, R.; Heads, M.; Rivas, G. Biogeographical analysis of two Polypodium species complexes (Polypodiaceae) in Mexico and Central America. Biol. J. Linn. Soc. 2012, 106, 940–955. [Google Scholar] [CrossRef]
- Tanaka, T.; Sato, T. Elevational patterns of fern species assemblages and richness in central Japan. Plant Ecol. 2013, 214, 1189–1197. [Google Scholar] [CrossRef]
- Hemp, A. Ecology of the pteridophytes on the southern slopes of Mt. Kilimanjaro, I. Altitudinal distribution. Plant Ecol. 2002, 159, 211–239. [Google Scholar] [CrossRef]
- Kessler, M.; Kluge, J.; Hemp, A.; Ohlemuller, R. A global comparative analysis of elevational species richness patterns of ferns. Glob. Ecol. Biogeogr. 2011, 20, 868–880. [Google Scholar] [CrossRef]
- Watkins, J.E., Jr.; Cardelus, C.; Colwell, R.K.; Moran, R.C. Species richness and distribution of ferns along an elevational gradient in Costa Rica. Am. J. Bot. 2006, 93, 73–83. [Google Scholar] [CrossRef]
- Jácome, J.; Kessler, M.; Smith, A.R. A human-induced downward-skewed elevational abundance distribution of pteridophytes in the Bolivian Andes. Glob. Ecol. Biogeogr. 2007, 16, 313–318. [Google Scholar] [CrossRef]
- Parra, M.J.; Rodriguez, R.; Cavieres, L.; Munoz-Tapia, L.; Atala, C. Latitudinal patterns in Pteridophyte distribution of Continental Chile. Gayana Bot. 2015, 72, 58–69. [Google Scholar] [CrossRef]
- Wei, R.; Ree, R.H.; Sundue, M.A.; Zhang, X. Polyploidy and elevation contribute to opposing latitudinal gradients in diversification and species richness in lady ferns (Athyriaceae). BioRxiv 2018. [Google Scholar] [CrossRef]
- Karger, D.N.; Kluge, J.; Kromer, T.; Hemp, A.; Lehnert, M.; Kessler, M. The effect of area on local and regional elevational patterns of species richness. J. Biogeogr. 2011, 38, 1177–1185. [Google Scholar] [CrossRef]
- Bhattarai, K.R.; Vetaas, O.R.; Grytnes, J.A. Fern species richness along a central Himalayan elevational gradient, Nepal. J. Biogeogr. 2004, 31, 389–400. [Google Scholar] [CrossRef]
- Kluge, J.; Kessler, M. Fern endemism and its correlates: Contribution from an elevational transect in Costa Rica. Divers. Distrib. 2006, 12, 535–545. [Google Scholar] [CrossRef]
- Syfert, M.M.; Brummitt, N.A.; Coomes, D.A.; Bystriakova, N.; Smith, M.J. Inferring diversity patterns along an elevation gradient from stacked SDMs: A case study on Mesoamerican ferns. Glob. Ecol. Conserv. 2018, 16. [Google Scholar] [CrossRef]
- Kluge, J.; Kessler, M. Influence of niche characteristics and forest type on fern species richness, abundance and plant size along an elevational gradient in Costa Rica. Plant Ecol. 2011, 212, 1109–1121. [Google Scholar] [CrossRef][Green Version]
- Karst, J.; Gilbert, B.; Lechowicz, J. Fern community assembly: The roles of chance and the environment at local and intermediate scales. Ecology 2005, 96, 2473–2486. [Google Scholar] [CrossRef]
- Qian, H.; Wang, S.; Li, Y.; Xiao, M.; Wang, X. Disentangling the relative effects of ambient energy, water availability, and energy-water balance on pteridophyte species richness at a landscape scale in China. Plant Ecol. 2012, 213, 749–756. [Google Scholar] [CrossRef]
- Nagalingum, N.S.; Knerr, N.; Laffan, S.W.; Gonzalez-Orozco, C.E.; Thornhill, A.H.; Miller, J.T.; Mishler, B.D. Continental scale patterns and predictors of fern richness and phylogenetic diversity. Front. Genet. 2015, 6, 132. [Google Scholar] [CrossRef]
- Kluge, J.; Bach, K.; Kessler, M. Elevational distribution and zonation of tropical pteridophyte assemblages in Costa Rica. Basic Appl. Ecol. 2008, 9, 35–43. [Google Scholar] [CrossRef]
- Acebey, A.R.; Kromer, T.; Kessler, M. Species richness and vertical distribution of ferns and lycophytes along an elevational gradient in Los Tuxtlas, Veracruz, Mexico. Flora 2017, 235, 83–91. [Google Scholar] [CrossRef]
- Kluge, J.; Kessler, M.; Dunn, R.R. What drives elevational patterns of diversity? A test of geometric constraints, climate and species pool effects for pteridophytes on an elevational gradient in Costa Rica. Glob. Ecol. Biogeogr. 2006, 15, 358–371. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Z.; Ji, M.; Fan, Z.; Deng, J. Patterns of species richness in relation to temperature, taxonomy and spatial scale in eastern China. Acta Oecologica Int. J. Ecol. 2011, 37, 307–313. [Google Scholar] [CrossRef]
- Marini, L.; Bona, E.; Kunin, W.E.; Gaston, K.J. Exploring anthropogenic and natural processes shaping fern species richness along elevational gradients. J. Biogeogr. 2011, 38, 78–88. [Google Scholar] [CrossRef]
- Nervo, M.H.; Andrade, B.O.; Tornquist, C.G.; Mazurana, M.; Windisch, P.G.; Overbeck, G.E. Distinct responses of terrestrial and epiphytic ferns and lycophytes along an elevational gradient in Southern Brazil. J. Veg. Sci. 2019, 30, 55–64. [Google Scholar] [CrossRef]
- Tanaka, T.; Sato, T. Species richness of seed plants and ferns along a temperate elevational gradient in central Japan. Plant Ecol. 2014, 215, 1299–1311. [Google Scholar] [CrossRef]
- Schneider, H.; He, L.; Marquardt, J.; Wang, L.; Heinrichs, J.; Hennequin, S.; Zhang, X. Exploring the origin of the latitudinal diversity gradient: Contrasting the sister fern genera Phegopteris and Pseudophegopteris. J. Syst. Evol. 2013, 51, 61–70. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, S.; John, R.; Li, R.; Liu, H.; Ye, Q. Habitat filtering and exclusion of weak competitors jointly explain fern species assemblage along a light and water gradient. Sci. Rep. 2017, 7, 298. [Google Scholar] [CrossRef]
- Creese, C.; Lee, A.; Sack, L. Drivers of morphological diversity and distribution in the Hawaiian fern flora: Trait associations with size, growth form, and environment. Am. J. Bot. 2011, 98, 956–966. [Google Scholar] [CrossRef]
- Kessler, M.; Gudel, R.; Salazar, L.; Homeier, J.; Kluge, J. Impact of mycorrhization on the abundance, growth and leaf nutrient status of ferns along a tropical elevational gradient. Oecologia 2014, 175, 887–900. [Google Scholar] [CrossRef]
- Hietz, P.; Briones, O. Correlation between water relations and within-canopy distribution of epiphytic ferns in a Mexican cloud forest. Oecologia 1998, 114, 305–316. [Google Scholar] [CrossRef]
- Paixão, E.C.; Noronha, J.d.C.d.; Nunes da Cunha, C.; Arruda, R. More than light: Distance-dependent variation on riparian fern community in Southern Amazonia. Braz. J. Bot. 2013, 36, 25–30. [Google Scholar] [CrossRef]
- Zuquim, G.; Tuomisto, H.; Jones, M.M.; Prado, J.; Figueiredo, F.O.G.; Moulatlet, G.M.; Costa, F.R.C.; Quesada, C.A.; Emilio, T.; Rocchini, D. Predicting environmental gradients with fern species composition in Brazilian Amazonia. J. Veg. Sci. 2014, 25, 1195–1207. [Google Scholar] [CrossRef]
- Jones, M.M.; Cicuzza, D.; van Straaten, O.; Veldkamp, E.; Kessler, M.; Pugnaire, F. Determinants of fern and angiosperm herb community structure in lower montane rainforest in Indonesia. J. Veg. Sci. 2014, 25, 1216–1224. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, X.; Liu, J.; Zhan, X. The relationship between biodiversity and canopy openness of tropical ground ferns under three different forest types. China For. Sci. Technol. 2015, 29, 23–27. [Google Scholar]
- Kessler, M.; Lehnert, M. Do ridge habitats contribute to pteridophyte diversity in tropical montane forests? A case study from southeastern Ecuador. J. Plant Res. 2009, 122, 421–428. [Google Scholar] [CrossRef]
- Yan, Y.; He, Z.; Yuan, H.; Xing, F. The ecological response of fern diversity to different slopes in Gudoushan Nature Reserve, Guangdong. Biodivers. Sci. 2011, 19, 41–47. [Google Scholar]
- Nettesheim, F.C.; Damasceno, E.R.; Sylvestre, L.S. Different slopes of a mountain can determine the structure of ferns and lycophytes communities in a tropical forest of Brazil. An. Acad. Bras. Cienc. 2014, 86, 199–210. [Google Scholar] [CrossRef]
- Xu, C.; Li, X.; Feng, J. Relationships between epiphyte ferns species diversity and their phorophytes in Mt. Ailao National Nature Reserve. Chin. J. Ecol. 2011, 30, 1858–1862. [Google Scholar]
- Ceballos, G.; Ehrlich, P.R.; Barnosky, A.D.; Garcia, A.; Pringle, R.M.; Palmer, T.M. Accelerated modern human-induced species losses: Entering the sixth mass extinction. Sci. Adv. 2015, 1, e1400253. [Google Scholar] [CrossRef] [PubMed]
- Oldekop, J.A.; Bebbington, A.J.; Truelove, N.K.; Tysklind, N.; Villamarin, S.; Preziosi, R.F. Co-occurrence patterns of common and rare leaf-litter frogs, epiphytic ferns and dung beetles across a gradient of human disturbance. PLoS ONE 2012, 7, e38922. [Google Scholar] [CrossRef] [PubMed]
- Bystriakova, N.; Bader, M.; Coomes, D.A. Long-term tree fern dynamics linked to disturbance and shade tolerance. J. Veg. Sci. 2011, 22, 72–84. [Google Scholar] [CrossRef]
- Carvajal-Hernández, C.I.; Krömer, T.; López-Acosta, J.C.; Gómez-Díaz, J.A.; Kessler, M.; Wulf, M. Conservation value of disturbed and secondary forests for ferns and lycophytes along an elevational gradient in Mexico. Appl. Veg. Sci. 2017, 20, 662–672. [Google Scholar] [CrossRef]
- Rodríguez, M.R.; Zavala, J.H.; Pacheco, L. Presence, abundance and reproductive strategies of ferns in disturbed areas of Sierra Nevada, México. Rev. Biol. Trop. 2011, 59, 417–433. [Google Scholar]
- Castello, A.C.D.; Coelho, S.; Cardoso-Leite, E. Lianas, tree ferns and understory species: Indicators of conservation status in the Brazilian Atlantic Rainforest remnants, southeastern Brazil. Braz. J. Biol. 2017, 77, 213–226. [Google Scholar] [CrossRef]
- Silva, I.A.A.; Pereira, A.F.N.; Barros, I.C.L. Edge effects on fern community in an Atlantic forest remnant of Rio Formoso, PE, Brazil. Braz. J. Biol. 2011, 71, 421–430. [Google Scholar] [CrossRef]
- Kessler, M. Plant species richness and endemism during natural landslide succession in a perhumid montane forest in the Bolivian Andes. Ecotropica 1999, 5, 123–136. [Google Scholar]
- Kessler, M. Maximum plant community endemism at intermediate intensities of anthropogenic disturbance in Bolivian montane forests. Conserv. Biol. 2001, 15, 634–641. [Google Scholar] [CrossRef]
- Blair, D.P.; McBurney, L.M.; Blanchard, W.; Banks, S.C.; Lindenmayer, D.B. Disturbance gradient shows logging affects plant functional groups more than fire. Ecol. Appl. 2016, 26, 2280–2301. [Google Scholar] [CrossRef] [PubMed]
- Ough, K.; Murphy, A. Decline in tree-fern abundance after clearfell harvesting. For. Ecol. Manag. 2004, 199, 153–163. [Google Scholar] [CrossRef]
- Paciencia, M.L.B.; Prado, J. Effects of forest fragmentation on pteridophyte diversity in a tropical rain forest in Brazil. Plant Ecol. 2005, 180, 87–104. [Google Scholar] [CrossRef]
- da Silva, I.A.A.; Pereira, A.F.D.N.; Barros, I.C.L. Fragmentation and loss of habitat: Consequences for the fern communities in Atlantic forest remnants in Alagoas, north-eastern Brazil. Plant Ecol. Divers. 2014, 7, 509–517. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef]
- Gray, J.S. Effects of environmental stress on species rich assemblages. Biol. J. Linn. Soc. 1989, 37, 19–32. [Google Scholar] [CrossRef]
- Magura, T.; Tothmeresz, B.; Molnar, T. Changes in carabid beetle assemblages along an urbanisation gradient in the city of Debrecen, Hungary. Landsc. Ecol. 2004, 19, 747–759. [Google Scholar] [CrossRef]
- Tanaka, T.; Sato, T. Taxonomic, phylogenetic and functional diversities of ferns and lycophytes along an elevational gradient depend on taxonomic scales. Plant Ecol. 2015, 216, 1597–1609. [Google Scholar] [CrossRef]
- Kluge, J.; Kessler, M. Phylogenetic diversity, trait diversity and niches: Species assembly of ferns along a tropical elevational gradient. J. Biogeogr. 2011, 38, 394–405. [Google Scholar] [CrossRef]
- Hill, M.O. Diversity and evenness: A unifying notation and its consequences. Ecology 1973, 54, 427–432. [Google Scholar] [CrossRef]
- Chao, A.; Chiu, C.; Jost, L. Phylogenetic diversity measures based on Hill numbers. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 3599–3609. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.; Sander, E.L.; Ma, K.; Colwell, R.K.; Ellison, A.M. Rarefaction and extrapolation with Hill numbers: A framework for sampling and estimation in species diversity studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef]
- Hsieh, T.; Ma, K.; Chao, A. iNEXT: An R package for rarefaction and extrapolation of species diversity (Hill numbers). Methods Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- Hsieh, T.; Chao, A. Rarefaction and extrapolation: Making fair comparison of abundance-sensitive phylogenetic diversity among multiple assemblages. Syst. Biol. 2017, 66, 100–111. [Google Scholar] [CrossRef]
- Chao, A.; Chiu, C.; Jost, L. Unifying species diversity, phylogenetic diversity, functional diversity, and related similarity and differentiation measures through Hill numbers. Annu. Rev. Ecol. Evol. Syst. 2014, 45, 297–324. [Google Scholar] [CrossRef]
- Vega-Álvarez, J.; García-Rodríguez, J.A.; Cayuela, L.; Swenson, N. Facilitation beyond species richness. J. Ecol. 2019, 107, 722–734. [Google Scholar] [CrossRef]
- Ebihara, A.; Nitta, J.H. An update and reassessment of fern and lycophyte diversity data in the Japanese Archipelago. J. Plant Res. 2019, 132, 723–738. [Google Scholar] [CrossRef]
- Sessa, E.B.; Chambers, S.M.; Li, D.; Trotta, L.; Endara, L.; Burleigh, J.G.; Baiser, B. Community assembly of the ferns of Florida. Am. J. Bot. 2018, 105, 549–564. [Google Scholar] [CrossRef] [PubMed]
- Link-Pérez, M.A.; Laffan, S.W. Fern and lycophyte diversity in the Pacific Northwest: Patterns and predictors. J. Syst. Evol. 2018, 56, 498–522. [Google Scholar] [CrossRef]
- Lehtonen, S.; Jones, M.M.; Zuquim, G.; Prado, J.; Tuomisto, H. Phylogenetic relatedness within Neotropical fern communities increases with soil fertility. Glob. Ecol. Biogeogr. 2015, 24, 695–705. [Google Scholar] [CrossRef]
- Kessler, M.; Karger, D.N.; Kluge, J. Elevational diversity patterns as an example for evolutionary and ecological dynamics in ferns and lycophytes. J. Syst. Evol. 2016, 54, 617–625. [Google Scholar] [CrossRef]
- The Editorial Committee of China National Geographic Atlas. China National Geographic Atlas; Encyclopedia of China Publishing House: Beijing, China, 2011. [Google Scholar]
- Steinshouer, D.W.; Qiang, J.; McCabe, P.J.; Ryder, R.T. Maps Showing Geology, Oil and Gas Fields, and Geologic Provinces of the Asia Pacific Region; 97-470F; USGA: Reston, VA, USA, 1999. [Google Scholar]
- Liu, P.; Liao, C.; Xu, J.; Staines, C.L.; Dai, X. The Cassidinae beetles of Longnan County (Jiangxi, China): Overview and community composition. Biodivers. Data J. 2019, 7, e39053. [Google Scholar] [CrossRef]
- Dai, X.; Xu, J.; Guo, Q.; Lai, S.; Liu, P.; Fan, J.; Tang, P. Density effect and intraspecific competition in a leaf-mining moth on bamboo leaves. J. For. Res. 2018, 30, 689–697. [Google Scholar] [CrossRef]
- Dai, X.; Xu, J.; Ding, X. Circular distribution pattern of plant modulars and endophagous herbivory within tree crowns: The impact of roadside light conditions. J. Insect Sci. 2013, 13, 141. [Google Scholar] [CrossRef]
- Dai, X.; Xu, J.; Cai, L. Effects of roads on Castanopsis carlesii seedlings and their leaf herbivory in a subtropical forest in China. J. Insect Sci. 2014, 14, 17. [Google Scholar] [CrossRef]
- QGIS Development Team. QGIS Geographic Information System; Open Source Geospatial Foundation Project. Available online: http://qgis.osgeo.org (accessed on 4 February 2020).
- Yan, Y.; Yi, Q.; Huang, Z.; Xing, F. The ecological response of fern diversity to vegetation succession in Gudoushan Nature Reserve, Guangdong. Biodivers. Sci. 2004, 12, 339–347. [Google Scholar]
- Bystriakova, N.; Peregrym, M.; Dragicevic, S. Effect of environment on distributions of rock ferns in the Mediterranean climate: The case of the genus Asplenium in Montenegro. Flora Morphol. Distrib. Funct. Ecol. Plants 2015, 215, 84–91. [Google Scholar] [CrossRef]
- Pryer, K.M.; Schneider, H.; Smith, A.R.; Cranfill, R.; Wolf, P.G.; Hunt, J.S.; Sipes, S.D. Horsetails and ferns are a monophyletic group and the closest living relatives to seed plants. Nature 2001, 409, 618–622. [Google Scholar] [CrossRef]
- Zhang, J. Plantlist: Looking up the Status of Plant Scientific Names Based on the Plant List Database. R Package Version 0.5. 5. Available online: https://github.com/helixcn/plantlist (accessed on 15 November 2018).
- Jin, Y.; Qian, H.V. PhyloMaker: An R package that can generate very large phylogenies for vascular plants. Ecography 2019, 42, 1353–1359. [Google Scholar] [CrossRef]
- Zanne, A.E.; Tank, D.C.; Cornwell, W.K.; Eastman, J.M.; Smith, S.A.; FitzJohn, R.G.; McGlinn, D.J.; O’Meara, B.C.; Moles, A.T.; Reich, P.B.; et al. Three keys to the radiation of angiosperms into freezing environments. Nature 2014, 506, 89–92. [Google Scholar] [CrossRef]
- Smith, S.A.; Brown, J.W. Constructing a broadly inclusive seed plant phylogeny. Am. J. Bot. 2018, 105, 302–314. [Google Scholar] [CrossRef]
- Kembel, S.W.; Cowan, P.D.; Helmus, M.R.; Cornwell, W.K.; Morlon, H.; Ackerly, D.D.; Blomberg, S.P.; Webb, C.O. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 2010, 26, 1463–1464. [Google Scholar] [CrossRef]
- Laliberte, E.; Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 2010, 91, 299–305. [Google Scholar] [CrossRef]
- Li, D. hillR: Taxonomic, functional, and phylogenetic diversity and similarity through Hill Numbers. J. Open Source Softw. 2018, 3, 1041. [Google Scholar] [CrossRef]
- Chiu, C.; Chao, A. Distance-based functional diversity measures and their decomposition: A framework based on Hill numbers. PLoS ONE 2014, 9, e100014. [Google Scholar] [CrossRef]
- Morris, E.K.; Caruso, T.; Buscot, F.; Fischer, M.; Hancock, C.; Maier, T.S.; Meiners, T.; Muller, C.; Obermaier, E.; Prati, D.; et al. Choosing and using diversity indices: Insights for ecological applications from the German Biodiversity Exploratories. Ecol. Evol. 2014, 4, 3514–3524. [Google Scholar] [CrossRef]
- Faith, D.P. Conservation evaluation and phylogenetic diversity. Biol. Conserv. 1992, 61, 1–10. [Google Scholar] [CrossRef]
- Walker, B.; Kinzig, A.; Langridge, J. Plant attribute diversity, resilience, and ecosystem function: The nature and significance of dominant and minor species. Ecosystems 1999, 2, 95–113. [Google Scholar] [CrossRef]
- Guiasu, R.C.; Guiasu, S. The weighted quadratic index of biodiversity for pairs of species: A generalization of Rao’s index. Nat. Sci. 2011, 3, 795–801. [Google Scholar] [CrossRef]
- Guiasu, R.C.; Guiasu, S. The weighted Gini-Simpson index: Revitalizing an old index of biodiversity. Int. J. Ecol. 2012, 2012, 1–10. [Google Scholar] [CrossRef]
- Caceres, M.D.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- RStudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2019. [Google Scholar]
- Yusuf, F.B.; Tan, B.C.; Turner, I.M. What is the minimum area needed to estimate the biodiversity of pteridophytes in natural and man-made lowland forests in Malaysia and Singapore? Fern Gaz. 2003, 17, 1–9. [Google Scholar]
- Barthlott, W.; Schmit-Neuerburg, V.; Nieder, J.; Engwald, S. Diversity and abundance of vascular epiphytes: A comparison of secondary vegetation and primary montane rain forest in the Venezuelan Andes. Plant Ecol. 2001, 152, 145–156. [Google Scholar] [CrossRef]
- De Palma, A.; Kuhlmann, M.; Bugter, R.; Ferrier, S.; Hoskins, A.J.; Potts, S.G.; Roberts, S.P.M.; Schweiger, O.; Purvis, A. Dimensions of biodiversity loss: Spatial mismatch in land-use impacts on species, functional and phylogenetic diversity of European bees. Divers. Distrib. 2017, 23, 1435–1446. [Google Scholar] [CrossRef]
- Giehl, E.L.H.; Jarenkow, J.A.; Prinzing, A. Disturbance and stress gradients result in distinct taxonomic, functional and phylogenetic diversity patterns in a subtropical riparian tree community. J. Veg. Sci. 2015, 26, 889–901. [Google Scholar] [CrossRef]
- Marchant, R. The use of taxonomic distinctness to assess environmental disturbance of insect communities from running water. Freshw. Biol. 2007, 52, 1634–1645. [Google Scholar] [CrossRef]
- Sobral, F.L.; Jardim, L.; Lemes, P.; Machado, N.; Loyola, R.; Cianciaruso, M.V. Spatial conservation priorities for top predators reveal mismatches among taxonomic, phylogenetic and functional diversity. Nat. Conserv. 2014, 12, 150–155. [Google Scholar] [CrossRef]
- Li, Y.; Lu, Q.; Wu, B.; Zhu, Y.; Liu, D.; Zhang, J.; Jin, Z. A review of leaf morphology plasticity linked to plant response and adaptation characteristics in arid ecosystems. Chin. J. Plant Ecol. 2012, 36, 88–98. [Google Scholar] [CrossRef]
- Kluge, J.; Kessler, M. Morphological characteristics of fern assemblages along an elevational gradient: Patterns and causes. Ecotropica 2007, 13, 27–43. [Google Scholar]
- Gonzales, J.; Kessler, M. A synopsis of the Neotropical species of Sticherus (Gleicheniaceae), with descriptions of nine new species. Phytotaxa 2011, 31, 1–54. [Google Scholar]
- Pang, C.; Ma, X.K.; Hung, T.T.; Hau, B.C. Early ecological succession on landslide trails, Hong Kong, China. Ecoscience 2018, 25, 153–161. [Google Scholar] [CrossRef]
- Ranker, T.A.; Gemmill, C.E.C.; Trapp, P.G. Microevolutionary patterns and processes of the native Hawaiian colonizing fern Odontosoria chinensis (Lindsaeaceae). Evolution 2000, 54, 828–839. [Google Scholar] [CrossRef]
- Lynch, R.L.; Chen, H.; Brandt, L.A.; Mazzotti, F.J. Old World climbing fern (Lygodium microphyllum) invasion in hurricane caused treefalls. Nat. Areas J. 2009, 29, 210–215. [Google Scholar] [CrossRef]
- Hou, X.; Liu, S.; Zhao, S.; Beazley, R.; Cheng, F.; Wu, X.; Xu, J.; Dong, S. Selection of suitable species as a key factor for vegetation restoration of degraded areas in an open-pit manganese-ore mine in Southern China using multivariate-analysis methods. Land Degrad. Dev. 2019, 30, 942–950. [Google Scholar] [CrossRef]
- Ramadhanil, R.; Tjitrosoedirdjo, S.S.; Setiadi, D. Structure and composition of understory plant assemblages of six land use types in the Lore Lindu National Park, Central Sulawesi, Indonesia. Bangladesh J. Plant Taxon. 2008, 15, 1–12. [Google Scholar] [CrossRef]
- Kang, B.; Liu, S.; Cai, D.; Lu, L. Effects of Pinus massoniana plantation stand density on understory vegetation and soil properties. Chin. J. Appl. Ecol. 2009, 20, 2323–2331. [Google Scholar]
- He, J.; Chen, W.; Xie, Z.; Hu, D. Types of the evergreen broadleaf forest and their community species diversity in Longqishan Nature Reserve, Fujian Province. Chin. J. Ecol. 1998, 17, 1–6. [Google Scholar]
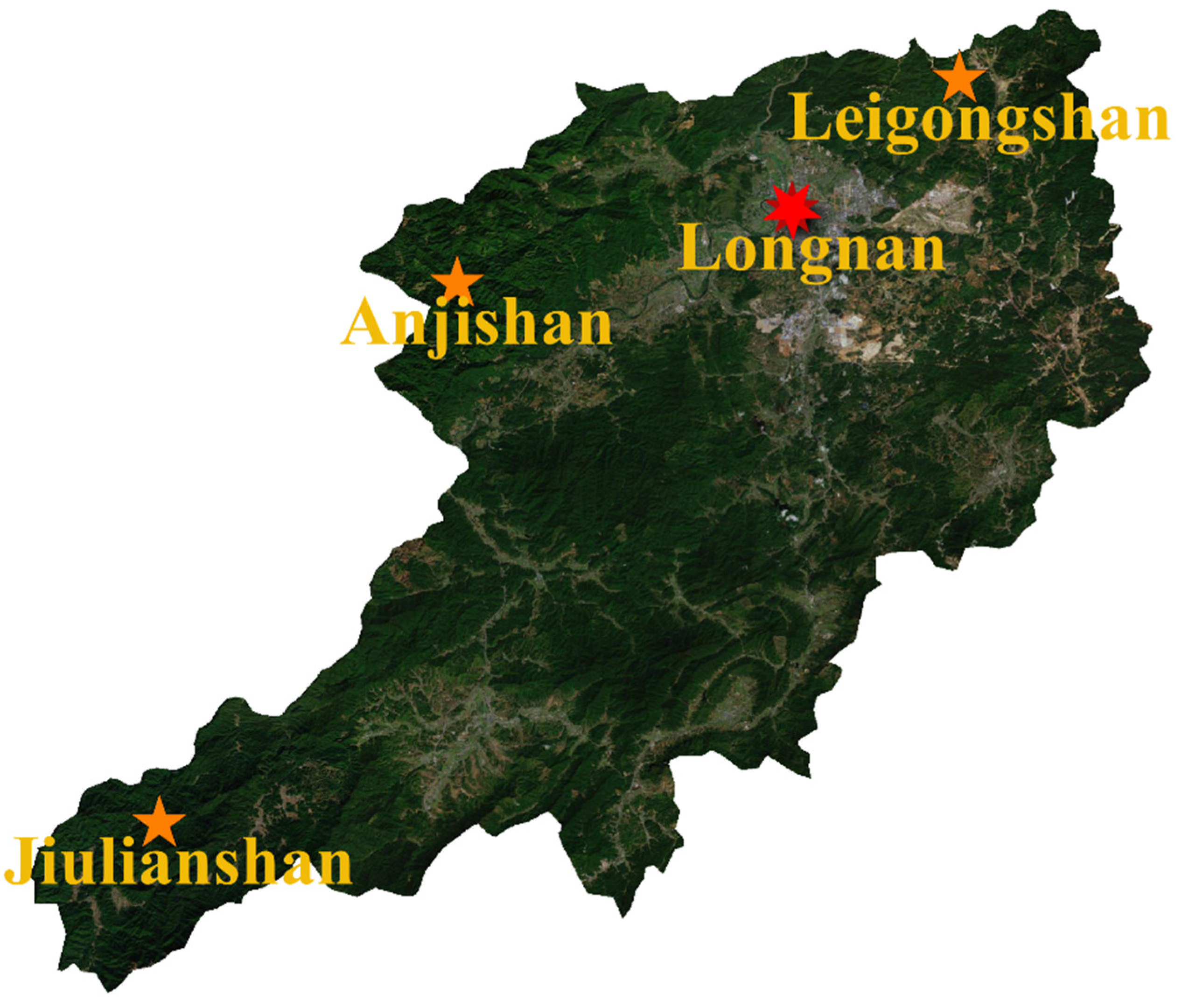
| Study Sites | Specific Sample Routes | Distance to County Town (km) | Main Vegetation | Disturbance Level | Elevational Range of Samples (m) |
|---|---|---|---|---|---|
| Jiulianshan National Nature Reserve (JLS) | Daqiutian-Hengkengshui-Xiagongtang | 80 | Primary evergreen broad-leaved forests | low | approximately 490 ± 80 |
| Anjishan Provincial Forest Park (AJS) | Qingchahu- Anjishan- Zhongping | 30 | Secondary evergreen broad-leaved forests and economic forests | intermediate | approximately 410 ± 50 |
| Leigongshan Family Farm (LGS) | Around Leigongshan orchards | 10 | Chinese fir forests and navel-orange orchards | high | approximately 310 ± 30 |
| Diversity Component | Hill Number Series | All Species (q = 0) | Common Species (q = 1) | Dominant Species (q = 2) | References |
|---|---|---|---|---|---|
| taxonomic diversity @ | the effective number of species | S | 1/λ | [73,74,101,102] | |
| phylogenetic diversity # | the effective total phylogenetic distance between species | FPD/T | 1/(1-Q/T) | [73,103] | |
| functional diversity $ | the effective total functional distance between species | FAD | - | [101,104,105,106] |
| Taxonomic Diversity Index | JLS 1 | AJS | LGS |
|---|---|---|---|
| Species richness (S = 0TD) | 79 | 61 | 39 |
| Exponential of Shannon’s entropy (eH = 1TD) | 54 | 39 | 21 |
| Inverse of Simpson’s index (1/λ = 2TD) | 22 | 15 | 9 |
| Genus richness | 47 | 38 | 23 |
| Family richness | 20 | 17 | 11 |
| Order richness | 9 | 7 | 4 |
| Subclass richness | 4 | 4 | 1 |
| Individual number | 3250 | 2905 | 3604 |
| Functional Traits | JLS 1 | AJS | LGS |
|---|---|---|---|
| Lamina length | 66.048 | 68.277 | 88.670 |
| Lamina width | 21.373 | 17.624 | 17.748 |
| Petiole length | 36.720 | 33.146 | 43.481 |
| Frond heterophylly | 1.096 | 1.080 | 1.084 |
| Lamina texture | 2.178 | 2.149 | 2.087 |
| Frond arrangement | 2.066 | 2.386 | 2.119 |
| Rhizome type | 2.228 | 2.444 | 2.148 |
| Rhizome position | 1.069 | 1.094 | 1.000 |
| Lamina dissection | 2.617 | 2.351 | 3.131 |
| Lamina shape | linear-lanceolate | linear-lanceolate | linear-lanceolate |
| Sori shape | orbicular | linear | orbicular |
| Sori position | medial_on_veins | 1_row_on_either_side_of_veins | 1_line_on_each_side_of_costule |
| Indusia | 0.677 | 0.682 | 0.878 |
| Reproduction type | 1.021 | 1.023 | 1.002 |
| Phenology | 1.014 | 1.030 | 1.031 |
| Habit | 1.109 | 1.068 | 1.000 |
| Scales or hair density | 1.186 | 1.155 | 1.259 |
| Disturbance Level | Indicator Species | Indicator Value | P-Value | Significance Code 1 |
|---|---|---|---|---|
| High | Dicranopteris pedata | 0.692 | 0.0001 | *** |
| Blechnum orientale | 0.612 | 0.0001 | *** | |
| Odontosoria chinensis | 0.579 | 0.0001 | *** | |
| Lygodium microphyllum | 0.376 | 0.0004 | *** | |
| Cyclosorus parasiticus | 0.366 | 0.0015 | ** | |
| Christella dentata | 0.274 | 0.016 | * | |
| Pteris semipinnata | 0.264 | 0.0165 | * | |
| Intermediate | Leptochilus hemitomus | 0.429 | 0.0001 | *** |
| Diplazium virescens | 0.331 | 0.0034 | ** | |
| Arachniodes chinensis | 0.330 | 0.0005 | *** | |
| Coniogramme japonica | 0.248 | 0.0001 | *** | |
| Low | Ctenitis subglandulosa | 0.536 | 0.0001 | *** |
| Angiopteris fokiensis | 0.434 | 0.0002 | *** | |
| Cyathea hancockii | 0.378 | 0.0001 | *** | |
| Cornopteris opaca | 0.333 | 0.0001 | *** | |
| Dictyocline wilfordii | 0.314 | 0.0039 | ** | |
| Arachniodes amabilis | 0.300 | 0.0057 | ** | |
| Plagiogyria adnata | 0.261 | 0.0007 | *** | |
| Intermediate + low | Osmunda vachellii | 0.243 | 0.047 | * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, X.; Chen, C.; Li, Z.; Wang, X. Taxonomic, Phylogenetic, and Functional Diversity of Ferns at Three Differently Disturbed Sites in Longnan County, China. Diversity 2020, 12, 135. https://doi.org/10.3390/d12040135
Dai X, Chen C, Li Z, Wang X. Taxonomic, Phylogenetic, and Functional Diversity of Ferns at Three Differently Disturbed Sites in Longnan County, China. Diversity. 2020; 12(4):135. https://doi.org/10.3390/d12040135
Chicago/Turabian StyleDai, Xiaohua, Chunfa Chen, Zhongyang Li, and Xuexiong Wang. 2020. "Taxonomic, Phylogenetic, and Functional Diversity of Ferns at Three Differently Disturbed Sites in Longnan County, China" Diversity 12, no. 4: 135. https://doi.org/10.3390/d12040135
APA StyleDai, X., Chen, C., Li, Z., & Wang, X. (2020). Taxonomic, Phylogenetic, and Functional Diversity of Ferns at Three Differently Disturbed Sites in Longnan County, China. Diversity, 12(4), 135. https://doi.org/10.3390/d12040135





