Middle Eocene Rhodoliths from Tropical and Mid-Latitude Regions
Abstract
1. Introduction
2. Materials and Methods
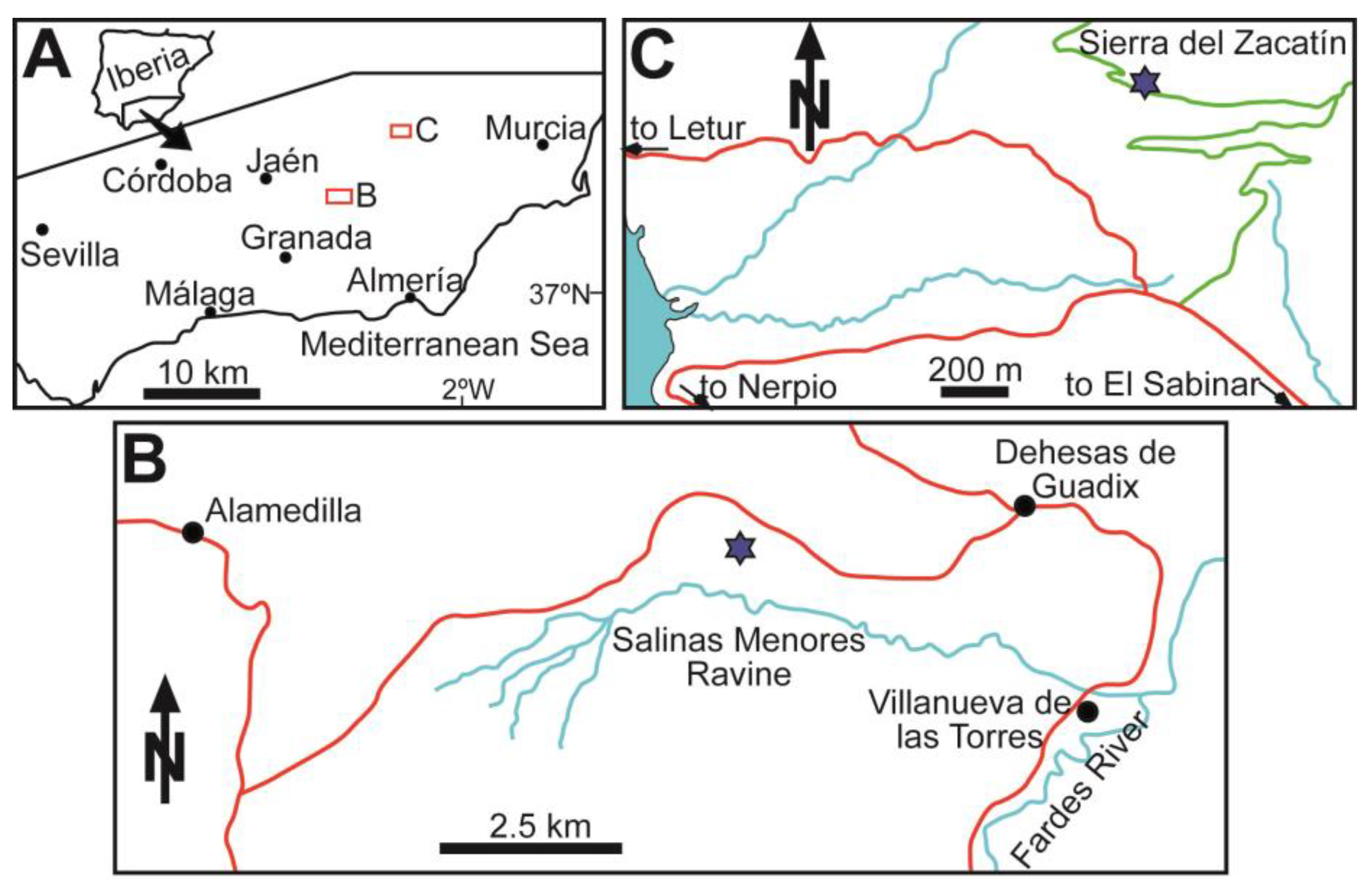
3. Study Areas: Location and Geological Settings
3.1. Southern Spain
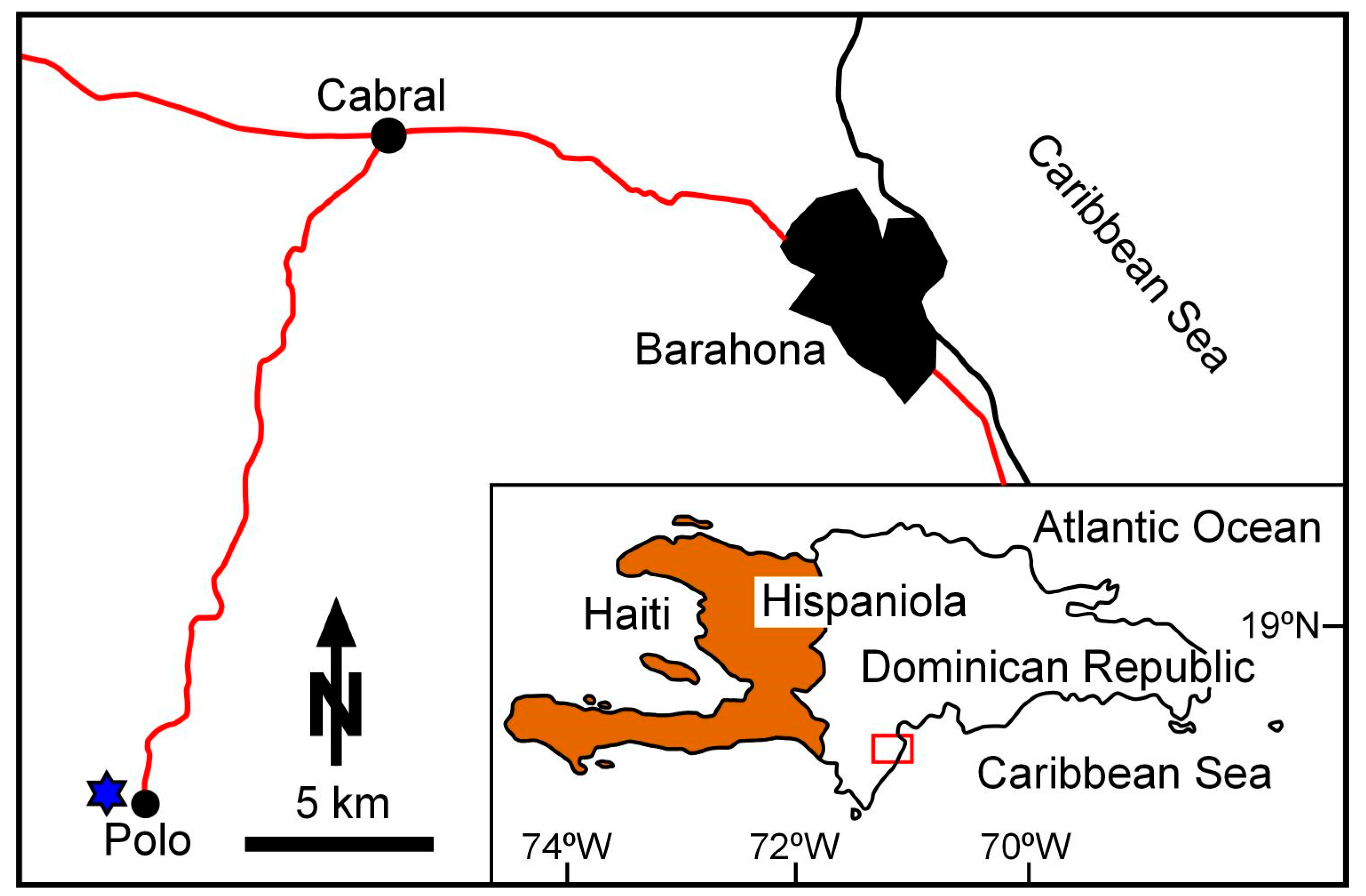
3.2. Dominican Republic
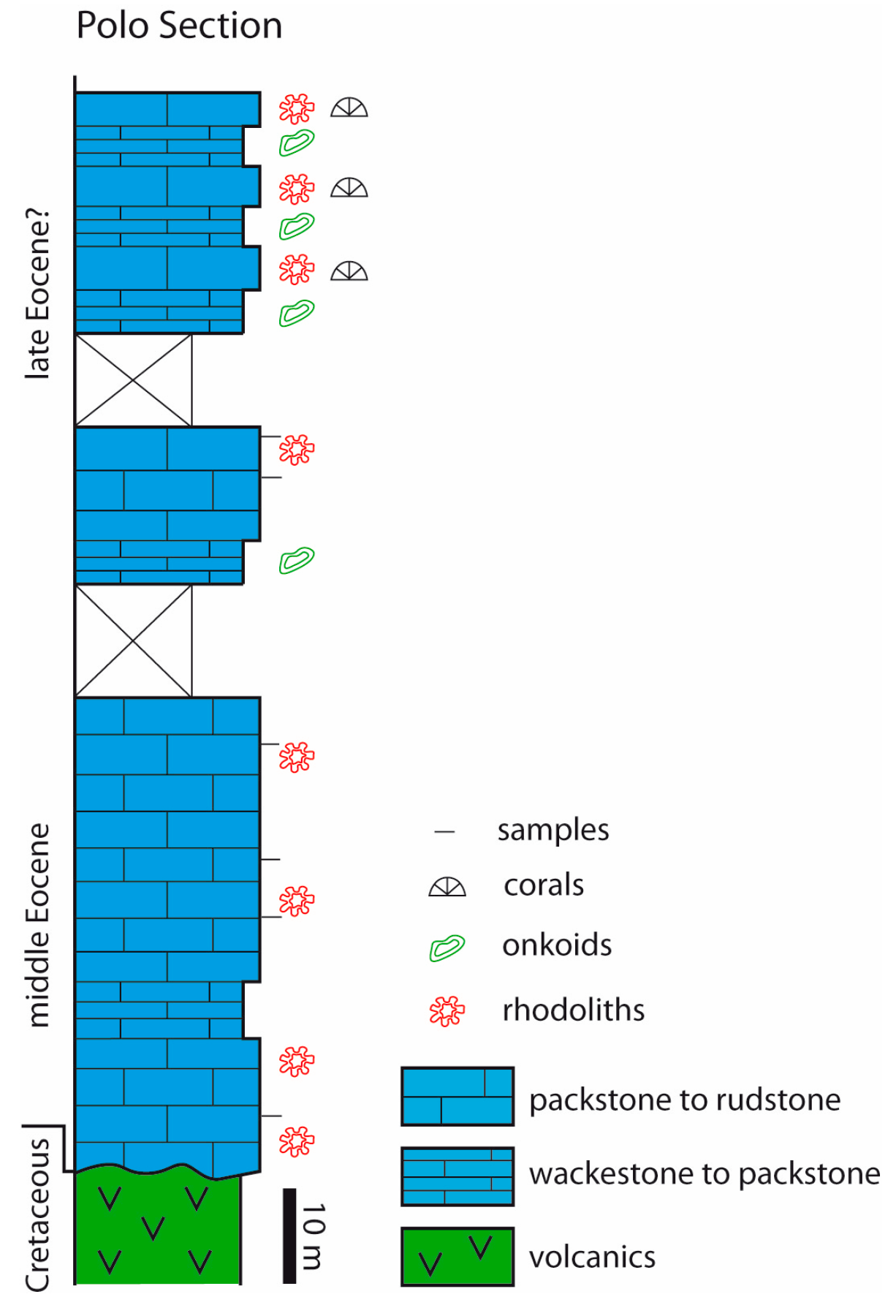
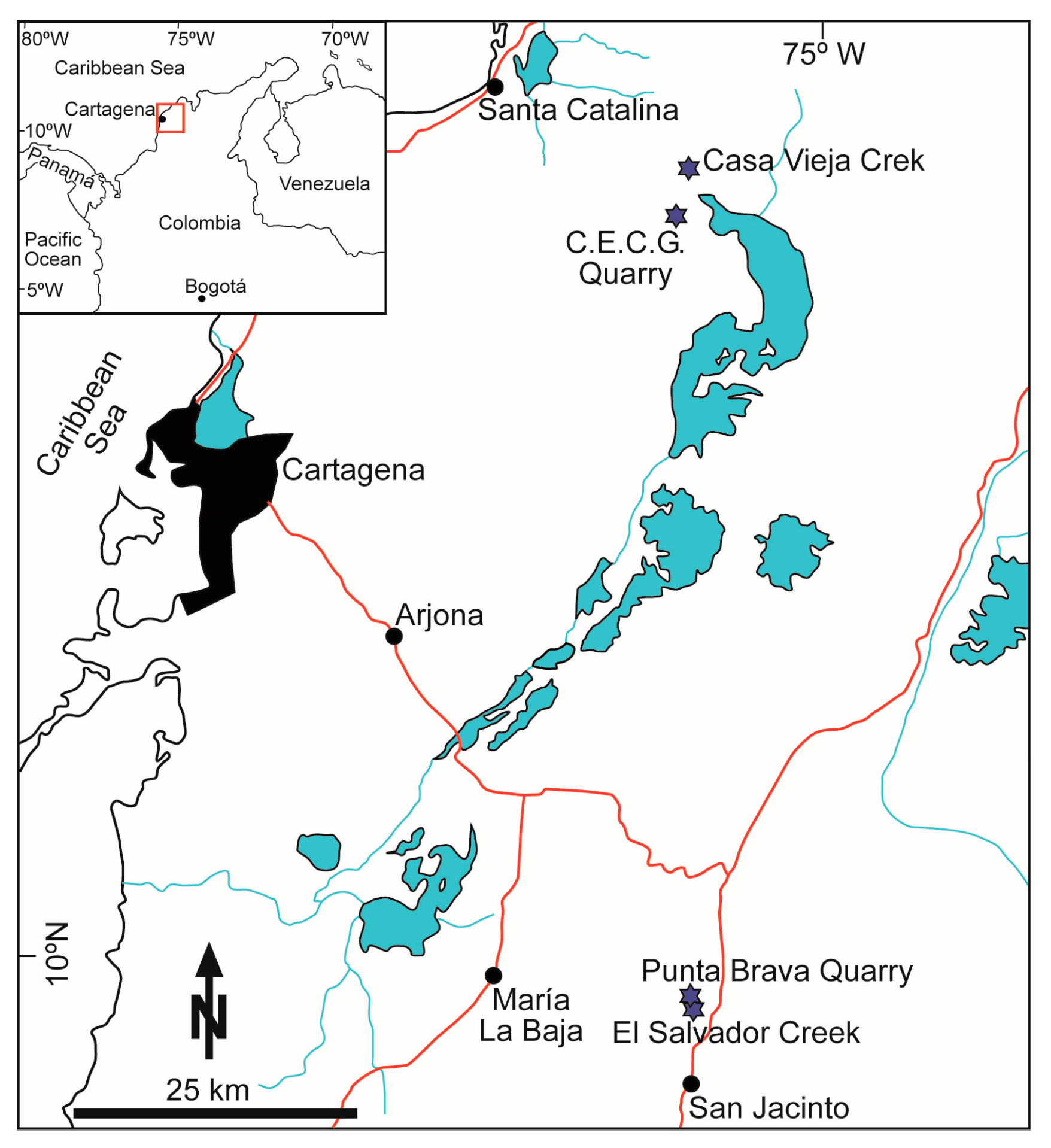
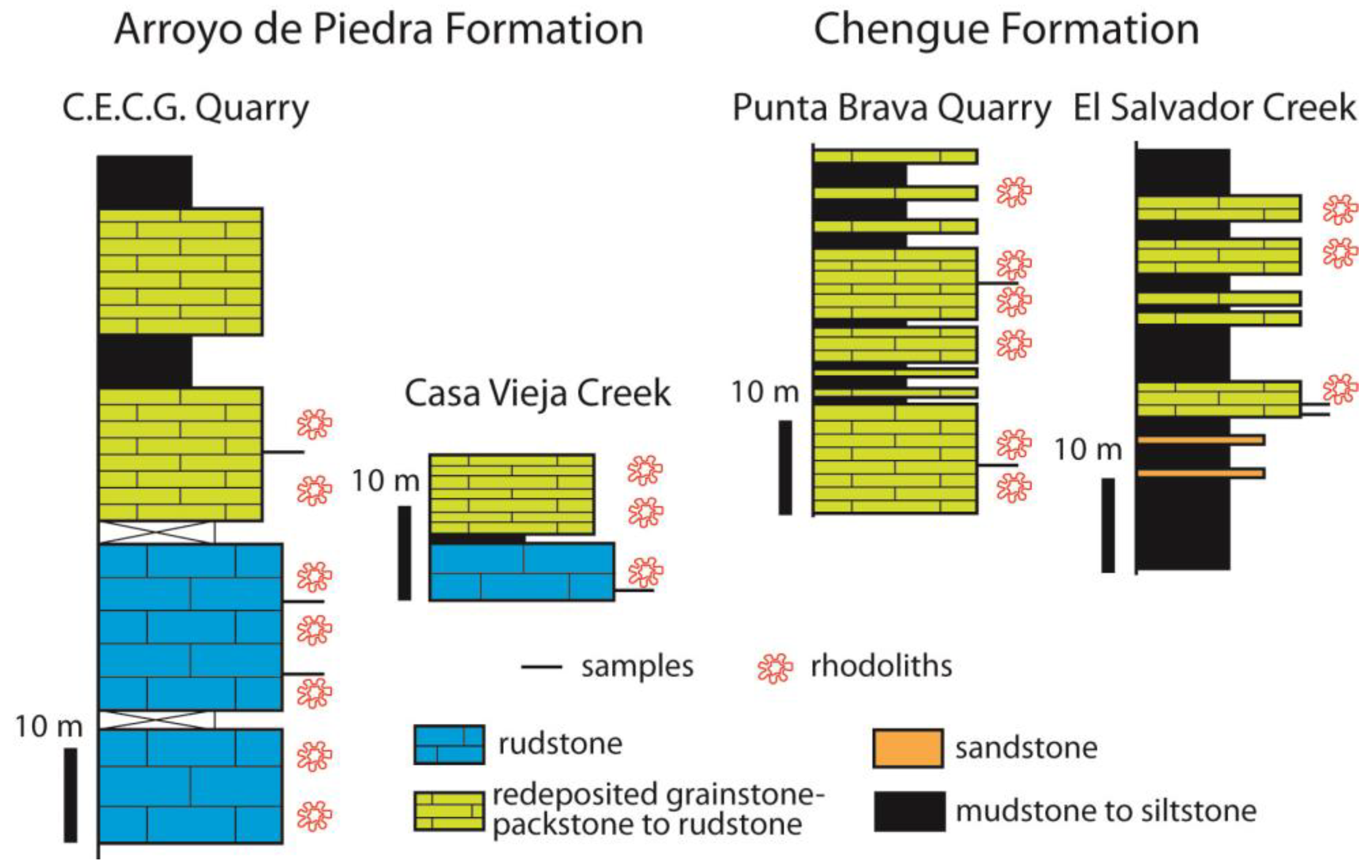
3.3. Colombia
4. Results
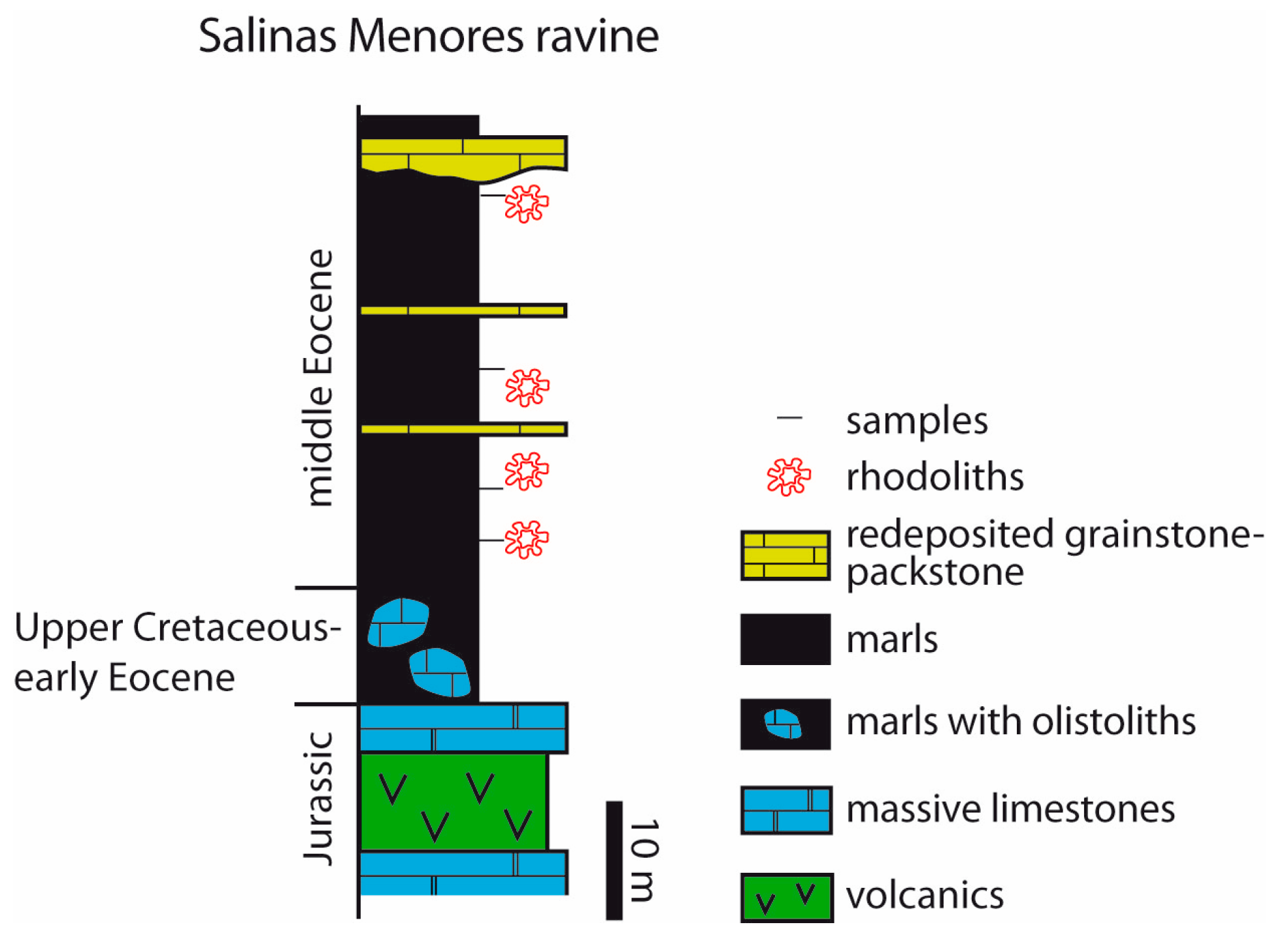
4.1. Salinas Menores Ravine
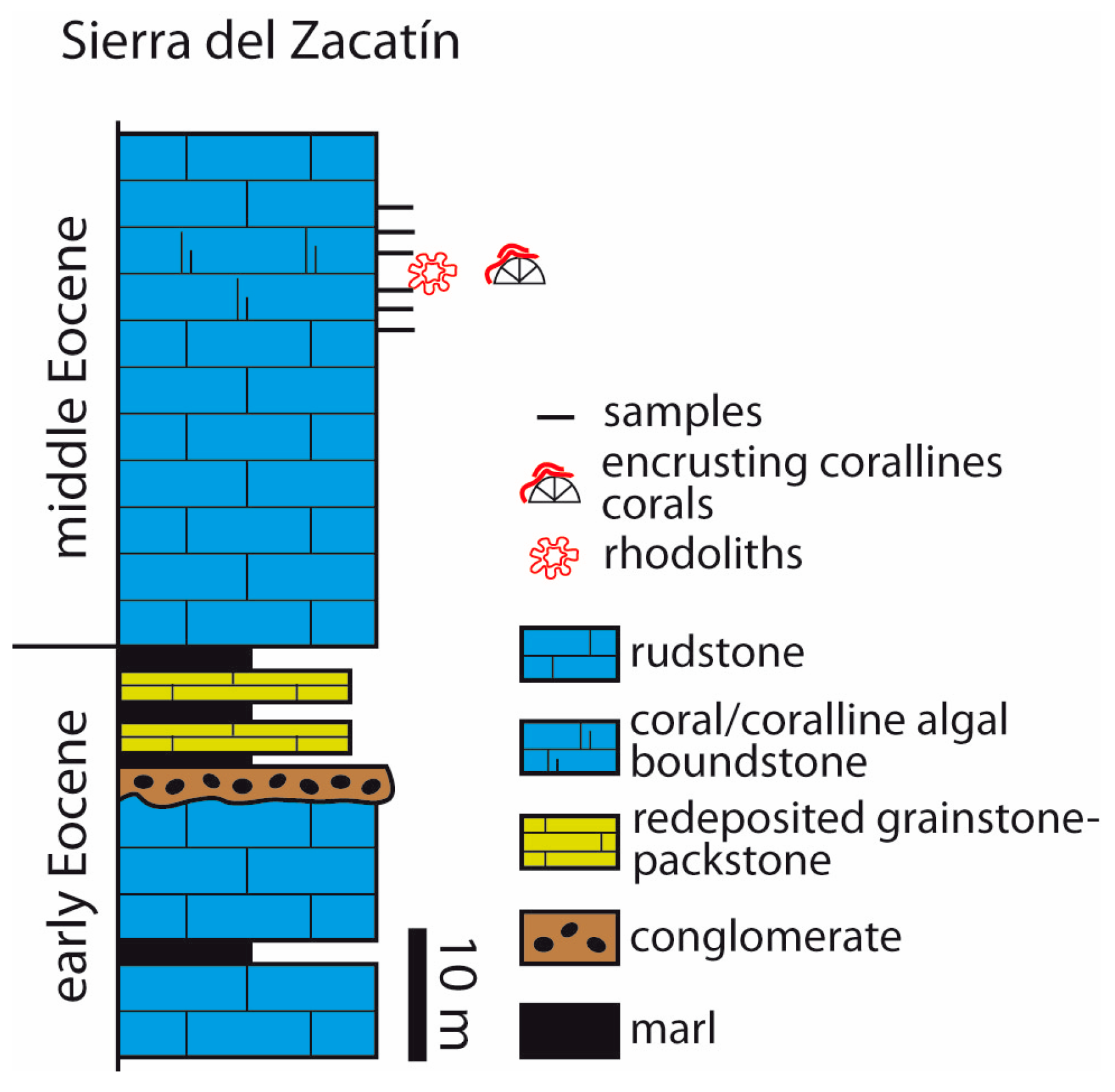
4.2. Sierra del Zacatín
4.3. Dominican Republic
4.4. Colombia
5. Discussion
5.1. Paleoenvironmental Interpretation
5.2. Taxonomic Composition
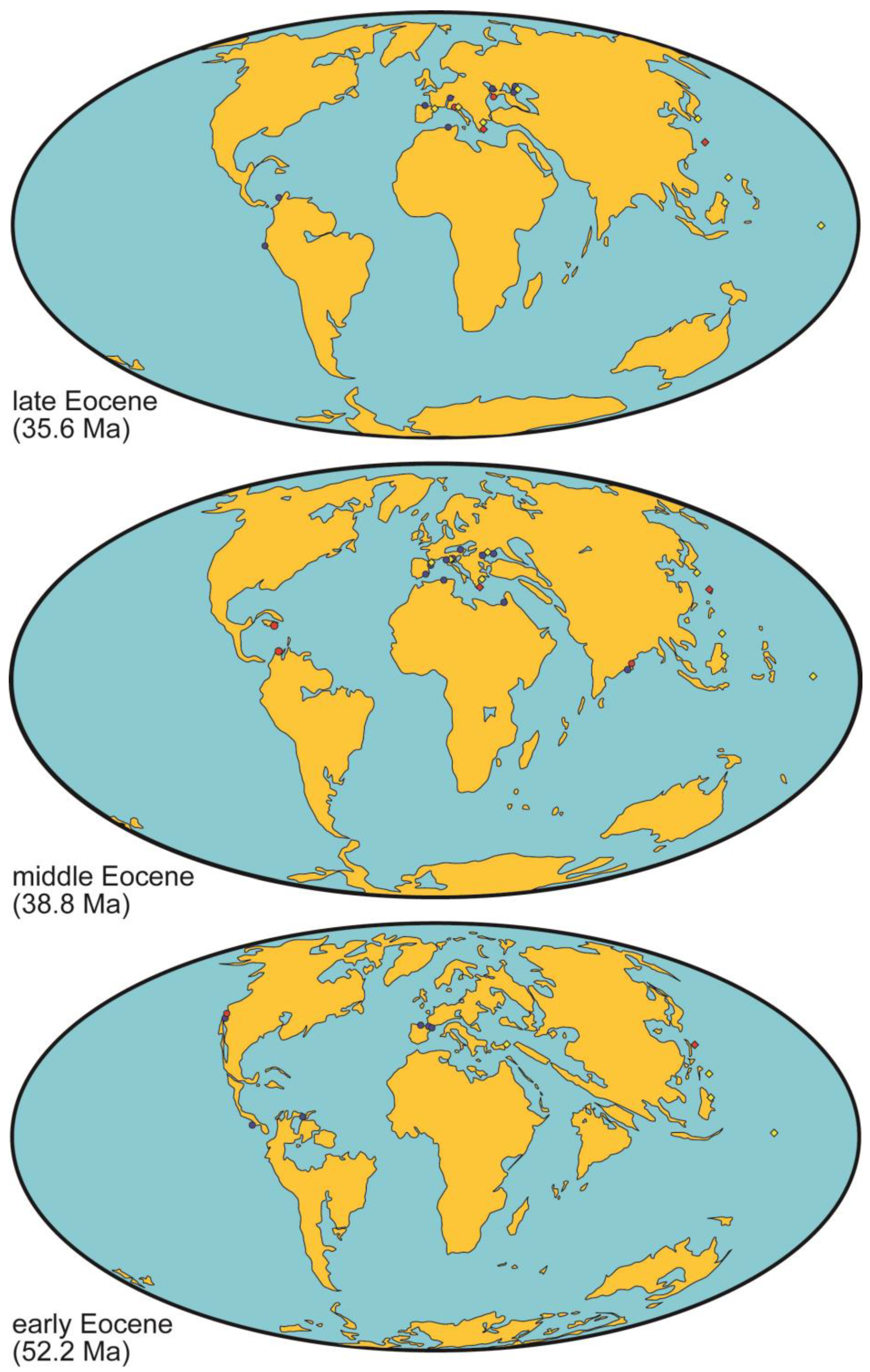
5.3. Rhodolith Beds during the Eocene
6. Conclusions
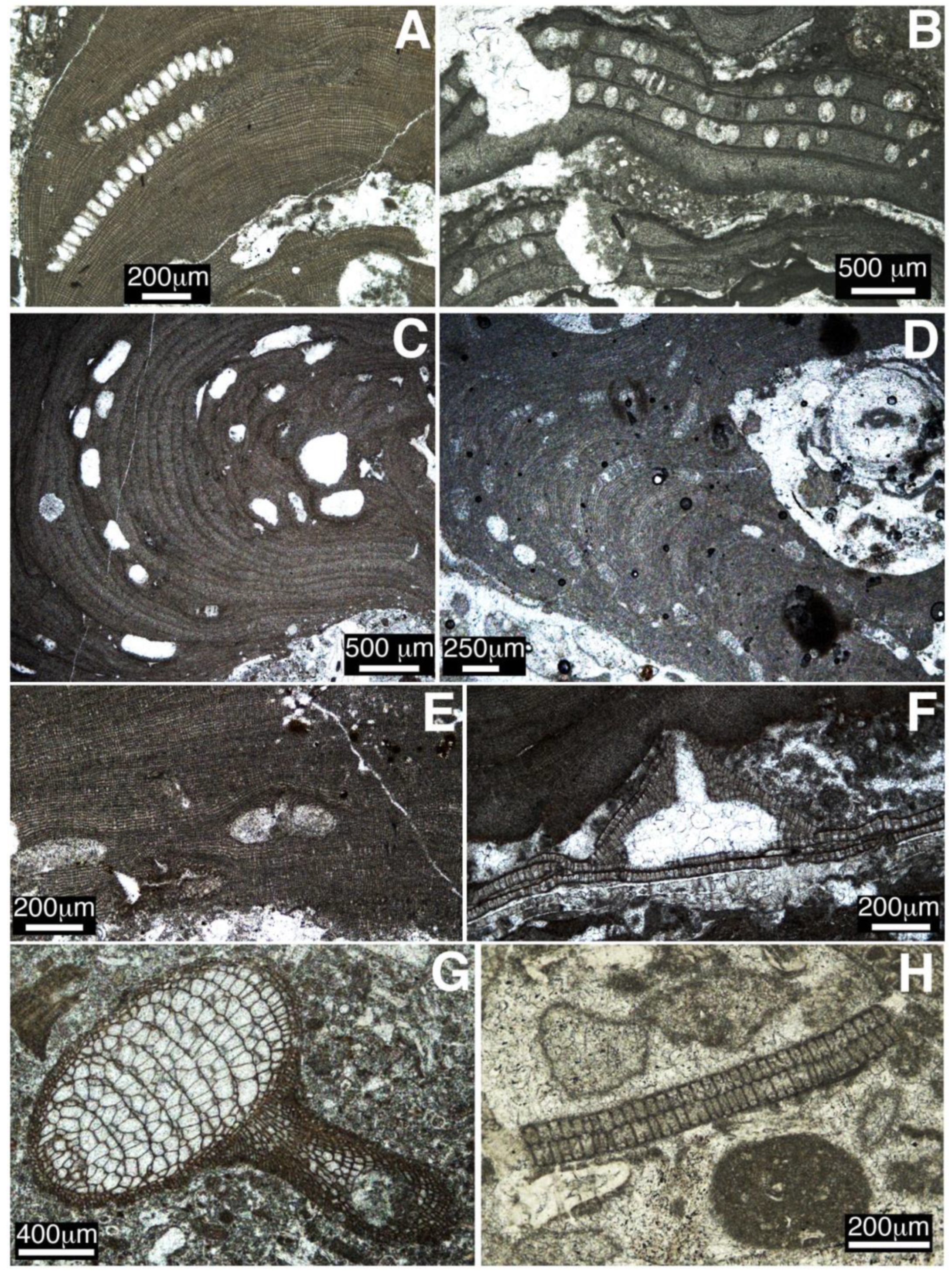
- In two tropical settings, the Dominican Republic and Colombia, middle Eocene coralline algae occur as dense concentrations of rhodoliths in rhodolith rudstone lithofacies. Rhodoliths are ellipsoidal to spheroidal in shape and are composed by encrusting to warty coralline algal thalli in association with benthic foraminifers, bryozoans, corals, and other calcareous red algae such as Parachaetetes asvapatii and Polystrata alba.
- In mid-latitude areas in Southern Spain, coralline algae occur in two different contexts. In Salinas Menores ravine, rhodoliths are spheroidal to ellipsoidal and occur dispersed in planktic foraminifer-rich marls. In the Sierra del Zacatín, LBF dominate the middle Eocene carbonate deposits and rhodoliths are scarce. Coralline algae mostly occur attached to and intergrown with corals. Rhodoliths consist of encrusting and warty (occasionally fruticose) algal thalli intergrown with bryozoans, corals, and benthic foraminifers.
- In all the study areas, coralline algal assemblages are dominated by Hapalidiales and Sporolithales. The order Corallinales is scarcely represented, except in Salinas Menores, where its members are relatively abundant and diverse. Within this group, Subterraniphyllum thomasii is found in the Salinas Menores section. The oldest previously known record of this species is from the late Eocene; therefore, our finding extends the occurrence of S. thomasii back to the middle Eocene.
- Rhodoliths in the two tropical areas developed in relatively deep platform settings (tens of meters of water depth) as shown by the LBF and coralline algal assemblages. In the Salinas Menores section, miliolids in the internal voids indicate that rhodoliths grew in a shallow-water inner platform or lagoon and were re-deposited in deep outer-shelf settings.
- During the greenhouse conditions in the early–middle Eocene, shallow-water carbonate platforms from the tropics to intermediate latitudes were depauperate in rhodolith beds. The key paleoenvironmental factors accounting for this decline remain elusive up to now. Extremely high global temperatures due to high atmospheric CO2 concentrations could negatively affect coralline algae. In addition, distribution of oceanic productivity might account for the main carbonate producers in marine shelves: mesotrophic conditions associated with upwelling areas in tropical regions could have favored the development of rhodolith beds, such as those in the Dominican Republic and Colombia, while oligotrophic conditions in mid–high latitudes catalyzed the widespread dominance by larger benthic foraminifer assemblages, as observed in Southern Spain.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adey, W.H.; Macintyre, I.G. Crustose coralline algae: A re-evaluation in the geological sciences. Geol. Soc. Am. Bull. 1971, 84, 883–904. [Google Scholar] [CrossRef]
- Foster, M.S. Rhodoliths: Between rocks and soft places. J. Phycol. 2001, 87, 659–667. [Google Scholar] [CrossRef]
- Kamenos, N.A.; Burdett, H.L.; Darrenougue, N. Coralline algae as recorders of past climatic and environmental conditions. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 27–53. [Google Scholar]
- Aguirre, J.; Braga, J.C.; Bassi, D. Rhodoliths and rhodolith beds in the rock record. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 105–138. [Google Scholar]
- Johnson, M.E.; Ledesma-Vázquez, J.; Ramalho, R.S.; da Silva, C.M.; Rebelo, A.C.; Santos, A.; Baarli, G.; Mayoral, E.; Cachão, M. Taphonomic range and sedimentary dynamics of modern and fossil rhodolith beds: Macaronesian realm (North Atlantic Ocean). In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 221–261. [Google Scholar]
- Bassi, D.; Simone, L.; Nebelsik, J.H. Re-sedimented rhodoliths in channelized depositional systems. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 139–167. [Google Scholar]
- Moussavian, E.; Salas, R.; Martín-Closas, C. Evidence of modern red algae (Corallinaceae, Peyssonneliaceae) in the prae-Barremian Cretaceous. In Proceedings of the Abstracts, 2nd Regional Symposium on Fossil Algae, Alpine Algae, Munich, Germany, 29 August–5 September 1993; p. 42. [Google Scholar]
- Arias, C.; Masse, J.P.; Vilas, L. Hauterivian shallow marine calcareous biogenic mounds: SE Spain. Palaeogeog. Palaeoclimatol. Palaeoecol. 1995, 119, 3–17. [Google Scholar] [CrossRef]
- Tomás, S.; Aguirre, J.; Braga, J.C.; Martín-Closas, C. Late Hauterivian coralline algae (Rhodophyta, Corallinales) from the Iberian Chain (E Spain). Taxonomy and the evolution of multisporangial reproductive structures. Facies 2007, 53, 79–95. [Google Scholar] [CrossRef]
- Chatalov, A.; Bonev, N.; Ivanova, D. Depositional characteristics and constraints on the mid-Valanginian demise of a carbonate platform in the intra-Tethyan domain, Circum-Rhodope Belt, northern Greece. Cretac. Res. 2015, 55, 84–115. [Google Scholar] [CrossRef]
- Pfender, J. Sur les organismes du Nummulitique de la colline de San Salvador près Camarasa. Bol. R. Acad. Esp. Hist. Nat. 1926, 26, 321–330. [Google Scholar]
- Beckmann, J.P.; Beckmann, R. Cretaceous algae from the Cretaceous and Tertiary of Cuba. Schweiz. Paläont. Abh. 1966, 85, 1–45. [Google Scholar]
- Lemoine, P.M. Les algues floridées calcaires du Crétacé du Sud de la France. Arch. Mus. Nat. Hist. Nat. Paris 1970, 7, 129–240. [Google Scholar]
- Poignant, A.F. Sur des formes nouvelles d’algues rouges crétacées. Cret. Res. 1981, 2, 187–195. [Google Scholar] [CrossRef]
- Misra, P.K.; Kumar, P. Fossil algae from the Cretaceous of Varagur, Tiruchirapalli District, Tamil Nadu. Palaeobotanist 1988, 37, 36–51. [Google Scholar]
- Simone, L.; Bassi, D.; Carannante, G.; Cherchi, A. Rudist-bearing rhodalgal facies in the post-Turonian recovery of the peri-Tethyan carbonate systems: The case history from the Nurra Region (northwestern Sardinia, Italy). Geodiversitas 2012, 34, 167–187. [Google Scholar] [CrossRef]
- Halfar, J.; Mutti, M. Global dominance of coralline red-algal facies: A response to Miocene oceanographic events. Geology 2005, 33, 481–484. [Google Scholar] [CrossRef]
- Braga, J.C.; Bassi, D.; Piller, W. Palaeoenvironmental significance of Oligocene-Miocene coralline red algae—A review. In Carbonate Systems During the Oligocene-Miocene Climatic Transition; Mutti, M., Piller, W.E., Betzler, C., Eds.; IAS Spec. Publ.: Oxford, UK, 2010; pp. 165–182. [Google Scholar]
- Braga, J.C. Neogene rhodoliths in the Mediterranean basins. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 169–193. [Google Scholar]
- Aguirre, J.; Riding, R.; Braga, J.C. Diversity of coralline red algae: Origination and extinction patterns from the Early Cretaceous to the Pleistocene. Paleobiology 2000, 26, 651–667. [Google Scholar] [CrossRef]
- Norris, R.D.; Kirtland Turner, S.; Hull, P.M.; Ridgwell, A. Marine ecosystem responses to Cenozoic global change. Science 2013, 341, 492–498. [Google Scholar] [CrossRef]
- Mudelsee, M.; Bickert, T.; Lear, C.H.; Lohmann, G. Cenozoic climate changes: A review based on time series analysis of marine benthic δ18O records. Rev. Geophys. 2014, 52, 333–374. [Google Scholar] [CrossRef]
- Anagnostou, E.; John, E.; Edgar, K.; Foster, G.; Ridgwell, A.; Inglis, G.; Pancost, R.; Lunt, D.; Pearson, P. Changing atmospheric CO2 concentration was the primary driver of early Cenozoic climate. Nature 2016, 533, 380–384. [Google Scholar] [CrossRef]
- Miller, K.G.; Fairbanks, R.G.; Mountain, G.S. Tertiary oxygen isotope synthesis, sea-level history/continental margin erosion. Paleoceanography 1988, 2, 1–19. [Google Scholar] [CrossRef]
- Kennett, J.P.; Stott, L.D. Abrupt deep-sea warming, palaeoceanographic changes and benthic extinctions at the end of the Palaeocene. Nature 1990, 353, 225–229. [Google Scholar] [CrossRef]
- Zachos, J.C.; Pagani, M.; Sloan, L.; Thomas, E.; Billups, K. Trends, rhythms, and aberrations in global climate 65 Ma to present. Science 2001, 292, 686–693. [Google Scholar] [CrossRef]
- Miller, K.G.; Mountain, G.S.; Wright, J.D.; Browning, J.V. A 180-million-year record of sea level and ice volume variations from continental margin and deep-sea isotopic records. Oceanography 2011, 24, 40–53. [Google Scholar] [CrossRef]
- Scheibner, C.; Speijer, R.P. Late Paleocene-early Eocene Tethyan carbonate platform evolution—A response to long- and short-term paleoclimatic change. Earth-Sc. Rev. 2008, 90, 71–102. [Google Scholar] [CrossRef]
- Kiessling, W.; Kocsis, Á.T. Biodiversity dynamics and environmental occupancy of fossil azooxanthellate and zooxanthellate scleractinian corals. Paleobiology 2015, 41, 402–414. [Google Scholar] [CrossRef]
- Perrin, C.; Kiessling, W. Latitudinal trends in Cenozoic reef patterns and their relationship to climate. In Carbonate Systems During the Oligocene-Miocene Climatic Transition; Mutti, M., Piller, W.E., Betzler, C., Eds.; IAS Spec. Publ.: Oxford, UK, 2010; pp. 17–34. [Google Scholar]
- Nebelsick, J.H.; Rasser, M.; Bassi, D. Facies dynamics in Eocene to Oligocene circumalpine carbonates. Facies 2005, 51, 197–216. [Google Scholar] [CrossRef]
- Scotese, C.R. Atlas of Paleogene Paleogeographic Maps (Mollweide Projection), Maps 8-15, Volume 1, The Cenozoic, PALEOMAP Atlas for ArcGIS; PALEOMAP Project: Evanston, IL, USA, 2014. [Google Scholar]
- Comas, M.C.; Martínez-Gallego, J.; Molina, E. Litofacies y sucesión estratigráfica del Eoceno y Oligoceno al norte del Cerro Mencal (Zona Subbética, prov. de Granada). Cuad. Geol. 1984, 12, 145–155. [Google Scholar]
- Wade, B.S.; Pearson, P.N.; Berggren, W.A.; Pälike, H. Review and revision of Cenozoic tropical planktonic foraminiferal biostratigraphy and calibration to the geomagnetic polarity and astronomical time scale. Earth-Sc. Rev. 2011, 104, 111–142. [Google Scholar] [CrossRef]
- IGME. Mapa Geológico de España E. 1:50.000 Moratalla; Geological and Mining Institute of Spain: Madrid, Spain, 1981; p. 54. [Google Scholar]
- Serra-Kiel, J.; Hottinger, L.; Caus, E.; Drobne, K.; Ferràndez, C.; Jauhri, A.K.; Less, G.; Pavlovec, R.; Pignatti, J.; Samsó, J.M.; et al. Larger foraminiferal biostratigraphy of the Tethyan Paleocene and Eocene. Bull. Soc. Géol. Fr. 1998, 169, 281–299. [Google Scholar]
- Papazzoni, C.A.; Fornaciari, E.; Giusberti, L.; Vescogni, A.; Fornaciari, B. Integrating shallow benthic and calcareous nannofossil zones: The lower Eocene of the Monte Postale section (northern Italy). Palaios 2017, 32, 6–17. [Google Scholar] [CrossRef]
- Maurrasse, F.J.M.; Husler, G.; Georges, J.; Schmitt, G.; Damond, R.; Upraised, P. Caribbean sea-floor below acoustic reflector B” and the Southern Peninsula of Haiti. Geol. Mijn. 1979, 8, 71–83. [Google Scholar]
- Pérez-Valera, F.; Abad, M. Informe Estratigráfico y Sedimentológico. Informe interno, Proyecto SYSMIN. Consorcio IGME-BRGM-INYPSA; Dirección General de Minería: Santo Domingo, Dominican Republic, 2011; pp. 1–230. [Google Scholar]
- Cardona, A.; Montes, C.; Ayala, C.; Bustamante, C.; Hoyos, N.; Montenegro, O.; Ojeda, C.; Niño, H.; Ramírez, V.; Valencia, V.; et al. From arc-continent collision to continuous convergence, clues from Paleogene conglomerates along the southern Caribbean–South America plate boundary. Tectonophysics 2012, 580, 58–87. [Google Scholar] [CrossRef]
- Salazar-Ortiz, E.; Rincón-Martínez, D.; Páez, L.A.; Restrepo, S.M.; Barragán, S. Middle Eocene mixed carbonate-siliciclastic systems in the southern Caribbean (NW Colombian margin). J. South Am. Earth Sci. 2020. [Google Scholar] [CrossRef]
- Parra, D.; Rincón, D. Foraminiferal assemblages and paleoenvironments of the Early–Middle Eocene succession in the Sinú–San Jacinto basin, northern south America. In Proceedings of the 4th International Paleontological Congress, Mendoza, Argentina, 28 September–3 October 2014; p. 773. [Google Scholar]
- Granier, B.; Dias-Brito, D. On the fossil alga Elianella elegans PFENDER & BASSE, 1948, and its so-called lookalikes, with description of Elianella brasiliana n.sp. Revision of the Juliette PFENDER Collection. Part 1. Carnets Geol. 2016, 16, 213–229. [Google Scholar]
- Murray, J.W. Ecology and Applications of Benthic Foraminifera; Cambridge University Press: Cambridge, UK, 1996; p. 426. [Google Scholar]
- BouDagher-Fadel, M.K. Evolution and Geological Significance of Larger Benthic Foraminifera; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1–544. [Google Scholar]
- BouDagher-Fadel, M.K. Evolution and Geological Significance of Larger Benthic Foraminifera, 2nd ed.; University College London Press: London, UK, 2018; p. 639. [Google Scholar]
- Adey, W.H. Crustose coralline algae as microenvironmental indicators in the tertiary. In Historical Biogeography, Plate Tectonics and the Changing Environment; Gray, J., Boucot, A.J., Eds.; Oregon State University Press: Corvallis, OR, USA, 1979; pp. 459–464. [Google Scholar]
- Braga, J.C.; Martín, J.M. Neogene coralline-algal growth-forms and their palaeoenvironments in the Almanzora River Valley (Almeria, S.E. Spain). Palaeogeogr., Palaeoclimatol., Palaeoecol. 1988, 67, 285–303. [Google Scholar] [CrossRef]
- Bosence, D.W.J. Coralline algae: Mineralization, taxonomy, and palaeoecology. In Calcareous Algae and Stromatolites; Riding, R., Ed.; Springer-Verlag: Berlin, Germany, 1991; pp. 98–113. [Google Scholar]
- Braga, J.C.; Aguirre, J. Coralline algal assemblages in upper Neogene reef and temperate carbonates in Southern Spain. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2001, 175, 27–41. [Google Scholar] [CrossRef]
- Braga, J.C.; Vescogni, A.; Bosellini, F.; Aguirre, J. Coralline algae (Corallinales, Rhodophyta) in western and central Mediterranean Messinian reefs. Palaeogeogr., Palaeoclimatol. Palaeoecol. 2009, 275, 113–128. [Google Scholar] [CrossRef]
- Aguirre, J.; Braga, J.C.; Martín, J.M. Algal nodules in the upper Pliocene deposits at the coast of Cadiz (S Spain). In Studies on Fossil Benthic Algae; Barattolo, F., de Castro, P., Parente, M., Eds.; Boll. Soc. Paleontol. Italia: Modena, Italy, 1993; pp. 1–7. [Google Scholar]
- Aguirre, J.; Braga, J.C.; Martín, J.M.; Betzler, C. Palaeoenvironmental and stratigraphic significance of Pliocene rhodolith beds and coralline algal bioconstructions from the Carboneras Basin (SE Spain). Geodiversitas 2012, 34, 115–136. [Google Scholar] [CrossRef]
- Iryu, Y.; Nakimori, T.; Matsuda, S.; Abe, O. Distribution of marine organisms and its geological significance in the modern reef complex of the Ryukyu Islands. Sediment. Geol. 1995, 99, 243–258. [Google Scholar] [CrossRef]
- Lund, M.; Davies, P.J.; Braga, J.C. Coralline algal nodules off Fraser Island, eastern Australia. Facies 2000, 42, 25–34. [Google Scholar] [CrossRef]
- Rasser, M.W. Influence of bottom stability and sediment input on growth forms of Polystrata alba (red algae) from the Late Eocene Alpine Foreland: A new tool for the reconstruction of sedimentary environments. Palaios 2001, 16, 532–538. [Google Scholar] [CrossRef]
- Prager, E.J.; Ginsburg, R.N. Carbonate nodule growth on Florida’s outer shelf and its implications for fossil interpretations. Palaios 1989, 4, 10–317. [Google Scholar] [CrossRef]
- Rasser, M.W.; Piller, W.E. Crustose algal frameworks from the Eocene Alpine Foreland. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2004, 206, 21–39. [Google Scholar] [CrossRef]
- Bassi, D. Larger foraminiferal and coralline algal facies in an Upper Eocene storm influenced, shallow water carbonate platform (Colli Berici, north-eastern Italy). Palaeogeog. Palaeoclimatol. Palaeoecol. 2005, 226, 17–35. [Google Scholar] [CrossRef]
- Bassi, D.; Nebelsick, J.H.; Checconi, A.; Hohenegger, J.; Iryu, Y. Present-day and fossil rhodolith pavements compared: Their potential for analysing shallow-water carbonate deposits. Sediment. Geol. 2009, 214, 74–84. [Google Scholar] [CrossRef]
- Bassi, D.; Carannante, G.; Checconi, A.; Simone, L.; Vigorito, M. Sedimentological and palaeoecological integrated analysis of a Miocene canalized coralline red algal carbonate margin (Matese Mountains, Central-Southern Apennines, Italy). Sediment. Geol. 2010, 230, 105–122. [Google Scholar] [CrossRef]
- Bassi, D.; Nebelsick, J.H. Components, facies and ramps: Redefining Upper Oligocene shallow water carbonates using coralline red algae and larger foraminifera (Venetian area, northeast Italy). Palaeogeog. Palaeoclimatol. Palaeoecol. 2010, 295, 258–280. [Google Scholar] [CrossRef]
- Brandano, M.; Vannucci, G.; Pomar, L.; Obrador, A. Rhodolith assemblages from the lower Tortonian carbonate ramp of Menorca (Spain): Environmental and paleoclimatic implications. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2005, 226, 307–323. [Google Scholar] [CrossRef]
- Brandano, M.; Frezza, V.; Tomassetti, L.; Cuffaro, M. Heterozoan carbonates in oligotrophic tropical waters: The Attard member of the lower coralline limestone formation (Upper Oligocene, Malta). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2009, 274, 54–63. [Google Scholar] [CrossRef]
- Payros, A.; Pujalte, V.; Tosquella, J.; Orue-Etxebarria, X. The Eocene storm-dominated foralgal ramp of the western Pyrenees (Urbasa-Andia Formation): An analogue of future shallow-marine carbonate systems? Sediment. Geol. 2010, 228, 184–204. [Google Scholar] [CrossRef]
- Puga-Bernabéu, Á.; Braga, J.C.; Martín, J.M. High-frequency cycles in Upper-Miocene ramp-temperate carbonates (Sorbas Basin, SE Spain). Facies 2007, 53, 329–345. [Google Scholar] [CrossRef]
- Adey, W.H.; Townsend, R.; Boykins, W. The crustose coralline algae of the Hawaiian Islands. Smithson. Contrib. Mar. Sci. 1982, 15, 1–74. [Google Scholar] [CrossRef]
- Verheij, E. The genus Sporolithon (Sporolithaceae fam. nov., Corallinales, Rhodophyta) from the Spermonde Archipelago, Indonesia. Phycologia 1993, 32, 184–196. [Google Scholar] [CrossRef]
- Braga, J.C.; Bassi, D. Neogene history of Sporolithon Heydrich (Corallinales, Rhodophyta) in the Mediterranean region. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2007, 243, 189–203. [Google Scholar] [CrossRef]
- Basso, D.; Nalin, R.; Nelson, C.S. Shallow-water Sporolithon rhodoliths from north island (New Zealand). Palaios 2009, 24, 92–103. [Google Scholar] [CrossRef]
- Badve, R.M.; Kundal, P. Distichoplax Pia from Baratang Island, Andaman, India. Biovigyanum 1988, 14, 95–102. [Google Scholar]
- Elliott, G.F. Subterraniphyllum, a new Tertiary calcareous alga. Palaeontology 1957, 1, 73–75. [Google Scholar]
- Sarkar, S. The enigmatic Palaeocene-Eocene coralline Distichoplax: Approaching structural complexities, ecological affinities and extinction hypotheses. Mar. Micropaleontol. 2018, 139, 72–83. [Google Scholar] [CrossRef]
- Riosmena-Rodríguez, R. Natural history of rhodolith/mäerl beds: Their role in near-shore biodiversity and management. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 3–26. [Google Scholar]
- Takayanagi, H.; Iryu, Y.; Oda, M.; Sato, T.; Chiyonobu, S.; Nishimura, A.; Nakazawa, T.; Ishikawa, T.; Nagaishi, K. Temporal changes in biotic and abiotic composition of shallow-water carbonates on submerged seamounts in the northwestern Pacific Ocean and their controlling factors. Geodiversitas 2012, 34, 189–217. [Google Scholar] [CrossRef]
- Hallock, P. Why are larger foraminifera large? Paleobiology 1985, 11, 195–208. [Google Scholar] [CrossRef]
- Hallock, P.; Schlager, W. Nutrient excess and the demise of coral reefs and carbonate platforms. Palaios 1986, 1, 389–398. [Google Scholar] [CrossRef]
- Hallock, P.; Premoli Silva, I.; Boersma, A. Similarities between planktonic and larger foraminiferal evolutionary trends through Paleogene paleoceanographic changes. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1991, 83, 49–64. [Google Scholar] [CrossRef]
- Brassier, M.D. Fossil indicators of nutrient levels. 2: Evolution and extinction in relation to oligotrophy. In Marine Palaeoenvironmental Analysis from Fossils; Bosence, D.W.J., Allison, P.A., Eds.; The Geological Society: Oxford, UK, 1995; pp. 133–150. [Google Scholar]
- Wilson, M.E.J.; Vecsei, A. The apparent paradox of abundant foramol facies in low latitudes: Their environmental significance and effect on platform development. Earth-Sci. Rev. 2005, 69, 133–168. [Google Scholar] [CrossRef]
- Halfar, J.; Strasser, M.; Riegl, B.; Godinez-Orta, L. Oceanography, sedimentology and acoustic mapping of a bryomol carbonate factory in the northern Gulf of California, Mexico. In Cold-Water Carbonates: Depositional systems and Palaeoenvironmental Controls; Pedley, H.M., Carannante, G., Eds.; The Geological Society: London, UK, 2006; pp. 197–215. [Google Scholar]
- Aguilera, J.; Bischof, K.; Karsten, U.; Hanelt, D.; Wiencke, C. Seasonal variation in ecophysiological patterns in macroalgae from an Arctic fjord: II. Pigment accumulation and biochemical defense systems. Mar. Biol. 2002, 140, 1087–1095. [Google Scholar]
- Teichert, S.; Woelkerling, W.; Rüggeberg, A.; Wisshak, M.; Piepenburg, D.; Meyerhöfer, M.; Form, A.; Büdenbender, J.; Freiwald, A. Rhodolith beds (Corallinales, Rhodophyta) and their physical and biological environment at 80º 31’N in Nordkappbukta (Nordaustlander, Svalbard Archipelago, Norway). Phycologia 2012, 51, 371–390. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Moura, R.L.; Bastos, A.C.; Salgado, L.T.; Sumida, P.Y.; Guth, A.Z.; Francini-Filho, R.B.; Pereira-Filho, G.H.; Abrantes, D.P.; Brasileiro, P.S.; et al. Thompson, F.L. Rhodolith beds are major CaCO3 bio-factories in the tropical south west Atlantic. PLoS ONE 2012, 7, e35171. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Bahia, R.G.; Pereira-Filho, G.H.; Longo, L.L. South Atlantic rhodolith beds: Latitudinal distribution, species composition, structure and ecosystem functions, threats and conservation status. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 299–317. [Google Scholar]
- Moura, R.L.; Secchin, N.A.; Amado-Filho, G.M.; Francini-Filho, R.B.; Freitas, M.O.; Minte-Vera, C.V.; Teixeira, J.B.; Thompson, F.L.; Dutra, G.F.; Sumida, P.Y.G.; et al. Spatial patterns of benthic megahabitats and conservation planning in the Abrolhos Bank. Cont. Shelf Res. 2013, 70, 109–117. [Google Scholar] [CrossRef]
- Bahia, R.G.; Abrantes, D.P.; Brasileiro, P.S.; Pereira-Filho, G.H.; Amado-Filho, G.M. Rhodolith bed structure along a depth gradient on the northern coast of Bahia State, Brazil. Braz. J. Oceanogr. 2010, 58, 323–337. [Google Scholar] [CrossRef]
- Brasileiro, P.; Braga, J.C.; Amado-Filho, G.M.; Leal, R.N.; Bassi, D.; Franco, T.; Bastos, A.; Moura, R.L. Burial rate determines Holocene rhodolith development on the Brazilian shelf. Palaios 2018, 33, 464–477. [Google Scholar] [CrossRef]
- Pereira-Filho, G.H.; Francini-Filho, R.B.; Pierozzi, I., Jr.; Pinheiro, H.T.; Bastos, A.C.; Moura, R.L.; Moraes, F.C.; Matheus, Z.; Bahia, R.G.; Amado-Filho, G.M. Sponges and fish facilitate succession from rhodolith beds to reefs. Bull. Mar. Sci. 2014, 91, 45–46. [Google Scholar] [CrossRef]
- Ghisolfi, R.D.; Silva, M.P.; Santos, F.T.; Servino, R.N.; Thompson, F.L. Physical forcing mechanisms controlling the variability of chlorophyll-a over the Royal-Charlotte and Abrolhos Banks-Eastern Brazilian Shelf. PLoS ONE 2015, 10, e0117082. [Google Scholar] [CrossRef]
- D’Agostini, D.P.; Bastos, A.C.; Dos Reis, A.T. The modern mixed carbonate-siliciclastic Abrolhos Shelf: Implications for a mixed depositional model. J. Sediment. Res. 2015, 85, 124–139. [Google Scholar] [CrossRef]
- Moura, R.L.; Amado-Filho, G.M.; Moraes, F.C.; Brasileiro, P.S.; Salomon, P.S.; Mahiques, M.M.; Bastos, A.C.; Almeida, M.G.; Silva, J.M., Jr.; Araujo, B.F.; et al. An extensive reef system at the Amazon River mouth. Sci. Adv. 2016, 2, e1501252. [Google Scholar] [CrossRef]
- Vale, N.F.L.; Amado-Filho, G.M.; Braga, J.C.; Brasileiro, P.S.; Karez, C.S.; Moraes, F.C.; Bahia, R.G.; Bastos, A.C.; Moura, R.L. Structure and composition of rhodoliths from the Amazon River mouth, Brazil. J. South Am. Earth Sc. 2018, 84, 149–159. [Google Scholar] [CrossRef]
- Steller, D.L.; Foster, M.S.; Riosmena-Rodríguez, R. Living rhodolith bed ecosystems in the Gulf of California. In Atlas of Coastal Ecosystems in the Gulf of California: Past and Present; Johnson, J.M., Ledesma-Vázquez, J., Eds.; University of Arizona Press: Tucson, AZ, USA, 2009; pp. 72–82. [Google Scholar]
- Riosmena-Rodríguez, R.; Steller, D.L.; Hinojosa.Arango, G.; Foster, M.S. Reefs that rock and roll: Biology and conservation of rhodolith beds in the Gulf of California. In The Gulf of California Biodiversity and Conservation; Brusca, R.C., Ed.; The University of Arizona Press and The Arizona-Sonora Desert Museum: Tucson, AZ, USA, 2010; pp. 49–71. [Google Scholar]
- Halfar, J.; Eisele, M.; Riegl, B.; Hetzinger, S.; Godinez-Orta, L. Modern rhodolith-dominated carbonates at Punta Chivato, Mexico. Geodiversitas 2012, 34, 99–113. [Google Scholar] [CrossRef]
- Steller, D.L.; Foster, M.S. Environmental factors influencing distribution and morphology of rhodoliths in Bahía Concepción, B.C.S., México. J. Exp. Mar. Biol. Ecol. 1995, 194, 201–212. [Google Scholar] [CrossRef]
- Betzler, C.; Braga, J.C.; Jaramillo-Vogel, D.; Römers, M.; Hübscher, C.; Schmiedl, G.; Lindhorst, S. Late Pleistocene and Holocene cool-water carbonates of the western Mediterranean Sea. Sedimentology 2011, 58, 643–669. [Google Scholar] [CrossRef]
- Basso, D.; Babbini, L.; Ramos-Esplá, Á.A.; Salomidi, M. Mediterranean rhodolith beds. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 281–298. [Google Scholar]
- Kato, A.; Baba, M.; Matsuda, S.; Iryu, Y. Western Pacific. In Rhodolith/Maërl Beds: A Global Perspective; Riosmena-Rodríguez, R., Nelson, W., Aguirre, J., Eds.; Springer Intern. Publ.: Basel, Switzerland, 2017; pp. 335–347. [Google Scholar]
- Mutti, M.; Hallock, P. Carbonate systems along nutrient and temperature gradients: Some sedimentological and geochemical constraints. Int. J. Earth Sci. (Geol. Rundsch.) 2003, 92, 465–475. [Google Scholar] [CrossRef]
- Moore, T.C., Jr.; Backman, J.; Raffi, I.; Nigrini, C.; Sanfilippo, A.; Pälike, H.; Lyle, M. Paleogene tropical Pacific: Clues to circulation, productivity, and plate motion. Paleoceanography 2004, 19, PA3013:1–PA3013:16. [Google Scholar] [CrossRef]
- Lyle, M.; Barron, J.; Bralower, T.J.; Huber, M.; Lyle, A.O.; Ravelo, A.C.; Rea, D.K.; Wilson, P.A. Pacific Ocean and Cenozoic evolution of climate. Rev. Geophys. 2008, 46, RG2002:1–RG2002:47. [Google Scholar] [CrossRef]
- Boscolo Galazzo, F.; Thomas, E.; Pagani, M.; Warren, C.; Luciani, V.; Giusberti, L. The middle Eocene climatic optimum (MECO): A multiproxy record of paleoceanographic changes in the southeast Atlantic (ODP Site 1263, Walvis Ridge). Paleoceanography 2014, 29, 1143–1161. [Google Scholar] [CrossRef]
- Short, J.; Foster, T.; Falter, J.; Kendricj, G.A.; McCulloch, M.T. Crustose coralline algal growth, calcification and mortality following a marine heatwave in Western Australia. Cont. Shelf Res. 2015, 106, 38–44. [Google Scholar] [CrossRef]

| Species | Salinas Menores (Southern Spain) | Sierra del Zacatín (Southern Spain) | San Jacinto Fold Belt (Colombia) | Bahoruco Peninsula (Dominican Republic) |
|---|---|---|---|---|
| Sporolithales | 12.5% | 72% | 21.4% | 54.3% |
| S. liberum | X | X | X | X |
| S. cf. oulianovi | - | X | - | X |
| S. lugeonii | X | X | X | X |
| S. nummuliticum | X | X | X | X |
| Sporolithon sp. | - | X | - | - |
| Hapalidiales | 50.8% | 8.6% | 43.3% | 40% |
| Melobesia sp. | - | - | X | - |
| Lithothamnion camarasae | X | - | X | - |
| L. corallinaeforme | X | - | - | - |
| ‘Palaeothamnium’ kossovense | X | - | X | X |
| Lithothamnion sp. 1 | - | - | X | - |
| Lithothamnion sp. 2 | - | X | X | - |
| Lithothamnion sp. 3 | - | - | X | X |
| Lithothamnion sp. 4 | X | - | X | X |
| Lithothamnion sp. 5 | X | - | - | X |
| Mesophyllum sp. 1 | X | - | - | X |
| Mesophyllum sp. 2 | X | - | - | - |
| Corallinales | 19.4% | 2% | 1% | - |
| Lithoporella sp. | X | X | X | - |
| Subterraniphyllum thomasii | X | - | - | - |
| Karpathia sphaerocellulosa | X | - | - | - |
| Hydrolithon cf. lemoinei | X | - | - | - |
| Distichoplax biserialis | - | X | - | - |
| Spongites sp. 1 | X | - | - | - |
| Spongites sp. 2 | X | - | - | - |
| Spongites sp. 3 | X | - | - | - |
| Spongites sp. 4 | X | - | - | - |
| Neogoniolithon sp. 1 | X | - | - | - |
| Neogoniolithon sp. 2 | - | X | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguirre, J.; Braga, J.C.; Pujalte, V.; Orue-Etxebarria, X.; Salazar-Ortiz, E.; Rincón-Martínez, D.; Abad, M.; Pérez-Valera, F. Middle Eocene Rhodoliths from Tropical and Mid-Latitude Regions. Diversity 2020, 12, 117. https://doi.org/10.3390/d12030117
Aguirre J, Braga JC, Pujalte V, Orue-Etxebarria X, Salazar-Ortiz E, Rincón-Martínez D, Abad M, Pérez-Valera F. Middle Eocene Rhodoliths from Tropical and Mid-Latitude Regions. Diversity. 2020; 12(3):117. https://doi.org/10.3390/d12030117
Chicago/Turabian StyleAguirre, Julio, Juan C. Braga, Victoriano Pujalte, Xabier Orue-Etxebarria, Edward Salazar-Ortiz, Daniel Rincón-Martínez, Manuel Abad, and Fernando Pérez-Valera. 2020. "Middle Eocene Rhodoliths from Tropical and Mid-Latitude Regions" Diversity 12, no. 3: 117. https://doi.org/10.3390/d12030117
APA StyleAguirre, J., Braga, J. C., Pujalte, V., Orue-Etxebarria, X., Salazar-Ortiz, E., Rincón-Martínez, D., Abad, M., & Pérez-Valera, F. (2020). Middle Eocene Rhodoliths from Tropical and Mid-Latitude Regions. Diversity, 12(3), 117. https://doi.org/10.3390/d12030117






