Can Topographic Variation in Climate Buffer against Climate Change-Induced Population Declines in Northern Forest Birds?
Abstract
1. Introduction
2. Material and Methods
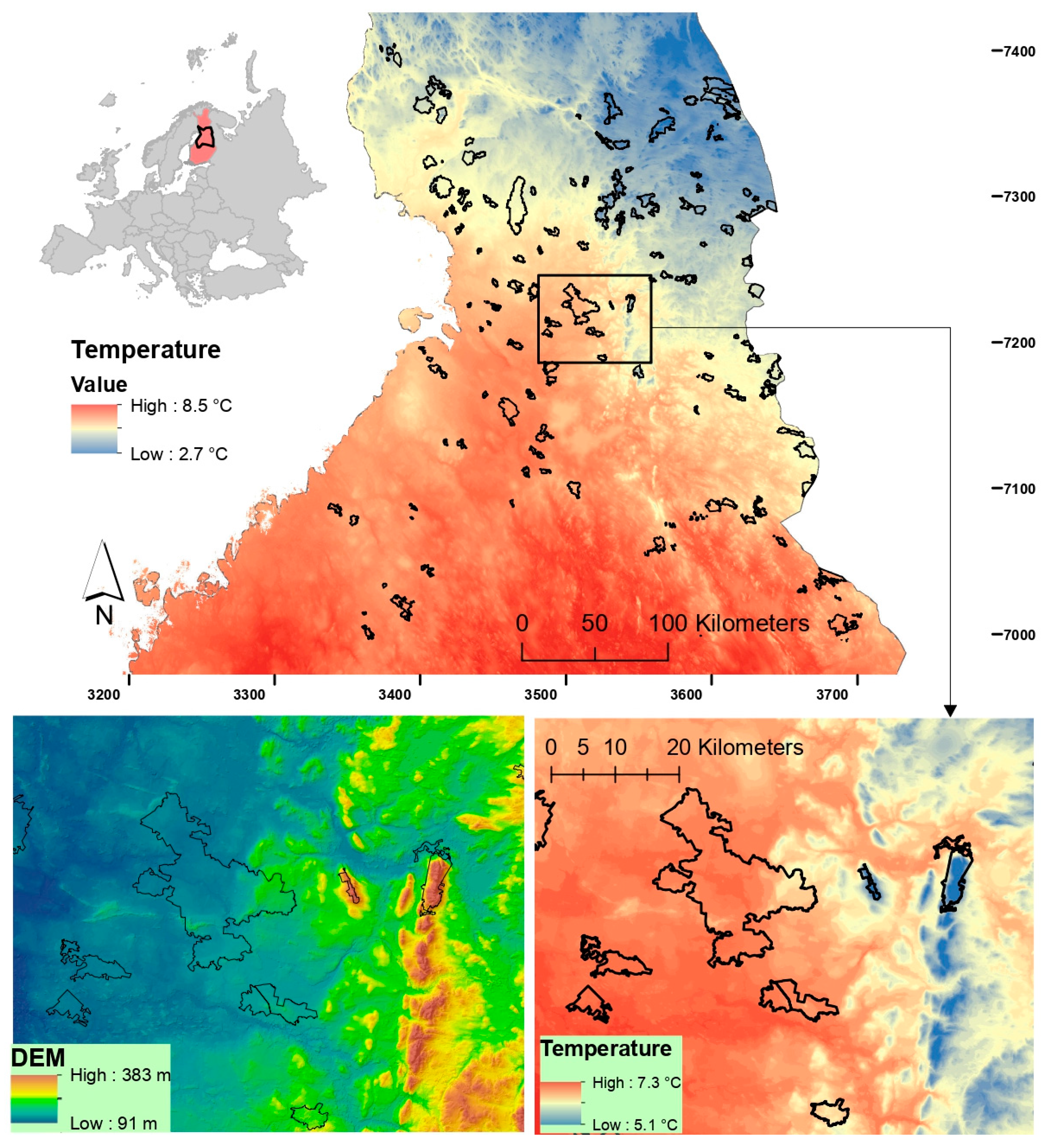
2.1. Study Areas
2.2. Bird Censuses
2.3. Climate Data
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pereira, H.M.; Leadley, P.W.; Proenca, V.; Alkemade, R.; Scharlemann, J.P.W.; Fernandez-Manjarrés, J.F.; Araújo, M.B.; Balvanera, P.; Biggs, R.; Cheung, W.W.L.; et al. Scenarios for Global Biodiversity in the 21st Century. Science 2010, 330, 1496–1501. [Google Scholar] [CrossRef]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15, 365–377. [Google Scholar] [CrossRef]
- Chen, I.-C.; Hill, J.K.; Ohleműller, R.; Roy, D.B.; Thomas, C.D. Rapid range shifts of species associated with high levels of climate warming. Science 2011, 333, 1024–1026. [Google Scholar] [CrossRef] [PubMed]
- Stephens, P.A.; Mason, L.R.; Green, R.E.; Gregory, R.D.; Sauer, J.R.; Alison, J.; Aunins, A.; Brotons, L.; Butchart, S.H.M.; Campedelli, T.; et al. Consistent response of bird populations to climate change on two continents. Science 2016, 352, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Spooner, F.E.B.; Pearson, R.G.; Freeman, R. Rapid warming is associated with population decline among terrestrial birds and mammals globally. Glob. Chang. Biol. 2018, 24, 4521–4531. [Google Scholar] [CrossRef] [PubMed]
- Bowler, D.E.; Hof, C.; Haase, P.; Kröncke, I.; Schweiger, O.; Adrian, R.; Baert, L.; Bauer, H.-G.; Blick, T.; Brooker, R.W.; et al. Cross-realm assessment of climate change impacts on species’ abundance trends. Nat. Ecol. Evol. 2017, 1, 0067. [Google Scholar] [CrossRef]
- Auer, S.K.; King, D.I. Ecological and life-history traits explain recent boundary shifts in elevation and latitude of western North American songbirds. Glob. Ecol. Biogeogr. 2014, 23, 867–875. [Google Scholar] [CrossRef]
- Tingley, M.W.; Koo, M.S.; Moritz, C.; Rush, A.C.; Beissinger, S.R. The push and pull of climate change causes heterogeneous shifts in avian elevational ranges. Glob. Chang. Biol. 2012, 18, 3279–3290. [Google Scholar] [CrossRef]
- Freeman, B.G.; Freeman, A.M.C. Rapid upslope shifts in New Guinean birds illustrate strong distributional responses of tropical montane species to global warming. Proc. Natl. Acad. Sci. USA 2014, 111, 4490–4494. [Google Scholar] [CrossRef]
- Forero-Medina, G.; Terborgh, J.; Socolar, S.J.; Pimm, S.L. Elevational Ranges of Birds on a Tropical Montane Gradient Lag behind Warming Temperatures. PLoS ONE 2011, 6, e28535. [Google Scholar] [CrossRef]
- Barbet-Massin, M.; Thuiller, W.; Jiguet, F. The fate of European breeding birds under climate, land-use and dispersal scenarios. Glob. Chang. Biol. 2012, 18, 881–890. [Google Scholar] [CrossRef]
- Corlett, R.T.; Westcott, D.A. Will plant movements keep up with climate change? Trends Ecol. Evol. 2013, 28, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Burrows, M.T.; Schoeman, D.S.; Richardson, A.J.; Molinos, J.G.; Hoffmann, A.; Buckley, L.B.; Moore, P.J.; Brown, C.J.; Bruno, J.F.; Duarte, C.M.; et al. Geographical limits to species-range shifts are suggested by climate velocity. Nature 2014, 507, 492–495. [Google Scholar] [CrossRef] [PubMed]
- Ordonez, A.; Martinuzzi, S.; Radelo, V.C.; Williams, J.W. Combined speeds of climate and land-use change of the conterminous US until 2050. Nat. Clim. Chang. 2014, 4, 811–816. [Google Scholar] [CrossRef]
- Tingley, M.W.; Darling, E.S.; Wilcove, D.S. Fine- and coarse-filter conservation strategies in a time of climate change. In The Year in Ecology and Conservation Biology; Ostfeld, R.S., Power, A.G., Eds.; The New York Academy of Sciences: New York, NY, USA, 2014; Volume 1322, pp. 92–109. [Google Scholar]
- Nadeau, C.P.; Fuller, A.K.; Rosenblatt, D.L. Climate-smart management of biodiversity. Ecosphere 2015, 6, 91. [Google Scholar] [CrossRef]
- Ackerly, D.D.; Loarie, S.R.; Cornwell, W.K.; Weiss, S.B.; Hamilton, H.; Branciforte, R.; Kraft, N.J.B. The geography of climate change: Implications for conservation biogeography. Divers. Distrib. 2010, 16, 476–487. [Google Scholar] [CrossRef]
- Dobrowski, S.Z. A climatic basis for microrefugia: The influence of terrain on climate. Glob. Chang. Biol. 2011, 17, 1022–1035. [Google Scholar] [CrossRef]
- Greiser, C.; Ehrlén, J.; Meineri, E.; Hylander, K. Hiding from the climate: Characterizing microrefugia for boreal forest understory species. Glob. Chang. Biol. 2019, in press. [Google Scholar] [CrossRef]
- Hannah, L.; Flint, L.; Syphard, A.D.; Moritz, M.A.; Buckley, L.B.; McCullough, I.M. Fine-grain modeling of species’ response to climate change: Holdouts, stepping-stones, and microrefugia. Trends Ecol. Evol. 2014, 29, 390–397. [Google Scholar] [CrossRef]
- Lawler, J.J.; Ackerly, D.D.; Albano, C.M.; Anderson, M.G.; Dobrowski, S.Z.; Gill, J.L.; Heller, N.E.; Pressey, R.L.; Sanderson, E.W.; Weiss, S.B. The theory behind, and the challenges of, conserving nature’s stage in a time of rapid change. Conserv. Biol. 2015, 29, 618–629. [Google Scholar] [CrossRef]
- Thomas, C.D.; Gillingham, P.K. The performance of protected areas for biodiversity under climate change. Biol. J. Linn. Soc. 2015, 115, 718–730. [Google Scholar] [CrossRef]
- Lenoir, J.; Hattab, T.; Pierre, G. Climatic microrefugia under anthropogenic climate change: Implications for species redistribution. Ecography 2017, 40, 253–266. [Google Scholar] [CrossRef]
- Rapacciuolo, G.; Maher, S.P.; Schneider, A.C.; Hammond, T.T.; Jabis, M.D.; Walsh, R.E.; Iknayan, K.J.; Walden, G.K.; Oldfather, M.F.; Ackerly, D.D.; et al. Beyond a warming fingerprint: Individualistic biogeographic responses to heterogeneous climate change in California. Glob. Chang. Biol. 2014, 20, 2841–2855. [Google Scholar] [CrossRef] [PubMed]
- Oliver, T.H.; Morecroft, M.D. Interactions between climate change and land use change on biodiversity: Attribution problems, risks, and opportunities. Wiley Interdiscip. Rev. Clim. 2014, 5, 317–335. [Google Scholar] [CrossRef]
- Reside, A.E.; VanDerWal, J.J.; Kutt, A.S.; Perkins, G.C. Weather, Not Climate, Defines Distributions of Vagile Bird Species. PLoS ONE 2010, 5, e13569. [Google Scholar] [CrossRef] [PubMed]
- Suggitt, A.J.; Wilson, R.J.; Isaac, N.J.B.; Beale, C.M.; Auffret, A.G.; August, T.; Bennie, J.J.; Crick, H.Q.P.; Duffield, S.; Fox, R.; et al. Extinction risk from climate change is reduced by microclimatic buffering. Nat. Clim. Chang. 2018, 8, 713–717. [Google Scholar] [CrossRef]
- Newbold, T.; Hudson, L.N.; Hill, S.L.L.; Contu, S.; Lysenko, I.; Senior, R.A.; Borger, L.; Bennett, D.J.; Choimes, A.; Collen, B.; et al. Global effects of land use on local terrestrial biodiversity. Nature 2015, 520, 45–50. [Google Scholar] [CrossRef]
- Bowler, D.E.; Haase, P.; Kröncke, I.; Tackenberg, O.; Bauer, H.G.; Brendel, C.; Brooker, R.W.; Gerisch, M.; Henle, K.; Hickler, T.; et al. A cross-taxon analysis of the impact of climate change on abundance trends in central Europe. Biol. Conserv. 2015, 187, 41–50. [Google Scholar] [CrossRef]
- Maclean, I.M.D.; Hopkins, J.J.; Bennie, J.; Lawson, C.R.; Wilson, R.J. Microclimates buffer the responses of plant communities to climate change. Glob. Ecol. Biogeogr. 2015, 24, 1340–1350. [Google Scholar] [CrossRef]
- Freckleton, R.P.; Watkinson, A.R. Large-scale spatial dynamics of plants: Metapopulations, regional ensembles and patchy populations. J. Ecol. 2002, 90, 419–434. [Google Scholar] [CrossRef]
- Lindström, Å.; Green, M.; Paulson, G.; Smith, H.G.; Devictor, V. Rapid changes in bird community composition at multiple temporal and spatial scales in response to recent climate change. Ecography 2013, 36, 313–322. [Google Scholar] [CrossRef]
- Renwick, A.R.; Massimino, D.; Newson, S.E.; Chamberlain, D.E.; Pearce-Higgins, J.W.; Johnston, A. Modelling changes in species’ abundance in response to projected climate change. Divers. Distrib. 2012, 18, 121–132. [Google Scholar] [CrossRef]
- Howard, C.; Stephens, P.A.; Pearce-Higgins, J.W.; Gregory, R.D.; Willis, S.G. The drivers of avian abundance: Patterns in the relative importance of climate and land use. Glob. Ecol. Biogeogr. 2015, 24, 1249–1260. [Google Scholar] [CrossRef]
- Virkkala, R.; Pöyry, J.; Heikkinen, R.K.; Lehikoinen, A.; Valkama, J. Protected areas alleviate climate change effects on northern bird species of conservation concern. Ecol. Evol. 2014, 4, 2991–3003. [Google Scholar] [CrossRef] [PubMed]
- Gaüzère, P.; Jiguet, F.; Devictor, V. Can protected areas mitigate the impacts of climate change on bird’s species and communities? Divers. Distrib. 2016, 22, 625–637. [Google Scholar] [CrossRef]
- Santangeli, A.; Rajasärkkä, A.; Lehikoinen, A. Effects of high latitude protected areas on bird communities under rapid climate change. Glob. Chang. Biol. 2017, 23, 2241–2249. [Google Scholar] [CrossRef]
- Lehikoinen, P.; Santangeli, A.; Jaatinen, K.; Rajasärkkä, A.; Lehikoinen, A. Protected areas act as a buffer against detrimental effects of climate change-Evidence from large-scale, long-term abundance data. Glob. Chang. Biol. 2019, 25, 304–313. [Google Scholar] [CrossRef]
- Virkkala, R.; Rajasärkkä, A. Northward density shift of bird species in boreal protected areas due to climate change. Boreal Environ. Res. 2011, 16, 2–13. [Google Scholar]
- Virkkala, R.; Rajasärkkä, A.; Heikkinen, R.K.; Kuusela, S.; Leikola, N.; Pöyry, J. Birds in boreal protected areas shift northwards in the warming climate but show different rates of population decline. Biol. Conserv. 2018, 226, 271–279. [Google Scholar] [CrossRef]
- Loarie, S.R.; Duffy, P.B.; Hamilton, H.; Asner, G.P.; Field, C.B.; Ackerly, D.D. The velocity of climate change. Nature 2009, 462, 1052–1055. [Google Scholar] [CrossRef]
- Garcia, R.A.; Cabeza, M.; Rahbek, C.; Araújo, M.B. Multiple Dimensions of Climate Change and Their Implications for Biodiversity. Science 2014, 344, 1247579. [Google Scholar] [CrossRef] [PubMed]
- Boulinier, T.; Nichols, J.D.; Hines, J.E.; Sauer, J.R.; Flather, C.H.; Pollock, K.H. Higher temporal variability of forest breeding bird communities in fragmented landscapes. Proc. Natl. Acad. Sci. USA 1998, 95, 7497–7501. [Google Scholar] [CrossRef] [PubMed]
- Virkkala, R.; Rajasärkkä, A. Spatial variation of bird species in landscapes dominated by old-growth forests in northern boreal Finland. Biodivers. Conserv. 2006, 15, 2143–2162. [Google Scholar] [CrossRef]
- Väisänen, R.A.; Lammi, E.; Koskimies, P. Distribution, Numbers and Population Changes of Finnish Breeding Birds; Otava: Helsinki, Finland, 1998; p. 567, (In Finnish with an English Summary). [Google Scholar]
- Virkkala, R.; Korhonen, K.T.; Haapanen, R.; Aapala, K. Protected forests and mires in forest and mire vegetation zones in Finland based on the 8th National Forest Inventory. Finnish Environ. 2000, 395, 1–49, (In Finnish with an English Summary). [Google Scholar]
- Virkkala, R.; Lehikoinen, A. Patterns of climate-induced density shifts of species: Poleward shifts faster in northern boreal birds than in southern birds. Glob. Chang. Biol. 2014, 20, 2995–3003. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, O.; Väisänen, R.A. Correction coefficients for line transect censuses of breeding birds. Ornis Fenn. 1983, 60, 97–104. [Google Scholar]
- Väisänen, R.A. Changes in the distribution of species between two bird atlas surveys: The analysis of covariance after controlling for research activity. Ornis Fenn. 1998, 75, 53–67. [Google Scholar]
- Virkkala, R.; Lehikoinen, A. Birds on the move in the face of climate change: High species turnover in northern Europe. Ecol. Evol. 2017, 7, 8201–8209. [Google Scholar] [CrossRef]
- Aalto, J.; Pirinen, P.; Jylhä, K. New gridded daily climatology of Finland: Permutation-based uncertainty estimates and temporal trends in climate. JGR-A 2016, 121, 3807–3823. [Google Scholar] [CrossRef]
- Klok, E.J.; Tank, A. Updated and extended European dataset of daily climate observations. Int. J. Clim. 2009, 29, 1182–1191. [Google Scholar] [CrossRef]
- Wood, S.N. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J. R. Stat. Soc. Ser. 2011, 73, 3–36. [Google Scholar] [CrossRef]
- Aalto, J.; Riihimäki, H.; Meineri, E.; Hylander, K.; Luoto, M. Revealing topoclimatic heterogeneity using meteorological station data. Int. J. Clim. 2017, 37, 544–556. [Google Scholar] [CrossRef]
- Aalto, J.; le Roux, P.C.; Luoto, M. The meso-scale drivers of temperature extremes in high-latitude Fennoscandia. Clim. Dyn. 2014, 42, 237–252. [Google Scholar] [CrossRef]
- De Frenne, P.; Verheyen, K. Weather stations lack forest data. Science 2016, 351, 234. [Google Scholar] [CrossRef] [PubMed]
- Brooks, M.E.; Kristensen, K.; van Benthem, K.J.; Magnusson, A.; Berg, C.W.; Nielsen, A.; Skaug, H.J.; Machler, M.; Bolker, B.M. glmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling. R J. 2017, 9, 378–400. [Google Scholar] [CrossRef]
- R Development Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Gaüzère, P.; Prince, K.; Devictor, V. Where do they go? The effects of topography and habitat diversity on reducing climatic debt in birds. Glob. Chang. Biol. 2017, 23, 2218–2229. [Google Scholar] [CrossRef]
- Brommer, J.E. The range margins of northern birds shift polewards. Ann. Zool. Fenn. 2004, 41, 391–397. [Google Scholar]
- Virkkala, R.; Rajasärkkä, A. Climate change affects populations of northern birds in boreal protected areas. Biol. Lett. 2011, 7, 395–398. [Google Scholar] [CrossRef]
- Ruosteenoja, K.; Jylhä, K.; Kämäräinen, M. Climate projections for Finland under the RCP forcing scenarios. Geophysica 2016, 51, 17–50. [Google Scholar]
- Howard, C.; Stephens, P.A.; Pearce-Higgins, J.W.; Gregory, R.D.; Willis, S.G. Improving species distribution models: The value of data on abundance. Methods Ecol. 2014, 5, 506–513. [Google Scholar] [CrossRef]
- Crick, H.Q.P. The impact of climate change on birds. Ibis 2004, 146, 48–56. [Google Scholar] [CrossRef]
- Wegge, P.; Rolstad, J. Climate change and bird reproduction: Warmer springs benefit breeding success in boreal forest grouse. Proc. R. Soc. B Biol. Sci. 2017, 284, 20171528. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, G.X.; Alatalo, R.V.; Helle, P.; Lindén, H.; Lindström, J.; Siitari, H. Short- and long-term population dynamical consequences of asymmetric climate change in black grouse. Proc. R. Soc. B Biol. Sci 2006, 273, 2009–2016. [Google Scholar] [CrossRef] [PubMed]
- Seppälä, M. The Physical Geography of Fennoscandia; Oxford University Press: Oxford, UK, 2005; p. 432. [Google Scholar]
| Species | Density | Ranks (pos./neg./tied) | z | P | |
|---|---|---|---|---|---|
| 1981–1999 | 2000–2017 | ||||
| Rough-legged buzzard Buteo lagopus | 0.01 ± 0.01 | 0.01 ± 0.00 | 1/6/122 | −1.859 | 0.063 |
| Golden eagle Aquila chrysaetos | 0.00 ± 0.00 | 0.00 ± 0.00 | 10/6/113 | 1.189 | 0.235 |
| Merlin Falco columbarius | 0.04 ± 0.02 | 0.04 ± 0.02 | 10/14/105 | −0.943 | 0.346 |
| Northern hawk owl Surnia ulula | 0.04 ± 0.02 | 0.28 ± 0.22 | 14/8/107 | 1.185 | 0.236 |
| Great grey owl Strix nebulosa | 0.01 ± 0.01 | 0.01 ± 0.01 | 2/6/121 | −1.120 | 0.263 |
| Three-toed woodpecker Picoides tridactylus | 0.31 ± 0.05 | 0.65 ± 0.09 | 59/21/49 | 4.240 | <0.001 |
| Bohemian waxwing Bombycilla garrulus | 0.21 ± 0.05 | 0.79 ± 0.09 | 82/7/40 | 6.880 | <0.001 |
| Red-flanked bluetail Tarsiger cyanurus | 0.01 ± 0.00 | 0.14 ± 0.04 | 28/6/95 | 4.129 | <0.001 |
| Arctic warbler Phylloscopus borealis | 0.01 ± 0.00 | 0.00 ± 0.00 | 3/7/119 | −1.988 | 0.047 |
| Siberian tit Poecile cinctus | 0.14 ± 0.04 | 0.07 ± 0.03 | 8/17/104 | −1.682 | 0.093 |
| Great grey shrike Lanius excubitor | 0.21 ± 0.09 | 0.07 ± 0.02 | 17/18/94 | −0.541 | 0.589 |
| Siberian jay Perisoreus infaustus | 0.47 ± 0.07 | 0.43 ± 0.07 | 29/36/64 | −0.709 | 0.478 |
| Brambling Fringilla montifringilla | 16.73 ± 0.97 | 9.95 ± 0.83 | 23/106/0 | −7.229 | <0.001 |
| Common redpoll Carduelis flammea | 3.03 ± 0.33 | 1.42 ± 0.23 | 19/100/10 | −7.231 | <0.001 |
| Two-barred crossbill Loxia leucoptera | 0.56 ± 0.14 | 0.03 ± 0.01 | 10/46/73 | −4.968 | <0.001 |
| Pine grosbeak Pinicola enucleator | 0.10 ± 0.04 | 0.06 ± 0.02 | 7/9/113 | −0.310 | 0.756 |
| Rustic bunting Emberiza rustica | 3.43 ± 0.30 | 1.84 ± 0.28 | 32/82/15 | −5.365 | <0.001 |
| Combined | 25.29 ± 1.39 | 15.76 ± 1.23 | 20/109/0 | −7.154 | <0.001 |
| Term | Estimate | S.E. | z | P |
|---|---|---|---|---|
| Intercept | 0.221 | 0.532 | 0.416 | 0.677 |
| −0.579 | 0.041 | −14.232 | <0.001 | |
| 5.186 | 2.554 | 2.030 | 0.042 | |
| Forest proportion | 1.485 | 0.201 | 7.382 | <0.001 |
| Forest proportion × | −6.816 | 2.790 | −2.443 | 0.0146 |
| PA Size | 0.013 | 0.030 | 0.417 | 0.677 |
| Time | 0.195 | 0.577 | 0.337 | 0.736 |
| Term | Estimate | S.E. | z | P |
|---|---|---|---|---|
| Intercept | −1.057 | 0.348 | −3.041 | 0.002 |
| 0.191 | 0.301 | 0.634 | 0.526 | |
| −3.426 | 0.465 | −7.375 | <0.001 | |
| PA size | 0.078 | 0.035 | 2.213 | 0.027 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Virkkala, R.; Aalto, J.; Heikkinen, R.K.; Rajasärkkä, A.; Kuusela, S.; Leikola, N.; Luoto, M. Can Topographic Variation in Climate Buffer against Climate Change-Induced Population Declines in Northern Forest Birds? Diversity 2020, 12, 56. https://doi.org/10.3390/d12020056
Virkkala R, Aalto J, Heikkinen RK, Rajasärkkä A, Kuusela S, Leikola N, Luoto M. Can Topographic Variation in Climate Buffer against Climate Change-Induced Population Declines in Northern Forest Birds? Diversity. 2020; 12(2):56. https://doi.org/10.3390/d12020056
Chicago/Turabian StyleVirkkala, Raimo, Juha Aalto, Risto K. Heikkinen, Ari Rajasärkkä, Saija Kuusela, Niko Leikola, and Miska Luoto. 2020. "Can Topographic Variation in Climate Buffer against Climate Change-Induced Population Declines in Northern Forest Birds?" Diversity 12, no. 2: 56. https://doi.org/10.3390/d12020056
APA StyleVirkkala, R., Aalto, J., Heikkinen, R. K., Rajasärkkä, A., Kuusela, S., Leikola, N., & Luoto, M. (2020). Can Topographic Variation in Climate Buffer against Climate Change-Induced Population Declines in Northern Forest Birds? Diversity, 12(2), 56. https://doi.org/10.3390/d12020056





