More Than a Functional Group: Diversity within the Legume–Rhizobia Mutualism and Its Relationship with Ecosystem Function
Abstract
:1. Introduction
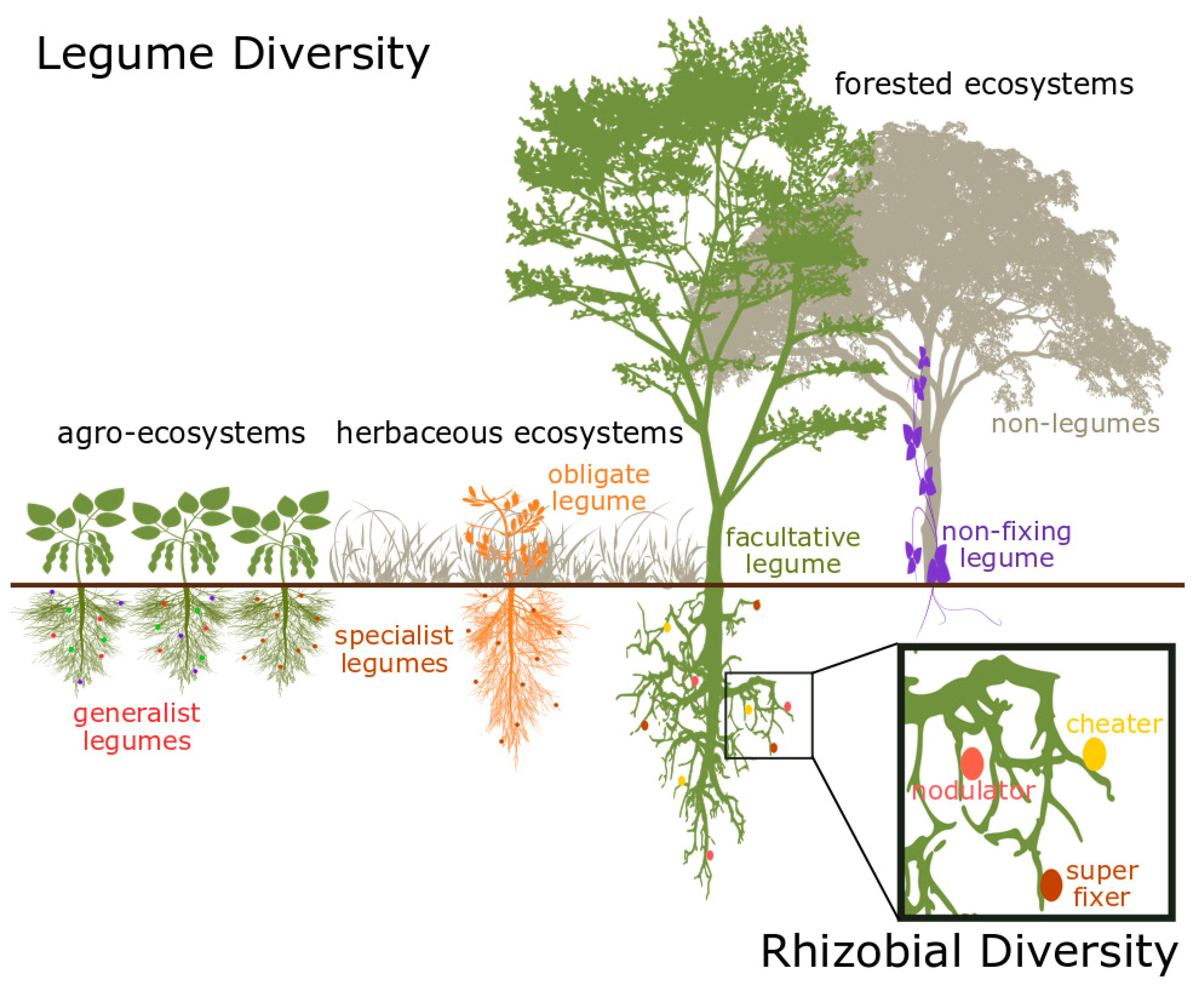
2. Diversity in the Legume–Rhizobia Mutualism
2.1. Species Diversity
2.2. Functional Diversity
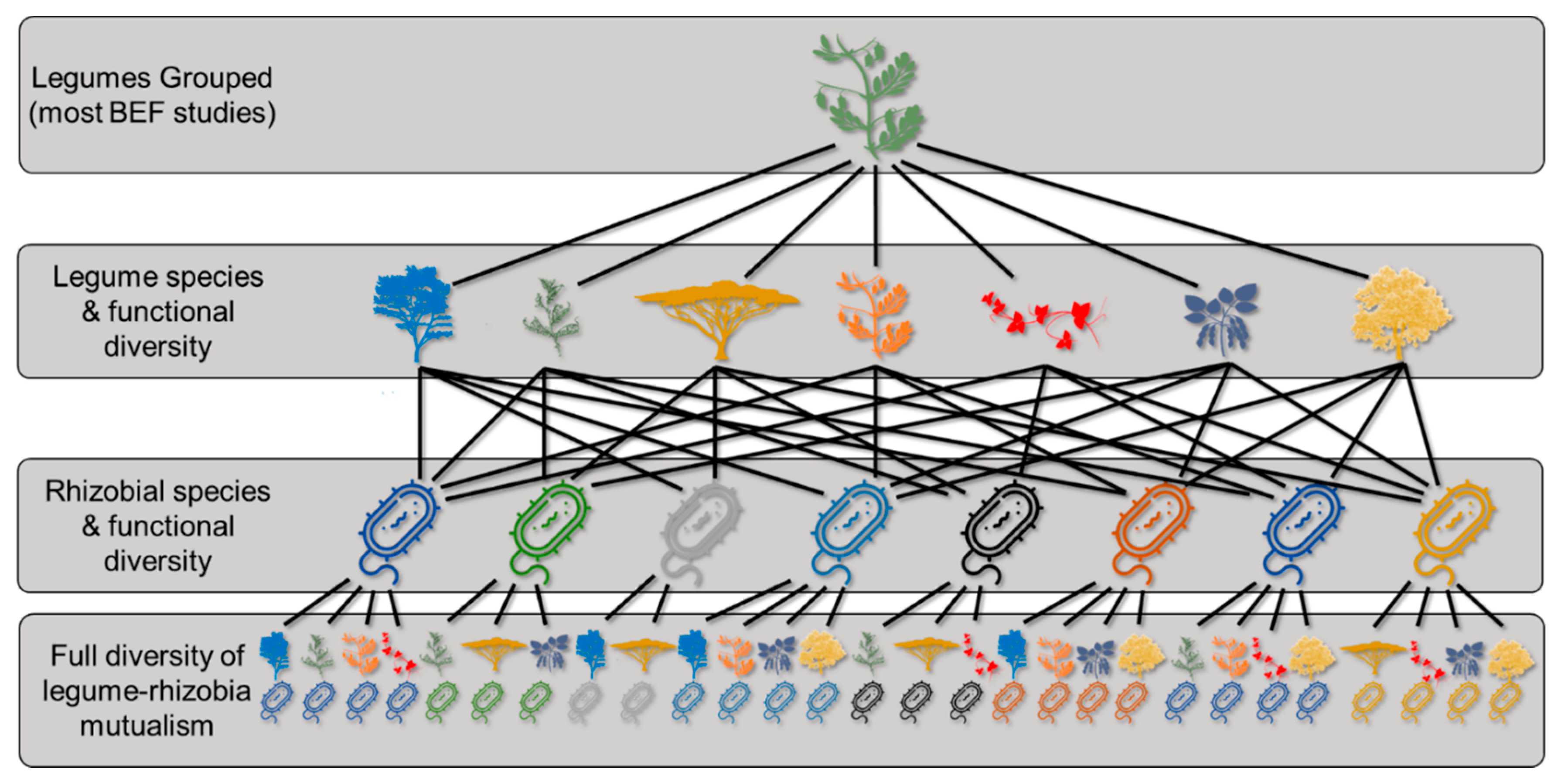
3. Diversity’s Influence on Ecosystem Function
3.1. Individual Legume Function
3.2. Community-Level Ecosystem Function
3.3. Variation in Function Across Space and Time
4. Function’s Influence on Diversity
5. Next Steps for the Legume–Rhizobia Mutualism in BEF Research
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cardinale, B.J.; Srivastava, D.S.; Duffy, J.E.; Wright, J.P.; Downing, A.L.; Sankaran, M.; Jouseau, C. Effects of biodiversity on the functioning of trophic groups and ecosystems. Nature 2006, 443, 989–992. [Google Scholar] [CrossRef]
- Hooper, D.; Chapin, F., III; Ewel, J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.; Lodge, D.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A concensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Cardinale, B.J.; Wright, J.P.; Cadotte, M.W.; Carroll, I.T.; Hector, A.; Srivastava, D.S.; Loreau, M.; Weis, J.J. Impacts of plant diversity on biomass production increase through time because of species complementarity. Proc. Natl. Acad. Sci. USA 2007, 104, 18123–18128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turnbull, L.A.; Levine, J.M.; Loreau, M.; Hector, A. Coexistence, niches and biodiversity effects on ecosystem functioning. Ecol. Lett. 2013, 16, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Carroll, I.T.; Cardinale, B.J.; Nisbet, R.M. Niche and fitness differences relate the maintenance of diversity to ecosystem function. Ecology 2011, 92, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Flynn, D.F.B.; Prager, C.M.; Hart, G.M.; Devan, C.M.; Ahrestani, F.S.; Palmer, M.I.; Bunker, D.E.; Knops, J.M.H.; Jouseau, C.F.; et al. The importance of rare species: A trait-based assessment of rare species contributions to functional diversity and possible ecosystem function in tall-grass prairies. Ecol. Evol. 2014, 4, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Loreau, M.; Hector, A. Partitioning selection and complementarity in biodiversity experiments. Nature 2001, 412, 72–76. [Google Scholar] [CrossRef]
- Weigelt, A.; Schumacher, J.; Roscher, C.; Schmid, B. Does biodiversity increase spatial stability in plant community biomass? Ecol. Lett. 2008, 11, 338–347. [Google Scholar] [CrossRef] [Green Version]
- Isbell, F.I.; Polley, H.W.; Wilsey, B.J. Biodiversity, productivity and the temporal stability of productivity: Patterns and processes. Ecol. Lett. 2009, 12, 443–451. [Google Scholar] [CrossRef] [Green Version]
- de Mazancourt, C.; Isbell, F.; Larocque, A.; Berendse, F.; De Luca, E.; Grace, J.B.; Haegeman, B.; Polley, H.W.; Roscher, C.; Schmid, B.; et al. Predicting ecosystem stability from community composition and biodiversity. Ecol. Lett. 2013, 16, 617–625. [Google Scholar] [CrossRef] [Green Version]
- Wilcox, K.R.; Tredennick, A.T.; Koerner, S.E.; Grman, E.; Hallett, L.M.; Avolio, M.L.; La Pierre, K.J.; Houseman, G.R.; Isbell, F.; Johnson, D.S.; et al. Asynchrony among local communities stabilises ecosystem function of metacommunities. Ecol. Lett. 2017, 20, 1534–1545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillebrand, H.; Matthiessen, B. Biodiversity in a complex world: consolidation and progress in function biodiversity research. Ecol. Lett. 2009, 12, 1405–1419. [Google Scholar] [CrossRef] [PubMed]
- Lefcheck, J.S.; Byrnes, J.E.K.; Isbell, F.; Gamfeldt, L.; Griffin, J.N.; Eisenhauer, N.; Hensel, M.J.S.; Hector, A.; Cardinale, B.J.; Duffy, J.E. Biodiversity enhances ecosystem multifunctionality across trophic levels and habitats. Nat. Commun. 2015, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Petchey, O.L.; Gaston, K.J. Functional diversity: Back to basics and looking forward. Ecol. Lett. 2006, 9, 741–758. [Google Scholar] [CrossRef]
- Roscher, C.; Schumacher, J.; Gubsch, M.; Lipowsky, A.; Weigelt, A.; Buchmann, N.; Schmid, B.; Schulze, E.D. Using plant functional traits to explain diversity-productivity relationships. PLoS ONE 2012, 7, e36760. [Google Scholar] [CrossRef]
- Sprent, J.I. The effects of water stress on nitrogen-fixing root nodules. New Phytol. 1972, 71, 443–450. [Google Scholar] [CrossRef]
- Mylona, P.; Pawlowski, K.; Bisseling, T. Symbiotic Nitrogen Fixation. Plant Cell 1995, 7, 869–885. [Google Scholar] [CrossRef]
- Vitousek, P.; Cassman, K.; Cleveland, C. Towards an ecological understanding of biological nitrogen fixation. Biogeochemistry 2002, 57, 1–45. [Google Scholar] [CrossRef]
- Binkley, D.; Giardina, C. Nitrogen fixation in tropical forest plantations. ACIAR Monogr. Ser. 1997, 43, 297–337. [Google Scholar]
- Schlesinger, W.; Bernhardt, E.S. Biogeochemistry: An Analysis Of Global Change, 3rd ed.; Elsevier: Oxford, UK, 2013. [Google Scholar]
- Peoples, M.B.; Brockwell, J.; Herridge, D.F.; Rochester, I.J.; Alves, B.J.R.; Urquiaga, S.; Boddey, R.M.; Dakora, F.D.; Bhattarai, S.; Maskey, S.L.; et al. The contributions of nitrogen-fixing crop legumes to the productivity of agricultural systems. Symbiosis 2009, 48, 1–17. [Google Scholar] [CrossRef]
- Tilman, D.; Knops, J.; Wedin, D.; Reich, P.; Ritchie, M.; Siemann, E. The influence of functional diversity and composition on ecosystem processes. Science (80-) 1997, 277, 1300–1302. [Google Scholar] [CrossRef] [Green Version]
- Vitousek, P.; Howarth, R. Nitrogen limitation on land and in the sea: how can it occur? Biogeochemistry 1991, 13, 87–115. [Google Scholar] [CrossRef]
- Temperton, V.M.; Mwangi, P.N.; Scherer-Lorenzen, M.; Schmid, B.; Buchmann, N. Positive interactions between nitrogen-fixing legumes and four different neighbouring species in a biodiversity experiment. Oecologia 2007, 151, 190–205. [Google Scholar] [CrossRef] [PubMed]
- Menge, D.N.L.; Chazdon, R.L. Higher survival drives the success of nitrogen-fixing trees through succession in Costa Rican rainforests. New Phytol. 2016, 209, 965–977. [Google Scholar] [CrossRef] [Green Version]
- Adams, M.A.; Turnbull, T.L.; Sprent, J.I.; Buchmann, N. Legumes are different: leaf nitrogen, photosynthesis, and water use efficiency. Proc. Natl. Acad. Sci. USA 2016, 113, 4098–4113. [Google Scholar] [CrossRef] [Green Version]
- Gei, M.; Rozendaal, D.M.A.; Poorter, L.; Bongers, F.; Sprent, J.I.; Garner, M.D.; Aide, T.M.; Andrade, J.L.; Balvanera, P.; Becknell, J.M.; et al. Legume abundance along successional and rainfall gradients in Neotropical forests. Nat. Ecol. Evol. 2018, 2, 1104–1111. [Google Scholar] [CrossRef]
- Pellegrini, A.F.A.; Staver, A.C.; Hedin, L.O.; Charles-Dominique, T.; Tourgee, A. Aridity, not fire, favors nitrogen-fixing plants across tropical savanna and forest biomes. Ecology 2016, 97, 2177–2183. [Google Scholar] [CrossRef]
- Batterman, S.A.; Hedin, L.O.; van Breugel, M.; Ransijn, J.; Craven, D.J.; Hall, J.S. Key role of symbiotic dinitrogen fixation in tropical forest secondary succession. Nature 2013, 502, 224–227. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Walker, L.R. Colonization, succession, and resource availability: Ecosystem-level interactions. In Colonization, Succession, and Stability; Gray, A., Crawley, M., Edwards, P., Eds.; Blackwell Scientific: Oxford, UK, 1987; pp. 207–223. [Google Scholar]
- Sprent, J.I.; Sutherland, J.; de Faria, S. Some aspects of the biology of nitrogen-fixing organisms. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1987, 317, 111–129. [Google Scholar]
- Oono, R.; Schmitt, I.; Sprent, J.I.; Denison, R.F. Multiple evolutionary origins of legume traits leading to extreme rhizobial differentiation. New Phytol. 2010, 187, 508–520. [Google Scholar] [CrossRef]
- Werner, G.D.A.; Cornwell, W.K.; Sprent, J.I.; Kattge, J.; Kiers, E.T. A single evolutionary innovation drives the deep evolution of symbiotic N2-fixation in angiosperms. Nat. Commun. 2014, 5, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tilman, D. Constraints and tradeoffs: toward a predictive theory of competition and succession. Oikos 1990, 58, 3–15. [Google Scholar] [CrossRef] [Green Version]
- Flynn, D.F.B.; Mirotchnick, N.; Jain, M.; Palmer, M.I.; Naeem, S. Functional and phylogenetic diversity as predictors of biodiversity- Ecosystem-function relationships. Ecology 2011, 92, 1573–1581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spehn, E.M.; Scherer-Lorenzen, M.; Schmid, B.; Hector, A.; Caldeira, M.C.; Dimitrakopoulos, P.G.; Finn, J.A.; Jumpponen, A.; O’Donnovan, G.; Pereira, J.S.; et al. The role of legumes as a component of biodiversity in a cross-European study of grassland biomass nitrogen. Oikos 2002, 98, 205–218. [Google Scholar] [CrossRef] [Green Version]
- Eisenhauer, N. Aboveground-belowground interactions as a source of complementarity effects in biodiversity experiments. Plant Soil 2012, 351, 1–22. [Google Scholar] [CrossRef]
- Huss-Danell, K. Actinorhizal symbioses and their N2 fixation. New Phytol. 1997, 136, 375–405. [Google Scholar] [CrossRef]
- Santi, C.; Bogusz, D.; Franche, C. Biological nitrogen fixation in non-legume plants. Ann. Bot. 2013, 111, 743–767. [Google Scholar] [CrossRef] [Green Version]
- de Lajudie, P.M.; Andrews, M.; Ardley, J.; Eardly, B.; Jumas-Bilak, E.; Kuzmanović, N.; Lassalle, F.; Lindström, K.; Mhamdi, R.; Martínez-Romero, E.; et al. Minimal standards for the description of new genera and species of rhizobia and agrobacteria. Int. J. Syst. Evol. Microbiol. 2019, 69, 1852–1863. [Google Scholar] [CrossRef]
- Doyle, J.J.; Luckow, M.A. The rest of the iceberg. Legume diversity and evolution in a phylogenetic context. Plant Physiol. 2003, 131, 900–910. [Google Scholar] [CrossRef] [Green Version]
- Azani, N.; Babineau, M.; Bailey, C.D.; Banks, H.; Barbosa, A.R.; Pinto, R.B.; Boatwright, J.S.; Borges, L.M.; Brown, G.K.; Bruneau, A.; et al. A new subfamily classification of the leguminosae based on a taxonomically comprehensive phylogeny. Taxon 2017, 66, 44–77. [Google Scholar] [CrossRef] [Green Version]
- Sprent, J.; Ardley, J.; James, E.K. Biogeography of nodulated legumes and their nitrogen fixing symbionts. New Phytol. 2017, 215, 40–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siefert, A.; Zillig, K.W.; Friesen, M.L.; Strauss, S.Y. Mutualists stabilize the coexistence of congeneric legumes. Am. Nat. 2019, 193, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Van Der Heijden, M.G.A.; Bakker, R.; Verwaal, J.; Scheublin, T.R.; Rutten, M.; Van Logtestijn, R.; Staehelin, C. Symbiotic bacteria as a determinant of plant community structure and plant productivity in dune grassland. FEMS Microbiol. Ecol. 2006, 56, 178–187. [Google Scholar] [CrossRef] [PubMed]
- De Queiroz, K. Species concepts and species delimitation. Syst. Biol. 2007, 56, 879–886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sprent, J.I. Evolving ideas of legume evolution and diversity: A taxonomic perspective on the occurrence of nodulation. New Phytol. 2007, 174, 11–25. [Google Scholar] [CrossRef]
- Bailly, X.; Giuntini, E.; Sexton, M.C.; Lower, R.P.J.; Harrison, P.W.; Kumar, N.; Young, J.P.W. Population genomics of Sinorhizobium medicae based on low-coverage sequencing of sympatric isolates. ISME J. 2011, 5, 1722–1734. [Google Scholar] [CrossRef] [Green Version]
- González, V.; Santamaría, R.I.; Bustos, P.; Pérez-Carrascal, O.M.; Vinuesa, P.; Juárez, S.; Martínez-Flores, I.; Cevallos, M.Á.; Brom, S.; Martínez-Romero, E.; et al. Phylogenomic Rhizobium species are structured by a continuum of diversity and genomic clusters. Front. Microbiol. 2019, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Lad, G.; Giuntini, E.; Kaye, M.E.; Udomwong, P.; Jannah Shamsani, N.; Peter W Young, J.; Bailly, X. Bacterial genospecies that are not ecologically coherent: Population genomics of rhizobium leguminosarum. Open Biol. 2015, 5, 140133. [Google Scholar] [CrossRef] [Green Version]
- Wielbo, J. Rhizobial communities in symbiosis with legumes: Genetic diversity, competition and interactions with host plants. Cent. Eur. J. Biol. 2012, 7, 363–372. [Google Scholar] [CrossRef]
- Willems, A. The taxonomy of rhizobia: An overview. Plant Soil 2006, 287, 3–14. [Google Scholar] [CrossRef]
- Aoki, S.; Ito, M.; Iwasaki, W. From β- To α-proteobacteria: The origin and evolution of rhizobial nodulation genes nodij. Mol. Biol. Evol. 2013, 30, 2494–2508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrison, S.P.; Jones, D.G.; Young, J.P.W. Rhizobium population genetics: Genetic variation within and between populations from diverse locations. Microbiology 1989, 135, 1061–1069. [Google Scholar] [CrossRef] [Green Version]
- Dresler-Nurmi, A.; Fewer, D.; Rasanen, L.; Lindstrom, K. The diversity and evolution of rhizobia. In Prokaryotic Symbionts in Plants; Pawlowski, K., Ed.; Springer-Verlag: Berlin/Heidelberg, Germany, 2009; pp. 3–41. [Google Scholar]
- Young, J.; Haukka, K. Diversity and phylogeny of rhizobia. New Phytol. 1996, 133, 87–94. [Google Scholar]
- Siler, E.; Friesen, M.L. Widespread negative frequency-dependent selection maintains diversity in the legume-rhizobia symbiosis: Balancing nodulation may explain the paradox of rhizobium diversity. bioRxiv 2017. [Google Scholar] [CrossRef]
- La Pierre, K.J.; Simms, E.L.; Tariq, M.; Zafar, M.; Porter, S.S. Invasive legumes can associate with many mutualists of native legumes, but usually do not. Ecol. Evol. 2017, 7, 8599–8611. [Google Scholar] [CrossRef] [Green Version]
- Keet, J.H.; Ellis, A.G.; Hui, C.; Le Roux, J.J. Legume-rhizobium symbiotic promiscuity and effectiveness do not affect plant invasiveness. Ann. Bot. 2017, 119, 1319–1331. [Google Scholar] [CrossRef]
- Jiao, Y.S.; Liu, Y.H.; Yan, H.; Wang, E.T.; Tian, C.F.; Chen, W.X.; Guo, B.L.; Chen, W.F. Rhizobial diversity and nodulation characteristics of the extremely promiscuous legume Sophora flavescens. Mol. Plant-Microbe Interact. 2015, 28, 1338–1352. [Google Scholar] [CrossRef] [Green Version]
- Simms, E.L.; Taylor, D.L. Partner choice in nitrogen-fixation mutualisms of legumes and rhizobia. Integr. Comp. Biol. 2002, 42, 369–380. [Google Scholar] [CrossRef]
- Marquard, E.; Weigelt, A.; Temperton, V.M.; Roscher, C.; Schumacher, J.; Buchmann, N.; Fischer, M.; Weisser, W.W.; Schmid, B. Plant species richness and functional composition drive overyielding in a six-year grassland experiment. Ecology 2009, 90, 3290–3302. [Google Scholar] [CrossRef] [Green Version]
- Menge, D.N.L.; Wolf, A.A.; Funk, J.L. Diversity of nitrogen fixation strategies in Mediterranean legumes. Nat. Plants 2015, 1, 15064. [Google Scholar] [CrossRef]
- Batterman, S.A.; Wurzburger, N.; Hedin, L.O. Nitrogen and phosphorus interact to control tropical symbiotic N2 fixation: A test in Inga punctata. J. Ecol. 2013, 101, 1400–1408. [Google Scholar] [CrossRef]
- Taylor, B.N.; Menge, D.N.L. Light regulates tropical symbiotic nitrogen fixation more strongly than soil nitrogen. Nat. Plants 2018, 4, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Myster, R.W. Light and nutrient effects on growth and allocation of Inga vera (Leguminosae), a successional tree of Puerto Rico. Can. J. For. Res. 2006, 36, 1121–1128. [Google Scholar] [CrossRef]
- Wurzburger, N.; Miniat, C.F. Drought enhances symbiotic dinitrogen fixation and competitive ability of a temperate forest tree. Oecologia 2014, 174, 1117–1126. [Google Scholar] [CrossRef]
- Dovrat, G.; Masci, T.; Bakhshian, H.; Mayzlish Gati, E.; Golan, S.; Sheffer, E. Drought-adapted plants dramatically downregulate dinitrogen fixation: Evidences from Mediterranean legume shrubs. J. Ecol. 2018, 106, 1534–1544. [Google Scholar] [CrossRef]
- Dovrat, G.; Sheffer, E. Symbiotic dinitrogen fixation is seasonal and strongly regulated in water-limited environments. New Phytol. 2019, 221, 1866–1877. [Google Scholar] [CrossRef]
- Crews, T.E. Phosphorus regulation of nitrogen fixation in a traditional Mexican agroecosystem. Biogeochemistry 1993, 21, 141–166. [Google Scholar] [CrossRef]
- Wilcots, M.E.; Menge, D.N.L.; Kuprewicz, E.K.; Taylor, B.N. Small traits with big consequences: How seed traits of nitrogen-fixing plants might influence ecosystem nutrient cycling. Oikos 2019, 128, 668–679. [Google Scholar] [CrossRef]
- Reid, D.E.; Ferguson, B.J.; Hayashi, S.; Lin, Y.H.; Gresshoff, P.M. Molecular mechanisms controlling legume autoregulation of nodulation. Ann. Bot. 2011, 108, 789–795. [Google Scholar] [CrossRef] [Green Version]
- Kiers, E.T.; Rousseau, R.A.; West, S.A.; Denison, R.F. Host sanctions and the legume-rhizobium mutualism. Nature 2003, 425, 78–81. [Google Scholar] [CrossRef]
- Menge, D.N.L.; Levin, S.A.; Hedin, L.O. Facultative versus obligate nitrogen fixation strategies and their ecosystem consequences. Am. Nat. 2009, 174, 465–477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Binkley, D.; Cromack, K.; Backer, D.D. Nitrogen fixation by red alder: Biology, rates, and controls. In The Biology and Managment of Red Alder; Hibbs, D., Tarrant, R., DeBell, D., Eds.; Oregon State University Press: Corvalis, OR, USA, 1994; pp. 57–72. [Google Scholar]
- Roscher, C.; Thein, S.; Weigelt, A.; Temperton, V.M.; Buchmann, N.; Schulze, E.D. N2 fixation and performance of 12 legume species in a 6-year grassland biodiversity experiment. Plant Soil 2011, 341, 333–348. [Google Scholar] [CrossRef]
- West, J.B.; HilleRisLambers, J.; Lee, T.D.; Hobbie, S.E.; Reich, P.B. Legume species identity and soil nitrogen supply determine symbiotic nitrogen-fixation responses to elevated atmospheric [CO2]. New Phytol. 2005, 167, 523–530. [Google Scholar] [CrossRef]
- Hagen, M.; Hamrick, J. A hierarchical analysis of population genetic structure in Rhizobium leguminosarum bv. trifolii. Mol. Ecol. 1996, 5, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Klock, M.M.; Barrett, L.G.; Thrall, P.H.; Harms, K.E. Host promiscuity in symbiont associations can influence exotic legume establishment and colonization of novel ranges. Divers. Distrib. 2015, 21, 1193–1203. [Google Scholar] [CrossRef]
- Futuyma, D.J.; Moreno, G. The evolution of ecological specialization. Annu. Rev. Ecol. Syst. 1988, 19, 207–233. [Google Scholar] [CrossRef]
- Wilson, D.S.; Yoshimura, J. On the coexistence of specialists and generalists. Am. Nat. 1994, 144, 692–707. [Google Scholar] [CrossRef]
- Ehinger, M.; Mohr, T.J.; Starcevich, J.B.; Sachs, J.L.; Porter, S.S.; Simms, E.L. Specialization-generalization trade-off in a Bradyrhizobium symbiosis with wild legume hosts. BMC Ecol. 2014, 14, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Devine, T.E.; Kuykendall, L.D. Host genetic control of symbiosis in soybean (Glycine max L.). Plant Soil 1996, 186, 173–187. [Google Scholar] [CrossRef]
- Materon, L.; Zibilske, L. Delayed inoculation and competition of nitrogen-fixing strains in Medicago noeana (Boiss.) and Medicago polymorpha (L.). Appl. Soil Ecol. 2001, 17, 175–181. [Google Scholar] [CrossRef]
- Oono, R.; Denison, R.F.; Kiers, E.T. Controlling the reproductive fate of rhizobia: How universal are legume sanctions? New Phytol. 2009, 183, 967–979. [Google Scholar] [CrossRef] [PubMed]
- Kiers, E.T.; Denison, R.F. Sanctions, cooperation, and the stability of plant-rhizosphere mutualisms. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 215–236. [Google Scholar] [CrossRef] [Green Version]
- West, S.A.; Kiers, E.T.; Simms, E.L.; Denison, R.F. Sanctions and mutualism stability: Why do rhizobia fix nitrogen? Proc. R. Soc. B Biol. Sci. 2002, 269, 685–694. [Google Scholar] [CrossRef] [Green Version]
- Bever, J.D.; Broadhurst, L.M.; Thrall, P.H. Microbial phylotype composition and diversity predicts plant productivity and plant-soil feedbacks. Ecol. Lett. 2013, 16, 167–174. [Google Scholar] [CrossRef] [Green Version]
- Thrall, P.H.; Slattery, J.F.; Broadhurst, L.M.; Bickford, S. Geographic patterns of symbiont abundance and adaptation in native Australian Acacia-rhizobia interactions. J. Ecol. 2007, 95, 1110–1122. [Google Scholar] [CrossRef]
- Finan, T.M. Evolving insights: Symbiosis islands and horizontal gene transfer. J. Bacteriol. 2002, 184, 2855–2856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacLean, A.M.; Finan, T.M.; Sadowsky, M.J. Genomes of the symbiotic nitrogen-fixing bacteria of legumes. Plant Physiol. 2007, 144, 615–622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peix, A.; Ramírez-Bahena, M.H.; Velázquez, E.; Bedmar, E.J. Bacterial associations with legumes. CRC. Crit. Rev. Plant Sci. 2015, 34, 17–42. [Google Scholar] [CrossRef]
- Drew, E.A.; Charman, N.; Dingemanse, R.; Hall, E.; Ballard, R.A. Symbiotic performance of Mediterranean Trifolium spp. with naturalised soil rhizobia. Crop Pasture Sci. 2011, 62, 903–913. [Google Scholar] [CrossRef]
- Lammel, D.R.; Cruz, L.M.; Carrer, H.; Cardoso, E.J.B.N. Diversity and symbiotic effectiveness of beta-rhizobia isolated from sub-tropical legumes of a Brazilian Araucaria forest. World J. Microbiol. Biotechnol. 2013, 29, 2335–2342. [Google Scholar] [CrossRef]
- Sachs, J.L.; Kembel, S.W.; Lau, A.H.; Simms, E.L. In situ phylogenetic structure and diversity of wild Bradyrhizobium communities. Appl. Environ. Microbiol. 2009, 75, 4727–4735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perret, X.; Staehelin, C.; Broughton, W.J. Molecular Basis of Symbiotic Promiscuity. Microbiol. Mol. Biol. Rev. 2000, 64, 180–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrett, L.G.; Bever, J.D.; Bissett, A.; Thrall, P.H. Partner diversity and identity impacts on plant productivity in Acacia-rhizobial interactions. J. Ecol. 2015, 103, 130–142. [Google Scholar] [CrossRef]
- Kaschuk, G.; Leffelaar, P.A.; Giller, K.E.; Alberton, O.; Hungria, M.; Kuyper, T.W. Responses of legumes to rhizobia and arbuscular mycorrhizal fungi: A meta-analysis of potential photosynthate limitation of symbioses. Soil Biol. Biochem. 2010, 42, 125–127. [Google Scholar] [CrossRef]
- Fox, J.W. Interpreting the “selection effect” of biodiversity on ecosystem function. Ecol. Lett. 2005, 8, 846–856. [Google Scholar] [CrossRef]
- Lechevalier, M.; Lechevalier, H. Systematics, isolation, and culture of Frankia. In The Biology of Frankia and Actinorhizal Plants; Schwintzer, C.R., Tjepkema, J.D., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 35–56. [Google Scholar]
- Friesen, M.L.; Mathias, A. Mixed infections may promote diversification of mutualistic symbionts: Why are there ineffective rhizobia? J. Evol. Biol. 2010, 23, 323–334. [Google Scholar] [CrossRef] [Green Version]
- Clark, T.J.; Friel, C.A.; Grman, E.; Friesen, M.L.; Shachar-Hill, Y. Unfair trade underground revealed by integrating data with Nash bargaining models. New Phytol. 2019, 222, 1325–1337. [Google Scholar] [CrossRef]
- Friesen, M.L. Widespread fitness alignment in the legume—Rhizobium symbiosis. New Phytol. 2012, 194, 1096–1111. [Google Scholar] [CrossRef]
- Goel, A.K.; Sindhu, S.S.; Dadarwal, K.R. Symbiotic effectiveness of bacteriocin producing and non-producing strains of Rhizobium in green gram (Vigna radiata). Indian J. Exp. Biol. 2001, 39, 821–823. [Google Scholar]
- Bala, A.; Giller, K.E. Relationships between rhizobial diversity and host legume nodulation and nitrogen fixation in tropical ecosystems. Nutr. Cycl. Agroecosystems 2006, 76, 319–330. [Google Scholar] [CrossRef]
- Lambers, J.H.R.; Harpole, W.S.; Tilman, D.; Knops, J.; Reich, P.B. Mechanisms responsible for the positive diversity-productivity relationship in Minnesota grasslands. Ecol. Lett. 2004, 7, 661–668. [Google Scholar] [CrossRef]
- Fargione, J.; Tilman, D.; Dybzinski, R.; Lambers, J.H.R.; Clark, C.; Harpole, W.S.; Knops, J.M.H.; Reich, P.B.; Loreau, M. From selection to complementarity: Shifts in the causes of biodiversity-productivity relationships in a long-term biodiversity experiment. Proc. R. Soc. B Biol. Sci. 2007, 274, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Mulder, C.P.H.; Jumpponen, A.; Högberg, P.; Huss-Danell, K. How plant diversity and legumes affect nitrogen dynamics in experimental grassland communities. Oecologia 2002, 133, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.D.; Reich, P.B.; Tjoelker, M.G. Legume presence increases photosynthesis and N concentrations of co-occurring non-fixers but does not modulate their responsiveness to carbon dioxide enrichment. Oecologia 2003, 137, 22–31. [Google Scholar] [CrossRef]
- Spehn, E.M.; Hector, A.; Joshi, J.; Scherer-Lorenzen, M.; Schmid, B.; Bazeley-White, E.; Beierkuhnlein, C.; Caldeira, M.C.; Diemer, M.; Dimitrakopoulos, P.G.; et al. Ecosystem effects of biodiversity manipulations in european grasslands. Ecol. Monogr. 2005, 75, 37–63. [Google Scholar] [CrossRef] [Green Version]
- Whittaker, R.H. Vegetation of the Siskiyou mountains, Oregon and California. Ecol. Monogr. 1960, 30, 279–338. [Google Scholar] [CrossRef]
- Grünzweig, J.M.; Körner, C. Growth, water and nitrogen relations in grassland model ecosystems of the semi-arid Negev of Israel exposed to elevated CO2. Oecologia 2001, 128, 251–262. [Google Scholar] [CrossRef]
- Ferreira da Silva, A.; Santiago de Freitas, A.D.; Costa, T.L.; Fernandes-Junior, P.I.; Martins, L.M.V.; de Rosalia e Silva Santos, C.E.; Menezes, K.A.S.; de Sa Barretto Sampaio, E.V. Biological nitrogen fixation in tropical dry forests with different legume diversity and abundance. Nutr. Cycl. Agroecosystems 2017, 107, 321–334. [Google Scholar] [CrossRef]
- Levine, J.M.; HilleRisLambers, J. The importance of niches for the maintenance of species diversity. Nature 2009, 461, 254–257. [Google Scholar] [CrossRef]
- Turnbull, L.A.; Rahm, S.; Baudois, O.; Eichenberger-Glinz, S.; Wacker, L.; Schmid, B. Experimental invasion by legumes reveals non-random assembly rules in grassland communities. J. Ecol. 2005, 93, 1062–1070. [Google Scholar] [CrossRef] [Green Version]
- Kéfi, S.; Domínguez-García, V.; Donohue, I.; Fontaine, C.; Thébault, E.; Dakos, V. Advancing our understanding of ecological stability. Ecol. Lett. 2019, 1349–1356. [Google Scholar] [CrossRef] [PubMed]
- Donohue, I.; Hillebrand, H.; Montoya, J.M.; Petchey, O.L.; Pimm, S.L.; Fowler, M.S.; Healy, K.; Jackson, A.L.; Lurgi, M.; McClean, D.; et al. Navigating the complexity of ecological stability. Ecol. Lett. 2016, 19, 1172–1185. [Google Scholar] [CrossRef] [PubMed]
- Menge, D.N.L.; Levin, S.A. Spatial heterogeneity can resolve the nitrogen paradox of tropical forests. Ecology 2017, 98, 1049–1061. [Google Scholar] [CrossRef] [PubMed]
- Menge, D.; Hedin, L. Nitrogen fixation in different biogeochemical niches along a 120,000-year chronosequence in New Zealand. Ecology 2009, 90, 2190–2201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boring, L.; Swank, W. The role of black locust (Robinia pseudo-acacia) in forest succession. J. Ecol. 1984, 72, 749–766. [Google Scholar] [CrossRef] [Green Version]
- Chapin, F.S., III; Walker, L.; Fastie, C.; Sharman, L. Mechanisms of primary succession following deglaciation at Glacier Bay, Alaska. Ecol. Monogr. 1994, 64, 149–175. [Google Scholar] [CrossRef]
- Oliver, T.H.; Heard, M.; Isaac, N.; Roy, D.; Procter, D.; Eigenbrod, F.; Freckleton, R.; Hector, A.; Orme, C.; Petchey, O.; et al. Biodiversity and resilience of ecosystem functions. Trends Ecol. Evol. 2015, 30, 673–684. [Google Scholar] [CrossRef] [Green Version]
- Tilman, D.; Reich, P.B.; Knops, J.M.H. Biodiversity and ecosystem stability in a decade-long grassland experiment. Nature 2006, 441, 629–632. [Google Scholar] [CrossRef]
- Denison, R.F.; Kiers, E.T. Life histories of symbiotic rhizobia and mycorrhizal fungi. Curr. Biol. 2011, 21, R775–R785. [Google Scholar] [CrossRef] [Green Version]
- McKey, D. Legumes and nitrogen: The evolutionary ecology of a nitrogen-demanding lifestyle. In Advances in Legume Systematics 5: The Nitrogen Factor; Sprent, J., McKey, D., Eds.; Royal Botanic Gardens: Kew, UK, 1994; pp. 211–228. [Google Scholar]
- Menge, D.N.L.; Batterman, S.A.; Liao, W.; Taylor, B.N.; Lichstein, J.W.; Ángeles-Pérez, G. Nitrogen-fixing tree abundance in higher-latitude North America is not constrained by diversity. Ecol. Lett. 2017, 20, 842–851. [Google Scholar] [CrossRef]
- Poorter, L.; Bongers, F.; Aide, T.M.; Almeyda Zambrano, A.M.; Balvanera, P.; Becknell, J.M.; Boukili, V.; Brancalion, P.H.S.; Broadbent, E.N.; Chazdon, R.L.; et al. Biomass resilience of Neotropical secondary forests. Nature 2016, 530, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.N.; Chazdon, R.L.; Menge, D.N.L. Successional dynamics of nitrogen fixation and forest growth in regenerating Costa Rican rainforests. Ecology 2019, 100, e02637. [Google Scholar] [CrossRef] [PubMed]
- Kiers, E.T.; Rousseau, R.A.; Denison, R.F. Measured sanctions: Legume hosts detect quantitative variation in rhizobium cooperation and punish accordingly. Evol. Ecol. Res. 2006, 8, 1077–1086. [Google Scholar]
- King, B.J.; Layzell, D.B. Effect of increases in oxygen concentration during the argon-induced decline in nitrogenase activity in root nodules of soybean. Plant Physiol. 1991, 96, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Wårlind, D.; Smith, B.; Hickler, T.; Arneth, A. Nitrogen feedbacks increase future terrestrial ecosystem carbon uptake in an individual-based dynamic vegetation model. Biogeosciences 2014, 11, 151–185. [Google Scholar] [CrossRef]
- Peng, J.; Wang, Y.-P.; Houlton, B.Z.; Dan, L.; Pak, B.; Tang, X. Global carbon sequestration is highly sensitive to model-based formulations of nitrogen fixation. Glob. Biogeochem. Cycles 2019. [Google Scholar] [CrossRef]
- Gerber, S.; Hedin, L.O.; Keel, S.G.; Pacala, S.W.; Shevliakova, E. Land use change and nitrogen feedbacks constrain the trajectory of the land carbon sink. Geophys. Res. Lett. 2013, 40, 5218–5222. [Google Scholar] [CrossRef] [Green Version]
- Akçay, E.; Roughgarden, J. Negotiation of mutualism: Rhizobia and legumes. Proc. R. Soc. B Biol. Sci. 2007, 274, 25–32. [Google Scholar] [CrossRef] [Green Version]
- Trierweiler, A.M.; Winter, K.; Hedin, L.O. Rising CO2 accelerates phosphorus and molybdenum limitation of N2-fixation in young tropical trees. Plant Soil 2018, 429, 363–373. [Google Scholar] [CrossRef]
- Weese, D.J.; Heath, K.D.; Dentinger, B.T.M.; Lau, J.A. Long-term nitrogen addition causes the evolution of less-cooperative mutualists. Evolution (N. Y.) 2015, 69, 631–642. [Google Scholar] [CrossRef]
- Houlton, B.Z.; Wang, Y.-P.; Vitousek, P.M.; Field, C.B. A unifying framework for dinitrogen fixation in the terrestrial biosphere. Nature 2008, 454, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.; Menge, D.N.L.; Lichstein, J.W.; Ángeles-Pérez, G. Global climate change will increase the abundance of symbiotic nitrogen-fixing trees in much of North America. Glob. Chang. Biol. 2017, 23, 4777–4787. [Google Scholar] [CrossRef] [PubMed]
- Hayman, D. Mycorrhizae of nitrogen-fixing legumes. Mircen J. Appl. Microbiol. Biotechnol. 1986, 2, 121–145. [Google Scholar] [CrossRef]
- Smith, S.; Nicholas, D.; Smith, F. Effect of early mycorrhizal infection on nodulation and nitrogen fixation in Trifolium subterraneum L. Funct. Plant Biol. 1979, 6, 305. [Google Scholar] [CrossRef]
- Diagne, N.; Thioulouse, J.; Sanguin, H.; Prin, Y.; Krasova-Wade, T.; Sylla, S.; Galiana, A.; Baudoin, E.; Neyra, M.; Svistoonoff, S.; et al. Ectomycorrhizal diversity enhances growth and nitrogen fixation of Acacia mangium seedlings. Soil Biol. Biochem. 2013, 57, 468–476. [Google Scholar] [CrossRef]
- Nasto, M.K.; Alvarez-Clare, S.; Lekberg, Y.; Sullivan, B.W.; Townsend, A.R.; Cleveland, C.C. Interactions among nitrogen fixation and soil phosphorus acquisition strategies in lowland tropical rain forests. Ecol. Lett. 2014, 17, 1282–1289. [Google Scholar] [CrossRef]
- Mayer, V.E.; Frederickson, M.E.; Mckey, D.; Blatrix, R. Current issues in the evolutionary ecology of ant-plant symbioses. New Phytol. 2014, 202, 749–764. [Google Scholar] [CrossRef]
- Tarnita, C.E.; Palmer, T.M.; Pringle, R.M. Colonisation and competition dynamics can explain incomplete sterilisation parasitism in ant-plant symbioses. Ecol. Lett. 2014, 17, 1290–1298. [Google Scholar] [CrossRef]
- Godschalx, A.L.; Schadler, M.; Trisel, J.; Balkan, M.; Ballhorn, D. Ants are less attracted to the extrafloral nectar of plants with symbiotic, nitrogen-fixing rhizobia. Ecology 2015, 96, 348–354. [Google Scholar] [CrossRef] [Green Version]
- Afkhami, M.E.; Rudgers, J.A.; Stachowicz, J.J. Multiple mutualist effects: Conflict and synergy in multispecies mutualisms. Ecology 2014, 95, 833–844. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taylor, B.N.; Simms, E.L.; Komatsu, K.J. More Than a Functional Group: Diversity within the Legume–Rhizobia Mutualism and Its Relationship with Ecosystem Function. Diversity 2020, 12, 50. https://doi.org/10.3390/d12020050
Taylor BN, Simms EL, Komatsu KJ. More Than a Functional Group: Diversity within the Legume–Rhizobia Mutualism and Its Relationship with Ecosystem Function. Diversity. 2020; 12(2):50. https://doi.org/10.3390/d12020050
Chicago/Turabian StyleTaylor, Benton N., Ellen L. Simms, and Kimberly J. Komatsu. 2020. "More Than a Functional Group: Diversity within the Legume–Rhizobia Mutualism and Its Relationship with Ecosystem Function" Diversity 12, no. 2: 50. https://doi.org/10.3390/d12020050
APA StyleTaylor, B. N., Simms, E. L., & Komatsu, K. J. (2020). More Than a Functional Group: Diversity within the Legume–Rhizobia Mutualism and Its Relationship with Ecosystem Function. Diversity, 12(2), 50. https://doi.org/10.3390/d12020050




