Conserving the Diversity of Ecological Interactions: The Role of Two Threatened Macaw Species as Legitimate Dispersers of “Megafaunal” Fruits
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Areas
2.2. Plant Species Dispersed
2.3. Foraging and Dispersal Behavior and Dispersal Rates
2.4. Dispersal Distances
2.5. The Proportion of Surviving Dispersed Seeds and Realized Dispersal
2.6. Statistical Analyses
2.7. Ethics Statement
3. Results
3.1. Plants Dispersed
3.2. Fruit Handling and Dispersal Behavior
3.3. Primary Dispersal Rates
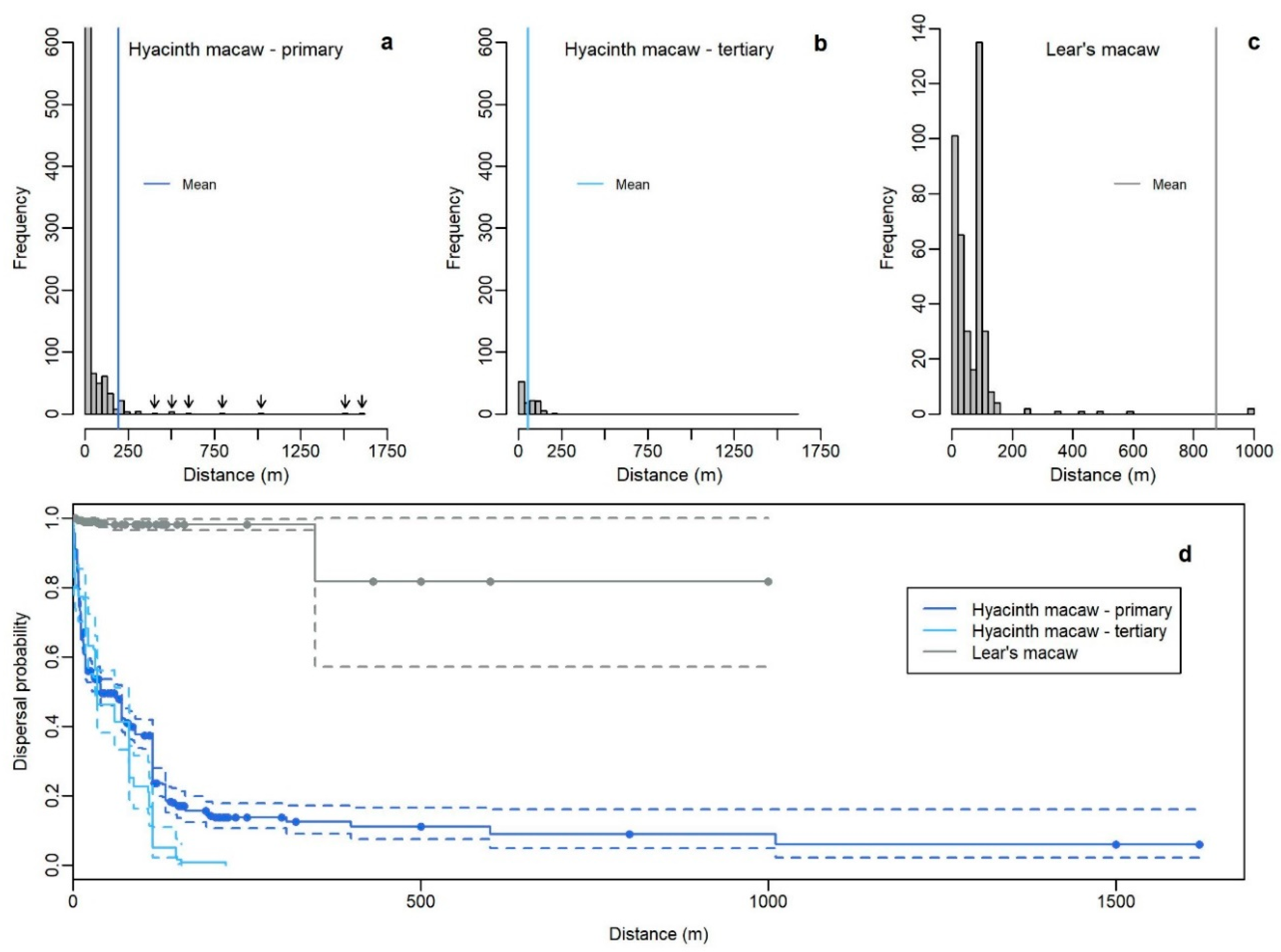
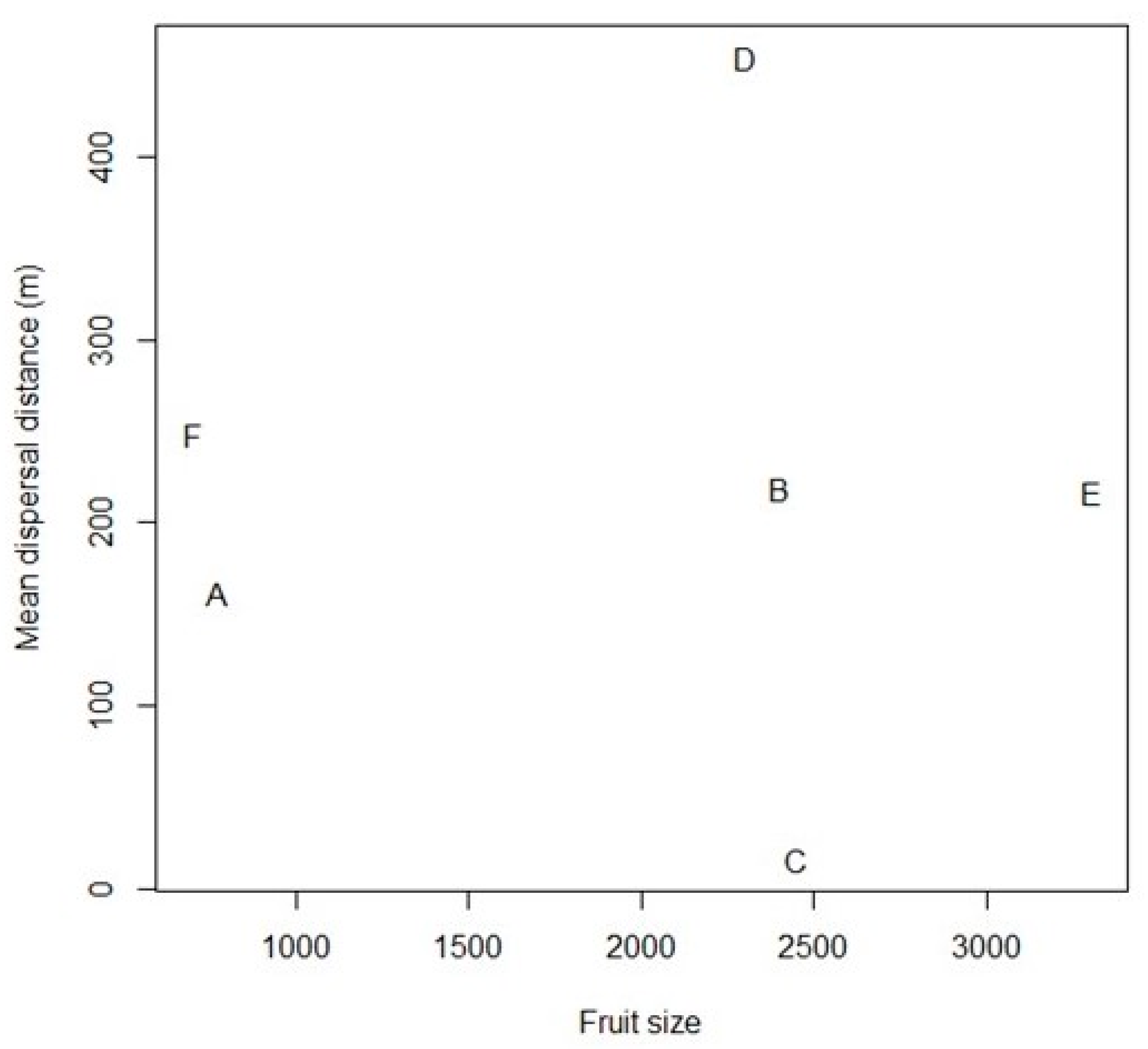
3.4. Dispersal Distances
3.5. The Proportion of Surviving Dispersed Seeds
3.6. Realized Seed Dispersal
4. Discussion
4.1. Anodorhynchus Macaws as Frequent, Long-distance Dispersers of Large-fruited Plants
4.2. Rates and Locations of Effective Seed Dispersal and Recruitment
4.3. Palm-Macaw Evolutionary Relationships and the Megafauna Syndrome
4.4. The Loss of Palm-Macaw Mutualisms
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Turvey, S.T.; Crees, J.J. Extinction in the Anthropocene. Cur. Biol. 2019, 29, R982-6. [Google Scholar] [CrossRef] [PubMed]
- The IUCN Red List of Threatened Species. Version 2019-3. Available online: https://www.iucnredlist.org (accessed on 28 December 2019).
- Johnson, C.N.; Balmford, A.; Brook, B.W.; Buettel, J.C.; Galetti, M.; Guangchun, L.; Wilmshurst, J.M. Biodiversity losses and conservation responses in the Anthropocene. Science 2017, 356, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Jordano, P. Chasing ecological interactions. PLoS Biol. 2016, 14, e1002559. [Google Scholar] [CrossRef] [PubMed]
- Valiente-Banuet, A.; Aizen, M.A.; Alcántara, J.M.; Arroyo, J.; Cocucci, A.; Galetti, M.; García, M.B.; García, D.; Gómez, J.M.; Jordano, P.; et al. Beyond species loss: the extinction of ecological interactions in a changing world. Funct. Ecol. 2015, 29, 299–307. [Google Scholar] [CrossRef]
- Akçakaya, H.R.; Rodrigues, A.S.; Keith, D.A.; Milner-Gulland, E.J.; Sanderson, E.W.; Hedges, S.; Mallon, D.P.; Grace, M.K.; Long, B.; Meijaard, E.; et al. Assessing ecological function in the context of species recovery. Conserv. Biol. 2019. [Google Scholar] [CrossRef]
- Jordano, P. Fruits and frugivory. In Seeds: The Ecology of Regeneration in Plant Communities, 2nd ed.; Fenner, M., Ed.; CABI: Wallingford, UK, 2000; pp. 125–166. [Google Scholar]
- Wisz, M.S.; Pottier, J.; Kissling, W.D.; Pellissier, L.; Lenoir, J.; Damgaard, C.F.; Dormann, C.F.; Forchhammer, M.C.; Grytnes, J.A.; Guisan, A.; et al. The role of biotic interactions in shaping distributions and realised assemblages of species: implications for species distribution modelling. Biol. Rev. 2013, 88, 15–30. [Google Scholar] [CrossRef]
- Wright, S.J.; Stoner, K.E.; Beckman, N.; Corlett, R.T.; Dirzo, R.; Muller-Landau, H.C.; Nuñez-Iturri, G.; Peres, C.A.; Wang, B.C. The plight of large animals in tropical forests and the consequences for plant regeneration. Biotropica 2007, 39, 289–291. [Google Scholar] [CrossRef]
- McConkey, K.R.; O’Farrill, G. Loss of seed dispersal before the loss of seed dispersers. Biol. Conserv. 2016, 201, 38–49. [Google Scholar] [CrossRef]
- Dirzo, R.; Mendoza, E.; Ortiz, P. Size-related differential seed predation in a heavily defaunated neotropical rain forest. Biotropica 2007, 39, 355–362. [Google Scholar] [CrossRef]
- Terborgh, J.; Nuñez-Iturri, G.; Pitman, N.C.; Valverde, F.H.C.; Alvarez, P.; Swamy, V.; Pringle, E.G.; Paine, C.E. Tree recruitment in an empty forest. Ecology 2008, 89, 1757–1768. [Google Scholar] [CrossRef]
- Peres, C.A.; Emilio, T.; Schietti, J.; Desmoulière, S.J.; Levi, T. Dispersal limitation induces long-term biomass collapse in overhunted Amazonian forests. Proc. Nat. Acad. Sci. USA 2016, 113, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Malhi, Y.; Doughty, C.E.; Galetti, M.; Smith, F.A.; Svenning, J.C.; Terborgh, J.W. Megafauna and ecosystem function from the Pleistocene to the Anthropocene. Proc. Nat. Acad. Sci. USA 2016, 113, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, P.R.; Galetti, M.; Jordano, P. Seed dispersal anachronisms: Rethinking the fruits extinct megafauna ate. PLoS ONE 2008, 3, e1745. [Google Scholar]
- Janzen, D.H.; Martin, P.S. Neotropical anachronisms: the fruits the gomphotheres ate. Science 1982, 215, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Blanco, G.; Tella, J.L.; Díaz-Luque, J.A.; Hiraldo, F. Multiple external seed dispersers challenge the megafaunal syndrome anachronism and the surrogate ecological function of livestock. Front. Ecol. Evol. 2019, 7, 328. [Google Scholar] [CrossRef]
- Fragoso, J.M.V.; Huffman, J.M. Seed-dispersal and seedling recruitment patterns by the last Neotropical megafaunal element in Amazonia, the tapir. J. Trop. Ecol. 2000, 16, 369–385. [Google Scholar] [CrossRef]
- Sebastián-González, E. Drivers of species’ role in avian seed-dispersal mutualistic networks. J. Anim. Ecol. 2017, 86, 878–887. [Google Scholar] [CrossRef]
- Onstein, R.E.; Baker, W.J.; Couvreur, T.L.; Faurby, S.; Svenning, J.C.; Kissling, W.D. Frugivory-related traits promote speciation of tropical palms. Nature Ecol. Evol. 2017, 1, 1903. [Google Scholar] [CrossRef]
- Onstein, R.E.; Baker, W.J.; Couvreur, T.L.; Faurby, S.; Herrera-Alsina, L.; Svenning, J.C.; Kissling, W.D. To adapt or go extinct? The fate of megafaunal palm fruits under past global change. Proc. R. Soc. B 2018, 285, 20180882. [Google Scholar] [CrossRef]
- Muñoz, G.; Trøjelsgaard, K.; Kissling, W.D. A synthesis of animal-mediated seed dispersal of palms reveals distinct biogeographical differences in species interactions. J. Biogeogr. 2019, 46, 466–484. [Google Scholar] [CrossRef]
- Tella, J.L.; Baños-Villalba, A.; Hernández-Brito, D.; Rojas, A.; Pacífico, E.; Díaz-Luque, J.A.; Carrete, M.; Blanco, G.; Hiraldo, F. Parrots as overlooked seed dispersers. Front. Ecol. Environ. 2015, 13, 338–339. [Google Scholar] [CrossRef]
- Baños-Villalba, A.; Blanco, G.; Díaz-Luque, J.A.; Dénes, F.V.; Hiraldo, F.; Tella, J.L. Seed dispersal by macaws shapes the landscape of an Amazonian ecosystem. Sci. Rep. 2017, 7, 7373. [Google Scholar] [CrossRef] [PubMed]
- Toft, C.A.; Wright, T.F. Parrots of the Wild: A Natural History of the World’s Most Captivating Birds; University of California Press: Oakland, CA, USA, 2015. [Google Scholar]
- Forshaw, J.M. Parrots of the World; Christopher Helm: London, UK, 2010. [Google Scholar]
- Yamashita, C. Anodorhynchus macaws as followers of extinct megafauna: An hypothesis. Ararajuba 1997, 5, 176–182. [Google Scholar]
- Montesinos-Navarro, A.; Hiraldo, F.; Tella, J.L.; Blanco, G. Network structure embracing mutualism-antagonism continuums increases community robustness. Nature Ecol. Evol. 2017, 1, 1661. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, C.; de Paula Valle, M. On the linkage between Anodorhynchus macaws and palm nuts, and the extinction of the glaucous macaw. Bull. Br. Ornithol. Club 1993, 113, 53–60. [Google Scholar]
- Bullock, J.M.; Shea, K.; Skarpaas, O. Measuring plant dispersal: An introduction to field methods and experimental design. Plant Ecol. 2006, 186, 217–234. [Google Scholar] [CrossRef]
- Nathan, R.; Perry, G.; Cronin, J.T.; Strand, A.E.; Cain, M.L. Methods for estimating long-distance dispersal. Oikos 2003, 103, 261–273. [Google Scholar] [CrossRef]
- Tella, J.L.; Blanco, G.; Dénes, F.V.; Hiraldo, F. Overlooked parrot seed dispersal in Australia and South America: insights on the evolution of dispersal syndromes and seed size in Araucaria trees. Front. Ecol. Evol. 2019, 7, 82. [Google Scholar] [CrossRef]
- BirdLife International. Anodorhynchus hyacinthinus. The IUCN Red List of Threatened Species 2016. Available online: http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22685516A93077457.en. (accessed on 25 December 2019).
- BirdLife International. Anodorhynchus leari. The IUCN Red List of Threatened Species 2019. Available online: https://www.iucnredlist.org/species/22685521/141364064 (accessed on 25 December 2019).
- Silva, J.M.C.; Leal, I.; Tabarelli, M. Caatinga: The Largest Tropical Dry Forest Region in South America; Springer: New York, NY, USA, 2018. [Google Scholar]
- Silva, A.C.; Souza, A.F. Aridity drives plant biogeographical sub regions in the Caatinga, the largest tropical dry forest and woodland block in South America. PLoS ONE 2018, 13, e0196130. [Google Scholar] [CrossRef]
- Silva, J.M.C.; Bates, J.M. Biogeographic patterns and conservation in the South American Cerrado: A tropical savanna hotspot. BioScience 2002, 52, 225. [Google Scholar] [CrossRef]
- Silva, J.F.; Felfili, J.M.; Klink, C.A.; Farin, M.R.; Farinas, M.R. Spatial heterogeneity, land use and conservation in the Cerrado region of Brazil. J. Biogeog. 2006, 33, 536–548. [Google Scholar] [CrossRef]
- Global Forest Watch. Available online: http://data.globalforestwatch.org/datasets/54ec099791644be4b273d9d8a853d452_4 (accessed on 15 June 2019).
- Landsat-4 images. Available online: http://glovis.usgs.gov/ (accessed on 15 June 2019).
- Lorenzi, H.; Noblyck, L.; Kahn, F.; Ferreira, E. Flora Brasileira: Arecaceae (palmeiras); Instituto Plantarum: Nova Odessa, SP, Brazil, 2010. [Google Scholar]
- Queiroz, L.P. Leguminosas da Caatinga; Universidad Estadual de Feirea de Santana: Bahía, Brazil, 2009. [Google Scholar]
- Moreno, L.R.; Moreno, O.I. Colecciones de las palmeras de Bolivia, 2nd ed; Palmae-Arecaceae Editorial Fundación Amigos de la Naturaleza: Santa Cruz de la Sierra, Bolivia, 2013. [Google Scholar]
- Lorenzi, H. Arvores brasileiras: Manual de identificaçao e cultivo de plantas arbóreas nativas do Brasil., 4th ed.; Instituto Plantarum: Nova Odessa, SP, Brazil, 2013; Volume 2. [Google Scholar]
- Blanco, G.; Hiraldo, F.; Rojas, A.; Dénes, F.V.; Tella, J.L. Parrots as key multilinkers in ecosystem structure and functioning. Ecol. Evol. 2015, 18, 4141–4160. [Google Scholar] [CrossRef] [PubMed]
- Tella, J.L.; Dénes, F.V.; Zulian, V.; Prestes, N.P.; Martínez, J.; Blanco, G.; Hiraldo, F. Endangered plant-parrot mutualisms: Seed tolerance to predation makes parrots pervasive dispersers of the Parana pine. Sci. Rep. 2016, 6, 31709. [Google Scholar] [CrossRef] [PubMed]
- Klein, J.P.; Moeschberger, M.L. Survival Analysis: Techniques for Censored and Truncated Data; Springer: New York, NY, USA, 2003. [Google Scholar]
- Harrington, D.P.; Fleming, T.R. A class of rank test procedures for censored survival data. Biometrika 1982, 69, 553–566. [Google Scholar] [CrossRef]
- Therneau, T.A. Package for Survival Analysis in S. version 2.38. Available online: http://CRAN.R-project.org/package=survival (accessed on 15 January 2019).
- R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available online: https://www.R-project.org/ (accessed on 15 January 2019).
- Grime, J.P. Plant Strategies, Vegetation Processes, and Ecosystem Properties; John Wiley & Sons: Hongkong, China, 2006. [Google Scholar]
- Vender Wall, S.B. How plants manipulate the scatter-hoarding behaviour of seed-dispersing animals. Phil. Trans. R. Soc. Lond. B 2010, 365, 989–997. [Google Scholar] [CrossRef]
- Jansen, P.A.; Visser, M.D.; Wright, J.S.; Rutten, G.; Muller-Landau, H.C. Negative density dependence of seed dispersal and seedling recruitment in a Neotropical palm. Ecol. Lett. 2014, 17, 1111–1120. [Google Scholar] [CrossRef]
- Blanco, G.; Hiraldo, G.; Tella, J.L. Ecological functions of parrots: An integrative perspective from plant life cycle to ecosystem functioning. Emu 2018, 118, 36–49. [Google Scholar] [CrossRef]
- Boehning-Gaese, K.; Gaese, B.H.; Rabemanantsoa, S.B. Importance of primary and secondary seed dispersal in the Malagasy tree Commiphora guillaumini. Ecology 1999, 80, 821–832. [Google Scholar] [CrossRef]
- Blanco, G.; Bravo, C.; Pacífico, E.; Chamorro, D.; Speziale, K.; Lambertucci, S.; Hiraldo, F.; Tella, J.L. Internal seed dispersal by parrots: An overview of a neglected mutualism. PeerJ 2016, 4, e1688. [Google Scholar] [CrossRef]
- Filardi, C.E.; Tewksbury, J. Ground-foraging palm cockatoos (Probosciger aterrimus) in lowland New Guinea: fruit flesh as a directed deterrent to seed predation? J. Trop. Ecol. 2005, 21, 355–361. [Google Scholar] [CrossRef]
- Jansen, P.A.; Hirsch, B.T.; Emsens, W.J.; Zamora-Gutierrez, V.; Wikelski, M.; Kays, R. Thieving rodents as substitute dispersers of megafaunal seeds. Proc. Nat. Acad. Sci. USA 2012, 109, 12610–12615. [Google Scholar] [CrossRef]
- Pires, M.M.; Guimarães, P.R.; Galetti, M.; Jordano, P. Pleistocene megafaunal extinctions and the functional loss of long-distance seed-dispersal services. Ecography 2017, 41, 153–163. [Google Scholar] [CrossRef]
- Cain, M.L.; Milligan, B.G.; Strand, A.E. Long-distance seed dispersal in plant populations. Am. J. Bot. 2000, 87, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Nathan, R.; Muller-Landau, H.C. Spatial patterns of seed dispersal, their determinants and consequences for recruitment. Trends Ecol. Evol. 2000, 15, 278–285. [Google Scholar] [CrossRef]
- Howe, H.F.; Miriti, M.N. When seed dispersal matters. BioScience 2004, 54, 651–660. [Google Scholar] [CrossRef]
- Jordano, P. What is long-distance dispersal? And a taxonomy of dispersal events. J. Ecol. 2017, 105, 75–84. [Google Scholar] [CrossRef]
- Bartel, R.D.; Sheppard, J.L.; Lovas-Kiss, Á.; Green, A.J. Endozoochory by mallard in New Zealand: what seeds are dispersed and how far? PeerJ 2018, 6, e4811. [Google Scholar] [CrossRef]
- Sebastián-González, E.; Hiraldo, F.; Blanco, G.; Hernández-Brito, D.; Romero-Vidal, P.; Carrete, M.; Gómez-Llanos, E.; Pacífico, E.C.; Díaz-Luque, J.A.; Dénes, F.V.; et al. The extent, frequency and ecological functions of food wasting by parrots. Sci. Rep. 2019, 9, 15280. [Google Scholar] [CrossRef]
- Tella, J.L.; Lambertucci, S.; Speziale, K.; Hiraldo, F. Large-scale impacts of multiple co-occurring invaders on monkey puzzle forest regeneration, native seed predators and their ecological interactions. Global Ecol. Conserv. 2016, 6, 1–15. [Google Scholar] [CrossRef]
- Dénes, F.; Tella, J.L.; Zulian, V.; Prestes, N.M.; Martínez, J.; Hiraldo, F. Combined impacts of multiple non-native mammals on two life stages of a critically endangered Neotropical tree. Biol. Invas. 2018, 20, 3055–3068. [Google Scholar] [CrossRef]
- Göldel, B.; Araujo, A.C.; Kissling, W.D.; Svenning, J.C. Impacts of large herbivores on spinescence and abundance of palms in the Pantanal, Brazil. Bot. J. Linn. Soc. 2016, 182, 465–479. [Google Scholar] [CrossRef]
- Thompson, J.N. The Coevolutionary Process; University of Chicago Press: Chicago, IL, USA, 1994. [Google Scholar]
- Borsari, A.; Ottoni, E.B. Preliminary observations of tool use in captive hyacinth macaws (Anodorhynchus hyacinthinus). Anim. Cogn. 2005, 8, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.; Serbena, A.L.; Guedes, N.M.R. Behavioral categories of hyacinth macaws (Anodorhynchus hyacinthinus) during the reproductive period, at South Pantanal. Brazil. Rev. Etol. 2006, 8, 71–80. [Google Scholar]
- Moura, A.C.deA.; Lee, P.C. Capuchin stone tool use in Caatinga dry forest. Science 2004, 306, 1909. [Google Scholar] [CrossRef] [PubMed]
- Litchfield, C. Taxonomic patterns in the fat content, fatty acid composition, and triglyceride composition of palmae seeds. Chem. Phy. Lipids 1970, 4, 96–103. [Google Scholar] [CrossRef]
- Crepaldi, I.C.; Almeida-Muradian, L.D.; Rios, M.D.G.; Penteado, M.V.C.; Salatino, A. Composição nutricional do fruto de licuri (Syagrus coronata (Martius) Beccari). Rev. Bras. Bot. 2004, 24, 155–159. [Google Scholar] [CrossRef]
- Karasov, W.H.; Martínez del Río, C. Physiological Ecology: How Animals Process Energy, Nutrients and Toxins; Princeton Univ. Press: Princeton, NJ, USA, 2007. [Google Scholar]
- Henderson, A. Evolution and ecology of palms; The New York Botanical Garden Press: New York, NY, USA, 2002. [Google Scholar]
- Baker, W.J.; Couvreur, T.L. Global biogeography and diversification of palms sheds light on the evolution of tropical lineages. II. Diversification history and origin of regional assemblages. J. Biogeogr. 2013, 40, 286–298. [Google Scholar] [CrossRef]
- Mayr, G. The origins of crown group birds: Molecules and fossils. Palaeontology 2014, 57, 231–242. [Google Scholar] [CrossRef]
- Wright, T.F.; Schirtzinger, E.E.; Matsumoto, T.; Eberhard, J.R.; Graves, G.R.; Sanchez, J.J.; Capelli, S.; Müller, H.; Scharpegge, J.; Chambers, G.K.; et al. A multilocus molecular phylogeny of the parrots (Psittaciformes): support for a Gondwanan origin during the Cretaceous. Mol. Biol. Evol. 2008, 25, 2141–2156. [Google Scholar] [CrossRef]
- Schweizer, M.; Seehausen, O.; Hertwig, S.T. Macroevolutionary patterns in the diversification of parrots – effects of climate change, geological events and key innovations. J. Biogeogr. 2011, 38, 2176–2194. [Google Scholar] [CrossRef]
- Claramunt, S.; Cracraft, J. A new time tree reveals Earth history’s imprint on the evolution of modern birds. Sci. Adv. 2015, 1, e1501005. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, O. Evolution of angiosperm seed disperser mutualisms: the timing of origins and their consequences for coevolutionary interactions between angiosperms and frugivores. Biol. Rev. 2016, 91, 168–186. [Google Scholar] [CrossRef] [PubMed]
- McGill, B.J.; Dornelas, M.; Gotelli, N.J.; Magurran, A.E. Fifteen forms of biodiversity trend in the Anthropocene. Trends Ecol. Evol. 2015, 30, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, C.H.A.; Tella, J.L.; Green, A.J. Editorial: Animal-mediated dispersal in understudied systems. Front. Ecol. Evol. 2019. [Google Scholar] [CrossRef]
- Faria, P.J.; Guedes, N.M.; Yamashita, C.; Martuscelli, P.; Miyaki, C.Y. Genetic variation and population structure of the endangered Hyacinth Macaw: Implications for conservation. Biodivers. Conserv. 2008, 17, 765–779. [Google Scholar] [CrossRef]
- Pacífico, E.C.; Barbosa, E.A.; Filadelfo, T.; Oliveira, K.G.; Silveira, L.F.; Tella, J.L. Breeding to non-breeding population ratio and breeding performance of the globally endangered Lear’s Macaw (Anodorhynchus leari): conservation and monitoring implications. Bird Conserv. Int. 2014, 24, 466–476. [Google Scholar]
- Roque, F.O.; Ochoa-Quintero, J.; Ribeiro, D.B.; Sugai, L.S.; Costa-Pereira, R.; Lourival, R.; Bino, G. Upland habitat loss as a threat to Pantanal wetlands. Conserv. Biol. 2016, 30, 1131–1134. [Google Scholar] [CrossRef]
- Carvalho, F.M.V.; De Marco, P.; Ferreira, L.G. The Cerrado into-pieces: habitat fragmentation as a function of landscape use in the savannas of central Brazil. Biol. Conserv. 2009, 142, 1392–1403. [Google Scholar] [CrossRef]
- Ribeiro, E.; Arroyo-Rodríguez, V.; Santos, B.A.; Tabarelli, M.; Leal, I.R. Chronic anthropogenic disturbance drives the biological impoverishment of the Brazilian Caatinga vegetation. J. Appl. Ecol. 2015, 52, 611–620. [Google Scholar] [CrossRef]
- Antogionvani, M.; Venticique, E.M.; Fonseca, C.R. Fragmentation patterns of the Caatinga drylands. Lands. Ecol. 2018, 33, 1353–1367. [Google Scholar] [CrossRef]
- Lugarini, C.; Barbosa, A.E.A.; Oliveira, K.G. Plano de açao nacional para a conservaçao da arara-azul-de-Lear; Série Espécies Amenaçadas nº 4. ICMBio: Brasília, Brazil, 2012. [Google Scholar]
- de Andrade, W.M.; Ramos, M.A.; Souto, W.M.S.; Bento-Silva, J.S. Knowledge, uses and practices of the licuri palm (Syagrus coronata (Mart.) Becc.) around protected areas in northeastern Brazil holding the endangered species Lear’s Macaw (Anodorhynchus leari). Trop. Conserv. Sci. 2015, 8, 893–911. [Google Scholar] [CrossRef]
- Luna, A.; Romero-Vidal, P.; Hiraldo, F.; Tella, J.L. Cities may save some threatened species but not their ecological functions. PeerJ 2018, 6, e4908. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Méndez, N.; Jordano, P.; García, C.; Valido, A. The signatures of Anthropocene defaunation: cascading effects of the seed dispersal collapse. Sci. Rep. 2016, 6, 24820. [Google Scholar] [CrossRef] [PubMed]
- Lanes, É.C.; Motoike, S.Y.; Kuki, K.N.; Resende, M.D.; Caixeta, E.T. Mating system and genetic composition of the macaw palm (Acrocomia aculeata): Implications for breeding and genetic conservation programs. J. Hered. 2016, 107, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.E.A.; Tella, J.L. How much does it cost to save a species from extinction? Costs and rewards of conserving the Lear’s macaw. R. Soc. Open Sci. 2019, 6, 190190. [Google Scholar] [CrossRef] [PubMed]
- Genes, L.; Fernandez, F.A.; Vaz-de-Mello, F.Z.; da Rosa, P.; Fernandez, E.; Pires, A.S. Effects of howler monkey reintroduction on ecological interactions and processes. Conserv. Biol. 2019, 33, 88–98. [Google Scholar] [CrossRef] [PubMed]
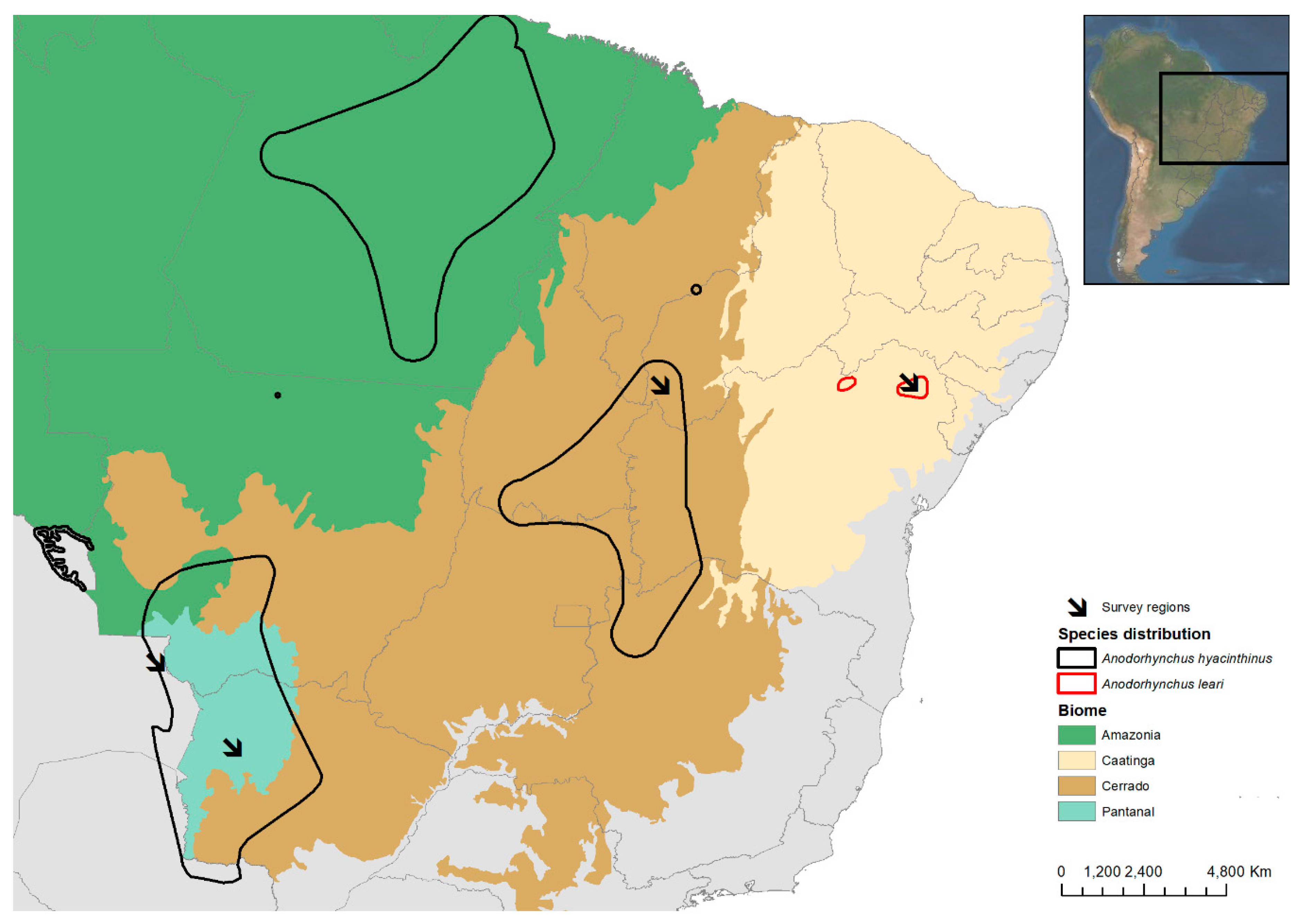


| Biome | Study Areas | Coordinates | Country | Fieldwork Dates | Macaw Species | Palm Species |
|---|---|---|---|---|---|---|
| Caatinga | Raso da Catarina | 10° 17.089′ S, 38° 42.419′ W | Brazil | August, September 2014; February, | A. leari | Syagrus coronata |
| April, May 2015, March-May 2016 | ||||||
| Cerrado | São Gonçalo | 10° 06.023′ S, 45° 22.228′ W | Brazil | June 2015, October 2016, | A. hyacinthinus | Attalea barreirensis |
| da Gurguéia | January 2017 | Attalea eichleri | ||||
| Mauritia flexuosa | ||||||
| Pantanal | Fazenda Caiman | 19° 57.263′ S, 56°18.258′ W | Brazil | November 2015 | A. hyacinthinus | Acrocomia totai |
| Attalea phalerata | ||||||
| Pantanal | San Matías | 17° 13.437′ S, 58° 36.700′ W | Bolivia | November 2017 | A. hyacinthinus | Acrocomia totai |
| Attalea phalerata |
| Plant species | Family | Biome | Fruit Size | Seed Size | N Seeds | Source | MF | PD | TD | Distance Range |
|---|---|---|---|---|---|---|---|---|---|---|
| Lear’s macaw | ||||||||||
| Anacardium occidentale | Anacardiaceae | Caatinga | 90 × 40 | 40 × 25 | 1 | [15] | Yes | 3 | 160–600 | |
| Cereus jamacaru | Cactaceae | Caatinga | 82.3 × 62.6 | 2.62 × 1.73 | 1400 | own | 2 | 50–127 | ||
| Colicodendron (Capparis) yco | Capparaceae | Caatinga | 67.6 × 42.4 | 42.4 × 12.9 | 17 | own | Yes | 2 | 10–10 | |
| Dioclea grandiflora | Fabacedae | Caatinga | 115 × 40 | 25 × 25 | 2–5 | [42] | 1 | 150 | ||
| Jatropha sp. | Euphorbiaceae | Caatinga | 4 | 119–348 | ||||||
| Jatropha mollisima | Euphorbiaceae | Caatinga | 19.3 × 18.7 | 9 × 6 | 3 | own | 10 | 4–25 | ||
| Manihot sp. | Euphorbiaceae | Caatinga | 1 | 4 | ||||||
| Pilosocereus pachycladus | Cactaceae | Caatinga | 50.5 × 38.1 | 1.89 × 1.35 | 3800 | own | 7 | 6–1000 | ||
| Spondias tuberosa | Anacardiaceae | Caatinga | 45 × 38 | 28 × 20 | 1 | [15]; own | Yes? | 1 | 2 | 12–432 |
| Syagrus coronata | Arecaceae | Caatinga | 28.8 × 24.3 | 22.3 × 15.5 | 1 | own | Yes? | 362 | 3–250 | |
| Zea mays | Poaceae | Caatinga | 167.6 × 47.4 | 1.09 × 0.93 | >100 | own | 2 | 33–1000 | ||
| Hyacinth macaw | ||||||||||
| Acrocomia totai | Arecaceae | Pantanal | 27.8 × 27.8 | 16.1 × 16.1 | 1 | [41]; own | Yes? | 300 | 114 | 1–400 |
| Attalea barreirensis | Arecaceae | Cerrado | 59.1 × 40.7 | 32 × 11.5 | 1–4 | own | Yes | 352 | 3–1620 | |
| Attalea eichleri | Arecaceae | Cerrado | 60.5 × 40.5 | 37.5 × 11.5 | 1–5 | [41,43] | Yes | 409 | 1–223 | |
| Attalea phalerata | Arecaceae | Pantanal | 57.5 × 40 | 45 × 25 | 2–4 | [41,43] | Yes | 132 | 3 | 4–1011 |
| Mauritia flexuosa | Arecaceae | Cerrado | 57.5 × 57.5 | 35 × 25 | 1 | [15] | Yes | 11 | 40–234 | |
| Spondias mombin | Anacardiaceae | Pantanal | 30 × 22.5 | 25.5 × 15.5 | 4–5 | own | 1 | 2 | 1–210 | |
| Vitex cymosa | Lamiaceae | Pantanal | 29 × 25 | 17 × 10 | 1 | [44] | 1 | 40 |
| Plant | Nt | Ne | Mean | SE | Median | 95% CI | UL | |
|---|---|---|---|---|---|---|---|---|
| Hyacinth macaw primary | all dispersions | 1074 | 553 | 195.3 | 32.4 | 40 | 39–70 | 1620 |
| Acrocomia totai | 300 | 28 | 162.4 | 19.7 | 150 | 85–161 | 400 | |
| Attalea barreirensis | 244 | 116 | 218.9 | 46.2 | 115 | 115–115 | 1620 | |
| Attalea eichleri | 385 | 368 | 17.1 | 1.6 | 9 | 9–10 | 223 | |
| Attalea phalerata | 132 | 40 | 453.6 | 59.4 | 90 | 70–NA | 1011 | |
| Mauritia flexuosa | 11 | 1 | 216.4 | 16.8 | NA | NA–NA | 234 | |
| Hyacinth macaw tertiary | all dispersions | 119 | 119 | 55.2 | 4.33 | 35 | 32–80 | 220 |
| Acrocomia totai | 114 | 114 | 56.5 | 4.4 | 35 | 32–80 | 220 | |
| Lear’s macaw primary | all dispersions | 397 | 7 | 874.5 | 97.5 | NA | NA–NA | 1000 |
| Syagrus coronata | 362 | 2 | 248.6 | 10.1 | NA | NA–NA | 250 |
| Species. | Biome | % Perching Sites | N | % Undamaged Nuts | N |
|---|---|---|---|---|---|
| Lear’s macaw | |||||
| Syagrus coronatus | Caatinga | 57.4 | 47 | 13.4 | 231 |
| Hyacinth macaw | |||||
| Acrocomia totai | Pantanal | 30.8 | 13 | 18.6 | 220 |
| Attalea barreirensis | Cerrado | 57.7 | 26 | 15.6 | 224 |
| Attalea eichleri | Cerrado | 73.2 | 71 | 10.9 | 357 |
| Attalea phalerata | Pantanal | 55 | 20 | 44 | 75 |
| Mauritia flexuosa | Cerrado | 100 | 4 | 75 | 8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tella, J.L.; Hiraldo, F.; Pacífico, E.; Díaz-Luque, J.A.; Dénes, F.V.; Fontoura, F.M.; Guedes, N.; Blanco, G. Conserving the Diversity of Ecological Interactions: The Role of Two Threatened Macaw Species as Legitimate Dispersers of “Megafaunal” Fruits. Diversity 2020, 12, 45. https://doi.org/10.3390/d12020045
Tella JL, Hiraldo F, Pacífico E, Díaz-Luque JA, Dénes FV, Fontoura FM, Guedes N, Blanco G. Conserving the Diversity of Ecological Interactions: The Role of Two Threatened Macaw Species as Legitimate Dispersers of “Megafaunal” Fruits. Diversity. 2020; 12(2):45. https://doi.org/10.3390/d12020045
Chicago/Turabian StyleTella, José L., Fernando Hiraldo, Erica Pacífico, José A. Díaz-Luque, Francisco V. Dénes, Fernanda M. Fontoura, Neiva Guedes, and Guillermo Blanco. 2020. "Conserving the Diversity of Ecological Interactions: The Role of Two Threatened Macaw Species as Legitimate Dispersers of “Megafaunal” Fruits" Diversity 12, no. 2: 45. https://doi.org/10.3390/d12020045
APA StyleTella, J. L., Hiraldo, F., Pacífico, E., Díaz-Luque, J. A., Dénes, F. V., Fontoura, F. M., Guedes, N., & Blanco, G. (2020). Conserving the Diversity of Ecological Interactions: The Role of Two Threatened Macaw Species as Legitimate Dispersers of “Megafaunal” Fruits. Diversity, 12(2), 45. https://doi.org/10.3390/d12020045








