Seaweed Biodiversity of India: Reviewing Current Knowledge to Identify Gaps, Challenges, and Opportunities
Abstract
1. Introduction
2. Current Knowledge on Seaweeds of India
2.1. History of Seaweed Research in India
2.2. Estimates of the Conservation Status of Indian Seaweeds
2.3. Estimates of Seaweed Biomass in Indian Waters
3. Seaweed Biodiversity Research Gaps and Challenges
3.1. Filling Geographic Gaps to Comprehensively Document Species Diversity and Biomass
3.2. Improving Data Access to the Scientific Community
3.3. Expertise Short-Cut and the Need for Capacity-Building and Nationwide Expertise
3.4. Developing Integrated Taxonomy
4. Opportunities to Develop Seaweed Cultivation and Sustainable Exploitation in India
4.1. Cultivation of Ulva flexuosa Wulfen and U. lactuca Linnaeus
4.2. Cultivation of Hydropuntia edulis (as Gracilaria edulis)
4.3. Cultivation of Gelidiella acerosa (Forsskål) G. Feldmann et Hamel
4.4. Cultivation of Hypnea musciformis (Wulfen) Lamouroux
4.5. Issue related to Kappaphycus cultivation in India
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barbier, M.; Charrier, B.; Araujo, R.; Holdt, S.L.; Jacquemin, B.; Rebours, C. Pegasus-Phycomorph European Guidelines for a Sustainable Aquaculture of Seaweeds, COST Action FA1406; Barbier, M., Charrier, B., Eds.; Station Biologique de Roscoff: Roscoff, France, 2019. [Google Scholar]
- Tiwari, B.K.; Troy, D.J. World seaweed utilization. In Seaweed Sustainability Food and Non-Food Applications; Tiwari, B.K., Troy, D.J., Eds.; Academic Press: New York, NY, USA, 2015; pp. 7–25. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture-Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018. [Google Scholar]
- Convention on Biological Diversity. Article 2. Use of Terms; United Nations; Convention on Biological Diversity: Montreal, QC, Canada, 1992. [Google Scholar]
- Mora, C.; Tittensor, D.P.; Adl, S.; Simpson, A.G.B.; Worm, B. How Many Species Are There on Earth and in the Ocean? PLoS Biol. 2011, 9, e1001127. [Google Scholar] [CrossRef] [PubMed]
- Roskov, Y.; Ower, G.; Orrell, T.; Nicolson, D.; Bailly, N.; Kirk, P.M.; Bourgoin, T.; DeWalt, R.E.; Decock, W.; van Nieukerken, E.; et al. (Eds.) Species 2000 & ITIS Catalogue of Life, 2019 Annual Checklist. Species 2000: Naturalis, Leiden, The Netherlands. ISSN 2405-884X. Available online: www.catalogueoflife.org/annual-checklist/2019 (accessed on 9 December 2019).
- Appeltans, W.; Bouchet, P.; Boxshall, G.A.; Fauchald, K.; Gordon, D.P.; Hoeksema, B.W.; Poore, G.C.B.; van Soest, R.W.M.; Stöhr, S.; Walter, T.C.; et al. World Register of Marine Species (WoRMS). Available online: http://www.marinespecies.org (accessed on 12 July 2011).
- Maddison, D.R.; Guralnick, R.; Hill, A.; Reysenbach, A.L.; McDade, L.A. Ramping up biodiversity discovery via online quantum contributions. Trends Ecol. Evol. 2012, 27, 72–77. [Google Scholar] [CrossRef] [PubMed]
- UNEP-WCMC; I.UCN; N.GS. Protected Planet Report 2018; UNEP-WCMC, IUCN and NGS: Cambridge, UK; Gland, Switzerland; Washington, DC, USA, 2018. [Google Scholar]
- Sixth National Report for the Convention of Biological Diversity. Available online: https://chm.cbd.int/database/record?documentID=241351 (accessed on 25 July 2019).
- Chitale, V.S.; Behera, M.D.; Roy, P.S. Future of Endemic Flora of Biodiversity Hotspots in India. PLoS ONE 2014, 9, e115264. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, D.; Sahu, N.; Sahoo, D. Seaweeds of Indian Coast; A. P.H. Publication: New Delhi, India, 2001; p. 283. [Google Scholar]
- Guiry, M.D. How many species of algae are there? J. Phycol. 2012, 48, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- Oza, R.M.; Zaidi, S.H. A Revised Checklist of Indian Marine Algae; Central Salt and Marine Chemicals Research Institute: Bhavnagar, India, 2001; p. 296. [Google Scholar]
- Kaliaperumal, N. Studies on phycocolloids from Indian marine algae – A review. Seaweed Res. Utilin. 2017, 39, 1–8. [Google Scholar]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. World-Wide Electronic Publication; National University of Ireland: Galway, Ireland, 2008; Available online: http://www.algaebase.org (accessed on 26 July 2019).
- Linnaeus, C. Species plantarum, exhibentes plantas rite cognitas, ad genera relatas, cum differentiis specificis, nominibus trivialibus, synonymis selectis, locis natalibus, secundum systema sexuale digestas. Nauk 1753, 2, 561–1200. [Google Scholar]
- Iyengar, M.O.P. Krusadai Island Flora. Bull. Madras Gov. Mus. New Ser. Nat. Hist. 1927, 1, 185–188. [Google Scholar]
- Børgesen, F. Some Indian green and brown algae, especially from the shores of the presidency of Bombay–III. J. Indian Bot. Soc. 1933, 12, 1–16. [Google Scholar]
- Børgesen, F. Some Indian Rhodophyceae, especially from the presidency of Bombay–III. Bull. Misc. Inform. Kew 1933, 3, 113–142. [Google Scholar] [CrossRef]
- Børgesen, F. Some Indian Rhodophyceae, especially from the shores of the presidency of Bombay. IV. Bull. Misc. Inform. Kew. 1934, 4, 1–30. [Google Scholar] [CrossRef]
- Børgesen, F. Some marine algae from the northern part of the Arabian Sea with remarks on their geographical distribution. Dansk. Vidensk. Selskab. Biol. Med. 1934, 11, 72. [Google Scholar]
- Børgesen, F. List of marine algae from Bombay. Dansk. Vidensk. Selskab. Biol. Med. 1935, 12, 64. [Google Scholar]
- Børgesen, F. Contribution to a south Indian marine algal flora-II. J. Indian Bot. Soc. 1937, 16, 311–357. [Google Scholar]
- Børgesen, F. Contribution to a south Indian marine algal flora–III. J. Indian Bot. Soc. 1938, 17, 205–242. [Google Scholar]
- Børgesen, F. Two species of Scinaia from south-India. Bot. Not. 1938, 18, 183–189. [Google Scholar]
- Dixit, S.C. Species list of Indian marine algae–II. J Univ. Bombay 1968, 36, 9–24. [Google Scholar]
- Krishnamurthy, V.K.; Joshi, H.V. A Check-List of Indian Marine Algae; Central Salt and Marine Chemicals Research Institute: Bhavnagar, India, 1970; p. 36. [Google Scholar]
- Untawale, A.G.; Dhargalkar, V.K.; Agadi, V.V. List of Marine Algae from India; Tech. Report National Institute of Oceanography: Goa, India, 1983; p. 42. [Google Scholar]
- Rao, P.S.N.; Gupta, R.K. Algae of India, A Checklist of Indian Marine Algae (Excluding Diatoms & Dinoflagellates); Botanical Survey of India: Kolkata, India, 2015; p. 93.
- Ganesan, M.; Trivedi, N.; Gupta, V. Seaweed resources in India – current status of diversity and cultivation: Prospects and challenges. Bot. Mar. 2019, 62, 463–482. [Google Scholar] [CrossRef]
- Jha, B.; Reddy, C.R.K.; Thakur, M.C.; Rao, M.U. Seaweeds of India. The Diversity and Distribution of Seaweeds of Gujarat Coast; Springer: Dordrecht, The Netherland, 2009; p. 215. [Google Scholar]
- Nayar, M.P.; Sastry, A.R.K. Red Data Book of Indian Plants; Botanical Survey of India: Calcutta, India, 1990.
- Jadeja, R.N.; Tewari, A. Effect of soda ash industry effluent on agarophytes, alginophytes and carrageenophyte of west coast of India. J. Hazard Mater. 2009, 162, 498–502. [Google Scholar] [CrossRef]
- Jadeja, R.N.; Tewari, A. Impact of discharge of soda ash industry effluent on abundance and community structure of rocky intertidal macroalgae of the Arabian Sea, Gujarat, India. Indian J. Mar. Sci. 2011, 40, 71–75. [Google Scholar]
- Børgesen, F. Some marine algae from Ceylon. Ceylon J. Sci. A Bot. 1936, 12, 57–96. [Google Scholar]
- Rao, M.U.; Chaugule, B.B. Endangered and extinct seaweeds of Indian shore. In Recent Advances on Applied Aspects of Indian Marine Algae with Reference to Global Scenario; Tewari, A., Ed.; Central Salt and Marine Chemicals Research Institute: Bhavnagar, India, 2006; pp. 141–146. [Google Scholar]
- Hornell, J. Report on the Further Development of Fishery Resources of Baroda State, Baroda State, Baroda. 1918.
- Chauhan, V.D.; Krishnamurthi, V. An estimate of algin-bearing seaweeds in the Gulf of Kutch. Curr. Sci. 1968, 37, 648. [Google Scholar]
- Thivy, F. Seaweed utilization in India. In Proceedings of the Symposium on Algology, India; Kachiroo, P., Ed.; ICAR: New Delhi, India, 1960; pp. 345–365. [Google Scholar]
- Subba Rao, P.V.; Mantri, V.A. Indian seaweed resources and sustainable utilization: Scenario at the dawn of new century. Curr. Sci. 2006, 91, 164–174. [Google Scholar]
- Krishnamurthy, V. Seaweed drift on the Indian coast. Proceedings of the Symposium on Indian Ocean. India Nat. Inst. Sci. Indian 1969, 38, 657–666. [Google Scholar]
- Thakur, M.C.; Reddy, C.R.K.; Jha, B. Seasonal variation in biomass and species composition of seaweeds stranded along Port Okha, northwest coast of India. J. Earth Syst. Sci. 2008, 117, 211–218. [Google Scholar] [CrossRef]
- Untawale, A.G.; Reddy, C.R.K.; Ambiye, V. Marine algal flora of submerged Angria Bank (Arabian Sea). Indian J. Mar. Sci 1989, 18, 207–209. [Google Scholar]
- Ambiye, V.; Untawale, A.G. Deep water marine algal flora of the submerged banks of west coast of India. In Oceanography of the Indian Ocean; Desai, B.N., Ed.; National Institute of Oceanography: Goa, India, 1992; pp. 247–252. [Google Scholar]
- Bhanderi, P.P.; Trivedi, Y.A. Seaweed resources of Hanumandandi reef and Vumani reef near Okha port, Gujarat. Indian J. Mar. Sci. 1975, 4, 97–99. [Google Scholar]
- Muthuvelan, B.; Chennubhotla, V.S.K.; Nair, K.V.K.; Sampath, V.; Ravindran, M. Standing crop biomass and comparative distribution of agarophytes, alginophytes and other algae in South Andaman. Indian Hydrobiol. 2001, 4, 130–138. [Google Scholar]
- Mukhopadhyay, A.; Pal, R. A report on biodiversity of algae from coastal West Bengal (South & North 24-parganas) and their cultural behavior in relation to mass cultivation programme. Indian Hydrobiol. 2002, 5, 97–107. [Google Scholar]
- Sahoo, D.; Sahu, N.; Sahoo, D. A Critical Survey of Seaweed Diversity of Chilika Lake, India. Algae 2003, 8, 1–13. [Google Scholar] [CrossRef]
- Mantri, V.A.; Subba Rao, P.V. Diu island: A paradise for tourists and seaweed biologists. Curr. Sci. 2005, 89, 1795–1797. [Google Scholar]
- Sonali, S.P. Marine Algal Flora of Maharashtra Coast, India. Botanical Survey of India, Western Regional Center; University of Pune: Pune, India, 2010. [Google Scholar]
- Kaladharan, P.; Zacharia, P.U.; Vijayakumaran, K. Coastal and marine floral biodiversity along the Karnataka coast. J. Mar. Biol. Ass. India 2011, 53, 121–129. [Google Scholar]
- Kamboj, R.D.; Das, L.; Palanisamy, M. Pictoral Guide to Seaweeds of Gulf of Kachh, Gujarat; Gujarat Ecological Education and Research Foundation: Gandhinagar, India, 2019; p. 337. [Google Scholar]
- Mantri, V.A.; Joshi, H.V.; Zaidi, S.H.; Jha, B. Seaweeds of Samiani Island. Seaweed Res. Utilin. 2003, 25, 143–146. [Google Scholar]
- Mantri, V.A.; Joshi, H.V.; Zaidi, S.H. Seaweeds of Samiani Island II Family: Caulerpaceae (Chlorophyta). Seaweed Res. Utilin. 2004, 26, 231–236. [Google Scholar]
- Krishnamurthy, V.; Baluswami, M. The species of Porphyra from the Indian region. Seaweed Res. Utilin. 1984, 7, 31–37. [Google Scholar]
- Census of Marine Life, Mapping & Visualization. Available online: http://comlmaps.org/ (accessed on 26 July 2019).
- Ocean Biogeographic Information System (OBIS). Available online: https://obis.org/about/ (accessed on 26 July 2019).
- Chavan, V.; Achuthankutty, C.T.; Berghe, E.V.; Wafer, M. IndoBIS, an Ocean Biogeographic Information System for assessment and conservation of Indian Ocean biodiversity. Indian J. Mar. Sci. 2005, 34, 120–127. [Google Scholar]
- Kakodkar, A.P.; Alornekar, A.; Dsouza, R.; Thomas, T.; Divekar, R.; Nath, I.; Kavlekar, D.P.; Ingole, B.S.; Bharathi, P.S. BioSearch: A glimpse into marine biodiversity of Indian coastal waters. Indian J. Mar. Sci. 2013, 42, 745–748. [Google Scholar]
- Chavan, V.; Krishnan, S. Natural history collections: A call for national information infrastructure. Curr. Sci. 2003, 84, 34–42. [Google Scholar]
- Bhushan, S.; Mehra, R.; Rani, P.; Bast, F. DbIndAlgae: An on-line resource of marine algae from India identified on the basis of molecular and morphological features. Curr. Sci. 2016, 111, 723–726. [Google Scholar] [CrossRef]
- Misra, J.N. Phaeophyceae in India; Volume 7 of I.C.A.R. monographs on algae; Indian Council of Agricultural Research: New Delhi, India, 1966; p. 203. [Google Scholar]
- Desikachari, T.V.; Krishnamurthy, V.; Balkrishnan, M.S. Rhodophyta-II; Taxonomy Part-II A; Madras Science Foundation Publication: Chennai, India, 1990; p. 279. [Google Scholar]
- Desikachari, T.V.; Krishnamurthy, V.; Balkrishnan, M.S. Rhodophyta-II; Taxonomy continued. Part-II B; Madras Science Foundation Publication: Chennai, India, 1998; p. 359. [Google Scholar]
- Krishnamurthy, V. Algae of India and Neighboring Countries I: Chlorophycota; Oxford and IBH publication Co Pvt Ltd.: New Delhi, India, 2000; p. 210. [Google Scholar]
- Tewari, A. (Ed.) Recent Advances on Applied Aspects of Indian Marine Algae with Reference to Global Scenario; Central Salt and Marine Chemicals Research Institute Publication: Bhavnagar, India, 2006; p. 385. [Google Scholar]
- Tewari, A. (Ed.) Recent Advances on Applied Aspects of Indian Marine Algae with Reference to Global Scenario; Central Salt and Marine Chemicals Research Institute Publication: Bhavnagar, India, 2006; p. 300. [Google Scholar]
- Dixon, P.S. The Rhodophyta: Some aspects of their biology. II. Oceanogr. Mar. Biol. Annu. Rev. 1970, 8, 307–352. [Google Scholar]
- Leliaert, F.; Verbruggen, H.; Vanormelingen, P.; Steen, F.; López-Bautista, J.M.; Zuccarello, G.C.; De Clerck, O. DNA-based species delimitation in algae. Eur. J. Phycol. 2014, 49, 179–196. [Google Scholar] [CrossRef]
- Joshi, H.V.; Krishnamurthy, V. The species of Enteromorpha from India. Bot. J. Linn. Soc. 1972, 65, 119–128. [Google Scholar] [CrossRef]
- Duraiswamy, R. Studies on Caulerpa Lamouroux from India, I. General. Seaweed Res. Utiln. 1988, 11, 83–106. [Google Scholar]
- Duraiswamy, R. Studies on Caulerpa Lamouroux from India II. Seaweed Res. Utiln. 1990, 12, 55–86. [Google Scholar]
- Duraiswamy, R. Studies on Caulerpa Lamouroux from India III. Seaweed Res. Utiln. 1990, 13, 29–76. [Google Scholar]
- Srinivasan, K.S. Conspectus of Sargassum species from Indian territorial waters. Phykos 1967, 5, 127–159. [Google Scholar]
- Umamaheswara Rao, M. On the Gracilariaceae of the seas around India. J. Mar. Biol. Assoc. India 1974, 14, 671–696. [Google Scholar]
- Parekh, M.; Mishra, A.; Jha, B. Molecular phylogeny of Gracilaria species inferred from molecular markers belonging to three different genomes. J. Phycol. 2010, 46, 1322–1328. [Google Scholar] [CrossRef]
- Kavale, M.G.; Kazi, M.A.; Sreenadhan, N. Pyropia acanthophora var. robusta var. nov. (Bangiales, Rhodophyta) from Goa, India. Indian J. Mar. Sci. 2015, 44, 866–873. [Google Scholar]
- Kavale, M.G.; Kazi, M.A.; Sreenadhan, N.; Singh, V.V. Morphological, ecological and molecular characterization of Pyropia vietnamensis (Bangiales, Rhodophyta) from the Konkan region, India. Phytotaxa 2015, 224, 45–58. [Google Scholar] [CrossRef]
- Kazi, M.A.; Reddy, C.R.K.; Jha, B. Molecular Phylogeny and Barcoding of Caulerpa (Bryopsidales) Based on the tufA, rbcL, 18S rDNA and ITS rDNA Genes. PLoS ONE 2013, 8, e82438. [Google Scholar] [CrossRef]
- Kazi, M.A. Molecular Characterization of Caulerpa (Bryopsidales) from Indian Waters; Academy of Scientific & Innovative Research (AcSIR): Gaziabad, India, 2014. [Google Scholar]
- Kazi, M.A.; Kavale, M.G.; Singh, V.V. Morphological and molecular characterization of Ulva chaugulii sp. nov., U. lactuca and U. ohnoi (Ulvophyceae, Chlorophyta) from India. Phycologia 2016, 55, 45–54. [Google Scholar] [CrossRef]
- Bast, F.; John, A.A.; Bhushan, S. Strong endemism of bloom-forming tubular Ulva in Indian west coast, with description of Ulva paschima Sp. Nov. (Ulvales, Chlorophyta). PLoS ONE 2014, 9, e109295. [Google Scholar] [CrossRef] [PubMed]
- Bast, F.; Bhushan, S.; John, A.A. DNA Barcoding of a new record of epi-endophytic green algae Ulvella leptochaete (Ulvellaceae, Chlorophyta) in India. J. Biosci. 2014, 39, 711–716. [Google Scholar] [CrossRef] [PubMed]
- O’kelly, C.J.; Kurihara, A.; Shipley, T.C.; Sherwood, A.R. Molecular assessment of Ulva spp. (Ulvophyceae, Chlorophyta) in the Hawaiian Islands. J. Phycol. 2010, 46, 728–735. [Google Scholar] [CrossRef]
- Ganesan, M.; Reddy, C.R.K.; Jha, B. Impact of cultivation on growth rate and agar content of Gelidiella acerosa (Gelidiales, Rhodophyta). Algal Res. 2015, 12, 398–404. [Google Scholar] [CrossRef]
- Meena, R.; Siddhanta, A.K.; Prasad, K.; Ramavat, B.K.; Eswaran, K.; Thiruppathi, S.; Ganesan, M.; Mantri, V.A.; Subba Rao, P.V. Preparation, characterization and benchmarking of agarose from Gracilaria dura of Indian waters. Carbohydr. Polym. 2007, 69, 179–188. [Google Scholar] [CrossRef]
- Siddhanta, A.K.; Meena, R.; Prasad, K.; Ramavat, B.K.; Ghosh, P.K.; Eswaran, K.; Thiruppathi, S.; Mantri, V.A. A cost-effective process for preparing agarose from Gracilaria spp. US Patent-US 2005/0267296 A1, 15 December 2005. [Google Scholar]
- Mantri, V.A.; Thakur, M.C.; Gupta, M.K.; Reddy, C.R.K.; Jha, B. The carpospore culture of industrially important red alga Gracilaria dura (Gracilariales, Rhodophyta). Aquaculture 2009, 297, 85–90. [Google Scholar] [CrossRef]
- Gupta, V.; Baghel, R.S.; Kumar, M.; Kumari, P.; Mantri, V.A.; Reddy, C.R.K.; Jha, B. Growth and agarose characteristics of isomorphic gametophyte (male and female) and sporophyte of Gracilaria dura and their marker assisted selection. Aquaculture 2011, 318, 389–396. [Google Scholar] [CrossRef]
- Mairh, O.P.; Parekh, G.; Chauhan, V.D.; Rao, P.S.; Mehta, D.J. Ecology, culture and chemical constituents of Enteromorpha from Gujarat coast. Seaweed Res. Utiln. 1983, 6, 19–22. [Google Scholar]
- Mairh, O.P.; Pandey, R.S.; Tewari, A. Culture of Enteromorpha flexuosa (Wulf) J. Ag. (Chlorophyceae) in outdoor pool. Indian J. Mar. Sci. 1986, 15, 212–218. [Google Scholar]
- Oza, R.M.; Joshi, H.V.; Mairh, O.P.; Tewari, A. Swarmer production and cultivation of Ulva fasciata Delile in intertidal region at Okha, west coast of India. Indian J. Mar. Sci. 1985, 14, 217–219. [Google Scholar]
- Oza, R.M.; Krishnakumar, G.R.; Mairh, O.P.; Zaidi, S.H. Cultivation of Ulva fasciata Delili on the coast of Diu, west cost of India. Seaweed Res. Utilin. 2001, 23, 5–12. [Google Scholar]
- Mantri, V.A.; Singh, R.P.; Bijo, A.J.; Kumari, P.; Reddy, C.R.K.; Jha, B. Differential response of varying salinity and temperature on zoospore induction, regeneration and daily growth rate in Ulva fasciata (Chlorophyta, Ulvales). J. Appl. Phycol. 2011, 23, 243–250. [Google Scholar] [CrossRef]
- Ganesan, M.; Sahoo, N.; Eswaran, K. Raft culture of Gracilaria edulis in open sea along the south-eastern coast of India. Aquaculture 2011, 321, 145–151. [Google Scholar] [CrossRef]
- Raju, P.V.; Thomas, P.C. Experimental field cultivation of Gracilaria edulis (Gmel.) Silva. Bot. Mar. 1971, 14, 71–75. [Google Scholar] [CrossRef]
- Subbaramaiah, K.; Thomas, P.C. Raft cultivation of Gracilaria edulis (Gmel.) Silva. Proc. Ind. Acad. Sci. (Plant. Sci.) 1990, 100, 123–127. [Google Scholar]
- Patel, J.B.; Gopal, B.V.; Nagulan, V.R.; Subbaramaiah, K.; Thomas, P.C. Experimental field cultivation of Gelidiella acerosa at Ervadi, India. In Proceedings of the Int. Symp. Marine Algae of the Indian Ocean Region, CSMCRI, Bhavnagar, India, 9–12 January 1979; pp. 24–25. [Google Scholar]
- Subba Rao, P.V.; Eswaran, K.; Ganesan, M. Cultivation of agarophytes in India: Retrospect, prospect and future strategies. Seaweed Res. Utiln. 2006, 26, 181–184. [Google Scholar]
- Ganesan, M.; Thiruppathi, S.; Eswaran, K.; Reddy, C.R.K.; Jha, B. Development of an improved method of cultivation to obtain high biomass of red alga of Gelidiella acerosa (Gelidiales, Rhodophyta) in the open sea. Biomass Bioener. 2011, 35, 2729–2736. [Google Scholar] [CrossRef]
- Ganesan, M.; Thiruppathi, S.; Jha, B. Mariculture of Hypnea musciformis (Wulfen) Lamouroux in South east coast of India. Aquaculture 2006, 256, 201–211. [Google Scholar] [CrossRef]
- Cai, J.; Hishamunda, N.; Ridler, N. Social and economic dimensions of carrageenan seaweed farming: A global synthesis. In Social and Economic Dimensions of Carrageenan Seaweed Farming; Valderrama, D., Cai, J., Hishamunda, N., Ridler, N., Eds.; FAO: Rome, Italy, 2013; pp. 5–59. [Google Scholar]
- Mairh, O.P.; Tewari, A. Studies on a new asexual propagules of Kappaphycus striatum (Solieriaceae, Rhodophyta). Phycologia 1994, 33, 62–64. [Google Scholar] [CrossRef]
- Mairh, O.P.; Zodape, S.T.; Tewari, A.; Rajyaguru, M.R. Culture of marine red algae Kappaphycus striatum (Schmitz) Doty on the Saurashtra region, west coast of India. Indian J. Mar. Sci. 1995, 24, 24–31. [Google Scholar]
- Mairh, O.P.; Zodape, S.T.; Tewari, A.; Mishra, J.P. Effect of nitrogen sources on the growth and bioaccumulation of nitrogen in marine red algae Kappaphycus striatum (Rhodophyta, Solieriaceae) in culture. Indian J. Mar. Sci. 1999, 28, 55–59. [Google Scholar]
- Eswaran, K.; Ghosh, P.K.; Mairh, O.P. Experimental field cultivation of Kappaphycus alvarezii (Doty) Doty ex Silva at Mandapam region. Seaweed Res. Utiln. 2002, 24, 67–72. [Google Scholar]
- Reddy, C.R.K.; Mairh, O.P.; Krishna Kumar, G.R.; Eswaran, K.; Subba Rao, P.V.; Mody, K.H.; Ghosh, P.K. An improved process for cultivation of algae. US Patent no. WO2002017707A1, 7 March 2002. [Google Scholar]
- Rao, P.S.N.; Rao, M.U. On a species of Kappaphycus (Solieriaceae, Gigartinales) form Andaman and Nicobar Island, India. Phykos 1999, 38, 93–96. [Google Scholar]
- Mantri, V.A.; Eswaran, K.; Shanmugam, M.; Ganesan, M.; Veeragurunathan, V.; Thiruppathi, S.; Reddy, C.R.K.; Seth, A. An appraisal on commercial farming of Kappaphycus alvarezii in India: Success in diversification of livelihood and prospects. J. Appl. Phycol. 2017, 29, 335–357. [Google Scholar] [CrossRef]
- Aquaculture Foundation of India. Final Report of the DBT Project: Seaweed Farming to Rehabilitate Tsunami Affected Coastal Communities in Tamil Nadu; Department of Biotechnology, Ministry of Science and Technology: New Delhi, India, 2008; p. 41. [Google Scholar]
- Eswaran, K.; Ghosh, P.K.; Siddhanta, A.K.; Patolia, J.S.; Periyasami, C.; Mehta, A.S.; Mody, K.H.; Ramavat, B.K.; Prasad, K.; Rajyaguru, M.R.; et al. Integrated Method for Production of Carrageenan and Liquid Fertilizer from Fresh Sea Weeds. US Patent 6983479B2, 19 February 2004. [Google Scholar]
- Ghosh, P.K.; Mody, K.H.; Reddy, M.P.; Patolia, J.S.; Eswaran, K.; Shah, R.; Barot, B.; Gandhi, M.R.; Mehta, A.S.; Bhatt, A.M.; et al. Low Sodium Salt of Botanic Origin. US patent US20050220975A1, 6 October 2005. [Google Scholar]
- Ghosh, P.K.; Siddhanta, A.K.; Prasad, K.; Meena, R.; Bhattacharya, A. Process of Preparation of Biodegradable Films from Semi Refined Kappa Carrageenan. US Patent US7067568B1, 27 June 2006. [Google Scholar]
- Mody, K.H.; Ghosh, P.K.; Barindra, S.; Gnanasekaran, G.; Shukla, A.D.; Eswaran, K. A Process for Integrated Production of Ethanol and Seaweed Sap from Kappaphycus Alvarezii. European Patent EP2475776 A1, 18 July 2012. [Google Scholar]
- Zemke-White, W.L. Assessment of the current knowledge on the environmental impacts of seaweed farming in the tropics. In Proceedings of the Asia-Pacific Conference on Marine Science and Technology, Kuala Lumpur, Malaysia, 12–16 May 2002; Phang, S.M., Chong, V.C., Ho, S.C., Noraieni, M., Sim, J.O.L., Eds.; pp. 465–475. [Google Scholar]
- Pereira, N.; Verlecar, X.N. Is Gulf of Mannar heading for marine bioinvasion? Curr. Sci. 2005, 89, 1309–1310. [Google Scholar]
- Chandrasekaran, S.; Nagendran, N.A.; Pandiaraja, D.; Krishnankutty, N.; Kamalakannan, B. Bioinvasion of Kappaphycus alvarezii on corals in the Gulf of Mannar, India. Curr. Sci. 2008, 94, 1167–1172. [Google Scholar]
- Bagla, P. Seaweed invader elicits angst in India. Science 2008, 320, 1271. [Google Scholar] [CrossRef]
- Mandal, S.K.; Mantri, V.A.; Haldar, S.; Eswaran, K.; Ganesan, M. Invasion potential of Kappaphycus alvarezii on corals at Kurusadai island, Gulf of Mannar, India. Algae 2010, 25, 205–216. [Google Scholar] [CrossRef]
- Mandal, S.K.; Ajay, G.; Monisha, N.; Malarvizhi, J.; Temkar, G.; Mantri, V.A. Differential response of varying temperature and salinity regimes on nutrient uptake of drifting fragments of Kappaphycus alvarezii: Implication on survival and growth. J. Appl. Phycol. 2015, 27, 1571–1581. [Google Scholar] [CrossRef]
- Ask, E.I.; Batibasaga, A.; Zertuche-González, J.A.; de San, M. Three Decades of Kappaphycus Alvarezii (Rhodophyta) Introduction to Non-Endemic Locations. 17th International Seaweed Symposium, Cape Town, South Africa, 28 January–2 February 2001; Chapman, A.R.O., Anderson, R.J., Vreeland, V.J., Davison, I.R., Eds.; Oxford University Press: New York, NY, USA, 2003; pp. 49–57. [Google Scholar]
- Ganesan, M.; Thiruppathi, S.; Sahu, N.; Rengarajan, N.; Veeragurunathan, V.; Jha, B. In situ observations on preferential grazing of seaweeds by some herbivores. Curr. Sci. 2006, 91, 1256–1260. [Google Scholar]
- Bindu, M.S.; Levine, I.A. The commercial red seaweed Kappaphycus alvarezii—an overview on farming and environment. J. Appl. Phycol. 2011, 23, 789–796. [Google Scholar] [CrossRef]

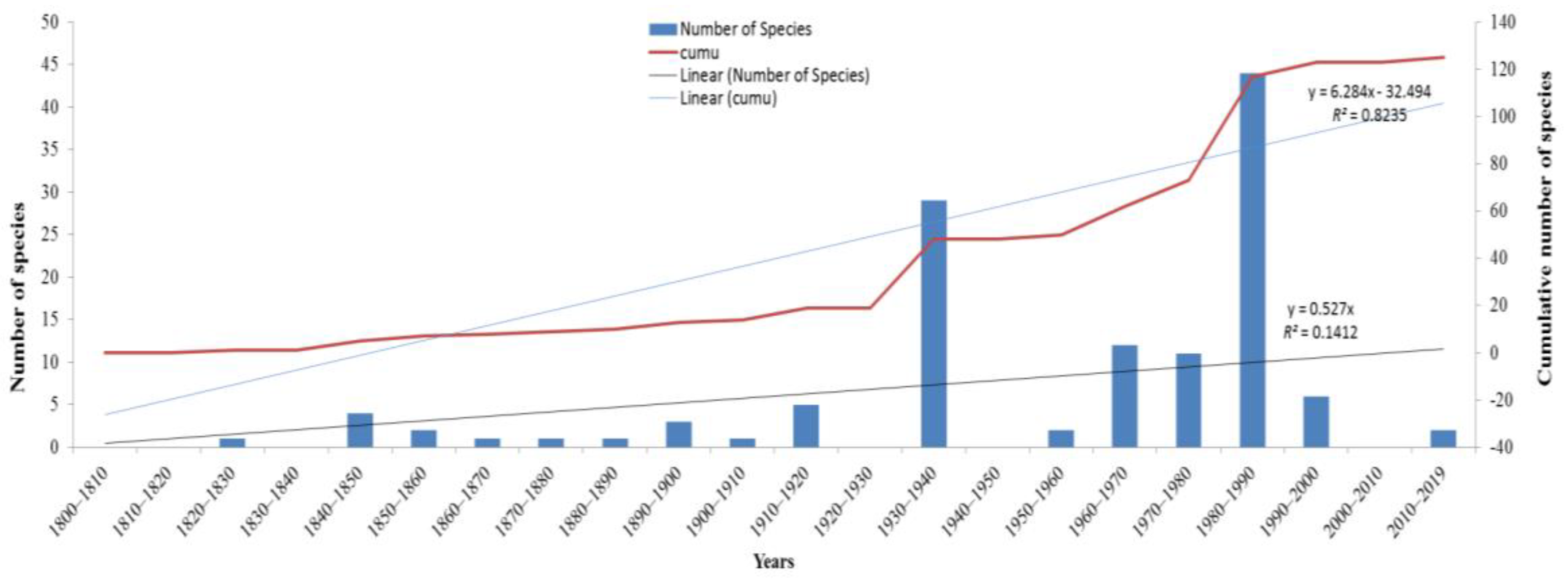
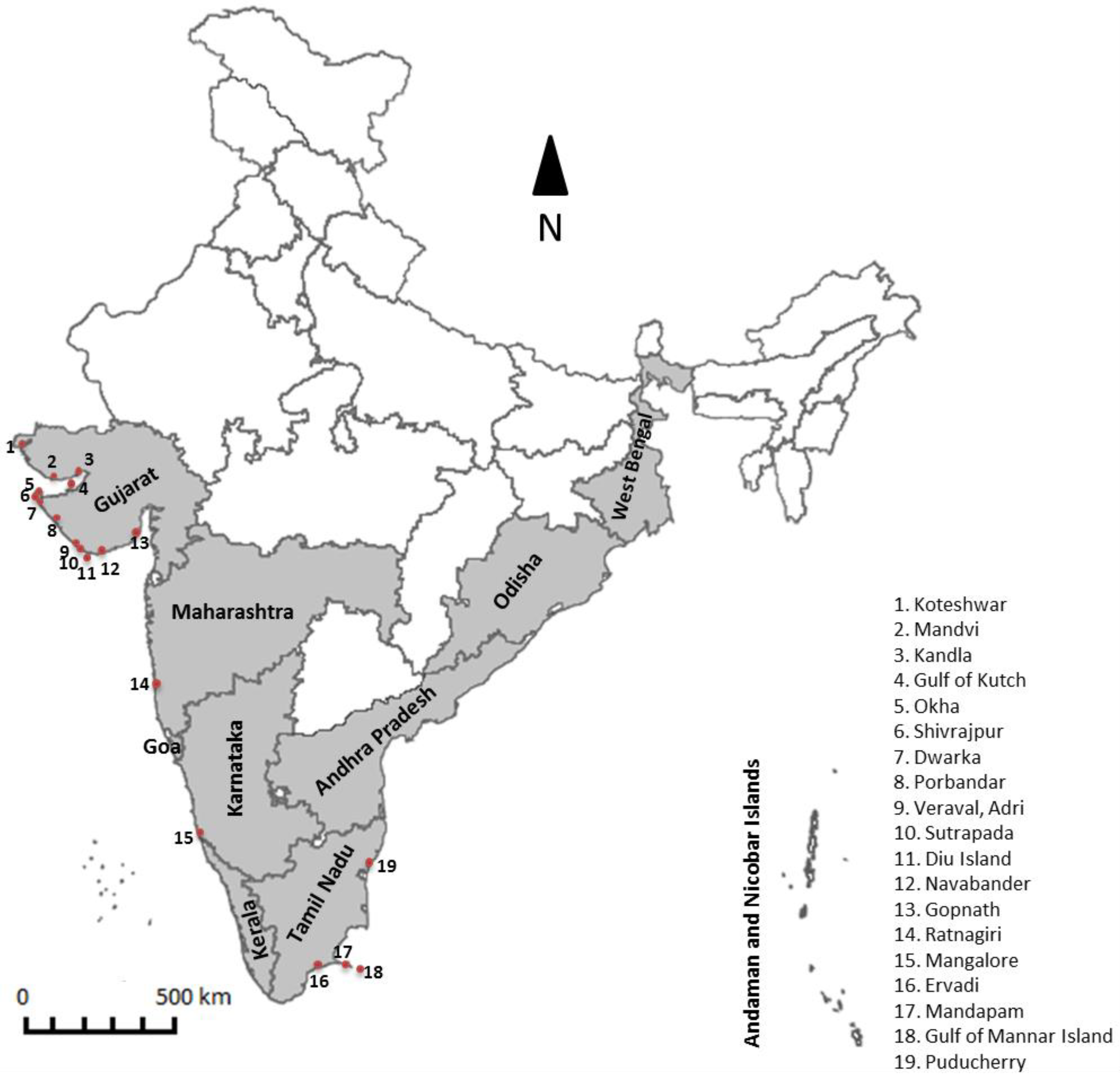


| Class | Order | Family | Genera | Species + Varieties | Total |
|---|---|---|---|---|---|
| Rhodophyta | 16 | 36 | 136 | 406 + 28 | 434 |
| Ochrophyta | 6 | 13 | 37 | 159 + 32 | 191 |
| Chlorophyta | 7 | 19 | 43 | 179 + 37 | 216 |
| Total | 29 | 68 | 216 | 746 + 97 | 841 |
| Check list | Title | Chlorophyta | Ochrophyta | Rhodophyta | Total |
|---|---|---|---|---|---|
| Krishnamurthy and Joshi [28] | Genera | 36 (21.56) | 33 (19.76) | 98 (58.68) | 167 |
| Species | 130 (24.90) | 136 (26.05) | 256 (49.04) | 522 | |
| Untawale et al. [29] | Genera | 44 (21.67) | 39 (19.21) | 120 (59.11) | 203 |
| Species | 156 (25.82) | 141 (23.34) | 307 (50.82) | 604 | |
| Oza and Zaidi [14] | Genera | 43 (19.90) | 37 (17.12) | 136 (62.96) | 216 |
| Species | 216 (25.68) | 191 (22.71) | 434 (51.60) | 841 | |
| Sahoo et al. [12] | Genera | 45 (20.36) | 38 (17.19) | 138 (62.44) | 221 |
| Species | 184 (23.89) | 166 (21.55) | 420 (54.54) | 770 | |
| Rao and Gupta [30] | Genera | 46 (19.65) | 50 (21.37) | 138 (58.97) | 234 |
| Species | 212 (24.51) | 211(24.39) | 442 (51.1) | 865 |
| Name of Marine Algae | Use | Method of Cultivation | Place of Cultivation | Year | DGR/Day (%) | Biomass Yield in g (Fresh Weight) m−2 | Reference |
|---|---|---|---|---|---|---|---|
| Ulva flexuosa | Antibacterial activity | In Plastic pools (attached to nylon threads) | Okha | 1981 | 21.6–26.5 | 996–1350 | [91] |
| Ulva lactuca | Food | Seeding of spore suspension on nets | Okha | 1983 | - | 829.9 | [93] |
| Ulva lactuca | Food, Antiviral activity | Seeding of swarmer suspension on nets | Diu | 1995–1996 | - | 183–1040 | [94] |
| Hydropuntia edulis | Food grade agar | Long-line rope method | Ervadi, southeast coast of India | 2006–2009 | 2.6–7.4 | 1500–2600 | [96] |
| Hydropuntia edulis | Food grade agar | Long-line rope method | Krusadai Island | 1967–1968 | - | 25.5–30.0 | [97] |
| Hydropuntia edulis | Food grade agar | Single rope floating raft technique | Krusadai Island | 1988–1989 | 4–5 | - | [98] |
| Gelidiella acerosa | Bacteriological grade agar | Coral stone | Ervadi | 1979 | - | 3.13 | [99] |
| Gelidiella acerosa | Bacteriological grade agar | Suspended stone method | Ervadi | 2004–2006 | 1.33–2.62 | 367 ± 45 | [101] |
| Hypnea musciformis | Carrageenan | Single Raft Floating Technique | 2001–2003 | 7.6–10.9 | 403 | [102] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantri, V.A.; Kavale, M.G.; Kazi, M.A. Seaweed Biodiversity of India: Reviewing Current Knowledge to Identify Gaps, Challenges, and Opportunities. Diversity 2020, 12, 13. https://doi.org/10.3390/d12010013
Mantri VA, Kavale MG, Kazi MA. Seaweed Biodiversity of India: Reviewing Current Knowledge to Identify Gaps, Challenges, and Opportunities. Diversity. 2020; 12(1):13. https://doi.org/10.3390/d12010013
Chicago/Turabian StyleMantri, Vaibhav A., Monica Gajanan Kavale, and Mudassar Anisoddin Kazi. 2020. "Seaweed Biodiversity of India: Reviewing Current Knowledge to Identify Gaps, Challenges, and Opportunities" Diversity 12, no. 1: 13. https://doi.org/10.3390/d12010013
APA StyleMantri, V. A., Kavale, M. G., & Kazi, M. A. (2020). Seaweed Biodiversity of India: Reviewing Current Knowledge to Identify Gaps, Challenges, and Opportunities. Diversity, 12(1), 13. https://doi.org/10.3390/d12010013




