Increased Peatland Nutrient Availability Following the Fort McMurray Horse River Wildfire
Abstract
1. Introduction
2. Materials and Methods
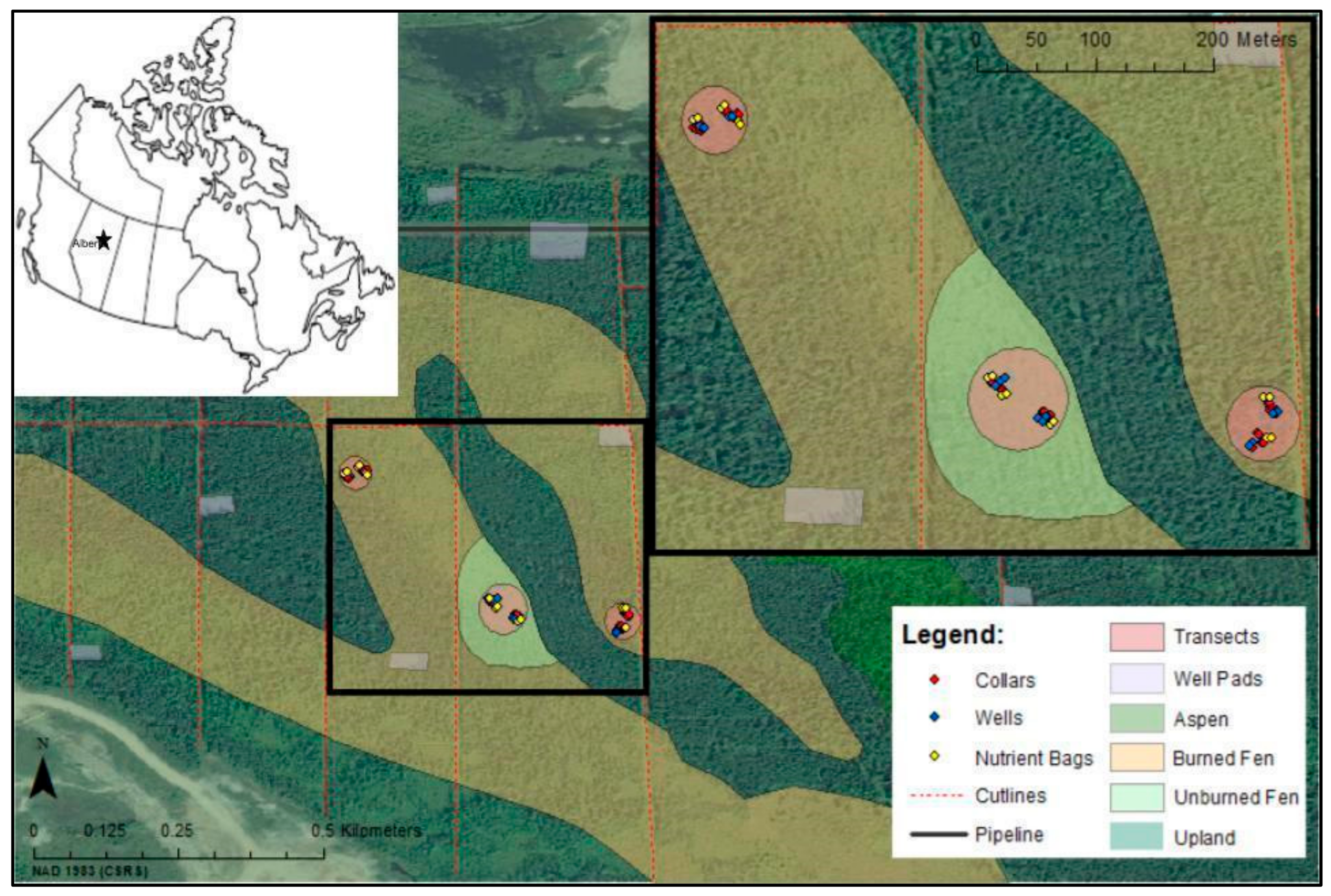
2.1. Study Area Pre-Fire
2.2. Study Area Post-Fire
2.3. Study Area Burn Severity
2.4. Soil Properties, Nutrient Dynamics and Stock Calculations
2.5. Groundwater and Shallow Leachate Dynamics
2.6. Vegetation Sampling and Nutrient Analysis
2.7. Statistical Analysis
3. Results
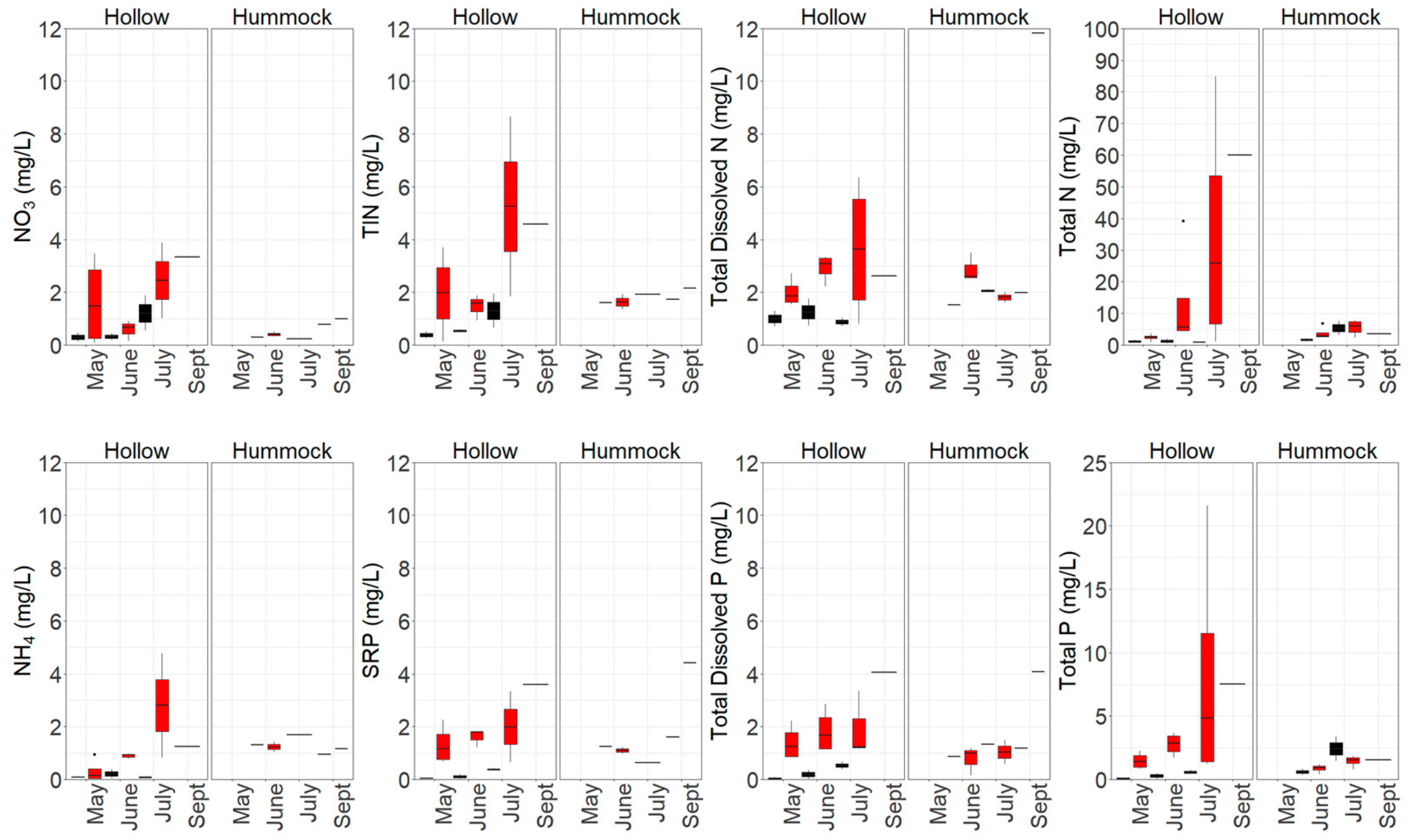
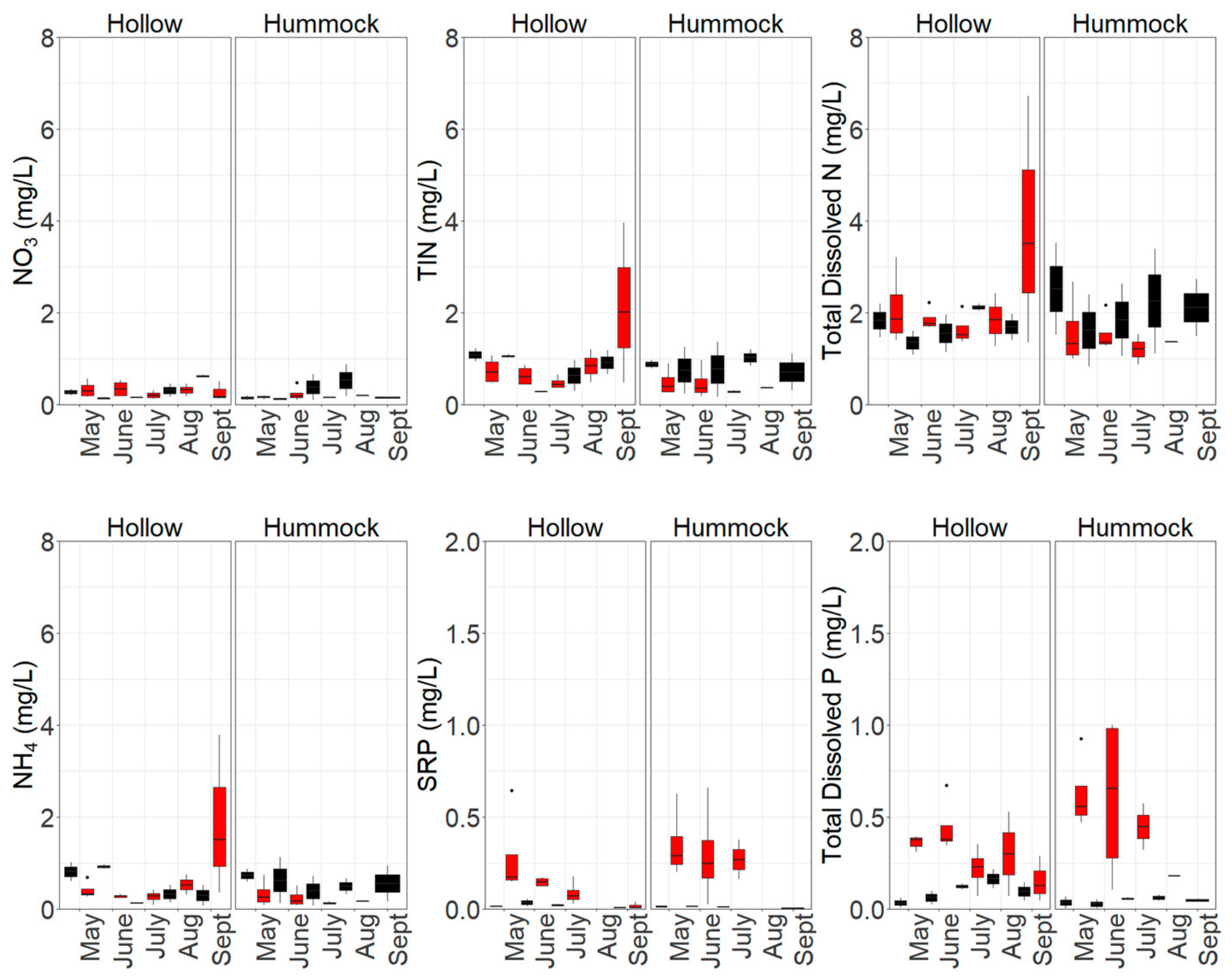
3.1. Nutrient Availability and Dynamics in Shallow Leachate, Groundwater and Peat
3.2. Carbon and Nutrients Stoichiometric Balance of Peat and Vegetation
4. Discussion
4.1. Increased Nutrient Availability Following Burning
4.2. Wildfire Disturbance on the Nutrient Stoichiometry Balance
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bridgham, S.D.; Megonigal, J.P.; Jason KKeller, J.K.; Bliss, N.B.; Trettin, C. The carbon balance of North American wetlands. Wetlands 2006, 26, 889–916. [Google Scholar] [CrossRef]
- Vitt, D.H.; Halsey, L.A.; Bauer, I.E.; Campbell, C. Spatial and temporal trends in carbon storage of peatlands of continental western Canada through the Holocene. Can. J. Eartth Sci. 2000, 37, 683–693. [Google Scholar] [CrossRef]
- Vitt, D.H.; Wieder, R.K.; Scott, K.D.; Faller, S. Decomposition and peat accumulation in rich fens of boreal Alberta, Canada. Ecosystems 2009, 12, 360373. [Google Scholar] [CrossRef]
- Tarnocai, C. The effect of climate change on carbon in Canadian peatlands. Glob. Planet. Chang. 2006, 53, 222–232. [Google Scholar] [CrossRef]
- Kettridge, N.; Turetsky, M.R.; Sherwood, J.H.; Thompson, D.K.; Miller, C.A.; Benscoter, B.W.; Flannigan, M.D.; Wotton, B.M.; Waddington, J.M. Moderate drop in water table increases peatland vulnerability to post-fire regime shift. Sci. Rep. 2015, 5, 8063. [Google Scholar] [CrossRef] [PubMed]
- Macrae, M.L.; Devito, K.J.; Strack, M.; Waddington, J.M. Effect of water table drawdown on peatland nutrient dynamics: Implications for climate change. Biogeochemistry 2013, 112, 661–676. [Google Scholar] [CrossRef]
- Rydin, H.; Jeglum, J. The biology of Peatlands. In Biology Habitats; Oxford University Press: Oxford, UK, 2013; p. 354. [Google Scholar]
- Nwaishi, F.; Petrone, R.M.; Price, J.S.; Andersen, R. Towards developing a functional-based approach for constructed peatlands evaluation in the Alberta oil sands region, Canada. Wetlands 2015, 35, 211–225. [Google Scholar] [CrossRef]
- Proemse, B.C.; Mayer, B.; Fenn, M.E.; Ross, C. A multi-isotope approach for estimating industrial contributions to atmospheric nitrogen deposition in the Athabasca oil sands region in Alberta, Canada. Environ. Pollut. 2013, 182, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.E.; Macrae, M.L.; Strack, M.; Price, J.S.; Osko, T.J.; Petrone, R.M. Spatial variation in nutrient dynamics among five different peatland types in the Alberta oil sands region. Ecohydrology 2015, 9, 688–699. [Google Scholar] [CrossRef]
- Borkenhagen, A.K. Reference Fens: Vegetation Community Types; Unpublished Report Prepared for NSERC CRD Suncore Energy Inc. Pilot Fen Project; Department of Forest and Rangeland Stewardship, Colorado State University: Fort Collins, CO, USA, 2013. [Google Scholar]
- Elmes, M. Hydrology of a Moderate–Rich Fen Watershed Prior to, and Following Wildfire in the Western Boreal Plain, Northern Alberta, Canada; UWSpace, University of Waterloo: Waterloo, ON, Canada, 2018; Available online: http://hdl.handle.net/10012/13740 (accessed on 1 April 2018).
- Nwaishi, F.; Petrone, R.M.; Macrae, M.L.; Price, J.S.; Strack, M.; Slawson, R.; Andersen, R. Above and below-ground nutrient cycling: A criteria for assessing the biogeochemical functioning of a constructed fen. Appl. Soil Ecol. 2016, 98, 177–194. [Google Scholar] [CrossRef]
- Kasischke, E.S.; Turetsky, M.R.; Ottmar, R.D.; French, N.H.F.; Hoy, E.E.; Kane, E.S. Evaluation of the composite burn index for assessing fire severity in Alaskan black spruce forests. Int. J. Wildland Fire 2008, 17, 515–526. [Google Scholar] [CrossRef]
- Lukenbach, M.C.; Devito, K.J.; Kettridge, N.; Petrone, R.M.; Waddington, J.M. Hydrogeological controls on post-fire moss recovery in peatlands. J. Hydrol. 2015, 530, 405–418. [Google Scholar] [CrossRef]
- Mack, M.; Schuur, E.; Syndonia Bret-Harte, M.; Shaver, G.; Chapin, F.S., III. Ecosystem C storage in Arctic tundra reduced by long-term nutrient fertilization. Nature 2004, 431, 440. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.R.; Caporn, S.J.; Nwaishi, F.; Nilsson, R.H.; Sen, R. Bacterial and fungal communities in a degraded ombrotrophic peatland undergoing natural and managed re-vegetation. PLoS ONE 2015, 10, e0124726. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.C.; Nason, G.E.; Myrold, D.D.; Perry, D.A. Dynamics of gross nitrogen transformations in an old-growth forest: The carbon connection. Ecology 1994, 75, 880–891. [Google Scholar] [CrossRef]
- Hart, S.C.; Stark, J.M.; Davidson, E.A.; Firestone, M.K. Nitrogen mineralization, immobilization, and nitrification. In Methods of Soil Analysis: Part 2—Microbiological and Biochemical Properties, SSSA Book Ser. 5.2; Bottomley, P.S., Angle, J.S., Weaver, R.W., Eds.; SSSA: Madison, WI, USA, 1994; pp. 985–1018. [Google Scholar] [CrossRef]
- Pote, D.H.; Daniel, T.C.; Moore, P.A.; Nichols, D.J.; Sharpley, A.N.; Edwards, D.R. Relating extractable soil phosphorus to phosphorus losses in runoff. Soil Sci. Soc. Am. J. 1996, 60, 855–859. [Google Scholar] [CrossRef]
- Plach, J.M.; Wood, M.E.; Macrae, M.L.; Osko, T.J.; Petrone, R.M. Effect of a semi-permanent road on N, P, and CO2 dynamics in a poor fen on the Western Boreal Plain, Canada. Ecohydrology 2017, 10, e1874. [Google Scholar] [CrossRef]
- Parkinson, J.A.; Allen, S.E. A wet oxidation procedure suitable for the determination of nitrogen and mineral nutrients in biological material. Commun. Soil Sci. Plant Anal. 1975, 6, 1–11. [Google Scholar] [CrossRef]
- Griffiths, N.A.; Sebestyen, S.D. Dynamic vertical profiles of peat porewater chemistry in a northern peatland. Wetlands 2016, 36, 1119–1130. [Google Scholar] [CrossRef]
- Badiou, P.; Page, B. Wetland Road Crossings Hydrological Monitoring Design and Data Summary; Ducks Unlimited Canada, Institute for Wetland and Waterfowl Research: Stonewall, MB, Canada, 2014; 49p. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Development Core Team: Vienna, Austria, 2013; Available online: ftp://ftp.uvigo.es/CRAN/web/packages/dplR/vignettes/intro-dplR.pdf (accessed on 1 April 2019).
- Hurlbert, S.H. Pseudoreplication and the design of ecological field experiments. Ecol. Monogr. 1984, 54, 187–211. [Google Scholar] [CrossRef]
- Davies, G.M.; Gray, A. Don’t let spurious accusations of pseudoreplication limit our ability to learn from natural experiments (and other messy kinds of ecological monitoring). Ecol. Evol. 2015, 5, 5295–5304. [Google Scholar] [CrossRef]
- Hokanson, K.J.; Moore, P.A.; Lukenbach, M.C.; Devito, K.J.; Kettridge, N.; Petrone, R.M.; Mendoza, C.A.; Waddington, J.M. A Hydrogeological landscape framework to identify peatland wildfire smouldering hotspots. Ecohydrol. Lett. 2018, 11. [Google Scholar] [CrossRef]
- Lukenbach, M.C.; Kettridge, N.; Devito, K.J.; Petrone, R.M.; Waddington, J.M. Burn severity alters peatland moss water availability: Implications for post-fire recovery. Ecohydrology 2016, 9, 341–353. [Google Scholar] [CrossRef]
- Benscoter, B.W.; Wieder, R.K. Variability in organic matter lost by combustion in a boreal bog during the 2001 Chisholm fire. Can. J. For. Res. 2003, 33, 2509–2513. [Google Scholar] [CrossRef]
- Shetler, G.; Turetsky, M.; Kane, E.S.; Kasischke, E.S. Sphagnum mosses limit total carbon consumption during fire in Alaskan black spruce forests. Can. J. For. Res. 2008, 38, 2328–2336. [Google Scholar] [CrossRef]
- Benscoter, B.W.; Thompson, D.K.; Waddington, J.M.; Flannigan, M.D.; Wotton, M.; DeGroot, W.; Turetsky, M.R. Interactive effects of vegetation, soil moisture, and bulk density on the burning of thick organic soils. Int. J. Wildfire 2011, 20, 418–429. [Google Scholar] [CrossRef]
- Hokanson, K.J.; Lukenbach, M.C.; Devito, K.J.; Kettridge, N.; Petrone, R.M.; Waddington, J.M. Groundwater connectivity controls peat burn severity in the Boreal Plains. Ecohydrology 2016, 9, 574–584. [Google Scholar] [CrossRef]
- Thompson, D.K.; Waddington, J.M. Peat properties and water retention in boreal forested peatlands subject to wildfire. Water Resour. Res. 2013, 49, 3651–3658. [Google Scholar] [CrossRef]
- Thompson, D.K.; Wotton, B.M.; Waddington, J.M. Estimating the heat transfer to an organic soil surface during crown fire. Int. J. Wildland Fire 2015, 24, 120–129. [Google Scholar] [CrossRef]
- Nwaishi, F.; Plach, J.M.; Petrone, R.M.; Morison, M.Q.; Macrae, M.L. Nutrient balance and carbon storage in the soil-plant system of boreal peatlands within the vicinity of active industrial development in Western Canada. Sci. Total Environ. 2019. under revision. [Google Scholar]
- Mayner, K.M.; Moore, P.A.; Wilkinson, S.; Petrone, R.M.; Waddington, J.M. Delineating boreal plains bog margin ecotones across hydrogeological settings for wildfire risk management. Wetl. Ecol. Manag. 2019, in press. [Google Scholar] [CrossRef]
- Sulwiński, M.; Mętrak, M.; Suska-Malawska, M. Long-term fire effects of the drained open fen on organic soils. Arch. Environ. Prot. 2017, 43, 11–19. [Google Scholar] [CrossRef]
- Laubhan, M.K. Effects of prescribed fire on moist-soil vegetation and soil macronutrients. Wetlands 1995, 15, 159–166. [Google Scholar] [CrossRef]
- Neff, J.C.; Harden, J.W.; Gleixner, G. Fire effects on soil organic matter content, composition, and nutrients in boreal interior Alaska. Can. J. For. Res. 2005, 35, 2178–2187. [Google Scholar] [CrossRef]
- Mandic-Mulec, I.; Ausec, L.; Danevčič, T.; Levičnik-Höfferle, Š.; Jerman, V.; Kraigher, B. Microbial community structure and function in peat soil. Food Technol. Biotechnol. 2014, 52, 180–187. [Google Scholar]
- Wang, G.; Yu, X.; Bao, K.; Gao, C.; Lin, Q.; Lu, X. Effect of Fire on phosphorus forms in Sphagnum moss and peat soils of ombrotrophic bogs. Chemosphere 2014, 119, 1329–1334. [Google Scholar] [CrossRef]
- Manzoni, S.; Trofymow, J.A.; Jackson, R.B.; Porporato, A. Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol. Monogr. 2010, 80, 89–106. [Google Scholar] [CrossRef]
- Dikici, H.; Yilmaz, H.C. Peat Fire Effects on Some Properties of an Artificially Drained Peatland. J. Environ. Qual. 2006, 35, 866–870. [Google Scholar] [CrossRef]
- Benscoter, B.W.; Wieder, R.K.; Vitt, D.H. Linking microtopography with post-fire succession in bogs. J. Veg. Sci. 2005, 16, 453–460. [Google Scholar] [CrossRef]
- Grygoruk, M.; Bańkowska, A.; Jabłońska, E.; Janauer, G.A.; Kubrak, J.; Mirosław-Świątek, D.; Kotowski, W. Assessing habitat exposure to eutrophication in restored wetlands: Model supported ex-ante approach to rewetting drained mires. J. Environ. Manag. 2015, 152, 230–240. [Google Scholar] [CrossRef]
- Verhoeven, J.T.; Koerselman, W.; Meuleman, A.F. Nitrogen- or phosphorus-limited growth in herbaceous, wet vegetation: Relations with atmospheric inputs and management regimes. Trends Ecol. Evol. 1996, 11, 494–497. [Google Scholar] [CrossRef]
- Zak, D.; Gelbrecht, J.; Zerbe, S.; Shatwell, T.; Barth, M.; Cabezas, A.; Steffenhagen, P. How helophytes influence the phosphorus cycle in degraded inundated peat soils—Implications for fen restoration. Ecol. Eng. 2014, 66, 82–90. [Google Scholar] [CrossRef]
- Schrautzer, J.; Sival, F.; Breuer, M.; Runhaar, H.; Fichtner, A. Characterizing and evaluating successional pathways of fen degradation and restoration. Ecol. Eng. 2013, 25, 108–120. [Google Scholar] [CrossRef]
- Moore, T.R.; Trofymow, J.A.; Siltanen, M.; Prescott, C. Patterns of decomposition and carbon, nitrogen, and phosphorus dynamics of litter in upland forest and peatland sites in central Canada. Can. J. For. Res. 2005, 35, 133–142. [Google Scholar] [CrossRef]
- Güsewell, S.; Gessner, M.O. N:P ratios influence litter decomposition and colonization by fungi and bacteria in microcosms. Funct. Ecol. 2009, 23, 211–219. [Google Scholar] [CrossRef]
- Wang, M.; Moore, T.R.; Talbot, J.; Richard, P.J. The cascade of C:N:P stoichiometry in an ombrotrophic peatland: From plants to peat. Environ. Res. Lett. 2014, 9, 24003. [Google Scholar] [CrossRef]
- Koerselman, W.; Meuleman, A.F. The Vegetation N:P Ratio: A New Tool to Detect the 563 Nature of Nutrient Limitation. J. Appl. Ecol. 1996, 33, 1441–1450. [Google Scholar] [CrossRef]
- Aerts, R.; Wallen, B.; Malmer, N. Growth-Limiting Nutrients in Sphagnum-Dominated Bogs Subject to Low and High Atmospheric Nitrogen Supply. J. Ecol. 1992, 80, 131–140. [Google Scholar] [CrossRef]
- Gunnarsson, U.; Rydin, H. Nitrogen fertilization reduces Sphagnum production in bog communities. New Phytol. 2000, 147, 527–537. [Google Scholar] [CrossRef]
- Güsewell, S.; Koerselman, W. Variation in nitrogen and phosphorus concentrations of wetland plants. Perspect. Plant Ecol. Evol. Syst. 2002, 5, 37–61. [Google Scholar] [CrossRef]
- Bradford, M.A.; Fierer, N.; Reynolds, J.F. Soil carbon stocks in experimental mesocosms are dependent on the rate of labile carbon, nitrogen and phosphorus inputs to soils. Funct. Ecol. 2008, 510, 964–974. [Google Scholar] [CrossRef]
- Lavoie, M.; Mack, M.C.; Schuur, E.A. Effects of elevated nitrogen and temperature on carbon and nitrogen dynamics in Alaskan arctic and boreal soils. JGR Biogeosci. 2011, 116. [Google Scholar] [CrossRef]
- Limpens, J.; Granath, G.; Gunnarsson, U.; Aerts, R.; Bayley, S.; Bragazza, L.; Bubier, J.; Buttler, A.; Van den Berg, L.J.; Francez, A.J.; et al. Climatic modifiers of the response to nitrogen deposition in peat-forming sphagnum mosses: A meta-analysis. New Phytol. 2011, 191, 496–507. [Google Scholar] [CrossRef]
- Verhoeven, J.T.A. Nutrient dynamics in minerotrophic peat mires. Aquat. Bot. 1986, 25, 117–137. [Google Scholar] [CrossRef]


| Transect | Microform | Depth (cm) | Macronutrient Concentrations | Nutrient Ratios | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C | N | P | Fe | K | C:N | C:P | N:P | Fe:P | |||
| (mg/g) | Ratio | ||||||||||
| Unburned | Hollow | 0–5 | 415.8 (20.6) | 17.2 (5.2) | 1.8 (1.6) | 2.7 (1.1) | 1.6 (0.9) | 27 (10.4) | 393 (364.5) | 12 (6) | 3 (3.2) |
| 5–10 | 415 (13.4) | 19.2 (4.4) | 0.9 (0) | 3.2 (2.7) | 17.3 (3.7) | 23 (6.1) | 475 (29.9) | 24 (0.1) | 4 (3.1) | ||
| 10–15 | 434.9 (24.3) | 18.1 (5.3) | 0.7 (0.1) | 2.3 (1.4) | 14.8 (19.5) | 25 (5.6) | 597 (5.1) | 27 (7.6) | 3 (1.7) | ||
| 15–20 | 404.6 (40.9) | 16.6 (7.1) | 0.7 (0) | 2.9 (2.3) | 20.2 (26.3) | 27 (8.7) | 609 (38.1) | 26 (15.2) | 4 (3) | ||
| Unburned | Hummock | 0–5 | 445.5 (41.1) | 14.3 (4.9) | 0.6 (0.2) | 1.2 (0.3) | 18.6 (12) | 35 (13.9) | 722 (217.3) | 20 (5.2) | 2 (0) |
| 5–10 | 438.2 (10.8) | 17.7 (5.5) | 0.7 (0.2) | 1.3 (0.6) | n/d | 27 (11.5) | 653 (176.1) | 22 (6.6) | 2 (0.3) | ||
| 10–15 | 405.6 (88.2) | 19.8 (8.5) | 0.6 (0.2) | 1.4 (0.1) | n/d | 24 (10) | 586 (3.7) | 23 (11.5) | 2 (0.6) | ||
| 15–20 | 422.5 (17.7) | 21.7 (2.8) | 0.8 (0.1) | 1.2 (0.2) | n/d | 20 (2.5) | 539 (96.9) | 31 (7) | 2 (0.4) | ||
| Burned | Hollow | 0–5 | 457.4 (26.9) | 10.3 (1.3) | 0.8 (0.2) | 2.9 (1.7) | 80.5 (66.4) | 45 (8) | 673 (306.4) | 16 (6.1) | 4 (1.3) |
| 5–10 | 479.2 (31.4) | 12.2 (2.5) | 0.6 (0.2) | 1.4 (0.7) | 34.7 (20) | 42 (14.1) | 903 (397.1) | 20 (1.4) | 2 (0.7) | ||
| 10–15 | 461.7 (55.1) | 12.8 (3.1) | 0.5 (0.2) | 1.7 (1.7) | 25.5 (20.7) | 40 (18.6) | 977 (580.1) | 20 (4.2) | 3 (2.8) | ||
| 15–20 | 460.7 (10.5) | 14.3 (2.8) | 0.8 (0.3) | 2 (1.8) | 19.6 (18.3) | 34 (8.2) | 688 (275.9) | 20 (7) | 2 (1.3) | ||
| Burned | Hummock | 0–5 | 477 (17.7) | 9.5 (1.9) | 0.5 (0) | 0.6 (0.4) | 40.4 (15.4) | 52 (11.2) | 975 (79.7) | 17 (0.8) | 1 (0.9) |
| 5–10 | 478.3 (14) | 11 (3.8) | 0.6 (0.2) | 0.8 (0.5) | 24.3 (35) | 49 (20.3) | 812 (249.9) | 19 (2.3) | 1 (0.4) | ||
| 10–15 | 471.5 (14.3) | 13.4 (2.8) | 0.7 (0.3) | 0.8 (0.6) | 3.4 (4.1) | 38 (12.8) | 849 (460) | 20 (4.5) | 1 (0.4) | ||
| 15–20 | 462.4 (24.1) | 14.6 (2.5) | 0.7 (0.2) | 1.3 (1.3) | 12.8 (15.7) | 33 (8.8) | 715 (281.9) | 21 (3.3) | 2 (1.8) | ||
| Transect | Vegetation Type | Macronutrient Concentrations | Nutrient Ratios | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C | N | P | Fe | K | C:N | C:P | N:P | ||
| (mg/g) | Ratio | ||||||||
| Unburned | Moss | 443 (3.2) | 8.8 (1.4) | 1.1 (1.3) | 341.9 (333.5) | 1.4 (0.9) | 51.3 (8.5) | 380 (230.4) | 7.1 (3.3) |
| Unburned | Vascular | 512.9 (31.5) | 17.8 (5.4) | 0.8 (0.9) | 232 (280.5) | 1.7 (1.1) | 31.9 (12.4) | 445.9 (336.8) | 13.2 (7.1) |
| Burned | Moss | 449 (38.5) | 8.7 (2.4) | 0.9 (0.7) | 52.8 (101) | 2 (1.2) | 54.6 (12.7) | 345.1 (241.3) | 6.3 (4.4) |
| Burned | Vascular | 476.5 (47.2) | 17.4 (5.6) | 1.1 (0.8) | 12.1 (10.7) | 1.6 (0.4) | 30.9 (12.5) | 314.4 (78.8) | 11.7 (5.6) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Beest, C.; Petrone, R.; Nwaishi, F.; Waddington, J.M.; Macrae, M. Increased Peatland Nutrient Availability Following the Fort McMurray Horse River Wildfire. Diversity 2019, 11, 142. https://doi.org/10.3390/d11090142
van Beest C, Petrone R, Nwaishi F, Waddington JM, Macrae M. Increased Peatland Nutrient Availability Following the Fort McMurray Horse River Wildfire. Diversity. 2019; 11(9):142. https://doi.org/10.3390/d11090142
Chicago/Turabian Stylevan Beest, Christine, Richard Petrone, Felix Nwaishi, James Michael Waddington, and Merrin Macrae. 2019. "Increased Peatland Nutrient Availability Following the Fort McMurray Horse River Wildfire" Diversity 11, no. 9: 142. https://doi.org/10.3390/d11090142
APA Stylevan Beest, C., Petrone, R., Nwaishi, F., Waddington, J. M., & Macrae, M. (2019). Increased Peatland Nutrient Availability Following the Fort McMurray Horse River Wildfire. Diversity, 11(9), 142. https://doi.org/10.3390/d11090142





