Ecosystem Engineering by Thalassinidean Crustaceans: Response Variability, Contextual Dependencies and Perspectives on Future Research
Abstract
1. Introduction
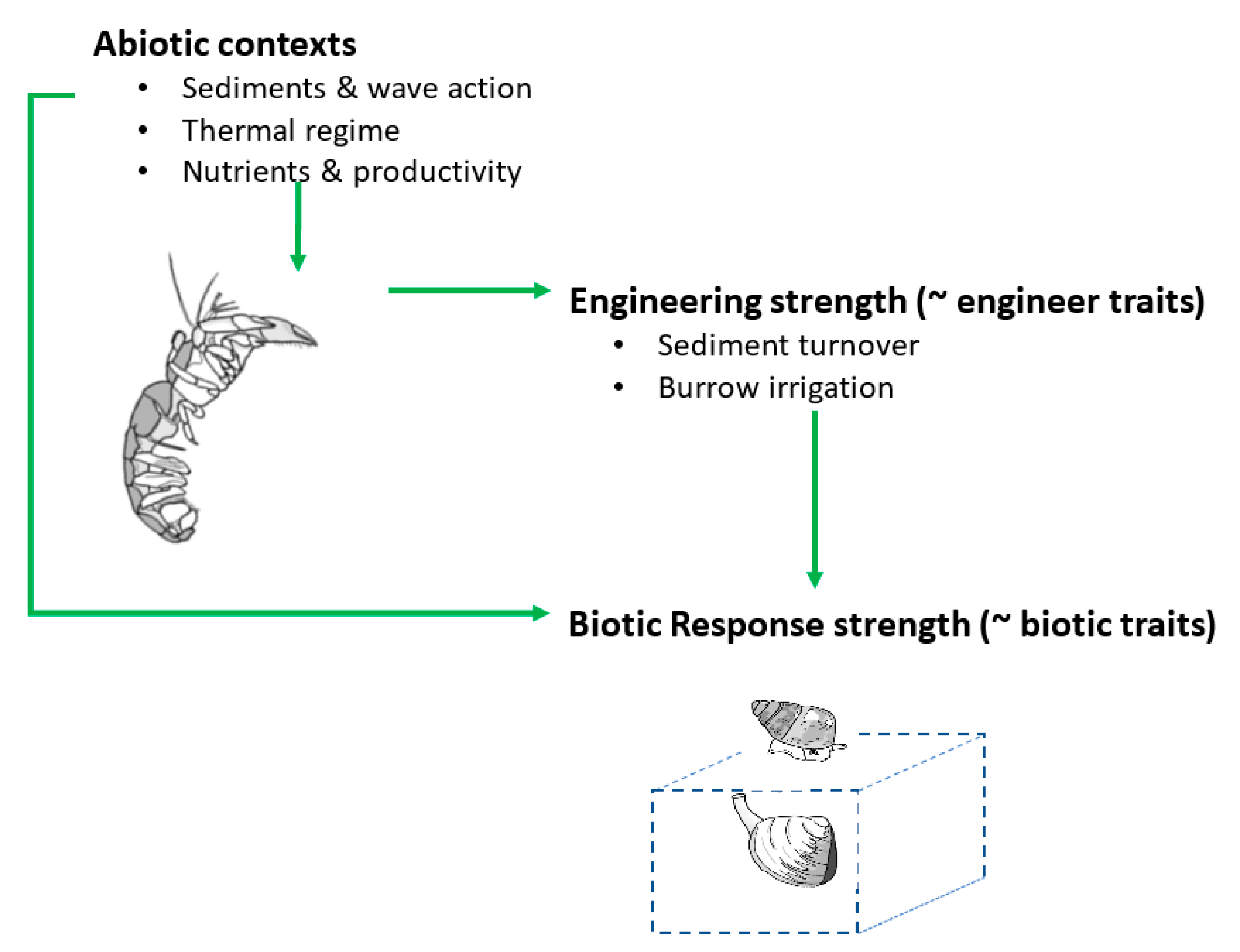
2. Contextual Processes/Variables
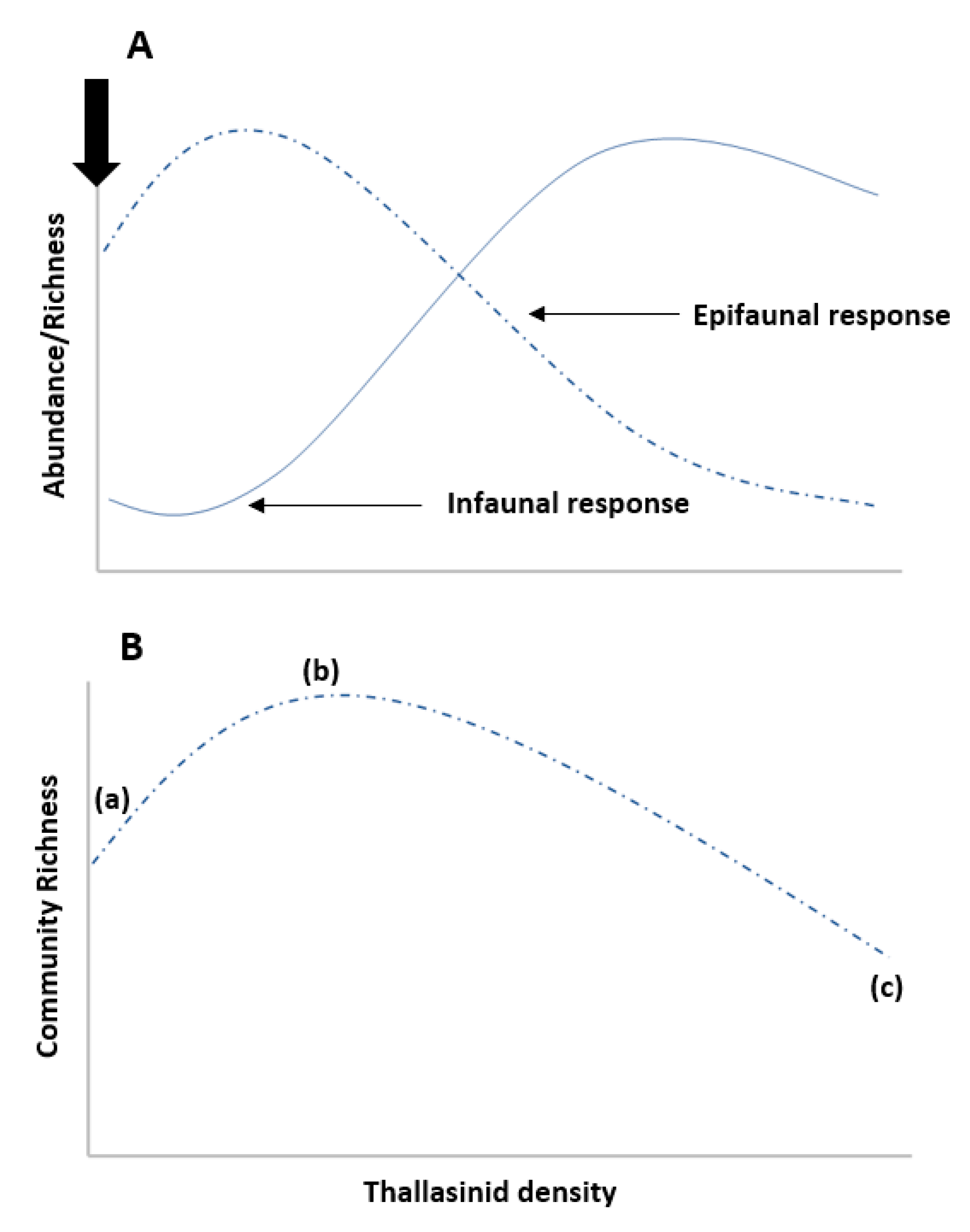
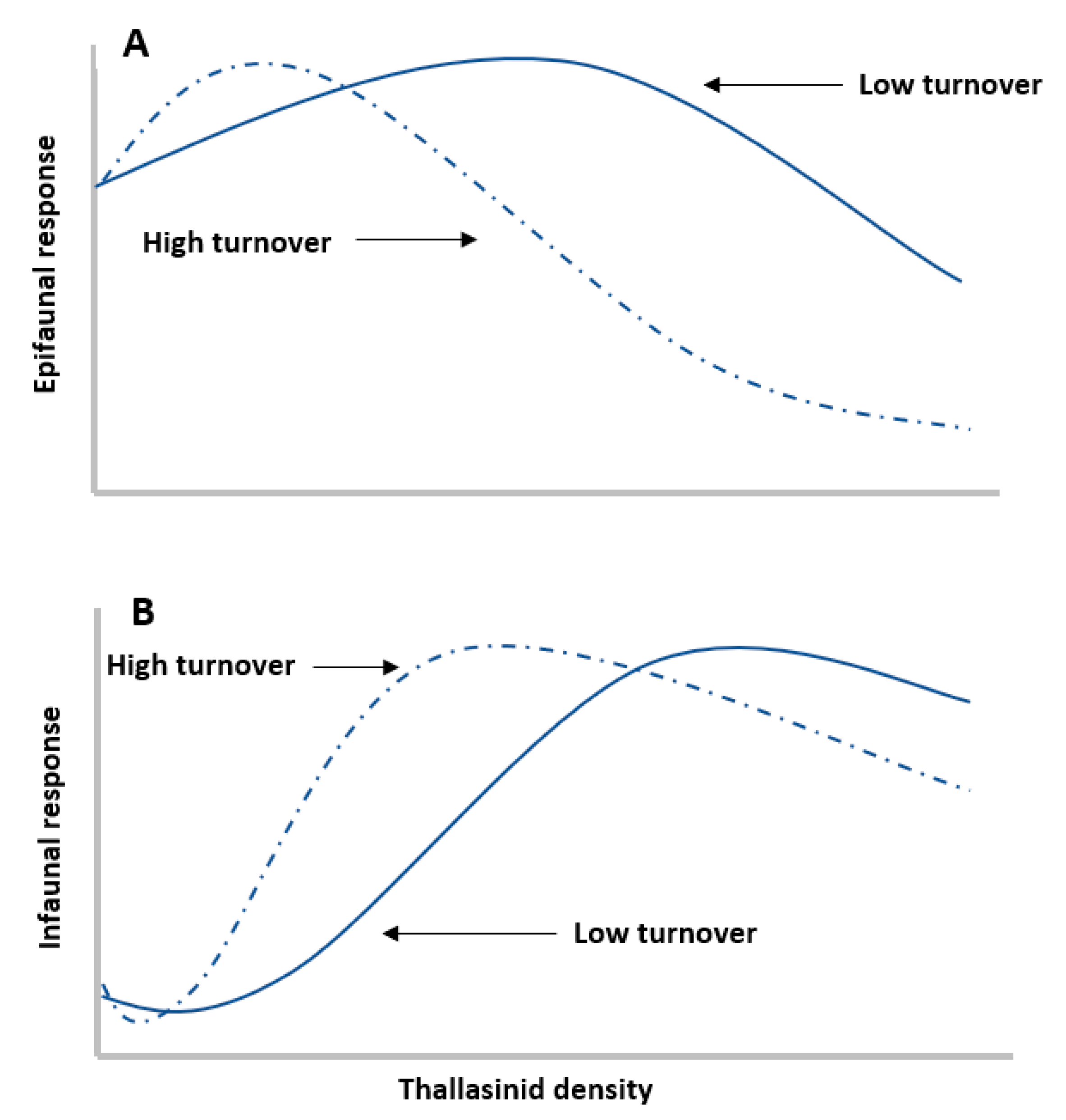
2.1. Engineer Density
2.2. Engineer Traits
2.3. Sediment Type and Wave Action
2.4. Temperature
2.5. Productivity and Nutrients
2.6. Community and Organismal-Level Traits
2.7. Novel/Emerging Determinants of Thalassinidean Ecosystem Engineering
3. Concluding Thoughts, Challenges and Directions for Future Research
Funding
Conflicts of Interest
References
- Jones, C.G.; Lawton, J.H.; Shachak, M. Organisms as ecosystem engineers. Oikos 1994, 69, 373–386. [Google Scholar] [CrossRef]
- Reise, K. Sediment mediated species interactions in coastal waters. J. Sea Res. 2002, 48, 127–141. [Google Scholar] [CrossRef]
- Bouma, T.J.; Olenin, S.; Reise, K.; Ysebaert, T. Ecosystem engineering and biodiversity in coastal sediments: Posing hypotheses. Helgoland Mar. Res. 2009, 63, 96–106. [Google Scholar] [CrossRef]
- Cadée, G.C. Sediment dynamics by bioturbating organisms. In Ecological Comparisons of Sedimentary Shores, Ecological Studies 151; Reise, K., Ed.; Springer: Berlin/Heidelberg, Germany, 2001; pp. 127–148. [Google Scholar]
- Berkenbusch, K.; Rowden, A.A. Ecosystem engineering—Moving away from ‘just-so’ stories. N. Z. J. Ecol. 2003, 27, 67–73. [Google Scholar]
- Pillay, D.; Branch, G.M. Bioengineering effects of burrowing thalassinidean shrimps on marine soft-bottom ecosystems. Oceanogr. Mar. Biol. Annu. Rev. 2011, 49, 137–192. [Google Scholar]
- Poore, G.C.B. A phylogeny of the families of Thalassinidea (Crustacea: Decapoda) with keys to families and genera. Mem. Mus. Vic. 1994, 54, 79–120. [Google Scholar] [CrossRef]
- Felder, D.L. Diversity and ecological significance of deep-burrowing macrocrustaceans in coastal tropical waters of the Americas (Decapoda: Thalassinidea). Intersciencia 2001, 26, 440–449. [Google Scholar]
- Bracken, H.D.; Toon, A.; Felder, D.L.; Martin, J.W.; Finley, M.; Rasmussen, J.; Palero, F.; Crandall, K.A. The decapod tree of life: Compiling the data and moving toward a consensus of decapod evolution. Arthropod. Syst. Phylogeny 2009, 67, 99–116. [Google Scholar]
- Robles, R.; Tudge, C.C.; Dworschak, P.C.; Poore, G.C.B.; Felder, D.L. Molecular phylogeny of the Thalassinidea based on nuclear and mitochondrial genes. In Decapod Crustacean Phylogenetics, Crustacean Issues 18; Martin, J.W., Crandall, K.A., Felder, D.L., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 309–326. [Google Scholar]
- Papaspyrou, S.; Gregersen, T.; Cox, R.P.; Thessalou-Legaki, M.; Kristensen, E. Sediment properties and bacterial community in burrows of the ghost shrimp Pestarella tyrrhena (Decapoda: Thalassinidea). Aquat. Microb. Ecol. 2005, 38, 181–190. [Google Scholar] [CrossRef]
- Pillay, D.; Branch, G.M.; Forbes, A.T. Habitat change in an estuarine embayment: Anthropogenic influences and a regime shift in biotic interactions. Mar. Ecol. Prog. Ser. 2008, 370, 19–31. [Google Scholar] [CrossRef][Green Version]
- Pillay, D.; Williams, C.; Whitfield, A.K. Indirect effects of bioturbation by the burrowing sandprawn Callichirus kraussi on a benthic foraging fish, Liza richardsonii. Mar. Ecol. Prog. Ser. 2012, 453, 151–158. [Google Scholar] [CrossRef]
- Koike, I.; Mukai, H. Oxygen and inorganic nitrogen contents and fluxes in burrows of the shrimps Callianassa japonica and Upogebia major. Mar. Ecol. Prog. Ser. 1983, 12, 185–190. [Google Scholar] [CrossRef]
- Ziebis, W.; Forster, S.; Huettel, M.; Jørgensen, B.B. Complex burrows of the mud shrimp Callianassa truncata and their geochemical impact in the sea bed. Nature 1996, 382, 619–622. [Google Scholar] [CrossRef]
- Rowden, A.A.; Jones, M.B.; Morris, A.W. The role of Callianassa subterranea (Montagu) (Thalassinidea) in sediment resuspension in the North Sea. Cont. Shelf Res. 1998, 18, 1365–1380. [Google Scholar] [CrossRef]
- Flach, E.; Tamaki, A. Competitive bioturbators on intertidal sand flats in the European Wadden Sea and Ariake Sound in Japan. In Ecological Comparisons of Sedimentary Shores, Ecological Studies 151; Reise, K., Ed.; Springer: Berlin, Germany, 2001; pp. 276–293. [Google Scholar]
- D’Andrea, A.F.; DeWitt, T.H. Geochemical ecosystem engineering by the mud shrimp Upogebia pugettensis (Crustacea: Thalassinidae) in Yaquina Bay, Oregon: Density-dependent effects on organic matter remineralization and nutrient cycling. Limnol. Oceanogr. 2009, 54, 1911–1932. [Google Scholar] [CrossRef]
- Volkenborn, N.; Polerecky, L.; Wethey, D.S.; DeWitt, T.H.; Woodin, S.A. Hydraulic activities by ghost shrimp Neotrypaea californiensis induce oxic− anoxic oscillations in sediments. Mar. Ecol. Progr. Ser. 2012, 455, 141–156. [Google Scholar] [CrossRef]
- Pascal, L.; Maire, O.; Volkenborn, N.; Lecroart, P.; Bichon, S.; de Montaudouin, X.; Grémare, A.; Deflandre, B. Influence of the mud shrimp Upogebia pusilla (Decapoda: Gebiidea) on solute and porewater exchanges in an intertidal seagrass (Zostera noltei) meadow of Arcachon Bay: An experimental assessment. J. Exp. Mar. Biol. Ecol. 2016, 477, 69–79. [Google Scholar] [CrossRef]
- Aller, R.C.; Dodge, R.E. Animal-sediment relations in a tropical lagoon Discovery Bay, Jamaica. J. Mar. Res. 1974, 32, 209–232. [Google Scholar]
- Peterson, C.H. Competitive organization of the softbottom macrobenthic communities of Southern California lagoons. Mar. Biol. 1977, 43, 343–359. [Google Scholar] [CrossRef]
- Brenchley, G.A. Disturbance and community structure: An experimental study of bioturbation in marine soft-bottom environments. J. Mar. Res. 1981, 39, 767–790. [Google Scholar]
- Murphy, R.C. Factors affecting the distribution of the introduced bivalve, Mercenaria mercenaria, in a California lagoon—The importance of bioturbation. J. Mar. Res. 1985, 43, 673–692. [Google Scholar] [CrossRef]
- Posey, M.H.; Dumbauld, B.R.; Armstrong, D.A. Effects of a burrowing mud shrimp, Upogebia pugettensis (Dana), on abundances of macro-infauna. J. Exp. Mar. Biol. Ecol. 1991, 148, 283–294. [Google Scholar] [CrossRef]
- Pillay, D.; Branch, G.M.; Forbes, A.T. The influence of bioturbation by the sandprawn Callianassa kraussi on feeding and survival of the bivalve Eumarcia paupercula and the gastropod Nassarius kraussianus. J. Exp. Mar. Biol. Ecol. 2007, 344, 1–9. [Google Scholar] [CrossRef]
- Pillay, D.; Branch, G.M.; Forbes, A.T. Experimental evidence for the effects of the thalassinidean sandprawn Callianassa kraussi on macrobenthic communities. Mar. Biol. 2007, 152, 611–618. [Google Scholar] [CrossRef]
- Flach, E.C. The distribution of the amphipod Corophium arenarium in the Dutch Wadden Sea: Relationships with sediment composition and the presence of cockles and lugworms. Neth. J. Sea Res. 1993, 31, 281–290. [Google Scholar] [CrossRef]
- Pillay, D.; Branch, G.M.; Forbes, A.T. Effects of Callianassa kraussi on microbial biofilms and recruitment of macrofauna: A novel hypothesis for adult-juvenile interactions. Mar. Ecol. Prog. Ser. 2007, 347, 1–14. [Google Scholar] [CrossRef]
- Rhoads, D.C.; Young, D.K. The influence of deposit-feeding organisms on sediment stability and community trophic structure. J. Mar. Res. 1970, 28, 150–178. [Google Scholar]
- Siebert, T.; Branch, G.M. Interactions between Zostera capensis and Callianassa kraussi: Influences on community composition of eelgrass beds and sandflats. Afr. J. Mar. Sci. 2005, 27, 357–373. [Google Scholar] [CrossRef]
- Siebert, T.; Branch, G.M. Interactions between Zostera capensis, Callianassa kraussi and Upogebia africana: Deductions from field surveys in Langebaan Lagoon, South Africa. Afr. J. Mar. Sci. 2005, 27, 345–356. [Google Scholar] [CrossRef]
- Siebert, T.; Branch, G.M. Ecosystem engineers: Interactions between eelgrass Zostera capensis and the sandprawn Callianassa kraussi and their indirect effects on the mudprawn Upogebia africana. J. Exp. Mar. Biol. Ecol. 2006, 338, 253–270. [Google Scholar] [CrossRef]
- Siebert, T.; Branch, G.M. Influences of biological interactions on community structure within seagrass beds and sandprawn-dominated sandflats. J. Exp. Mar. Biol. Ecol. 2007, 340, 11–24. [Google Scholar] [CrossRef]
- Pillay, D.; Branch, G.M.; Dawson, J.; Henry, D. Contrasting effects of ecosystem engineering by the cordgrass Spartina maritima and the sandprawn Callianassa kraussi in a marine-dominated lagoon. Est. Coast. Shelf Sci. 2011, 91, 169–176. [Google Scholar] [CrossRef]
- Branch, G.M.; Pringle, A. The impact of the sand prawn Callianassa kraussi Stebbing on sediment turnover and on bacteria, meiofauna, and benthic microflora. J. Exp. Mar. Biol. Ecol. 1987, 107, 219–235. [Google Scholar] [CrossRef]
- Dobbs, F.C.; Guckert, J.B. Callianassa trilobata (Crustacea: Thalassinidea) influences abundance of meiofauna and biomass, composition, and physiologic state of microbial communities within its burrow. Mar. Ecol. Prog. Ser. 1988, 45, 69–79. [Google Scholar] [CrossRef]
- Dworschak, P.C. The burrows of Callianassa tyrrhena (Petagna 1792) (Decapoda: Thalassinidea). Mar. Ecol. 2001, 22, 155–166. [Google Scholar] [CrossRef]
- Bird, F.L.; Boon, P.I.; Nichols, P.D. Physicochemical and microbial properties of burrows of the deposit-feeding thalassinidean ghost shrimp Biffarius arenosus (Decapoda: Callianassidae). Est. Coast. Shelf Sci. 2000, 51, 279–291. [Google Scholar] [CrossRef]
- Alongi, D.M. Effect of physical disturbance on population dynamics and trophic interactions among microbes and meiofauna. J. Mar. Res. 1985, 43, 351–364. [Google Scholar] [CrossRef]
- Dittmann, S. Effects of macrobenthic burrows on infaunal communities in tropical tidal flats. Mar. Ecol. Prog. Ser. 1996, 134, 119–130. [Google Scholar] [CrossRef]
- Suchanek, T.H. Control of seagrass communities and sediment distribution by Callianassa (Crustacea, Thalassinidea) bioturbation. J. Mar. Res. 1983, 41, 281–298. [Google Scholar] [CrossRef]
- Bird, F.L. The interaction between ghost shrimp activity and seagrass restoration. In Proceedings of the Symposium on Ecology of Large Bioturbators in Tidal Flats and Shallow Sublittoral Sediments—From Individual Behaviour to Their Role as Ecosystem Engineers; Tamaki, A., Ed.; Nagasaki University: Nagasaki, Japan, 2004; pp. 71–75. [Google Scholar]
- Henninger, T.O.; Froneman, P.W. Role of the sandprawn Callichirus kraussi as an ecosystem engineer in a South African temporarily open/closed estuary. Afr. J. Aquat. Sci. 2013, 38, 101–107. [Google Scholar] [CrossRef]
- Jones, C.G.; Lawton, J.H.; Shachak, M. Positive and negative effects of organisms as physical ecosystem engineers. Ecology 1997, 78, 1946–1957. [Google Scholar] [CrossRef]
- Lohrer, A.M.; Thrush, S.F.; Gibbs, M.M. Bioturbators enhance ecosystem function through complex biogeochemical interactions. Nature 2004, 431, 1092–1095. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, O.J.; Krivan, V.; Ovadia, O. Trophic cascades: The primacy of trait-mediated indirect interactions. Ecol. Lett. 2004, 7, 153–163. [Google Scholar] [CrossRef]
- Bouma, T.J.; DeVries, M.B.; Low, E.; Peralta, G.; Tánczos, I.V.; van de Koppel, J.; Herman, P.J. Trade-offs related to ecosystem engineering: A case study on stiffness of emerging macrophytes. Ecology 2005, 86, 2187–2199. [Google Scholar] [CrossRef]
- Nates, S.F.; Felder, D.L. Impact of burrowing ghost shrimp, Genus Lepidophthalmus Crustacea: Decapoda: Thalassinidea, on penaeid shrimp culture. J. World Aquac. Soc. 1998, 29, 188–210. [Google Scholar] [CrossRef]
- Shy, J.; Chan, T. Complete larval development of the edible mud shrimp Upogebia edulis Ngoc-Ho & Chan, 1992 (Decapoda, Thalassinidea, Upogebiidae) reared in the laboratory. Crustaceana 1996, 69, 175–186. [Google Scholar]
- Forbes, A.T. An unusual abbreviated larval life in the estuarine prawn Callianassa kraussi (Crustacea: Decapoda: Thalassinidea). Mar. Biol. 1973, 22, 361–365. [Google Scholar] [CrossRef]
- Coelho, V.R. Feeding behaviour, morphological adaptations and burrowing in thalassinidean crustaceans. In Proceedings of the Symposium on Ecology of Large Bioturbators in Tidal Flats and Shallow Sublittoral Sediments—From Individual Behaviour to Their Role as Ecosystem Engineers; Tamaki, A., Ed.; Nagasaki University: Nagasaki, Japan, 2004; pp. 1–6. [Google Scholar]
- Abed-Navandi, D.; Dworschak, P.C. Food sources of tropical thalassinidean shrimps: A stable-isotope study. Mar. Ecol. Prog. Ser. 2005, 291, 159–168. [Google Scholar] [CrossRef]
- Atkinson, R.J.A.; Taylor, A.C. Aspects of the physiology, biology and ecology of thalassinidean shrimps in relation to their burrow environment. Oceanogr. Mar. Biol. Annu. Rev. 2005, 43, 173–210. [Google Scholar]
- Shimoda, K.; Aramaki, Y.; Nasuda, J.; Yokoyama, H.; Ishihi, Y.; Tamaki, A. Food sources for three species of Nihonotrypaea (Decapoda: Thalassinidea: Callianassidae) from western Kyushu, Japan, as determined by carbon and nitrogen stable isotope analysis. J. Exp. Mar. Biol. Ecol. 2007, 342, 292–312. [Google Scholar] [CrossRef]
- Griffis, R.B.; Chavez, F.L. Effects of sediment type on burrows of Callianassa californiensis Dana and C. gigas Dana. J. Exp. Mar. Biol. Ecol. 1988, 117, 239–253. [Google Scholar] [CrossRef]
- Dworschak, P.C. Biology of Mediterranean and Caribbean Thalassinidea. In Proceedings of the Symposium on Ecology of Large Bioturbators in Tidal Flats and Shallow Sublittoral Sediments—From Individual Behaviour to Their Role as Ecosystem Engineers; Tamaki, A., Ed.; Nagasaki University: Nagasaki, Japan, 2004; pp. 15–22. [Google Scholar]
- Nickell, L.A.; Atkinson, R.J.A. Functional morphology of burrows and trophic modes of three thalassinidean shrimp species, and a new approach to the classification of thalassinidean burrow morphology. Mar. Ecol. Prog. Ser. 1995, 128, 181–197. [Google Scholar] [CrossRef]
- Berkenbusch, K.; Rowden, A.A. Factors influencing sediment turnover by the burrowing ghost shrimp Callianassa filholi (Decapoda: Thalassinidea). J. Exp. Mar. Biol. Ecol. 1999, 238, 283–292. [Google Scholar] [CrossRef]
- Rowden, A.A.; Jones, M.B. Critical evaluation of sediment turnover estimates for Callianassidae (Decapoda: Thalassinidea). J. Exp. Mar. Biol. Ecol. 1993, 173, 265–272. [Google Scholar] [CrossRef]
- Little, C.; Little, E.C.S. The Biology of Soft Shores and Estuaries; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Raffaelli, D.; Hawkins, S.J. Intertidal Ecology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Needham, H.R.; Pilditch, C.A.; Lohrer, A.M.; Thrush, S.F. Context-specific bioturbation mediates changes to ecosystem functioning. Ecosystems 2011, 14, 1096–1109. [Google Scholar] [CrossRef]
- Withers, P.C. Comparative Animal Physiology; Brooks/Cole: Boston, MA, USA, 1992. [Google Scholar]
- Carr, L.A.; Bruno, J.F. Warming increases the top-down effects and metabolism of a subtidal herbivore. PeerJ 2013, 1, e109. [Google Scholar] [CrossRef] [PubMed]
- White, D.S.; Klahr, P.C.; Robbins, J.A. Effects of temperature and density on sediment reworking by Stylodrilus heringianus (Oligochaeta: Lumbriculidae). J. Great Lakes Res. 1987, 13, 147–156. [Google Scholar] [CrossRef]
- Ouellette, D.; Desrosiers, G.; Gagne, J.P.; Gilbert, F.; Poggiale, J.C.; Blier, P.U.; Stora, G. Effects of temperature on in vitro sediment reworking processes by a gallery biodiffusor, the polychaete Neanthes virens. Mar. Ecol. Prog. Ser. 2004, 266, 185–193. [Google Scholar] [CrossRef]
- Hollertz, K.; Duchêne, J.C. Burrowing behaviour and sediment reworking in the heart urchin Brissopsis lyrifera Forbes (Spatangoida). Mar. Biol. 2001, 139, 951–957. [Google Scholar]
- Maire, O.; Duchene, J.C.; Gremare, A.; Malyuga, V.S.; Meysman, F.J.R. A comparison of sediment reworking rates by the surface deposit-feeding bivalve Abra ovata during summertime and wintertime, with a comparison between two models of sediment reworking. J. Exp. Mar. Biol. Ecol. 2007, 343, 21–36. [Google Scholar] [CrossRef]
- Canal, J.; Laffaille, P.; Gilbert, F.; Lauzeral, C.; Buisson, L. Influence of temperature on surface sediment disturbance by freshwater fish: A microcosm experiment. Ann. Limnol. Int. J. Lim. 2015, 51, 179–188. [Google Scholar] [CrossRef]
- Baranov, V.; Lewandowski, J.; Krause, S. Bioturbation enhances the aerobic respiration of lake sediments in warming lakes. Biol. Lett. 2016, 12, 20160448. [Google Scholar] [CrossRef] [PubMed]
- Sigman, D.M.; Hain, M.P. The Biological Productivity of the Ocean. Nat. Educ. Knowl. 2012, 3, 21. [Google Scholar]
- Bustamante, R.H.; Branch, G.M.; Eekhout, S.; Robertson, B.; Zoutendyk, P.; Schleyer, M.; Dye, A.; Hanekom, N.; Keats, D.; Jurd, M.; et al. Gradients of intertidal primary productivity around the coast of South Africa and their relationships with consumer biomass. Oecologia 1995, 102, 189–201. [Google Scholar] [CrossRef]
- Bustamante, R.H.; Branch, G.M.; Eekhout, S. Maintenance of an exceptional intertidal grazer biomass in South Africa: Subsidy by subtidal kelps. Ecology 1995, 76, 2314–2329. [Google Scholar] [CrossRef]
- Kemp, W.M.; Boynton, W.R.; Adolf, J.E.; Boesch, D.F.; Boicourt, W.C.; Brush, G.; Cornwell, J.C.; Fisher, T.R.; Glibert, P.M.; Hagy, J.D.; et al. Eutrophication of Chesapeake Bay: Historical trends and ecological interactions. Mar. Ecol. Prog. Ser. 2005, 303, 1–29. [Google Scholar] [CrossRef]
- Widbom, B.; Elmgren, R. Response of benthic meiofauna to nutrient enrichment of experimental marine ecosystems. Mar. Ecol. Prog. Ser. 1988, 24, 257–268. [Google Scholar] [CrossRef]
- Posey, M.H.; Alphin, T.D.; Cahoon, L.; Lindquist, D.; Becker, M.E. Interactive effects of nutrient additions and predation on infaunal communities. Estuaries 1999, 22, 785–792. [Google Scholar] [CrossRef]
- Posey, M.H.; Alphin, T.D.; Cahoon, L.B.; Lindquist, D.; Mallin, M.A.; Nevers, M.B. Top-down versus bottom-up limitation in benthic infaunal communities: Direct and indirect effects. Estuaries 2002, 25, 999–1014. [Google Scholar] [CrossRef]
- Hamilton, D.J.; Diamond, A.W.; Wells, P.G. Shorebirds, snails, and the amphipod Corophium volutator in the upper Bay of Fundy: Top–down vs. bottom–up factors, and the influence of compensatory interactions on mudflat ecology. Hydrobiologia 2006, 567, 285–306. [Google Scholar] [CrossRef]
- Armitage, A.R.; Fong, P. Upward cascading effects of nutrients: Shifts in a benthic microalgal community and a negative herbivore response. Oecologia 2004, 139, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Little, C.; Kitching, J.A. The Biology of Rocky Shores; Oxford University Press: New York, NY, USA, 1996. [Google Scholar]
- MacKay, F.; Cyrus, D.; Russell, K.L. Macrobenthic invertebrate responses to prolonged drought in South Africa’s largest estuarine lake complex. Est. Coast. Shelf Sci. 2010, 86, 553–567. [Google Scholar] [CrossRef]
- Pillay, D.; Bowns, S.; Nel, H. Benthic Fauna. In Ecology and Conservation of Estuarine Ecosystems: Lake St. Lucia as a Global Model; Perissinotto, R., Stretch, D., Taylor, R., Eds.; Cambridge University Press: Cambridge, UK, 2013; pp. 226–245. [Google Scholar]
- Dairain, A.; de Montaudouin, X.; Gonzalez, P.; Ciutat, A.; Baudrimont, M.; Maire, O.; Legeay, A. Do trace metal contamination and parasitism influence the activities of the bioturbating mud shrimp Upogebia cf. pusilla? Aquat. Toxicol. 2018, 204, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Moyo, R.; Pillay, D.; Baeza, J.A. Symbiont-mediated shifts in sandprawn behaviour: Implications for ecosystem functioning in marine soft-sediment ecosystems. J. Exp. Mar. Biol. Ecol. 2017, 486, 296–304. [Google Scholar] [CrossRef]
- Thrush, S.F.; Hewitt, J.E.; Parkes, S.; Lohrer, A.M.; Pilditch, C.; Woodin, S.A.; Wethey, D.S.; Chiantore, M.; Asnaghi, V.; De Juan, S.; et al. Experimenting with ecosystem interaction networks in search of threshold potentials in real-world marine ecosystems. Ecology 2014, 95, 1451–1457. [Google Scholar] [CrossRef] [PubMed]
- Dworschak, P.C. Global diversity in the Thalassinidea (Decapoda): An update (1998–2004). Nauplius 2005, 13, 57–63. [Google Scholar]
- Borer, E.T.; Seabloom, E.W.; Shurin, J.B.; Anderson, K.E.; Blanchette, C.A.; Broitman, B.; Cooper, S.D.; Halpern, B.S. What determines the strength of a trophic cascade? Ecology 2005, 86, 528–537. [Google Scholar] [CrossRef]
- Shurin, J.B.; Borer, E.T.; Seabloom, E.W.; Anderson, K.; Blanchette, C.A.; Broitman, B.; Cooper, S.D.; Halpern, B.S. A cross-ecosystem comparison of the strength of trophic cascades. Ecol. Lett. 2002, 5, 785–791. [Google Scholar] [CrossRef]
- Mackey, R.L.; Currie, D.J. The diversity–disturbance relationship: Is it generally strong and peaked? Ecology 2001, 82, 3479–3492. [Google Scholar]

© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pillay, D. Ecosystem Engineering by Thalassinidean Crustaceans: Response Variability, Contextual Dependencies and Perspectives on Future Research. Diversity 2019, 11, 64. https://doi.org/10.3390/d11040064
Pillay D. Ecosystem Engineering by Thalassinidean Crustaceans: Response Variability, Contextual Dependencies and Perspectives on Future Research. Diversity. 2019; 11(4):64. https://doi.org/10.3390/d11040064
Chicago/Turabian StylePillay, Deena. 2019. "Ecosystem Engineering by Thalassinidean Crustaceans: Response Variability, Contextual Dependencies and Perspectives on Future Research" Diversity 11, no. 4: 64. https://doi.org/10.3390/d11040064
APA StylePillay, D. (2019). Ecosystem Engineering by Thalassinidean Crustaceans: Response Variability, Contextual Dependencies and Perspectives on Future Research. Diversity, 11(4), 64. https://doi.org/10.3390/d11040064



