Environmental Variation and How its Spatial Structure Influences the Cross-Shelf Distribution of High-Latitude Coral Communities in South Africa
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Reef Community and Environmental Data
2.2.1. Reef Community Surveys
2.2.2. Environmental Variables Associated with Reef Communities
2.3. Data Analysis
2.3.1. Reef Communities
2.3.2. Environmental and Spatial Variables Associated with Reef Communities
2.3.3. Spatial Parameters and Distinguishing them from Environmental Effects
2.3.4. Environmental and Spatial Effects on Reef Communities
3. Results
3.1. Community Structure and Composition
3.2. Environmental and Spatial Characteristics of Reef Transects
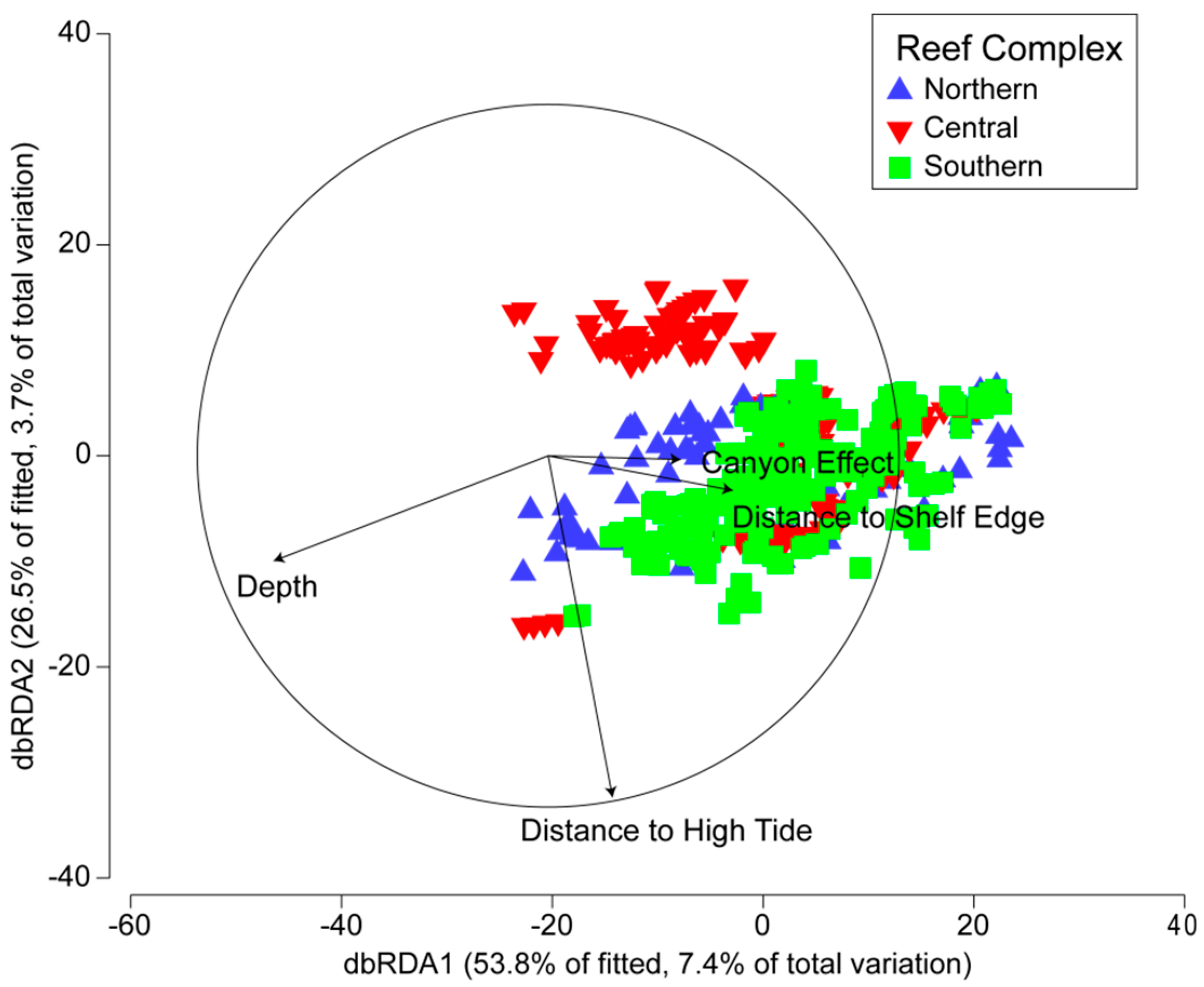
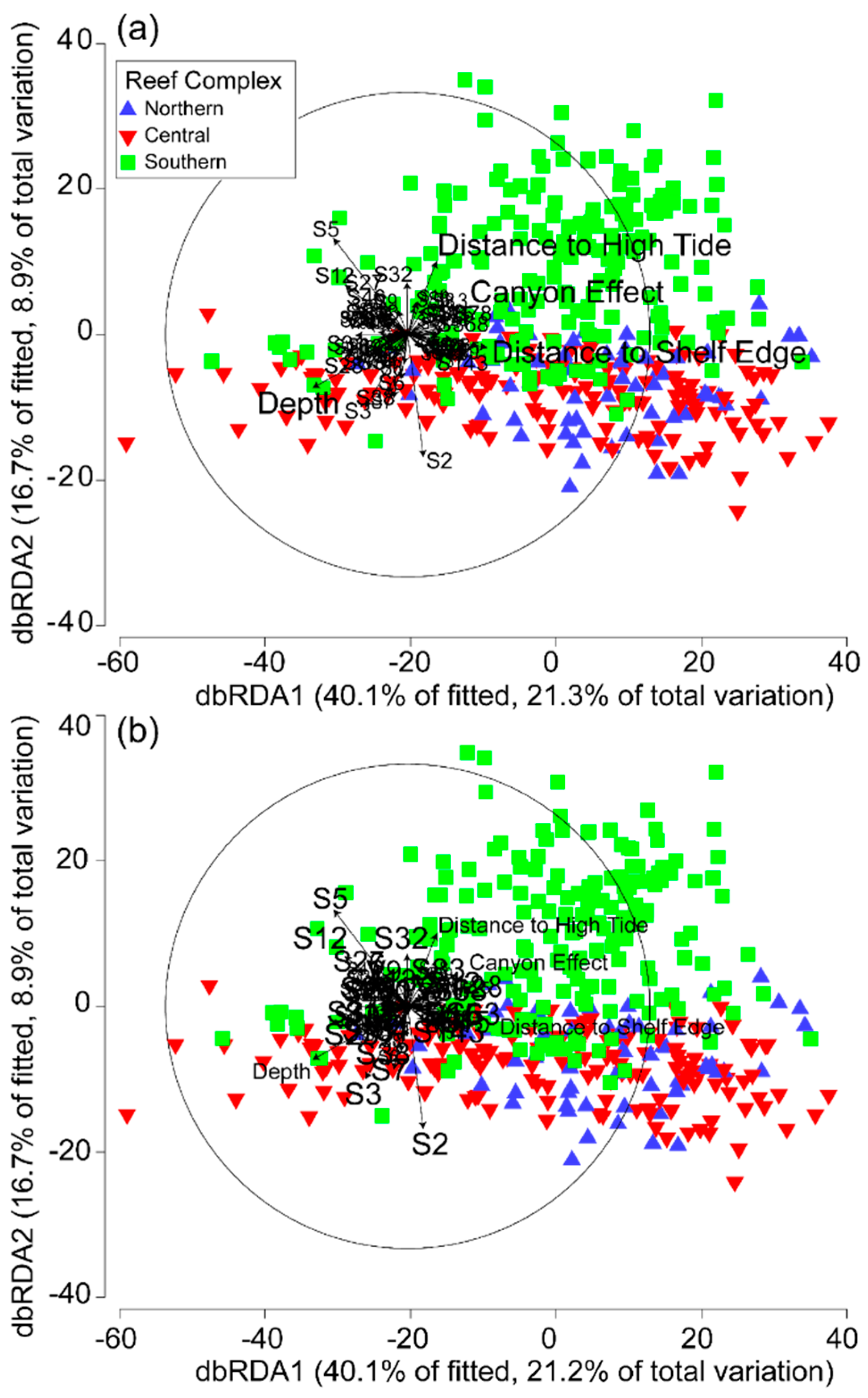
3.3. Environmental and Spatial Effects on Reef Communities
4. Discussion
4.1. Summary of Findings
4.2. Environmental and Spatial Effects
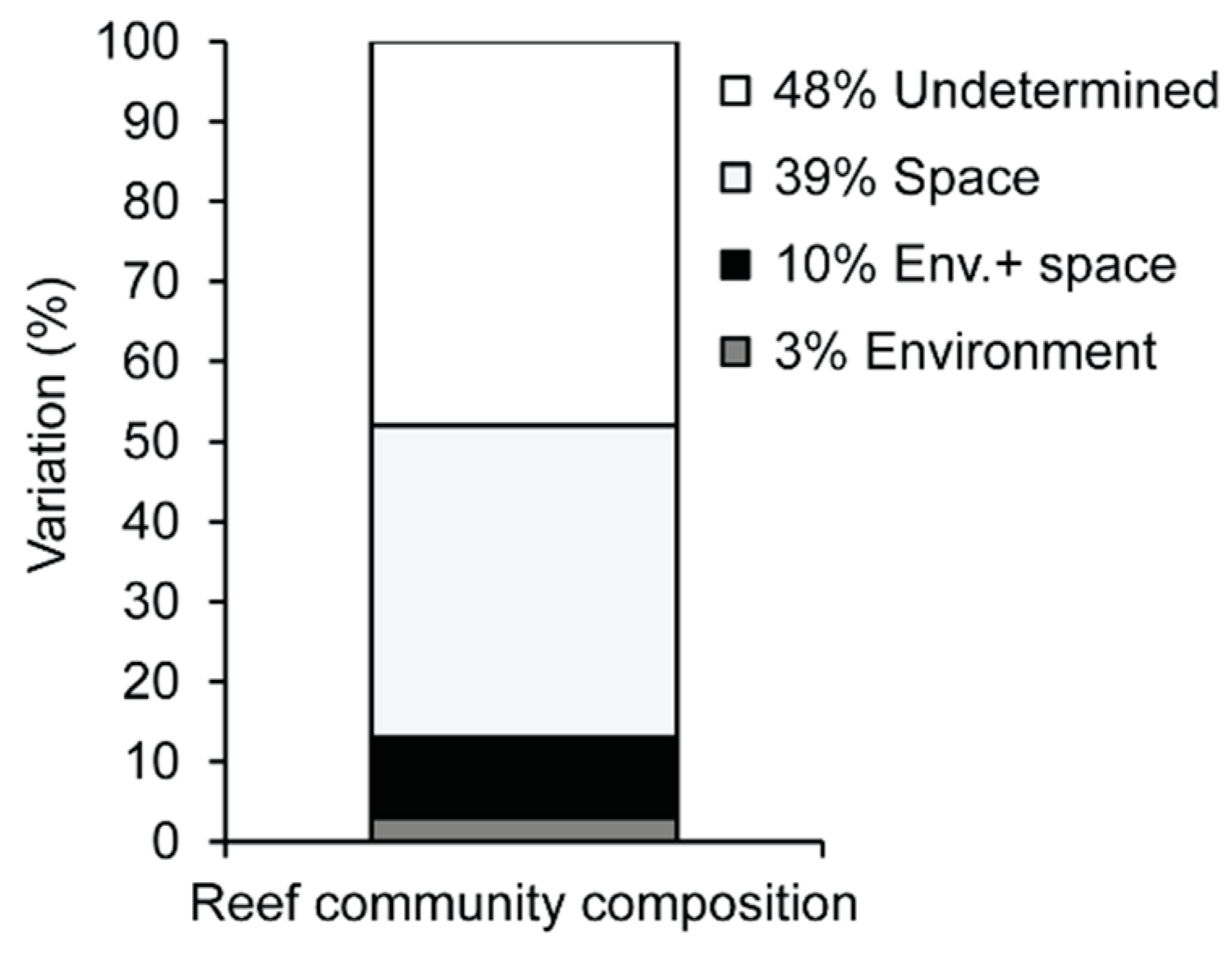
4.3. Unexplained Variation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Laing, S.C.S.; Turpie, J.K.; Schleyer, M.H. Using SCUBA diving and boat-based angling to estimate an economic value of the coral reefs of Sodwana Bay, South Africa. Afr. J. Mar. Sci. 2019. under review. [Google Scholar]
- Laing, S.C.S.; Turpie, J.K.; Schleyer, M.H. Ecosystem services and coral reefs: An estimate of the value of sand generation and entrapment at Sodwana Bay, South Africa. Afr. J. Mar. Sci. 2019. under review. [Google Scholar]
- Schleyer, M.H. South African Coral Communities. Coral Reefs of the Indian Ocean: Their Ecology and Conservation; McClanahan, T., Sheppard, C., Eds.; Oxford University Press: Oxford, UK, 2000; pp. 83–105. [Google Scholar]
- Schleyer, M.H.; Floros, C.; Laing, S.C.; Macdonald, A.H.; Montoya-Maya, P.H.; Morris, T.; Porter, S.N.; Seré, M.G. What can South African reefs tell us about the future of high-latitude coral systems? Mar. Pollut. Bull. 2018, 136, 491–507. [Google Scholar] [CrossRef]
- Celliers, L.; Schleyer, M.H. Coral community structure and risk assessment of high-latitude reefs at Sodwana Bay, South Africa. Biodivers. Conserv. 2008, 17, 3097–3117. [Google Scholar] [CrossRef]
- Monniot, C.; Monniot, F.; Griffiths, C.; Schleyer, M. A monograph of the South African ascidians. Ann. S. Afr. Mus. 2001, 108, 1–141. [Google Scholar]
- Anderson, R.J.; McKune, C.; Bolton, J.J.; DeClerck, O.; Tronchin, E. Patterns in subtidal seaweed communities on coral-dominated reefs at Sodwana Bay on the KwaZulu-Natal coast, South Africa. Afr. J. Mar. Sci. 2005, 27, 529–537. [Google Scholar] [CrossRef]
- Schleyer, M.H.; Celliers, L. Biodiversity on the marginal coral reefs of South Africa: What does the future hold? Zool. Verh. 2003, 345, 387–400. [Google Scholar]
- Schleyer, M.; Celliers, L. Modelling reef zonation in the Greater St. Lucia Wetland Park, South Africa. Estuar. Coast. Shelf Sci. 2005, 63, 373–384. [Google Scholar] [CrossRef]
- Samaai, T.; Gibbons, M.J.; Kerwath, S.; Yemane, D.; Sink, K. Sponge richness along a bathymetric gradient within the iSimangaliso Wetland Park, South Africa. Mar. Biodivers. 2010, 40, 205–217. [Google Scholar] [CrossRef]
- Milne, R.; Griffiths, C. Invertebrate biodiversity associated with algal turfs on a coral-dominated reef. Mar. Biodivers. 2014, 44, 181–188. [Google Scholar] [CrossRef]
- Chater, S.A.; Beckley, L.E.; Garratt, P.A.; Ballard, J.A.; Van der Elst, R.P. Fishes from offshore reefs in the St. Lucia and Maputaland marine reserves, South Africa. Lammergeyer 1993, 42, 1–17. [Google Scholar]
- Floros, C.; Schleyer, M.H.; Maggs, J.Q.; Celliers, L. Baseline assessment of high-latitude coral reef fish communities in southern Africa. Afr. J. Mar. Sci. 2012, 34, 55–69. [Google Scholar] [CrossRef]
- Schleyer, M.H.; Porter, S.N. Drivers of soft and stony coral community distribution on the high-latitude coral reefs of South Africa. Adv. Mar. Biol. 2018, 80, 1–55. [Google Scholar] [PubMed]
- Porter, S.N.; Branch, G.M.; Sink, K.J. Changes in shallow-reef community composition along environmental gradients on the East African coast. Mar. Biol. 2017, 164, 101. [Google Scholar] [CrossRef]
- Porter, S.N.; Schleyer, M.H. Long-term dynamics of a high-latitude coral reef community at Sodwana Bay, South Africa. Coral Reefs 2017, 36, 369–382. [Google Scholar] [CrossRef]
- Legendre, P. Spatial autocorrelation—Trouble or new paradigm? Ecology 1993, 74, 1659–1673. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology, 2nd ed.; Elsevier Science: Amsterdam, NL, USA, 1998. [Google Scholar]
- Schleyer, M.H.; Celliers, L. Coral dominance at the reef–sediment interface in marginal coral communities at Sodwana Bay, South Africa. Mar. Freshwater Res. 2003, 54, 967–972. [Google Scholar] [CrossRef]
- Porter, S.N.; Branch, G.M.; Sink, K.J. Sand-mediated divergence between shallow reef communities on horizontal and vertical substrata in the western Indian Ocean. Afr. J. Mar. Sci. 2017, 39, 121–127. [Google Scholar] [CrossRef]
- Montoya-Maya, P.H.; Schleyer, M.H.; Macdonald, A.H. Limited ecologically relevant genetic connectivity in the south-east African coral populations calls for reef-level management. Mar. Biol. 2016, 163, 171. [Google Scholar] [CrossRef]
- Montoya-Maya, P.H. Ecological Genetic Connectivity between and within Southeast African Marginal Coral Reefs. Ph.D. Thesis, University of KwaZulu-Natal, Durban, South Africa, 2014. [Google Scholar]
- Mitchell, J.; Jury, M.R.; Mulder, G.J. A study of Maputaland beach dynamics. S. Afr. Geogr. J. 2005, 87, 43–51. [Google Scholar] [CrossRef]
- Meyer, R.; Talma, A.S.; Duvenhage, A.W.A.; Eglington, B.M.; Taljaard, J.; Botha, J.F.; Verwey, J.; Van der Voort, I. Geohydrological Investigation and Evaluation of the Zululand Coastal Aquifer; Water Research Commission Report 221/1/01; Water Research Commision: Pretoria, South Africa, 2001. [Google Scholar]
- Smithers, J.C.; Gray, R.P.; Johnson, S.; Still, D. Modelling and water yield assessment of Lake Sibhayi. Water SA 2017, 43, 480–491. [Google Scholar] [CrossRef][Green Version]
- Porter, S.N.; Humphries, M.S.; Buah-Kwofie, A.; Schleyer, M.H. Accumulation of organochlorine pesticides in reef organisms from marginal coral reefs in South Africa and links with coastal groundwater. Mar. Pollut. Bull. 2018, 137, 295–305. [Google Scholar] [CrossRef]
- Morris, T.; Lamont, T.; Roberts, M.J. Effects of deep-sea eddies on the northern KwaZulu-Natal shelf, South Africa. Afr. J. Mar. Sci. 2013, 35, 343–350. [Google Scholar] [CrossRef]
- Ramsay, P.J. Marine geology of the Sodwana Bay shelf, southeast Africa. Mar. Geol. 1994, 120, 225–247. [Google Scholar] [CrossRef]
- Riegl, B.; Piller, W.E. Possible refugia for reefs in times of environmental stress. Int. J. Earth Sci. 2003, 92, 520–531. [Google Scholar] [CrossRef]
- Green, A. Sediment dynamics on the narrow, canyon-incised and current-swept shelf of the northern KwaZulu-Natal continental shelf, South Africa. Geo-Mar. Lett. 2009, 29, 201–219. [Google Scholar] [CrossRef]
- Morris, T. Physical Oceanography of Sodwana Bay and Its Effect on Larval Transport and Coral Bleaching. Master’s Thesis, Cape Peninsula University of Technology, Cape Town, South Africa, 2009. [Google Scholar]
- Riegl, B. Climate change and coral reefs: Different effects in two high-latitude areas (Arabian Gulf, South Africa). Coral Reefs 2003, 22, 433–446. [Google Scholar] [CrossRef]
- Porter, S.N.; Branch, G.M.; Sink, K.J. Biogeographic patterns on shallow subtidal reefs in the western Indian Ocean. Mar. Biol. 2013, 160, 1271–1283. [Google Scholar] [CrossRef]
- Ramsay, P.J.; Mason, T.R. Development of a type zoning model for Zululand coral reefs, Sodwana Bay, South Africa. J. Coast. Res. 1990, 6, 829–852. [Google Scholar]
- Grundling, A.T.; Van den Berg, E.C.; Price, J.S. Assessing the distribution of wetlands over wet and dry periods and land-use change on the Maputaland Coastal Plain, north-eastern KwaZulu-Natal, South Africa. S. Afr. J. Geomat. 2013, 2, 120–138. [Google Scholar]
- Schleyer, M.H.; Celliers, L. The status of South African coral reefs. In Coral Reef Degradation in the Indian Ocean: Status Report 2000; Souter, D., Obura, D., Linden, O., Eds.; CORDIO: Stockholm, Sweden, 2000; pp. 49–50. [Google Scholar]
- Celliers, L.; Schleyer, M.H. Coral bleaching on high-latitude marginal reefs at Sodwana Bay, South Africa. Mar. Pollut. Bull. 2002, 44, 1380–1387. [Google Scholar] [CrossRef]
- Sebastián, C.R.; Sink, K.J.; McClanahan, T.R.; Cowan, D.A. Bleaching response of corals and their Symbiodinium communities in southern Africa. Mar. Biol. 2009, 156, 2049–2062. [Google Scholar] [CrossRef]
- English, S.; Wilkinson, C.; Baker, V. Survey Manual for Tropical and Marine Resources. ASEAN—Australia Marine Science Project: Living Coastal Resources; Australian Institute of Marine Science: Townsville, Australia, 1994; 368p.
- Carleton, J.H.; Done, T.J. Quantitative video sampling of coral reef benthos: Large-scale application. Coral Reefs 1995, 14, 35–46. [Google Scholar] [CrossRef]
- Ezemvelo KwaZulu-Natal Wildlife (EKZNW). High Tide Line. Unpublished GIS Coverage; Biodiversity Conservation Planning Division, Ezemvelo KZN Wildlife: Cascades, Pietermaritzburg, South Africa, 2009. [Google Scholar]
- South African Navy Hydrographic Office (SAN-HO). South African Navy Hydrographer. South African Tide Tables; Hydrographer, South African Navy: Tokai, South Africa, 2019. [Google Scholar]
- Harris, J.M.; Livingstone, T.; Lombard, A.T.; Lagabrielle, E.; Haupt, P.; Sink, K.; Schleyer, M.; Mann, B.Q. Coastal and Marine Biodiversity Plan for KwaZulu-Natal. Spatial Priorities for the Conservation of Coastal and Marine Biodiversity in KwaZulu-Natal; Ezemvelo KZN Wildlife Scientific Services Technical Report; Ezemvelo KZN Wildlife: Pietermaritzburg, South Africa, 2012. [Google Scholar]
- Sink, K.J.; Holness, S.; Harris, L.; Majiedt, P.A.; Atkinson, L.; Robinson, T.; Kirkman, S.; Hutchings, L.; Leslie, R.; Lamberth, S.; et al. National Biodiversity Assessment 2011: Technical Report. Volume 4: Marine and Coastal Component; South African National Biodiversity Institute: Pretoria, South Africa, 2012. [Google Scholar]
- Dray, S.; Legendre, P.; Peres-Neto, P.R. Spatial modelling: A comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecol. Model. 2006, 196, 483–493. [Google Scholar] [CrossRef]
- Borcard, D.; Legendre, P.; Drapeau, P. Partialling out the spatial component of ecological variation. Ecology 1992, 73, 1045–1055. [Google Scholar] [CrossRef]
- Scarlett, M.J. Problems of Analysis of Spatial Distribution. Congrès International de Géographie, Montréal; Adams, W.P., Helleiner, F.M., Eds.; University of Toronto Press: Toronto, ON, Canada, 1972; pp. 928–931. [Google Scholar]
- Borcard, D.; Legendre, P. All-scale spatial analysis of ecological data by means of principal coordinates of neighbour matrices. Ecol. Model. 2002, 153, 51–68. [Google Scholar] [CrossRef]
- Legendre, P.; Anderson, M.J. Distance-based redundancy analysis: Testing multispecies responses in multifactorial ecological experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [CrossRef]
- McArdle, B.H.; Anderson, M.J. Fitting multivariate models to community data: A comment on distance-based redundancy analysis. Ecology 2001, 82, 290–297. [Google Scholar] [CrossRef]
- R Core Development Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Renka, R.; Gebhardt, A. Tripack: Triangulation of Irregularly Spaced Data. R package Version 1.3-6. 2013. Available online: http://CRAN.R-project.org/package=tripack (accessed on 5 October 2018).
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER. Guide to Software and Statistical Methods; PRIMER-E: Plymouth, UK, 2008. [Google Scholar]
- Burn, D.; Pratchett, M.S.; Heron, S.F.; Thompson, C.A.; Pratchett, D.J.; Hoey, A.S. Limited cross-shelf variation in the growth of three branching corals on Australia’s Great Barrier Reef. Diversity 2018, 10, 122. [Google Scholar] [CrossRef]
- Hamman, E.A. Aggregation patterns of two corallivorous snails and consequences for coral dynamics. Coral Reefs 2018, 37, 851–860. [Google Scholar] [CrossRef]
- Williams, G.B. Aggregation during settlement as a factor in the establishment of coelenterate colonies. Ophelia 1976, 15, 57–64. [Google Scholar] [CrossRef]
- Goreau, N.I.; Goreau, T.J.; Hayes, R.L. Settling, survivorship and spatial aggregation in planulae and juveniles of the coral Porites porites (Pallas). Bull. Mar. Sci. 1981, 31, 424–435. [Google Scholar]
- Karlson, R.H.; Cornell, H.V.; Hughes, T.P. Aggregation influences coral species richness at multiple spatial scales. Ecology 2007, 88, 170–177. [Google Scholar] [CrossRef]
- Chadwick, N.E.; Morrow, K.M. Competition among sessile organisms on coral reefs. In Coral Reefs: An Ecosystem in Transition; Dubinsky, Z., Stambler, N., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 347–371. [Google Scholar]
- Miller, K.J.; Ayre, D.J. Population structure is not a simple function of reproductive mode and larval type: Insights from tropical corals. J. Anim. Ecol. 2008, 77, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Gilmour, J.P.; Smith, L.D.; Brinkman, R.M. Biannual spawning, rapid larval development and evidence of self-seeding for scleractinian corals at an isolated system of reefs. Mar. Biol. 2009, 156, 1297–1309. [Google Scholar] [CrossRef]
- Keith, S.A.; Baird, A.H.; Hughes, T.P.; Madin, J.S.; Connolly, S.R. Faunal breaks and species composition of Indo-Pacific corals: The role of plate tectonics, environment and habitat distribution. Proc. R. Soc. B 2013, 280, 20130818. [Google Scholar] [CrossRef]
- Schleyer, M. Crown of thorns starfish in the western Indian Ocean. Reef Encounter 1998, 23, 25–27. [Google Scholar]
- Celliers, L.; Schleyer, M.H. Observations on the behaviour and the character of Acanthaster planci (L.) aggregation in a high latitude coral community in South Africa. Western Indian Ocean J. Mar. Sci. 2006, 5, 105–113. [Google Scholar] [CrossRef]
- Séré, M.G.; Schleyer, M.H.; Quod, J.P.; Chabanet, P. Porites white patch syndrome: An unreported coral disease on Western Indian Ocean reefs. Coral Reefs 2012, 31, 739. [Google Scholar] [CrossRef][Green Version]
- Séré, M.G.; Chabanet, P.; Turquet, J.; Quod, J.P.; Schleyer, M.H. Identification and prevalence of coral diseases on three Western Indian Ocean coral reefs. Dis. Aquat. Organ. 2015, 114, 249–261. [Google Scholar] [CrossRef]
- Glassom, D.; Celliers, L.; Schleyer, M.H. Coral recruitment patterns at Sodwana Bay, South Africa. Coral Reefs 2006, 25, 485–492. [Google Scholar] [CrossRef]
- Macdonald, A.H.H.; Schleyer, M.H.; Lamb, J.M. Acropora austera connectivity in the southwestern Indian Ocean assessed using nuclear intron sequence data. Mar. Biol. 2010, 158, 613–621. [Google Scholar] [CrossRef]
- Hart, J.R. Coral Recruitment on a High-Latitude Reef at Sodwana Bay, South Africa: Research Methods and Dynamics. Master’s Thesis, University of KwaZulu-Natal, Durban, South Africa, 2011. [Google Scholar]
- Sammarco, P.W.; Coll, J.C.; La Barre, S. Competitive strategies of soft corals (Coelenterata: Octocorallia). II. Variable defensive responses and susceptibility to scleractinian corals. J. Exp. Mar. Biol. Ecol. 1985, 91, 199–215. [Google Scholar] [CrossRef]
- La Barre, S.C.; Coil, J.C.; Sammarco, P.W. Competitive strategies of soft corals (Coelenterata: Octocorallia): 111. Spacing and aggressive interactions between alcyonaceana. Mar. Ecol. Prog. 1986, 28, 147–156. [Google Scholar] [CrossRef]
- McCook, L.; Jompa, J.; Diaz-Pulido, G. Competition between corals and algae on coral reefs: A review of evidence and mechanisms. Coral Reefs 2001, 19, 400–417. [Google Scholar] [CrossRef]
- Sammarco, P.W.; Coll, J.C.; La Barre, S.; Willis, B. Competitive strategies of soft corals (Coelenterata: Octocorallia): Allelopathic effects on selected scleractinian corals. Coral Reefs 1983, 1, 173–178. [Google Scholar] [CrossRef]
- Changyun, W.; Haiyan, L.; Changlun, S.; Yanan, W.; Liang, L.; Huashi, G. Chemical defensive substances of soft corals and gorgonians. Acta Ecol. Sin. 2008, 28, 2320–2328. [Google Scholar] [CrossRef]
- Fabricius, K.; De’ath, G. Environmental factors associated with the spatial distribution of crustose coralline algae on the Great Barrier Reef. Coral Reefs 2001, 19, 303–309. [Google Scholar] [CrossRef]
- Hsueh, Y.; O’Brien, J.J. Steady coastal upwelling induced by an along-shore current. J. Phys. Oceanogr. 1971, 1, 180–186. [Google Scholar] [CrossRef]
- Roberts, M.J.; Ribbink, A.J.; Morris, T.; Van den Berg, M.A.; Engelbrecht, D.C.; Harding, R.T. Oceanographic environment of the Sodwana Bay coelacanths (Latimeria chalumnae), South Africa: Coelacanth research. S. Afr. J. Sci. 2006, 102, 435–443. [Google Scholar]
- Graus, R.R.; Macintyre, I.G. The zonation patterns of Caribbean coral reefs as controlled by wave and light energy input, bathymetric setting and reef morphology: Computer simulation experiments. Coral Reefs 1989, 8, 9–18. [Google Scholar] [CrossRef]
- Birrell, C.L.; McCook, L.J.; Willis, B.L. Effects of algal turfs and sediment on coral settlement. Mar. Pollut. Bull. 2005, 51, 408–414. [Google Scholar] [CrossRef]
- Kench, P.S.; Brander, R.W. Wave processes on coral reef flats: Implications for reef geomorphology using Australian case studies. J. Coast. Res. 2006, 22, 209–223. [Google Scholar] [CrossRef]
- Cleary, D.F.; Becking, L.E.; de Voogd, N.J.; Renema, W.; de Beer, M.; van Soest, R.W.; Hoeksema, B.W. Variation in the diversity and composition of benthic taxa as a function of distance offshore, depth and exposure in the Spermonde Archipelago, Indonesia. Estuar. Coast. Shelf Sci. 2005, 65, 557–570. [Google Scholar] [CrossRef]
- McClanahan, T.R.; Ateweberhan, M.; Graham, N.A.J.; Wilson, S.K.; Sebastián, C.R.; Guillaume, M.M.; Bruggemann, J.H. Western Indian Ocean coral communities: Bleaching responses and susceptibility to extinction. Mar. Ecol. Prog. Ser. 2007, 337, 1–13. [Google Scholar] [CrossRef]
- Porter, S.N.; Schleyer, M.H.; Sink, K.J.; Pearton, D.J. South Africa. In Impact of the 3rd Global Coral Bleaching Event in the Western Indian Ocean in 2016; Gudka, M., Obura, D., Mwaura, J., Porter, S., Yahya, S., Mabwa, R., Eds.; Global Coral Reef Monitoring Network/Indian Ocean Commission: Port Louis, Mauritius, 2018. [Google Scholar]
- Guisan, A.; Zimmermann, N.E. Predictive habitat distribution models in ecology. Ecol. Model. 2000, 135, 147–186. [Google Scholar] [CrossRef]
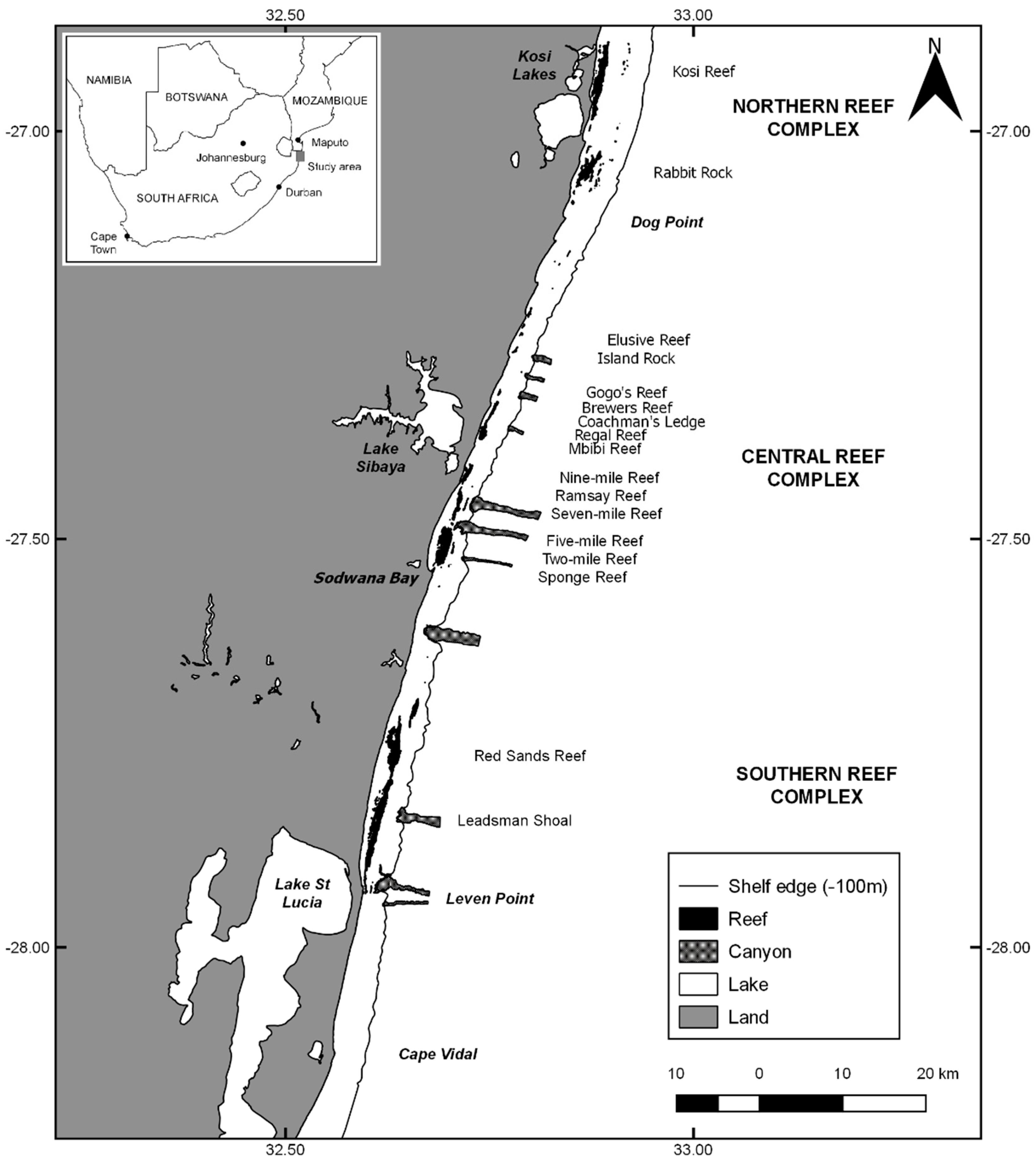
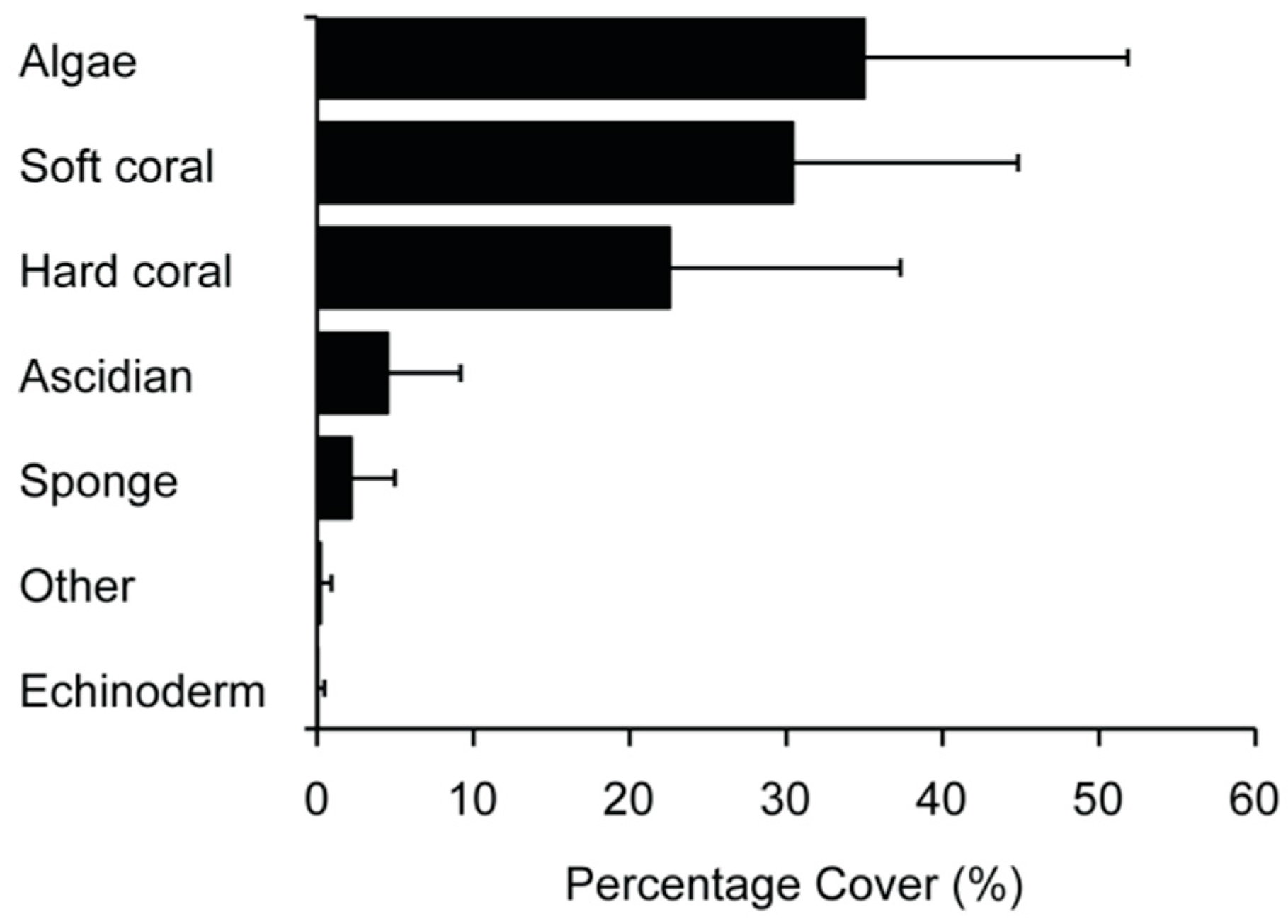
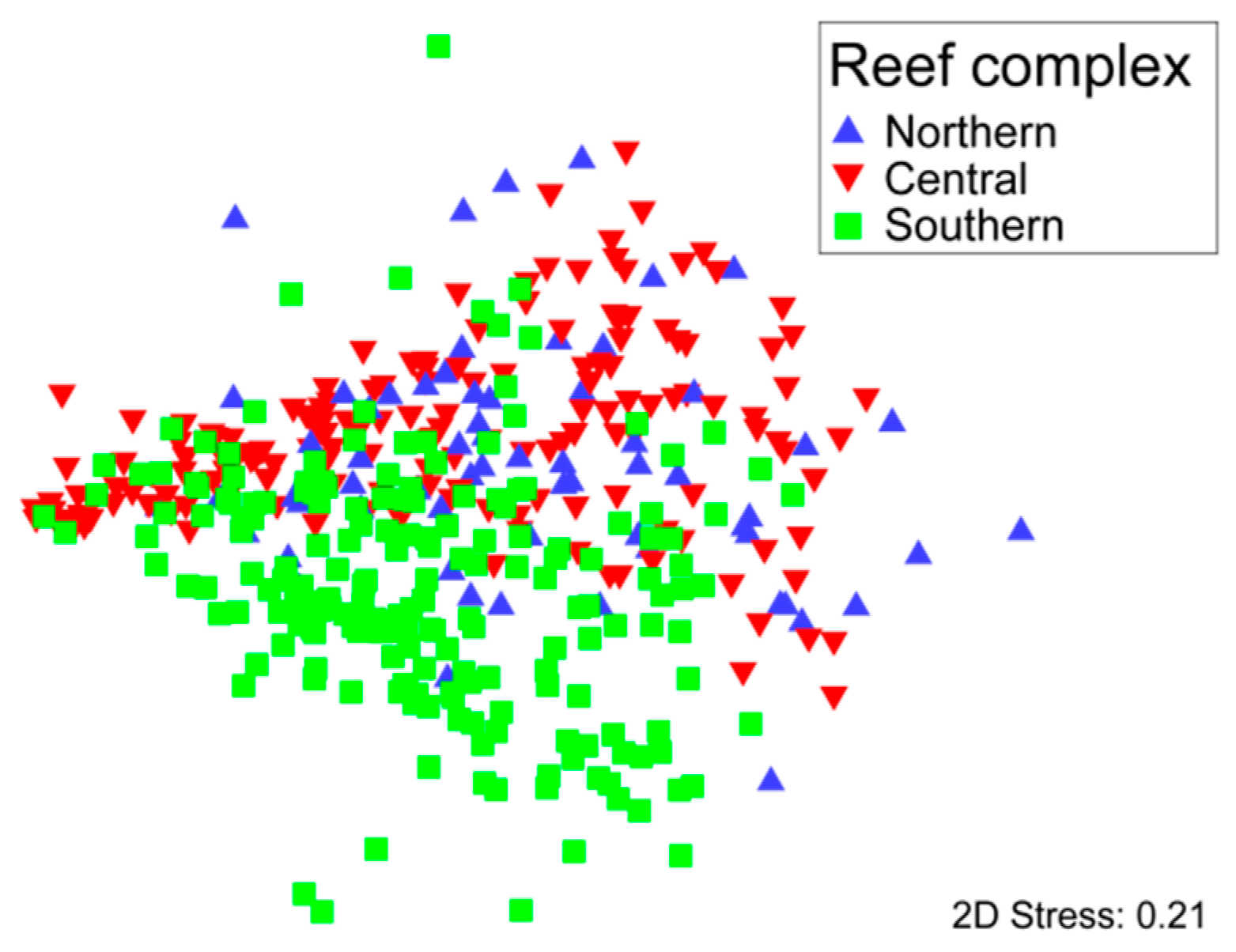

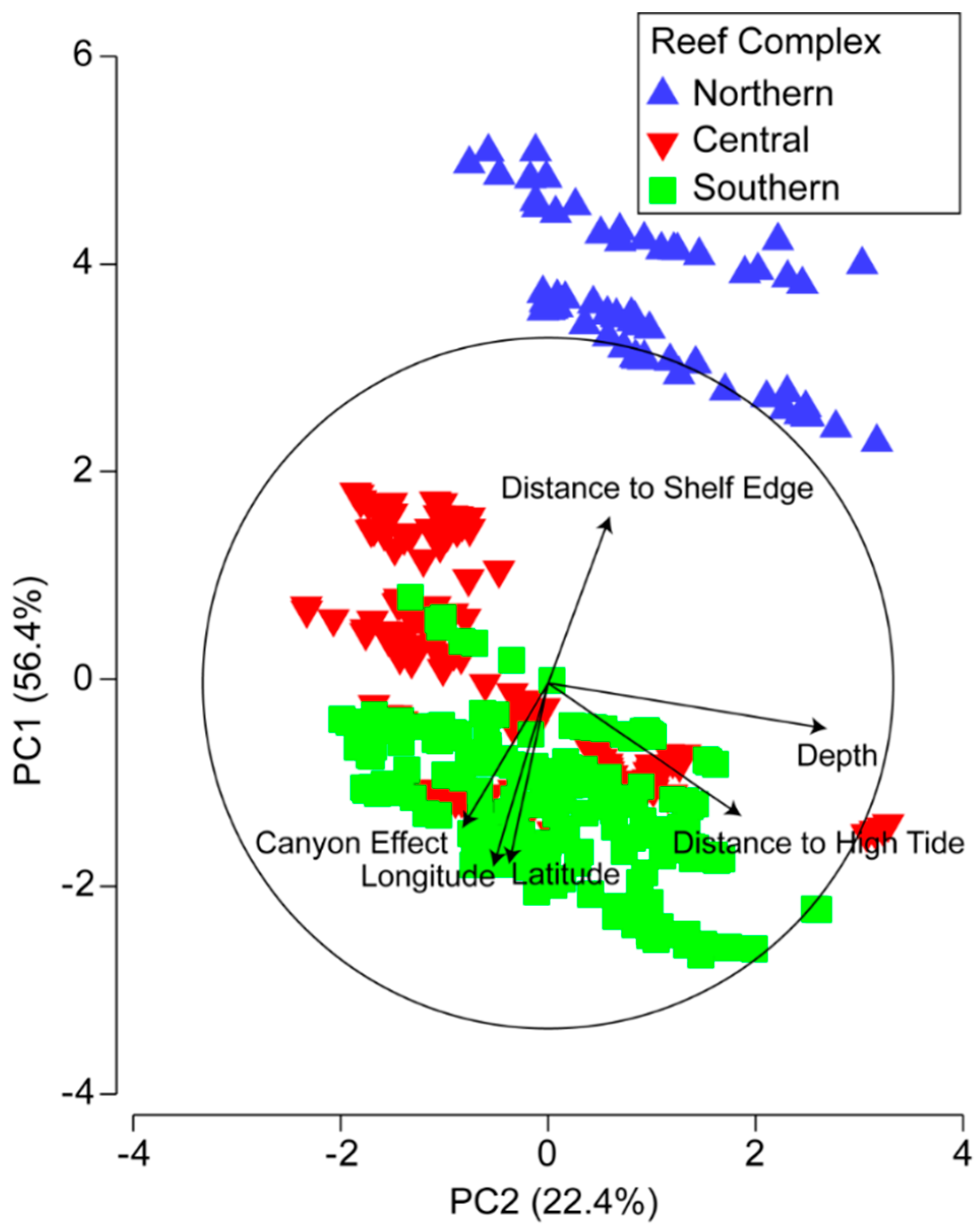
| Location | n | Canyon Effect | Depth (m) | Dist. to High Tide (m) | Dist. to Shelf Edge (m) | Latitude (km) | Longitude (km) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Min | Max | Ave ± SD | Min | Max | Ave ± SD | Min | Max | Ave ± SD | Min | Max | Ave ± SD | Min | Max | Ave ± SD | Min | Max | Ave ± SD | ||
| Region | 419 | 668 | 9842 | 4300 ± 2254 | 8 | 37 | 18 ± 5 | 512 | 708 | 1388 ± 474 | 1648 | 3543 | 2852 ± 888 | 2977 | 3086 | 3048 ± 32 | 11 | 39 | 30 ± 9 |
| Northern Complex | 64 | 668 | 907 | 812 ± 88 | 9 | 31 | 19 ± 5 | 516 | 1699 | 1079 ± 278 | 3543 | 6015 | 4563 ± 759 | 2977 | 2994 | 2988 ± 6 | 11 | 14 | 12 ± 1 |
| Central Complex | 159 | 2212 | 9842 | 5616 ± 2181 | 9 | 37 | 17 ± 5 | 512 | 2090 | 1181 ± 487 | 1648 | 3267 | 2445 ± 411 | 3015 | 3046 | 3037 ± 8 | 21 | 31 | 28 ± 3 |
| Southern Complex | 196 | 2716 | 6803 | 4372 ± 1274 | 8 | 31 | 18 ± 5 | 708 | 2385 | 1658 ± 355 | 1801 | 3949 | 2624 ± 466 | 3068 | 3086 | 3077 ± 5 | 36 | 39 | 37 ± 1 |
| Variable | PC1 | PC2 | PC3 | PC4 | PC5 | PC6 |
|---|---|---|---|---|---|---|
| Canyon effect | 0.391 | −0.231 | −0.629 | 0.462 | 0.427 | −0.056 |
| Depth | 0.126 | 0.775 | −0.294 | −0.428 | 0.337 | 0.019 |
| Dist. to high tide | 0.367 | 0.533 | 0.213 | 0.615 | −0.398 | 0.009 |
| Dist. to shelf edge | −0.453 | 0.168 | 0.405 | 0.411 | 0.659 | 0.009 |
| Latitude | 0.491 | −0.106 | 0.424 | −0.189 | 0.231 | −0.691 |
| Longitude | 0.500 | −0.149 | 0.359 | −0.147 | 0.243 | 0.720 |
| Eigenvalue | 3.38 | 1.34 | 0.807 | 0.243 | 0.218 | 0.00429 |
| % Variance explained | 56.4 | 22.4 | 13.5 | 4.1 | 3.6 | 0.1 |
| % Cumulative Variance | 56.4 | 78.8 | 92.2 | 96.3 | 99.9 | 100.0 |
| Environmental Variable | Adj. R2 | SS(Trace) | FPseudo | P(perm) | Proportion of Variance | Cumulative Variance Explained | Res. df |
|---|---|---|---|---|---|---|---|
| Marginal tests | |||||||
| Depth | 26422 | 19.364 | 0.0001 | 0.044376 | |||
| Distance to high tide | 19859 | 14.388 | 0.0001 | 0.033353 | |||
| Distance to shelf edge | 10190 | 7.2612 | 0.0001 | 0.017115 | |||
| Canyon effect | 10399 | 7.4126 | 0.0001 | 0.017466 | |||
| Sequential tests | |||||||
| Depth | 0.042085 | 26422 | 19.364 | 0.0001 | 0.044376 | 0.044376 | 417 |
| +Distance to high tide | 0.076918 | 22005 | 16.736 | 0.0001 | 0.036958 | 0.081335 | 416 |
| +Distance to shelf edge | 0.098627 | 14148 | 11.019 | 0.0001 | 0.023762 | 0.1051 | 415 |
| +Canyon Effect | 0.12949 | 19481 | 15.711 | 0.0001 | 0.032719 | 0.13782 | 414 |
| Variable | Adj. R2 | SS(Trace) | FPseudo | P(perm) | Proportion of Variance | Cumulative Variance Explained | Res. df | Regr. df |
|---|---|---|---|---|---|---|---|---|
| Group (Spatial parameters) | 0.39261 | 2.99E+05 | 4.6025 | 0.0001 | 0.50159 | 0.50159 | 343 | 76 |
| +Depth | 0.40781 | 8270.5 | 9.8047 | 0.0001 | 0.01389 | 0.51548 | 342 | 77 |
| +Distance to high tide | 0.41306 | 3394.6 | 4.0603 | 0.0001 | 0.005701 | 0.52118 | 341 | 78 |
| +Distance to shelf edge | 0.41735 | 2914.7 | 3.512 | 0.0002 | 0.004895 | 0.52608 | 340 | 79 |
| +Canyon effect | 0.42144 | 2804.6 | 3.4032 | 0.0002 | 0.00471 | 0.53079 | 339 | 80 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porter, S.N.; Schleyer, M.H. Environmental Variation and How its Spatial Structure Influences the Cross-Shelf Distribution of High-Latitude Coral Communities in South Africa. Diversity 2019, 11, 57. https://doi.org/10.3390/d11040057
Porter SN, Schleyer MH. Environmental Variation and How its Spatial Structure Influences the Cross-Shelf Distribution of High-Latitude Coral Communities in South Africa. Diversity. 2019; 11(4):57. https://doi.org/10.3390/d11040057
Chicago/Turabian StylePorter, Sean N., and Michael H. Schleyer. 2019. "Environmental Variation and How its Spatial Structure Influences the Cross-Shelf Distribution of High-Latitude Coral Communities in South Africa" Diversity 11, no. 4: 57. https://doi.org/10.3390/d11040057
APA StylePorter, S. N., & Schleyer, M. H. (2019). Environmental Variation and How its Spatial Structure Influences the Cross-Shelf Distribution of High-Latitude Coral Communities in South Africa. Diversity, 11(4), 57. https://doi.org/10.3390/d11040057





