New Evidence of Marine Fauna Tropicalization off the Southwestern Iberian Peninsula (Southwest Europe)
Abstract
1. Introduction
2. Materials and Methods
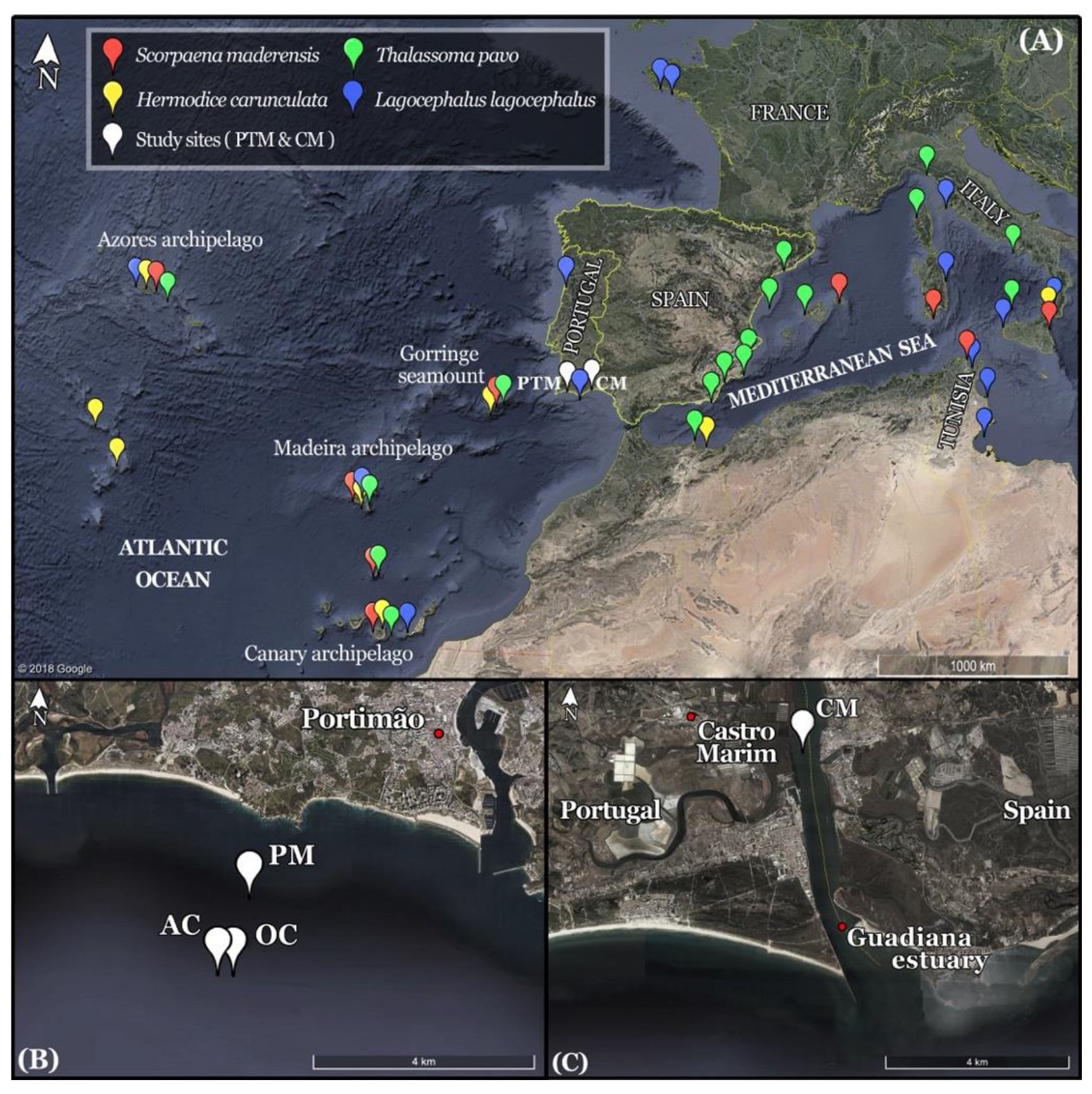
2.1. Study Area
2.2. Data Collection
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoegh-Guldberg, O.; Bruno, J.F. The impact of climate change on the world’s marine ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.N.; Morri, C. Global sea warming and “tropicalization” of the Mediterranean Sea: Biogeographic and ecological aspects. Biogeographia 2003, 24, 319–327. [Google Scholar] [CrossRef]
- Vergés, A.; Steinberg, P.D.; Hay, M.E.; Poore, A.G.; Campbell, A.H.; Ballesteros, E.; Heck, K.L., Jr.; Booth, D.J.; Coleman, M.A.; Feary, D.A.; et al. The tropicalization of temperate marine ecosystems: Climate-mediated changes in herbivory and community phase shifts. Proc. R. Soc. B 2014, 281, 20140846. [Google Scholar] [CrossRef] [PubMed]
- Wernberg, T.; Bennett, S.; Babcock, R.C.; de Bettignies, T.; Cure, K.; Depczynski, M.; Dufois, F.; Fromont, J.; Fulton, C.J.; Hovey, R.K.; et al. Climate driven regime shift of a temperate marine ecosystem. Science 2016, 353, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Vergés, A.; Doropoulos, C.; Malcolm, H.A.; Skye, M.; Garcia-Pizá, M.; Marzinelli, E.M.; Campbell, A.H.; Ballesteros, E.; Hoey, A.S.; Vila-Concejo, A.; et al. Long-term empirical evidence of ocean warming leading to tropicalization of fish communities, increased herbivory, and loss of kelp. Proc. Natl. Acad. Sci. USA 2016, 113, 13791–13796. [Google Scholar] [CrossRef] [PubMed]
- Zarco-Perello, S.; Wernberg, T.; Langlois, T.J.; Vanderklift, M.A. Tropicalization strengthens consumer pressure on habitat forming seaweeds. Sci. Rep. 2017, 7, 820. [Google Scholar] [CrossRef] [PubMed]
- Yamano, H.; Sugihara, K.; Nomura, K. Rapid poleward range expansion of tropical reef corals in response to rising sea surface temperatures. Geophys. Res. Lett. 2011, 38, L04601. [Google Scholar] [CrossRef]
- Nakamura, Y.; Feary, D.A.; Kanda, M.; Yamaoka, K. Tropical fishes dominate temperate reef fish communities within Western Japan. PLoS ONE 2013, 8, e81107. [Google Scholar] [CrossRef] [PubMed]
- Heck, K.L., Jr.; Fodrie, F.J.; Madsen, S.; Baillie, C.J.; Byron, D.A. Seagrass consumption by native and a tropically associated fish species: Potential impacts of the tropicalization of the northern Gulf of Mexico. Mar. Ecol. Prog. Ser. 2015, 520, 165–173. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Morri, C. Southern species in the Ligurian Sea (northern Mediterranean): New records and a review. Bollettino dei Musei e degli Istituti Biologici dell’Università di Genova 1994, 58/59, 181–197. [Google Scholar]
- Bianchi, C.N.; Caroli, F.; Guidetti, P.; Morri, C. Seawater warming at the northern reach for southern species: Gulf of Genoa, NW Mediterranean. J. Mar. Biol. Assoc. U. K. 2018, 98, 1–12. [Google Scholar] [CrossRef]
- Lima, F.P.; Ribeiro, P.A.; Queiroz, N.; Hawkins, S.J.; Santos, A.M. Do distributional shifts of northern and southern species of algae match the warming pattern? Glob. Chang. Biol. 2007, 13, 2592–2604. [Google Scholar] [CrossRef]
- Nicastro, K.R.; Zardi, G.I.; Teixeira, S.; Neiva, J.; Serrao, E.A.; Pearson, G.A. Shift happens: Trailing edge contraction associated with recent warming trends threatens a distinct genetic lineage in the marine macroalga Fucus vesiculosus. BMC Biol. 2013, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, C.R.; Nicastro, K.R.; Serrão, E.A.; Zardi, G.I. First record of the brown mussel (Perna perna) from the European Atlantic coast. Mar. Biodivers. Rec. 2012, 5, e39. [Google Scholar] [CrossRef]
- Abecasis, D.; Bentes, L.; Ribeiro, J.; Machado, D.; Oliveira, F.; Veiga, P.; Gonçalves, J.M.S.; Erzini, K. First record of the Mediterranean parrotfish, Sparisoma cretense in Ria Formosa (south Portugal). Mar. Biodivers. Rec. 2008, 1, e27. [Google Scholar] [CrossRef]
- Saldanha, L. Fauna Submarina Atlântica, 4th ed.; Publicações Europa-América: Lisboa, Portugal, 1995; 361p. [Google Scholar]
- Louisy, P. Europe and Mediterranean Marine Fish Identification Guide; Ulmer: Paris, France, 2015; 512p. [Google Scholar]
- Harmelin-Vivien, M.L.; Harmelin, G.; Almeida, A.J. Structure of fish assemblages on coastal rocky shores of the Azores. Boletim do Museu Municipal do Funchal 2001, 6, 127–138. [Google Scholar]
- Tuya, F.; Boyra, A.; Sanchez-Jerez, P.; Barbera, C.; Haroun, R.J. Relationships between rocky-reef fish assemblages, the sea urchin Diadema antillarum and macroalgae throughout the Canarian Archipelago. Mar. Ecol. Prog. Ser. 2004, 278, 157–169. [Google Scholar] [CrossRef]
- Yáñez-Rivera, B.; Salazar-Vallejo, S.I. Revision of Hermodice Kinberg, 1857 (Polychaeta: Amphinomidae). Sci. Mar. 2011, 75, 251–262. [Google Scholar] [CrossRef]
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, A.Z.; Finlayson, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A.; et al. Marine ecoregions of the world: A bioregionalization of coastal and shelf areas. BioScience 2007, 57, 573–583. [Google Scholar] [CrossRef]
- Hureau, J.C.; Litvinenko, N.I. Scorpaenidae. In Fishes of the North-Eastern Atlantic and the Mediterranean; Whitehead, P.J.P., Bauchot, M.L., Hureau, J.C., Nielsen, J., Tortonese, E., Eds.; UNESCO: Paris, France, 1986; Volume 3, pp. 1211–1229. [Google Scholar]
- Ahnelt, V.H. Erstnachweis von Scorpaena maderensis Val., 1833 (Pisces, Scorpaenidae) für Kreta und Rhodos (Griechenland) mit Angaben zum bisher einzigen Fund dieser Art aus der Adria. Annalen des Naturhistorischen Museums in Wien 1983, 85B, 9–11. [Google Scholar]
- La Mesa, M.; La Mesa, G.; Micalizzi, M. Age and growth of Madeira scorpionfish, Scorpaena maderensis Valenciennes, 1833, in the central Mediterranean. Fish. Res. 2005, 74, 265–272. [Google Scholar] [CrossRef]
- Falzon, M.A. First records of Scorpaena maderensis (Pisces Scorpaeniformes Scorpaenidae) in inshore Maltese waters. Naturalista Siciliano 2011, 35, 419–423. [Google Scholar]
- Cardona, L.; Elices, M. Datos sobre la presencia en el litoral de Menorca (islas Baleares, Mediterráneo Occidental) de Parablennius pilicornis (Cuvier, 1829) y Scorpaena maderensis Valenciennes 1833. Bolletí de la Societat d’Història Natural de les Balears 2000, 43, 33–38. [Google Scholar]
- Leitão, F.; Baptista, V.; Zeller, D.; Erzini, K. Reconstructed catches and trends for mainland Portugal fisheries between 1938 and 2009: Implications for sustainability, domestic fish supply and imports. Fish. Res. 2014, 155, 33–50. [Google Scholar] [CrossRef]
- Francour, P.; Boudouresque, C.F.; Harmelin, J.G.; Harmelin-Vivien, M.L.; Quignard, J.P. Are the Mediterranean waters becoming warmer? Information from biological indicators. Mar. Pollut. Bull. 1994, 28, 523–526. [Google Scholar] [CrossRef]
- Vacchi, M.; Morri, C.; Modena, M.; La Mesa, G.; Bianchi, C.N. Temperature changes and warm-water species in the Ligurian Sea: The case of the ornate wrasse Thalassoma pavo (Linnaeus, 1758). Archo Oceanogr. Limnol. 2001, 22, 149–154. [Google Scholar]
- Warner, R.R.; Hoffman, S. Local population size as a determinant of mating system and sexual composition in two tropical marine fishes (Thalassoma spp.). Evolution 1980, 34, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Witman, J.D. Effects of predation by the fireworm Hermodice carunculata on milleporid hydrocorals. Bull. Mar. Sci. 1988, 42, 446–458. [Google Scholar]
- Chatzigeorgiou, G.; Sarropoulou, E.; Vasileiadou, K.; Brown, C.; Faulwetter, S.; Kotoulas, G.; Arvanitidis, C.D. Community structure and population genetics of Eastern Mediterranean polychaetes. Front. Mar. Sci. 2014, 1, 1–8. [Google Scholar] [CrossRef]
- Verdes, A.; Simpson, D.; Holford, M. Are Fireworms Venomous? Evidence for the Convergent Evolution of Toxin Homologs in Three Species of Fireworms (Annelida, Amphinomidae). Genome Biol. Evol. 2018, 10, 249–268. [Google Scholar] [CrossRef]
- Gonçalves, J.M.S.; Bispo, J.; Silva, J. Underwater survey of ichthyofauna of eastern Atlantic seamounts: Gettysburg and Ormonde (Gorringe Bank). Arch. Fish. Mar. Res. 2004, 51, 233–240. [Google Scholar]
- Abecasis, D.; Cardigos, F.; Almada, F.; Gonçalves, J.M.S. New records on the ichthyofauna of the Gorringe seamount (Northeastern Atlantic). Mar. Biol. Res. 2009, 5, 605–611. [Google Scholar] [CrossRef]
- OCEANA. The Seamounts of the Gorringe Bank; Fondazione Ermenegildo Zegna: Trivero, Italy, 2005; 72p. [Google Scholar]
- Bianchi, C.N. Biodiversity issues for the forthcoming tropical Mediterranean Sea. Hydrobiologia 2007, 580, 7–21. [Google Scholar] [CrossRef]
- Barton, E.D. Encyclopedia of Ocean Sciences; Steele, J.H., Thorpe, S.A., Turekian, K.K., Eds.; Academic Press: Florence, Italy, 2001; pp. 380–389. [Google Scholar]
- Pingree, R. Ocean structure and climate (Eastern North Atlantic): In situ measurement and remote sensing (altimeter). J. Mar. Biol. Assoc. U. K. 2002, 82, 681–707. [Google Scholar] [CrossRef]
- Muñoz, M.; Casadevall, M.; Bonet, S. The ovarian morphology of Scorpaena notata shows a specialized mode of oviparity. J. Fish Biol. 2002, 61, 877–887. [Google Scholar] [CrossRef]
- Muñoz, M.; Sàbat, M.; Vila, S.; Casadevall, M. Annual reproductive cycle and fecundity of Scorpaena notata (Teleostei: Scorpaenidae). Sci. Mar. 2005, 69, 555–562. [Google Scholar] [CrossRef]
- Raventós, N.; Macpherson, E. Planktonic larval duration and settlement marks on the otoliths of Mediterranean littoral fishes. Mar. Biol. 2001, 138, 1115–1120. [Google Scholar]
- Ahrens, J.; Borda, E.; Barroso, R.; Paiva, P.C.; Campbell, A.M.; Wolf, A.; Nugues, M.M.; Rouse, G.W.; Schulze, A. The curious case of Hermodice carunculata (Annelida: Amphinomidae): Evidence for genetic homogeneity throughout the Atlantic Ocean and adjacent basins. Mol. Ecol. 2013, 22, 2280–2291. [Google Scholar] [CrossRef]
- Schulze, A.; Grimes, C.J.; Rudek, T.E. Tough, armed and omnivorous: Hermodice carunculata (Annelida: Amphinomidae) is prepared for ecological challenges. J. Mar. Biol. Assoc. U. K. 2017, 97, 1075–1080. [Google Scholar] [CrossRef]
- Matheson, C. Records of the Globefish and Filefish. Nature 1950, 165, 193. [Google Scholar] [CrossRef]
- Went, A.E.J. Recent Irish records of rare fish. Nature 1950, 165, 1025. [Google Scholar] [CrossRef]
- Wheeler, A.; Blacker, R.W. Rare and little-known fishes in British seas in 1968 and 1969. J. Fish Biol. 1972, 4, 141–170. [Google Scholar] [CrossRef]
- Erzini, K.; Monteiro, P.; Araújo, A.; Castro, M. Limited mid-water scavenging of trawl discards. J. Mar. Biol. Assoc. U. K. 2003, 83, 731–734. [Google Scholar] [CrossRef]
- Dulčić, J.; Pallaoro, A. First record of the oceanic puffer (Lagocephalus lagocephalus lagocephalus Linnaeus, 1758), for the Adriatic Sea. J. Appl. Ichthyol. 2006, 22, 94–95. [Google Scholar] [CrossRef]
- Hardy, G.; Jing, L.; Leis, J.L.; Liu, M.; Matsuura, K.; Shao, K. Lagocephalus lagocephalus. In The IUCN Red List of Threatened Species; IUCN: Gland, Switzerland, 2014; e.T190464A19929421. [Google Scholar]
- Farrag, M.; El-Haweet, A.A.K.; Akel, E.-S.k.A.; Moustafa, M.A. Occurrence of puffer fishes (Tetraodontidae) in the eastern Mediterranean, Egyptian coast—filling in the gap. BioInvasions Rec. 2016, 5, 47–54. [Google Scholar] [CrossRef]
- Erguden, D.; Gurlek, M.; Turan, C. First occurrence of the oceanic puffer, Lagocephalus lagocephalus (Linnaeus, 1758) in Iskenderun Bay, north-eastern Mediterranean, Turkey. J. Appl. Ichthyol. 2017, 33, 801–803. [Google Scholar] [CrossRef]
- Guardone, L.; Gasperetti, L.; Maneschi, A.; Ricci, E.; Susini, F.; Guidi, A.; Armani, A. Toxic invasive pufferfish (Tetraodontidae family) along Italian coasts: Assessment of an emerging public health risk. Food Control 2018, 91, 330–338. [Google Scholar] [CrossRef]
- Pombo, L.; Elliott, M.; Rebelo, J.E. Changes in the fish fauna of the Ria de Aveiro estuarine lagoon (Portugal) during the twentieth century. J. Fish Biol. 2002, 61, 167–181. [Google Scholar] [CrossRef]
- Yang, J. The dominant fish fauna in the North Sea and its determination. J. Fish Biol. 1982, 20, 635–643. [Google Scholar]
- Hansen, J.; Sato, M.; Ruedy, R.; Lo, K.; Lea, D.W.; Medina-Elizade, M. Global temperature change. Proc. Natl. Acad. Sci. USA 2006, 103, 14288–14293. [Google Scholar] [CrossRef] [PubMed]
- Quéro, P.; Bellail, R.; Casamajor, M.; Leaute, J.; Morandeau, G.; Morinière, P.; Spitz, J.; Vayne, J. Observations ichtyologiques effectuées en 2004. Ann. Soc. Sci. Nat. Charente-Maritime 2005, 9, 483–490. [Google Scholar]
- Saoudi, M.; Abdelmouleh, A.; Kammoun, W.; Ellouze, F.; Jamoussi, K.; El Feki, A. Toxicity assessment of the puffer fish Lagocephalus lagocephalus from the Tunisian coast. C. R. Biol. 2008, 331, 611–616. [Google Scholar] [CrossRef]
- Tsiamis, K.; Aydogan, O.; Bailly, N.; Balistreri, P.; Bariche, M.; Carden-Noad, S.; Corsini-Foka, M.; Crocetta, F.; Davidov, B.; Dimitriadis, C.; et al. New Mediterranean biodiversity records (July 2015). Mediterr. Mar. Sci. 2015, 16, 472–488. [Google Scholar] [CrossRef]
- Saoudi, M.; Messarah, M.; Boumendjel, A.; Abdelmouleh, A.; Kammoun, W.; Jamoussi, K.; Feki, A.E. Extracted tetrodotoxin from puffer fish Lagocephalus lagocephalus induced hepatotoxicity and nephrotoxicity to wistar rats. Afr. J. Biotechnol. 2011, 10, 8140–8145. [Google Scholar]
- Oliveira, F.; Monteiro, P.; Afonso, C.; Veiga, P.; Bentes, L.; Calado, R.; Gonçalves, J.M.S. First record of Calcinus tubularis on the southern coast of Portugal (Crustacea: Decapoda: Anomura: Diogenidae). Mar. Biodivers. Rec. 2011, 4, e21. [Google Scholar] [CrossRef]
- Morais, P.; Teodósio, M.A. The transatlantic introduction of weakfish Cynoscion regalis (Bloch & Schneider, 1801) (Sciaenidae, Pisces) into Europe. BioInvasions Rec. 2016, 5, 259–265. [Google Scholar]
- Morais, P.; Cerveira, I.; Teodósio, M.A. An Update on the Invasion of Weakfish Cynoscion regalis (Bloch & Schneider, 1801) (Actinopterygii: Sciaenidae) into Europe. Diversity 2017, 9, 47. [Google Scholar]
- Morais, P.; Gaspar, M.; Garel, E.; Baptista, V.; Cruz, J.; Cerveira, I.; Leitão, F.; Teodósio, M.A. The Atlantic blue crab Callinectes sapidus Rathbun, 1896 expands its non-native distribution into the Ria Formosa lagoon and the Guadiana estuary (SW-Iberian Peninsula, Europe). BioInvasions Rec. 2019, 8. in press. [Google Scholar]




| Morphometric Characteristics | Meristic Characteristics | ||
|---|---|---|---|
| Total length | 37.2 cm | Anal fin | 13 |
| Fork length | 34.5 cm | Caudal fin | II + 11 + II |
| Standard length | 30.6 cm | Dorsal fin | 16 |
| Body depth | 7.3 cm | Pectoral fin | 16 |
| Head length | 8.3 cm | ||
| Snout-eye length | 3.5 cm | Total fresh weight | |
| Eye diameter | 1.7 cm | Weight | 453 g |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Encarnação, J.; Morais, P.; Baptista, V.; Cruz, J.; Teodósio, M.A. New Evidence of Marine Fauna Tropicalization off the Southwestern Iberian Peninsula (Southwest Europe). Diversity 2019, 11, 48. https://doi.org/10.3390/d11040048
Encarnação J, Morais P, Baptista V, Cruz J, Teodósio MA. New Evidence of Marine Fauna Tropicalization off the Southwestern Iberian Peninsula (Southwest Europe). Diversity. 2019; 11(4):48. https://doi.org/10.3390/d11040048
Chicago/Turabian StyleEncarnação, João, Pedro Morais, Vânia Baptista, Joana Cruz, and Maria Alexandra Teodósio. 2019. "New Evidence of Marine Fauna Tropicalization off the Southwestern Iberian Peninsula (Southwest Europe)" Diversity 11, no. 4: 48. https://doi.org/10.3390/d11040048
APA StyleEncarnação, J., Morais, P., Baptista, V., Cruz, J., & Teodósio, M. A. (2019). New Evidence of Marine Fauna Tropicalization off the Southwestern Iberian Peninsula (Southwest Europe). Diversity, 11(4), 48. https://doi.org/10.3390/d11040048









