Conservation and Utilization of Livestock Genetic Diversity in the United States of America through Gene Banking
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Current Collection
3.2. Analyses of Breed Diversity
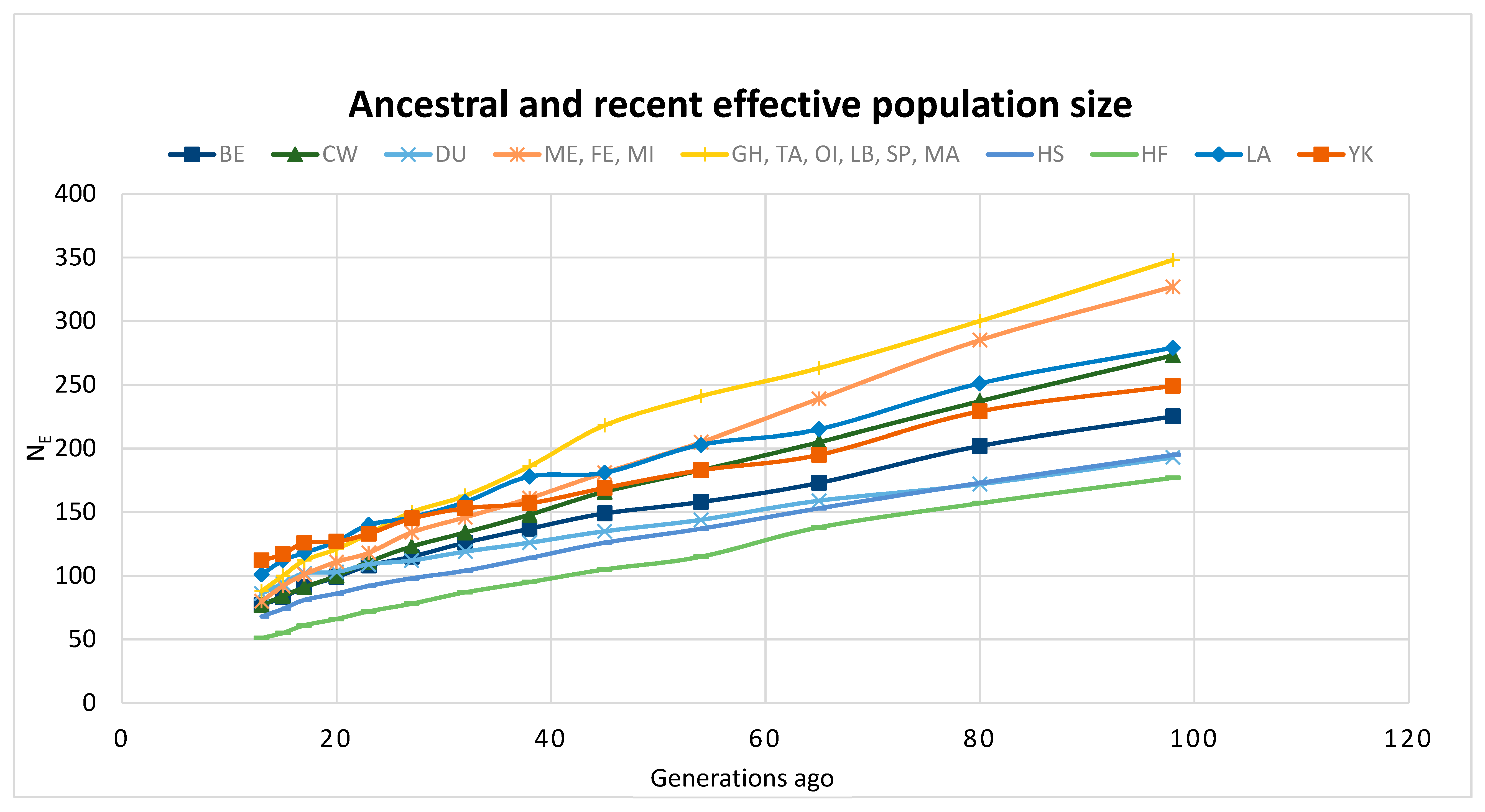
3.3. Future Work to Understand Genetic Diversity and Enhance Collections
3.4. Further Gene Bank Use
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization. World Watch List for Domestic Animal Diversity, 1st ed.; Loftus, R., Scherf, B., Eds.; FAO: Rome, Italy, 1993. [Google Scholar]
- Food and Agriculture Organization. The First Report on the State of the World’s Animal Genetic Resources for Food and Agriculture; Rischkowsky, B., Pilling, D., Eds.; FAO: Rome, Italy, 2007. [Google Scholar]
- FAO Commission on Genetic Resources for Food and Agriculture Assessments: Rome, Italy. 2015. Available online: http://www.fao.org/3/a-14787e/index.html (accessed on 30 November 2019).
- Falconer, D.; Mackay, T. Introduction to Quantitative Genetics, 4th ed.; Longman Group Ltd.: Harlow, UK, 1996. [Google Scholar]
- Eynard, S.E.; Windig, J.J.; Hulsegge, I.; Hiemstra, S.J.; Calus, M.P. The impact of using old germplasm on genetic merit and diversity—A cattle breed case study. J. Anim. Breed. Genet. 2018, 135, 311–322. [Google Scholar] [CrossRef] [PubMed]
- The Role of Genetic Drift and Selection Intensity on Small Populations. Available online: http://www.wcgalp.org/system/files/proceedings/2018/role-genetic-drift-and-selection-intensity-small-populations-sensitivity-and-genomic-analysis.pdf (accessed on 30 November 2019).
- Maiwashe, N.A.; Blackburn, H.D. Genetic diversity in and conservation strategy considerations for Navajo Churro sheep. Anim. Sci. 2004, 82, 2900–2905. [Google Scholar] [CrossRef] [PubMed]
- Welsh, C.S.; Stewart, T.S.; Schwab, C.; Blackburn, H.D. Pedigree analysis of 5 swine breeds in the United States and the implications for genetic conservation. J. Anim. Sci. 2010, 88, 1610–1618. [Google Scholar] [CrossRef] [PubMed]
- Norton, R. Agricultural Development Policy: Concepts and Experiences; FAO: Rome, Italy, 2004. [Google Scholar]
- Blackburn, H.D. Integrating policies for the management of animal genetic resources with demand for livestock products and environmental sustainability. Anim. Genet. Resour. Inf. 2007, 41, 53–64. [Google Scholar] [CrossRef]
- Blackburn, H.D. Assuring genetic diversity with reproductive technologies: Development of national animal genetic resource programs. Reprod. Fertil. Dev. 2004, 16, 27–32. [Google Scholar] [CrossRef]
- Blackburn, H.D. Genebank development for the conservation of livestock genetic resources in the United States of America. Livest. Sci. 2009, 120, 196–203. [Google Scholar] [CrossRef]
- Blackburn, H.D. Biobanking genetic material for agricultural animal species. Annu. Rev. Anim. Biosci. 2018, 6, 69–82. [Google Scholar] [CrossRef]
- Ouendeba, B.; Ejeta, G.; Hanna, W.; Kumar, A. Diversity among African pearl millet landrace populations. Crop Sci. 1995, 35, 919–924. [Google Scholar] [CrossRef]
- Animal Genetic Resources. Available online: https://agrin.ars.usda.gov/database_collaboration_page_dev) (accessed on 18 November 2019).
- Yue, X.-P.; Dechow, C.; Liu, W.-S. A limited number of Y chromosome lineages is present in North American Holsteins. J. Dairy Sci. 2015, 98, 2738–2745. [Google Scholar] [CrossRef]
- Dechow, C.D.; Liu, W.S.; Idun, J.S.; Maness, B. Short communication: Two dominant paternal lineages for North American Jersey artificial insemination sires. J. Dairy Sci. 2018, 101, 2281–2284. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Cryoconservation of Animal Genetic Resources; FAO Animal Production and Health Guidelines No. 12; FAO: Rome, Italy, 2012. [Google Scholar]
- Leroy, G.; Danchin-Burge, C.; Verrier, E. Impact of the use of cryobank samples in a selected cattle breed: A simulation study. Genet. Sel. Evol. 2011, 43, 36. [Google Scholar] [CrossRef]
- Dechow, C.D.; Liu, W.S.; Specht, L.W.; Blackburn, H. Reconstitution and modernization of lost Holstein male lineages using samples from a gene bank. J. Dairy Sci. under review.
- Blackburn, H.D.; Paiva, S.R.; Wildeus, S.; Getz, W.; Stobart, R.; Bixby, D.; Purdy, P.H.; Welsh, C.S.; Spiller, S.F.; Brown, M.A. Genetic Structure and Diversity among U.S. sheep breeds: Identification of the major gene pools. J. Anim. Sci. 2011, 89, 2336–2348. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, H.D.; Toishibekov, Y.; Toishibekov, M.; Welsh, C.S.; Spiller, S.F.; Brown, M.; Paiva, S.R. Genetic diversity of Ovis aries populations near domestication centers and in the New World. Genetica 2011, 139, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Do Prado, P.T.; Faria, D.A.; McManus, C.; Lanari, M.R.; Esquivel, L.C.; Cascante, M.I.; Alfaro, E.J.; Mendez, A.; Faco, O.; de Moraes, S.K.; et al. New world goat populations are a genetically diverse reservoir for future use. Sci. Rep. 2019, 9, 1476. [Google Scholar] [CrossRef]
- Faria, D.A.; Wilson, C.; Paiva, S.; Blackburn, H.D. Assessing Sus scrofa diversity among continental United States, and Pacific islands populations using molecular markers from a gene banks collection. Sci. Rep. 2019, 9, 3173. [Google Scholar] [CrossRef]
- Wilson, C.S.; Rohrer, G.A.; Newcom, D.W.; Blackburn, H.D. 217 Capturing genetic diversity—an assessment of the nation’s gene bank in securing Duroc pigs. J. Anim. Sci. 2019, 97, 46. [Google Scholar]
- Manel, S.; Holderegger, R. Ten years of landscape genetics. Trends Ecol. Evol. 2013, 28, 10. [Google Scholar] [CrossRef]
- Cartwright, T.C. Responses of beef cattle to high ambient temperatures. J. Anim. Sci. 1955, 14, 350–362. [Google Scholar] [CrossRef]
- Kroger, M.; Burns, W.; Pahnish, O.; Butts, W. Genotype by environment interactions in Hereford cattle: I. Reproductive Traits. J. Anim. Sci. 1979, 49, 396–402. [Google Scholar] [CrossRef]
- Bohmanova, J.; Misztal, I.; Tsuruta, S.; Norman, H.; Lawlor, T. Genotype by environment interaction due to heat stress. J. Dairy Sci. 2008, 91, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, H.D.; Pittroff, W. Biologically based coefficients for partitioning lamb and wool production costs. J. Anim. Sci. 1999, 77, 1353–1363. [Google Scholar] [CrossRef] [PubMed]
- Paiva, S.R.; Hermuche, P.; Costa Junior, J.; McManus, C.; Fontes Guimaraes, R.; Carvalho Junior, O.; Wislon, C.; Krehbiel, B.; Blackburn, H.B.; Auckland, N.Z. Gene bank collection strategies based upon geographic and environmental indicators. World Cong. Gen. Appl. Livstk Prod. 2017, 11, 747. [Google Scholar]
- Blackburn, H.D.; Krehbiel, B.; Ericsson, S.A.; Wilson, C.S.; Caetano, A.R.; Paiva, S.R. A fine structure genetic analysis evaluating ecoregional adaptability of a Bos taurus breed (Hereford). PLoS ONE 2017, 12, e0176474. [Google Scholar] [CrossRef] [PubMed]
| Registration /Census Category (Animals) | Beef Cattle | Dairy Cattle | Sheep | Goats | Pigs |
|---|---|---|---|---|---|
| <1000 | 23—Belgian Blue, Blonde d’ Aquitaine, Chianina, Criollo, Devon, Florida Cracker/Pineywoods, Galloway, Marchigiana, Masona, Murray Grey, Normande, Parthenais, Red Poll, Romagnola, Romosinuano, Senepol, South Devon, Tarentaise, Tuli, White Park, Nelore, Tabapua, Indu Brazil | 7—Dutch Belted, Herens, Kerry, Meuse-Rhine-Yssel, Milking Devon, Norwegian Red, Randall Lineback | 24—Barbados Blackbelly, Black Welsh Mountain, Bluefaced Leicester, Border Leicester, Cotswold, Delaine Merino, Finnsheep, Gulf Coast Native, Hog Island, Icelandic, Jacob, Karakul, Leicester Longwool, Lincoln, Merino, Navajo Churro, North Country Cheviot, Romadale, Santa Cruz Island, Scottish Blackface, Shetland, Soay, Saint Croix, Texel | 4—Kiko, Sable, San Clemente, Savanna | 10—Fengjing, Gloucestershire Old Spot, Guinea Hog, Mangalitsa, Meishan, Minzu, Mulefoot, Ossabaw Island, Red Wattle, Saddleback |
| <5,000 | 12—Belted Galloway, Braunvieh, British White, Highland, Piedmontese, Pinzgauer, Texas Longhorn, Wagyu, Braford, Simbrah, Red Brangus, Santa Gertrudis | 4—Ayrshire, Dexter, Guernsey, Milking Shorthorn | 6—Columbia, Corriedale, Polypay, Romanov, Shropshire, Targhee | 6—Lamancha, Myotonic, Nigerian Dwarf, Oberhasli, Saanen, Togenburg | 2—Tammworth, Hereford |
| <20,000 | 6—Limousin, Maine-Anjou, Salers, Shorthorn, Beefmaster, Brahman | 1—Brown Swiss | 7—Dorper, Dorset, Hampshire, Katahdin, Southdown, Suffolk, Rambouillet | 2—Alpine, Nubian | 3—Chester White, Pietrain, Poland China |
| >20,000 | 7—Angus, Charolais, Gelbvieh, Hereford, Red Angus, Brangus, Simmental | 2—Holstein, Jersey | 0 | 3—Angora, Boer, Spanish | 6—Berkshire, Duroc, Hampshire, Landrace, Spotted, Yorkshire |
| Registration /Census Category (Animals) | Collection Use | Frequency of Collection | Average Collection Completeness (Minimum Target Goal *), % | Collection Issues | Special Attributes |
|---|---|---|---|---|---|
| <1000 | C, G, E, R, B | Gaps in time between collections | 45.3 | Small population sizes, geographic separation of breeders | original imports, small farmer |
| <5000 | C, G, E, R, B | Gaps in time between collections | 75.1 | Small population sizes, geographic separation of breeders | original imports, small farmer |
| <20,000 | C, G, E, R, B | Continuous | 86.1 | Short generation intervals, fine scale sampling possible | Time span, original imports |
| >20,000 | C, G, E, R, B | Continuous | 97.7 | Short generation intervals, fine scale sampling possible | Time span, original imports |
| Registration/Census Category (Animals) | Breed | Animal Number | Units of Germplasm | Semen | Ovaries | Testes | Somatic Cells | Collection Completeness (%) |
|---|---|---|---|---|---|---|---|---|
| <1000 | Crevecoeur | 100 | 170 | 52 | 48 | 70 | 100 | |
| <1000 | Aseel | 97 | 161 | 56 | 41 | 64 | 100 | |
| <1000 | Transylvania Naked Neck | 2 | 46 | 46 | 6 | |||
| <1000 | Redcap | 4 | 8 | 2 | 2 | 4 | 9 | |
| <1000 | Spanish | 7 | 8 | 8 | 10 | |||
| <5000 | Sumatra | 2 | 11 | 11 | 4 | |||
| <5000 | Buttercup | 84 | 140 | 52 | 31 | 57 | 100 | |
| <5000 | Polish | 28 | 84 | 15 | 13 | 56 | 64 | |
| <5000 | Java | 40 | 80 | 20 | 20 | 40 | 92 | |
| <5000 | Jersey Giant | 40 | 77 | 32 | 18 | 27 | 100 | |
| <5000 | Andalusian | 70 | 109 | 46 | 24 | 39 | 100 | |
| <5000 | New Hampshire | 57 | 334 | 81 | 40 | 8 | 205 | 100 |
| <5000 | Old English Game | 8 | 252 | 252 | 31 | |||
| <5000 | Phoenix | 85 | 191 | 27 | 45 | 38 | 81 | 100 |
| <5000 | Buckeye | 74 | 118 | 52 | 22 | 44 | 100 | |
| <20,000 | Fayoumi | 68 | 135 | 108 | 19 | 8 | 100 | |
| >20,000 | Silkie | 3 | 37 | 37 | 7 | |||
| >20,000 | Leghorn | 4985 | 9415 | 4893 | 1959 | 1898 | 665 | 100 |
| >20,000 | Cornish | 777 | 1099 | 322 | 583 | 194 | 100 | |
| >20,000 | Rhode Island Red | 78 | 188 | 14 | 53 | 23 | 98 | 100 |
| >20,000 | Plymouth Rock | 178 | 896 | 562 | 88 | 34 | 212 | 100 |
| Species | Breed | Ne | Method of Computation |
|---|---|---|---|
| Goats | Angora | 121 | molecular |
| Boer | 59 | molecular | |
| LaMancha | 41 | molecular | |
| Spanish | 105 | molecular | |
| Pigs | Duroc | 86 | molecular |
| Berkshire | 77 | molecular | |
| Chester White | 77 | molecular | |
| Hampshire | 68 | molecular | |
| Hereford | 51 | molecular | |
| Landrace | 101 | molecular | |
| Yorkshire | 112 | molecular | |
| Cattle | Brangus | 90 | pedigree * |
| Hereford | 85 | pedigree * | |
| Limousin | 80 | pedigree * | |
| Holstein | 57 | CDCB * | |
| Ayrshire | 185 | CDCB * | |
| Brown Swiss | 72 | CDCB * | |
| Guernsey | 263 | CDCB * | |
| Jersey | 79 | CDCB * | |
| Sheep | Black Welsh Mountain | 32 | pedigree * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blackburn, H.D.; Wilson, C.S.; Krehbiel, B. Conservation and Utilization of Livestock Genetic Diversity in the United States of America through Gene Banking. Diversity 2019, 11, 244. https://doi.org/10.3390/d11120244
Blackburn HD, Wilson CS, Krehbiel B. Conservation and Utilization of Livestock Genetic Diversity in the United States of America through Gene Banking. Diversity. 2019; 11(12):244. https://doi.org/10.3390/d11120244
Chicago/Turabian StyleBlackburn, Harvey D., Carrie S. Wilson, and Bethany Krehbiel. 2019. "Conservation and Utilization of Livestock Genetic Diversity in the United States of America through Gene Banking" Diversity 11, no. 12: 244. https://doi.org/10.3390/d11120244
APA StyleBlackburn, H. D., Wilson, C. S., & Krehbiel, B. (2019). Conservation and Utilization of Livestock Genetic Diversity in the United States of America through Gene Banking. Diversity, 11(12), 244. https://doi.org/10.3390/d11120244





