Geographical Range Extension of the Spotfin burrfish, Chilomycterus reticulatus (L. 1758), in the Canary Islands: A Response to Ocean Warming?
Abstract
1. Introduction
2. Materials and Methods
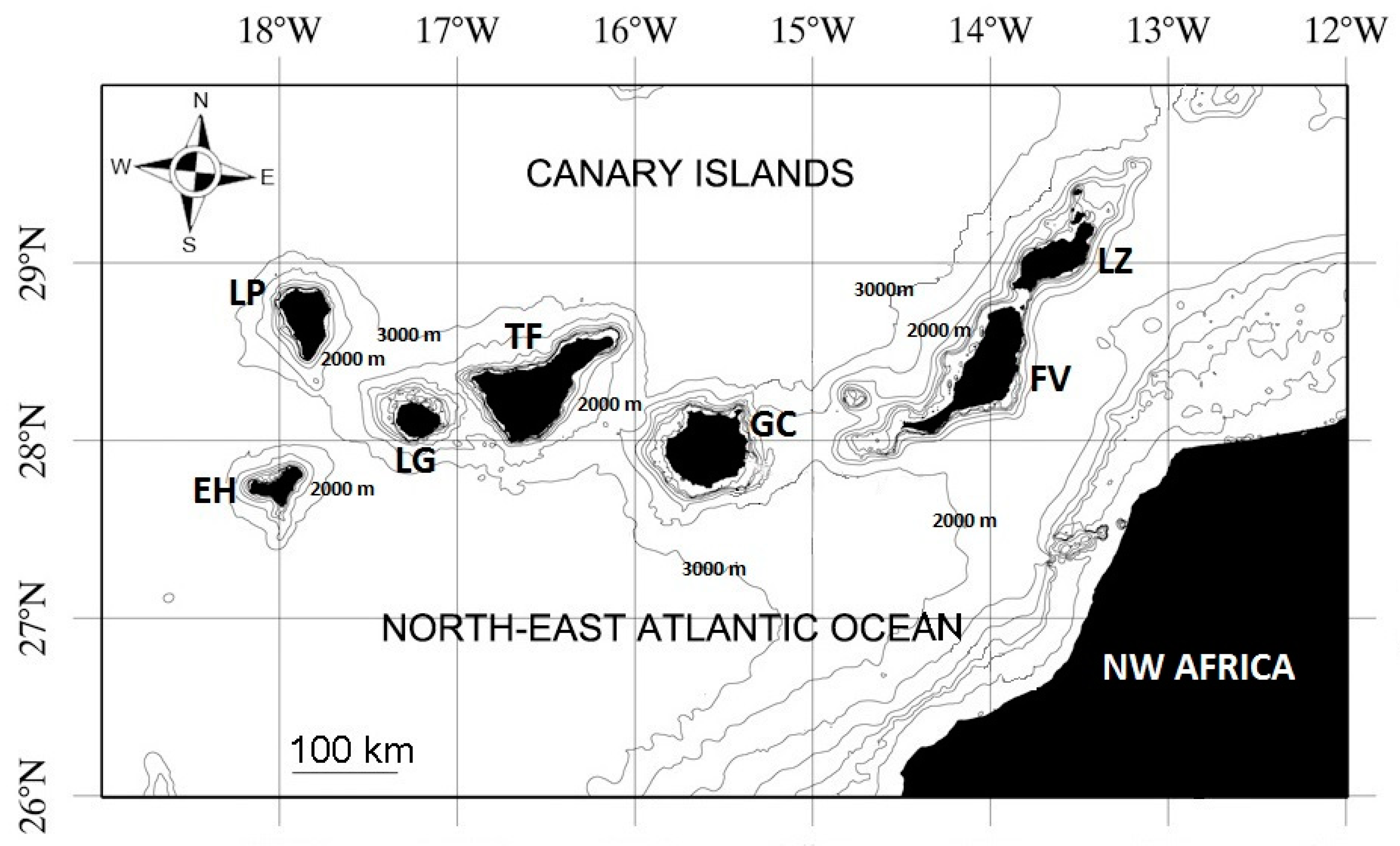
2.1. Study Region
2.2. Data Compilation and Statistical Analysis
2.3. Sea Surface Temperature and Anomalies Data
3. Results
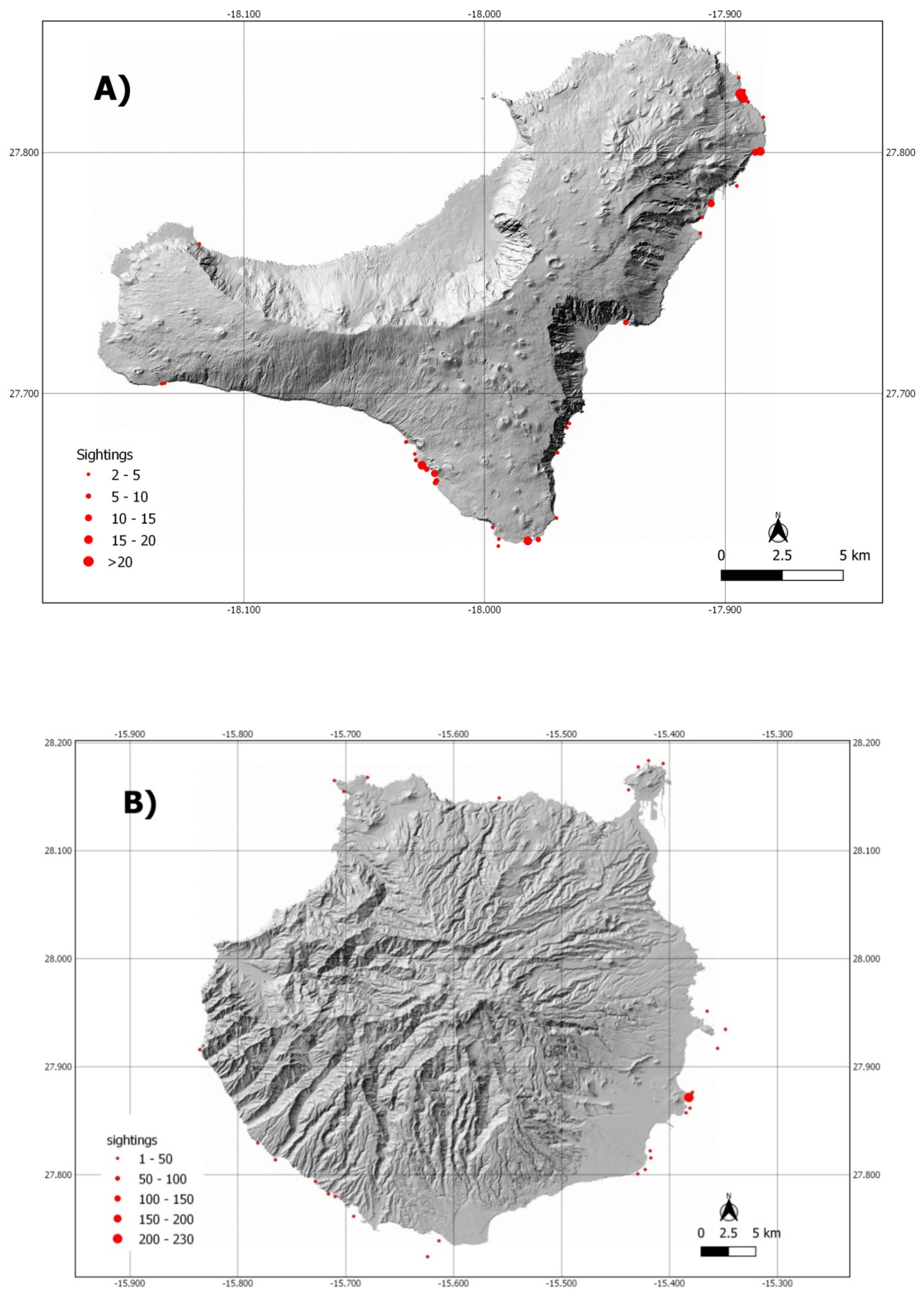
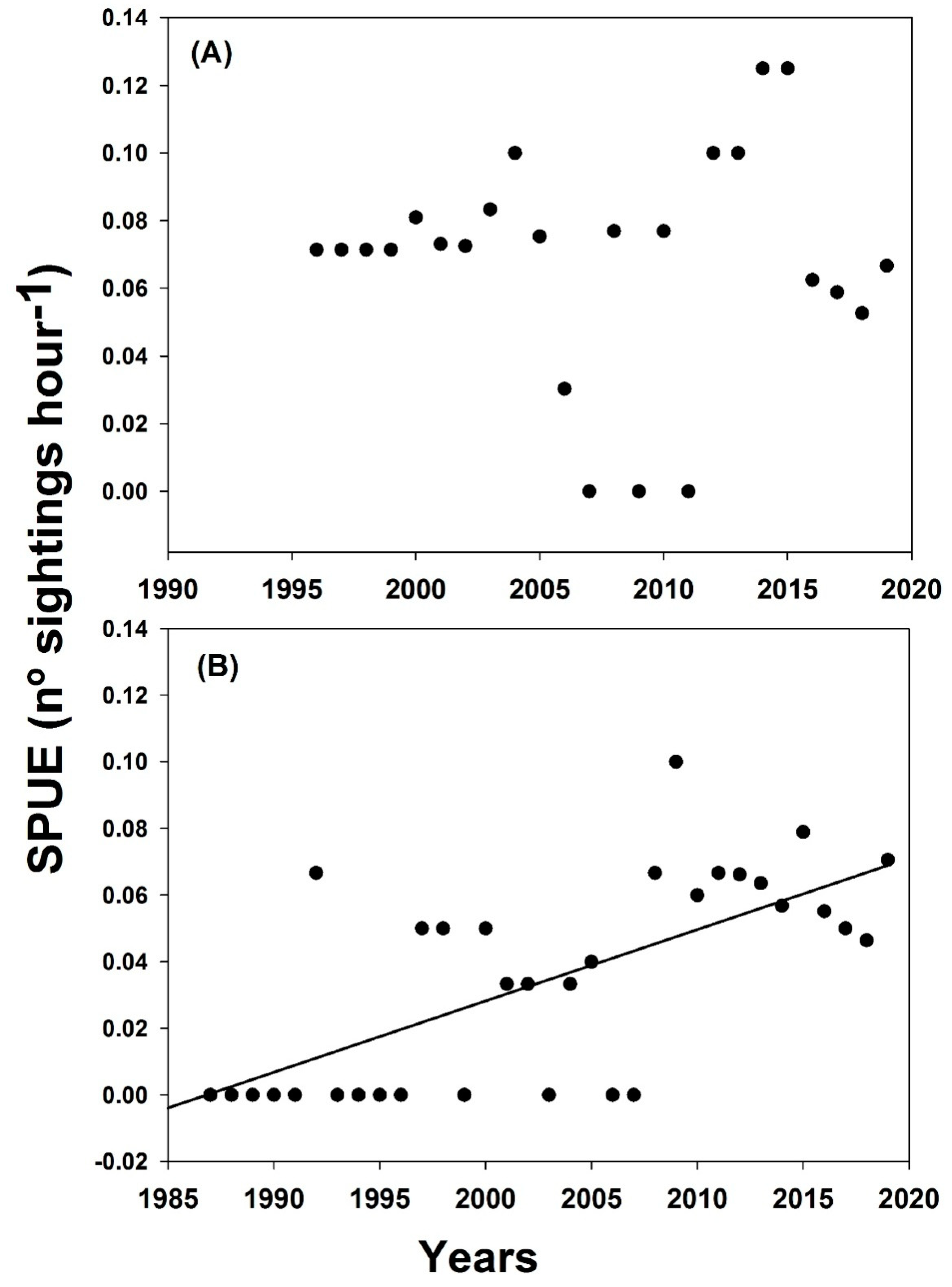
3.1. Sightings
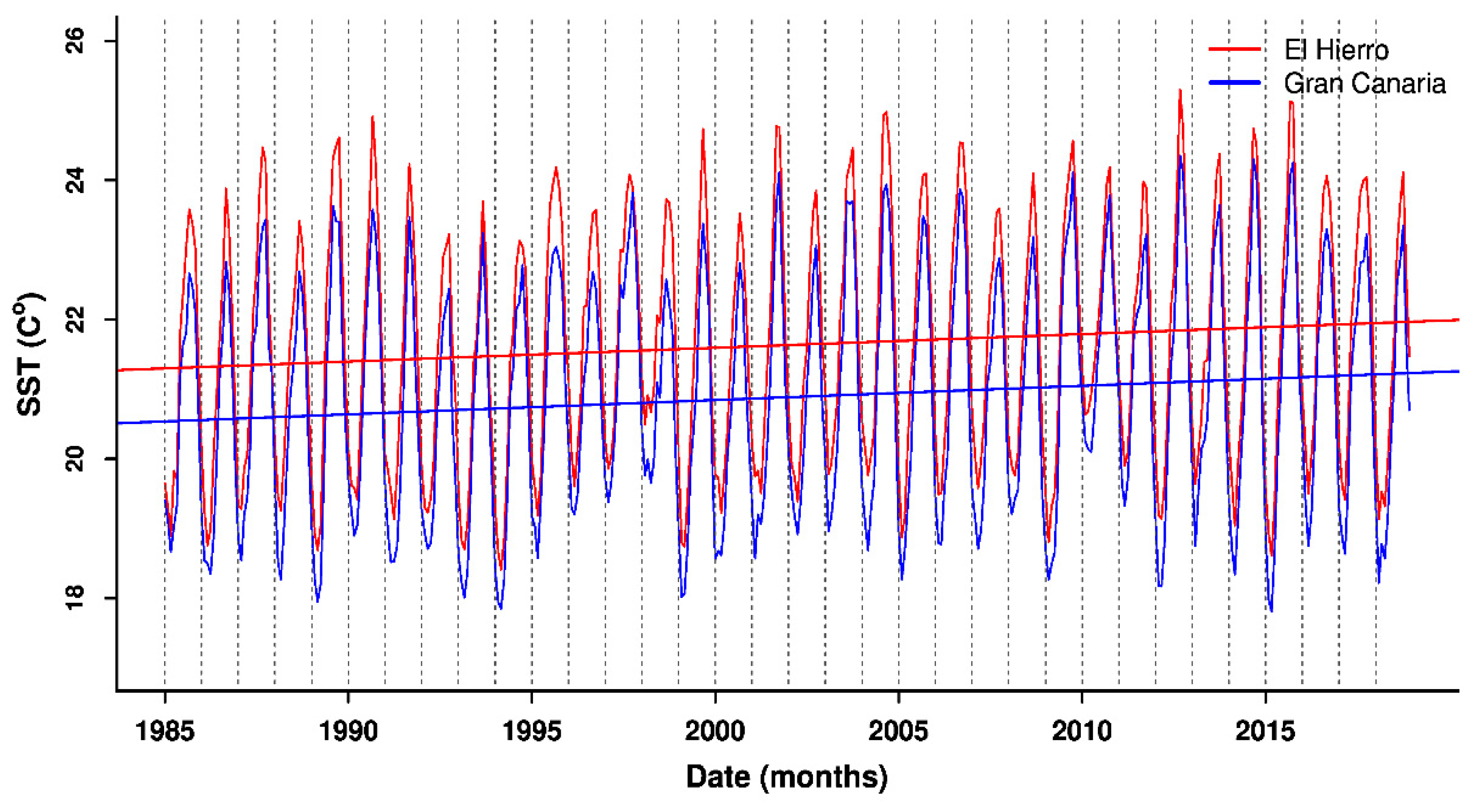
3.2. Patterns in Sea Surface Temperature and Anomalies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Durack, P.J.; Glecker, P.J.; Purkey, S.G.; Johnson, G.C.; Lyman, J.M.; Boyer, T.P. Ocean Warming: From the surface to the deep in observations and models. Oceanography 2018, 31, 41–51. [Google Scholar] [CrossRef]
- Poloczanska, E.S.; Burrows, M.T.; Brown, C.J.; Molinos, J.C.; Halpern, B.S.; Hoegh-Guldberg, O.; Kappel, C.V.; Moore, P.J.; Richardson, A.J.; Schoeman, D.S.; et al. Responses of marine organisms to climate change across oceans. Front. Mar. Sci. 2016, 3, 62. [Google Scholar] [CrossRef]
- Nagelkerken, I.; Connell, S.D. Global alteration of ocean ecosystem functioning due to increasing human CO2 emissions. PNAS 2015, 112, 13272–13277. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, P.K.; Bax, N.J. How far can marine species go? Influence of population biology and larval movement on future range limits. Mar. Ecol. Prog. Ser. 2007, 344, 15–28. [Google Scholar] [CrossRef]
- Feary, D.A.; Pratchett, M.S.; Emslie, M.J.; Fowler, A.M.; Figueira, W.F.; Luiz, O.J.; Nakamura, Y.; Booth, D.J. Latitudinal shifts in coral reef fishes: Why some species do and others do not shift. Fish Fish. 2014, 15, 593–615. [Google Scholar] [CrossRef]
- Poloczanska, E.S.; Brown, C.J.; Sydeman, W.J.; Kiessling, W.; Schoeman, D.S.; Moore, P.J.; Brander, K.; Bruno, J.F.; Buckley, L.B.; Burrows, M.T.; et al. Global imprint of climate change on marine life. Nat. Clim. Chang. 2013, 3, 919–925. [Google Scholar] [CrossRef]
- Perry, A.L.; Low, P.J.; Ellis, J.R.; Reynolds, J.D. Climate change and distribution shifts in marine fishes. Science 2005, 308, 1912–1915. [Google Scholar] [CrossRef]
- Nye, J.A.; Link, J.S.; Hare, J.A.; Overholtz, W.J. Changing spatial distribution of fish stocks in relation to climate and population size on the northeast United States continental shelf. Mar. Ecol. Prog. Ser. 2009, 393, 111–129. [Google Scholar] [CrossRef]
- Burrows, M.T.; Schoeman, D.S.; Buckley, L.B.; Moore, P.; Poloczanska, E.S.; Brander, K.M.; Brown, C.; Bruno, J.F.; Duarte, C.M.; Halpern, B.S.; et al. The pace of shifting climate in marine and terrestrial ecosystems. Science 2011, 334, 652–655. [Google Scholar] [CrossRef]
- Burrows, M.T.; Schoeman, D.S.; Richardson, A.J.; Molinos, J.G.; Hoffman, A.; Buckley, L.B.; Moore, P.; Brown, C.J.; Bruno, J.F.; Duarte, C.M.; et al. Geographical limits to species-range shifts are suggested by climate velocity. Nature 2014, 507, 492–495. [Google Scholar] [CrossRef]
- Pinsky, M.L.; Worm, B.; Fogarty, M.J.; Sarmiento, J.L.; Levin, S.A. Marine taxa track local climate velocity. Science 2013, 341, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Delgado, M.; Hidalgo, M.; Puerta, P.; Sánchez-Leal, R.; Rueda, L.; Sobrino, I. Concurrent changes in spatial distribution of the demersal community in response to climate variations in the southern Iberian coastal Large Marine Ecosystem. Mar. Ecol. Prog. Ser. 2018, 607, 19–36. [Google Scholar] [CrossRef]
- Wood, C.M.; McDonald, D.G. Global Warming: Implications for Freshwater and Marine Fish; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Brander, K.; Blom, G.; Borges, M.F.; Erzini, K.; Henderson, G.; MacKenzie, B.R.; Mendes, H.; Ribeiro, J.; Santos, A.M.P.; Toresen, R. Changes in fish distribution in the eastern North Atlantic: Are we seeing a coherent response to changing temperature? ICES Mar. Sci. Symp. 2003, 219, 261–270. [Google Scholar]
- Horta e Costa, B.; Assis, J.; Franco, G.; Erzini, K.; Henriques, M.; Gonçalves, E.J.; Caselle, J.E. Tropicalization of fish assemblages in temperate biogeographic transition zones. Mar. Ecol. Prog. Ser. 2014, 504, 241–252. [Google Scholar] [CrossRef]
- Azzurro, E. The advance of thermophilic fishes in the Mediterranean Sea: Overview and methodological questions. Comm. Int. Pour L’explor. Sci. Mediterr. CIESM Workshop Monogr. 2008, 35, 39–45. [Google Scholar]
- Ben Rais Lasram, F.; Mouillot, D. Increasing southern invasion enhances congruence between endemic and exotic Mediterranean fish fauna. Biol. Invasions 2009, 11, 697–711. [Google Scholar] [CrossRef]
- Heck, K.L., Jr.; Fodrie, F.J.; Madsen, S.; Baillie, C.J.; Byron, D.A. Seagrass consumption by native and a tropically associated fish species: Potential impacts of the tropicalization of the northern Gulf of Mexico. Mar. Ecol. Prog. Ser. 2015, 520, 165–173. [Google Scholar] [CrossRef]
- Pearce, A.F.; Feng, M. The rise and fall of the “marine heat wave” off Western Australia during the summer of 2010/2011. J. Mar. Syst. 2013, 111–112, 139–156. [Google Scholar] [CrossRef]
- Wernberg, T.; Smale, D.A.; Tuya, F.; Thomsen, M.S.; Langlois, T.J.; de Bettignies, T.; Bennett, S.; Rousseaux, C.S. An extreme climatic event alters marine ecosystem structure in a global hotspot. Nat. Clim. Chang. 2013, 3, 78–82. [Google Scholar] [CrossRef]
- Vergés, A.; Doropoulos, C.; Malcolm, H.A.; Skye, M.; Garcia-Pizá, M.; Marzinelli, E.M.; Campbell, A.H.; Ballesteros, E.; Hoey, A.S.; Vila-Concejo, A.; et al. Long-term empirical evidence of ocean warming leading to tropicalization of fish communities, increased herbivory, and loss of kelp. Proc. Natl. Acad. Sci. USA 2016, 113, 13791–13796. [Google Scholar] [CrossRef]
- Leis, J.L.; Matsuura, K.; Shao, K.-T.; Hardy, G.; Zapfe, G.; Liu, M.; Jing, L.; Robertson, R.; Tyler, J. Chilomycterus reticulatus (errata version published in 2017). IUCN Red List Threat. Species 2015. [Google Scholar] [CrossRef]
- Simon, T.; Macieira, R.M.; Joyeux, J.C. The shore fishes of the Trindade–Martin Vaz insular complex: An update. J. Fish Biol. 2013, 82, 2113–2127. [Google Scholar] [CrossRef] [PubMed]
- Leis, J.M. Diodontidae. Porcupine fishes (burrfishes, spiny puffers). In The Living Marine Resources of the Eastern Central Atlantic; FAO Species Identification Guide for Fishery Purposes, Bony fishes, Part 2 (Perciformes to Tetraodontiformes) and Sea Turtles; Carpenter, K.E., De Angelis, N., Eds.; FAO: Rome, Italy, 2016; Volume 4, pp. 3074–3079. [Google Scholar]
- Afonso, P.; Porteiro, F.M.; Fontes, J.; Tempera, F.; Morato, T.; Cardigos, F.; Santos, R.S. New and rare coastal fishes in the Azores islands: Occasional events or tropicalization process? J. Fish Biol. 2013, 83, 272–294. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, P.; Fricke, R.; Biscoito, M.J. The coastal fishes of Madeira Island-new records and an annotated check-list. Zootaxa 2008, 1715, 1–26. [Google Scholar] [CrossRef]
- Almada, F.; Abecasis, D.; Villegas-Ríos, D.; Henriques, S.; Pais, M.P.; Batista, M.; Horta e Costa, B.; Martins, J.; Tojeira, I.; Rodrigues, N.V.; et al. Ichthyofauna of the Selvagens Islands. Do small coastal areas show high species richness in the northeastern Atlantic? Mar. Biol. Res. 2015, 11, 49–61. [Google Scholar] [CrossRef]
- Brito, A.; Pascual, P.J.; Falcón, J.M.; Sancho, A.; González, G. Peces de Las Islas Canarias. Catálogo Comentado e Ilustrado; Lemus, F., Ed.; Editorial Lemus: La Laguna, Spain, 2002; p. 419. [Google Scholar]
- Wirtz, P.; Brito, A.; Falcón, J.M.; Freitas, R.; Fricke, R.; Monteiro, V.; Reiner, F.; Tariche, O. The coastal fishes of the Cape Verde Islands-new records and an annotated check-list. Spixiana 2013, 36, 113–142. [Google Scholar]
- Wirtz, P.; Ferreira, C.E.L.; Floeter, S.R.; Fricke, R.; Gasparini, J.L.; Iwamoto, T.; Rocha, L.; Sampaio, C.L.S.; Schliewen, U.K. Coastal fishes of Sâo Tomé and Príncipe Islands, Gulf of Guinea (Eastern Atlantic Ocean)-an update. Zootaxa 2007, 1523, 1–48. [Google Scholar] [CrossRef]
- Edwards, A.J.; Glass, C.W. The fishes of Saint Helena Island, South Atlantic Ocean. I. The shore fishes. J. Nat. Hist. 1987, 21, 617–686. [Google Scholar] [CrossRef]
- Wirtz, P.; Bingeman, J.; Bingeman, J.; Fricke, R. The fishes of Ascension Island, central Atlantic Ocean–new records and an annotated checklist. J. Mar. Biol. Assoc. UK 2017, 97, 783–798. [Google Scholar] [CrossRef]
- Follesa, M.C.; Mulas, A.; Porcu, C.; Cau, A. First record of Chilomycterus reticulatus (Osteichthyes: Diodontidae) in the Mediterranean Sea. J. Fish Biol. 2009, 74, 1677–1681. [Google Scholar] [CrossRef]
- Sanches, J.G. Catálogo dos Principais Peixes Marinhos da República de Guiné-Bissau; Publicações Avulsas Instituto Nacional de Investigação das Pescas: Lisboa, Portugal, 1991; Volume 16, p. 429. [Google Scholar]
- Sommer, C.; Schneider, W.; Poutiers, J.M. The Living Marine Resources of Somalia; FAO species identification field guide for fishery purposes; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 1996; p. 376. [Google Scholar]
- Leis, J.M. Nomenclature and distribution of the species of the porcupinefish family Diodontidae (pices, Teleostei). Mem. Mus. Vic. 2006, 63, 77–90. [Google Scholar] [CrossRef]
- Kaschner, K.; Kesner-Reyes, K.; Garilao, C.; Rius-Barile, J.; Rees, T.; Froese, R. AquaMaps: Predicted Range Maps for Aquatic Species; World Wide Web Electronic Publication, 2016. Available online: https://www.aquamaps.org/ (accessed on 15 August 2016).
- Brito, A.; Falcón, J.M. Contribution to the knowledge of the distribution and ecology of Chilomycterus atringa (Pisces, Diodontidae) in the Canary Islands. Vieraea 1990, 19, 271–275. [Google Scholar]
- Real Decreto 139/2011, de 4 de febrero, para el desarrollo del Listado de Especies Silvestres en Régimen de Protección Especial y del Catálogo Español de Especies Amenazadas. Ministerio de Medio Ambiente, y Medio Rural y Marino. BOE 2011, 46, 20912–20951.
- Ley 4/2010, de 4 de junio, del Catálogo Canario de Especies Protegidas. Gobierno de Canarias. BOC 2010, 112, 15200–15225.
- Haroun, R.J.; del Rosario, A. Contribución al conocimiento de la ecología del Tamboril espinoso, Chilomycterus atringa (Pisces, Diodontidae) y su distribución en la isla de El Hierro y La Gomera. En Comunicación presentada al XIII Simposio Ibérico del Bentos Marino, Las Palmas de Gran Canaria, Islas Canarias, Spain, 21–24 September 2004. [Google Scholar]
- Clemente, S.; Hernández, J.C.; Brito, A. Context-dependent effects of marine protected areas on predatory interactions. Mar. Ecol. Prog. Ser. 2011, 437, 119–133. [Google Scholar] [CrossRef][Green Version]
- Pendleton, L.H.; Beyer, H.; Grose, S.O.; Hoegh-Guldberg, O.; Karcher, D.B.; Kennedy, E.; Llewellyn, L.; Nys, C.; Shapiro, A.; Jain, R.; et al. Disrupting data sharing for a healthier ocean. ICES J. Mar. Sci. 2019. [Google Scholar] [CrossRef]
- Vianna, G.M.S.; Meekan, M.G.; Pannell, D.J.; Marsh, S.P.; Meeuwig, J.J. Socio-economic value and community benefits from shark-diving tourism in Palau: A sustainable use of reef shark populations. Biol. Conserv. 2012, 145, 267–277. [Google Scholar] [CrossRef]
- Stuart-Smith, R.D.; Edgar, G.J.; Bates, A.E. Thermal limits to the geographic distributions of shallow-water marine species. Nat. Ecol. Evol. 2017. [Google Scholar] [CrossRef]
- Meyers, E.K.M.; Tuya, F.; Barker, J.; Jiménez-Alvarado, D.; Castro-Hernández, J.J.; Haroun, R.; Rödder, D. Population structure, distribution and habitat use of the critically endangered Angelshark, Squatina squatina, in the Canary Islands. Aquat. Conserv. Mar. Freshw. Ecosyst. 2017, 27, 1133–1144. [Google Scholar] [CrossRef]
- Valdés, L.; Déniz-González, I. (Eds.) Oceanographic and Biological Features in the Canary Current Large Marine Ecosystem; IOC Technical Series; IOC-UNESCO: Paris, France, 2015; Volume 115, p. 383. [Google Scholar]
- Barton, E.D.; Arístegui, J.; Tett, P.; Cantón, M.; García-Braun, J.; Hernández-León, S.; Nykjaer, L.; Almeida, C.; Almunia, J.; Balleteros, S.; et al. The transition zone of the Canary Current upwelling region. Prog. Oceanogr. 1998, 41, 455–504. [Google Scholar] [CrossRef]
- Davenport, R.; Neuer, S.; Helmke, P.; Pérez-Marrero, J.; Llinás, O. Primary productivity in the northern Canary Islands region as inferred from SeaWiFS imagery. Deep Sea Res. Part II Top. Stud. Oceanogr. 2002, 49, 3481–3496. [Google Scholar] [CrossRef]
- Tuya, F.; Boyra, A.; Sánchez-Jérez, P.; Barbera, C.; Haroun, R.J. Relationships between rocky-reef fish assemblages, the sea urchin Diadema antillarum and macroalgae throughout the Canarian Archipelago. Mar. Ecol. Prog. Ser. 2004, 278, 157–169. [Google Scholar] [CrossRef]
- Tuya, F.; Ramírez, R.; Sánchez-Jérez, P.; Haroun, R.J.; González-Ramos, A.J.; Coca, J. Coastal resources exploitation can mask bottom-up mesoscale regulation of intertidal populations. Hydrobiologia 2006, 553, 337–344. [Google Scholar] [CrossRef]
- Fox, J. Getting started with the R Commander: A basic-statistics graphical user interface to R. J. Stat. Softw. 2005, 14, 1–42. [Google Scholar] [CrossRef]
- Harrison, X.A.; Donaldson, L.; Correa-Cano, M.E.; Evans, J.; Fisher, D.N.; Goodwin, C.E.D.; Robinson, B.S.; Hodgson, D.J.; Inger, R. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 2018, 6, e4794. [Google Scholar] [CrossRef] [PubMed]
- Patakamuri, S.K.; Modifiedmk: Modified Mann Kendall Trend Tests. R Package Version 1.1.0. 2018. Available online: https://CRAN.R-project.org/package=modifiedmk (accessed on 15 May 2019).
- Yue, S.; Wang, C.Y. The Mann-Kendall test modified by effective sample size to detect trend in serially correlated hydrological series. Water Resour. Manag. 2004, 18, 201–218. [Google Scholar] [CrossRef]
- Roesch, A.; Schmidbauer, H.; WaveletComp: Computational Wavelet Analysis. R Package Version 1.1. 2018. Available online: https://CRAN.R-project.org/package=WaveletComp (accessed on 15 May 2019).
- Brito, A.; Falcón, J.M.; Aguilar, N.; Pascual, P. Fauna Vertebrada Marina. In Naturaleza de Las Islas Canarias. Ecología y Conservación; Fernández-Palacios, J.M., Martín-Esquivel, J.L., Eds.; Editorial Publicaciones Turquesa: Santa Cruz de Tenerife, Spain, 2001; pp. 219–229. [Google Scholar]
- Brito, A.; Moreno-Borges, A.; Escánez, A.; Falcón, J.M.; Herrera, R. New records of Actinopterygian fishes from the Canary Islands: Tropicalization as the most important driving force increasing fish diversity. Rev. Acad. Canar. Cienc. 2017, 29, 31–44. [Google Scholar]
- Falcón, J.M.; Brito, A.; Herrera, R.; Monterroso, O.; Rodríguez, M.; Álvarez, O.; Ramos, E.; Miguel, A. New records of tropical littoral fishes from the Canary Islands as a result of two driving forces: Natural expansion and introduction by oil platforms. Rev. Acad. Canar. Cienc. 2018, 30, 39–56. [Google Scholar]
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, Z.A.; Finlayson, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A.; et al. Marine ecoregions of the world: A bioregionalization of coastal and shelf areas. BioScience 2007, 57, 573–583. [Google Scholar] [CrossRef]
- Peña-Izquierdo, J.; Pelegrí, J.L.; Pastor, M.V.; Castellanos, P.; Emelianov, M.; Gasser, M.; Salvador, J.; Vázquez-Domínguez, E. The continental slope current system between Cape Verde and the Canary Islands. Sci. Mar. 2012, 76, 65–78. [Google Scholar] [CrossRef]
- Luiz, O.J.; Allen, A.P.; Robertson, D.R.; Floeter, S.R.; Madin, J.S. Seafarers or castaways: Ecological traits associated with rafting dispersal in tropical reef fishes. J. Biogeogr. 2015, 42, 2323–2333. [Google Scholar] [CrossRef]
- Rodríguez, L.; López, C.; Casado-Amezua, P.; Ruiz-Ramos, D.V.; Martínez, B.; Banaszak, A.; Tuya, F.; García-Fernández, A.; Hernández, A. Genetic relationships of the hydrocoral Millepora alcicornis and its symbionts within and between locations across the Atlantic. Coral Reefs 2019, 38, 255–268. [Google Scholar] [CrossRef]
- Triay-Portella, R.; Pajuelo, J.G.; Manent, P.; Espino, F.; Ruiz-Díaz, R.; Lorenzo, J.M.; González, J.A. New records of non-indigenous fishes (Perciformes and Tetraodontiformes) from the Canary Islands (north-eastern Atlantic). Cybium 2015, 39, 163–174. [Google Scholar]
- Pajuelo, J.G.; González, J.A.; Triay-Portella, R.; Martín, J.A.; Ruiz-Díaz, R.; Lorenzo, J.M.; Luque, A. Introduction of non-native marine fish species to the Canary Islands waters through oil platforms as vectors. J. Mar. Syst. 2016, 163, 23–30. [Google Scholar] [CrossRef]
- García-Mederos, A.; Tuya, F.; Tuset, V.M. The structure of a nearshore fish assemblage at an oceanic island: Insight from small scale fisheries through bottom traps at Gran Canary Island (Canary Islands, eastern Atlantic). Aquat. Living Resour. 2015, 28, 1–10. [Google Scholar] [CrossRef][Green Version]
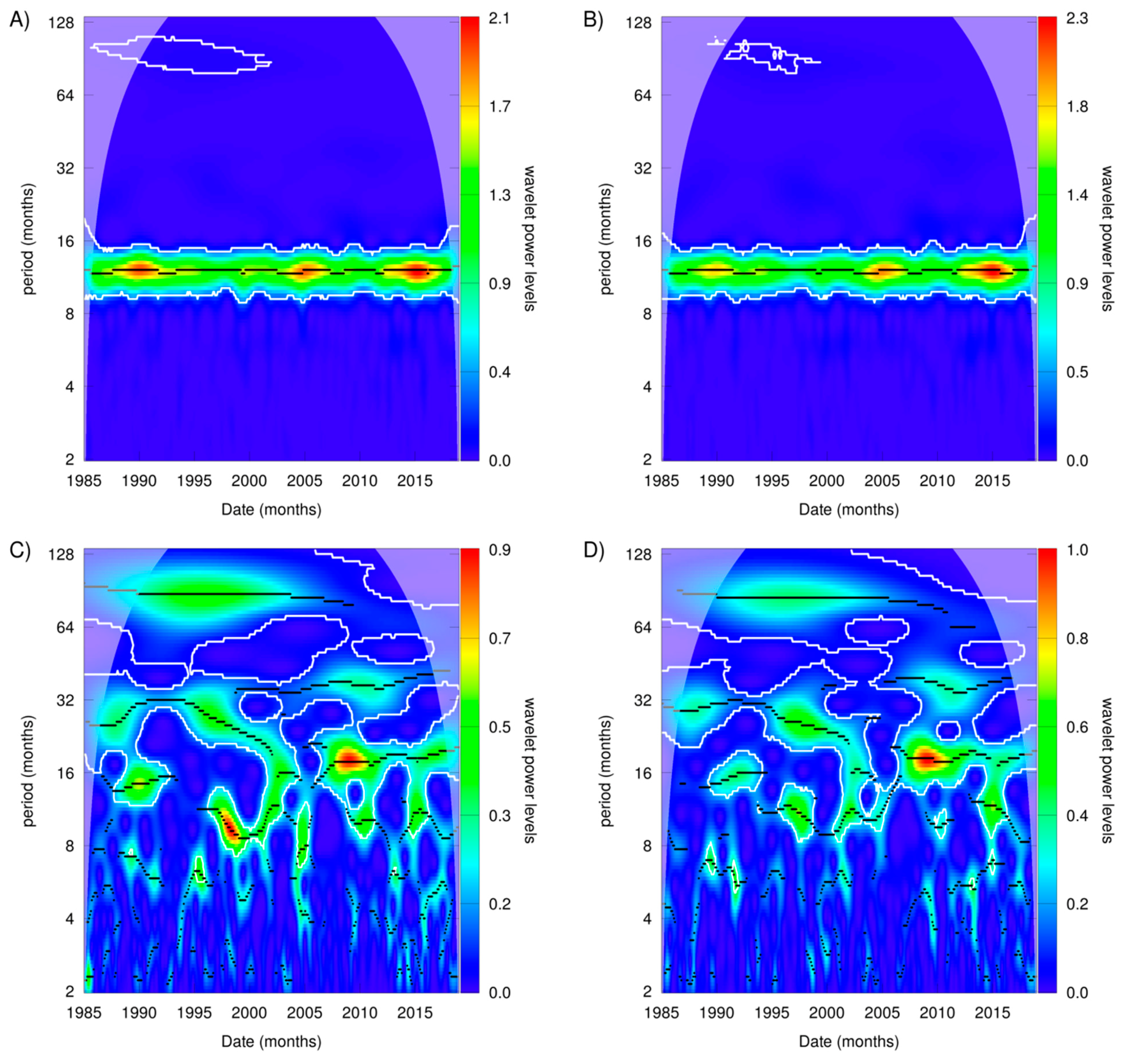
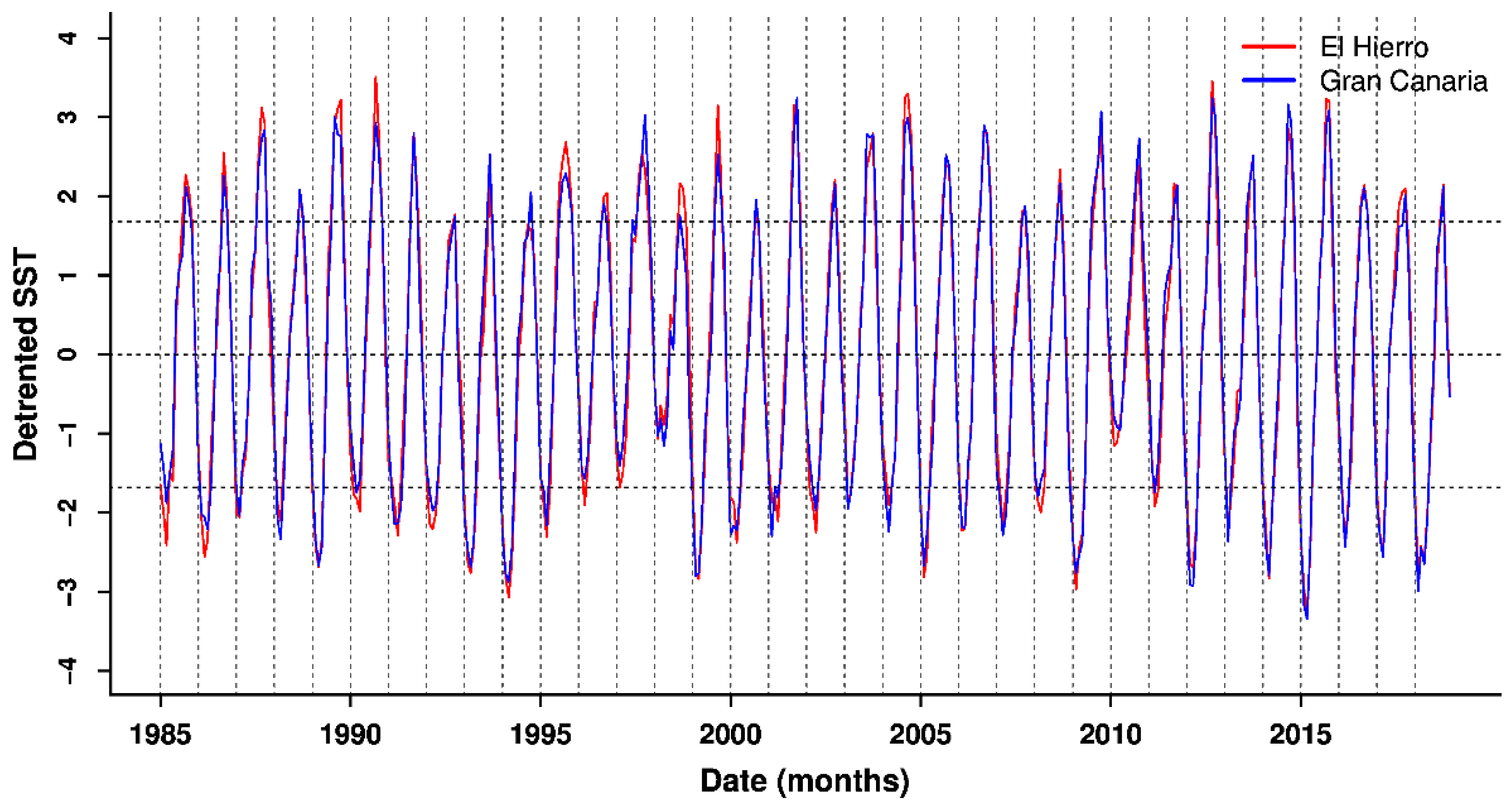
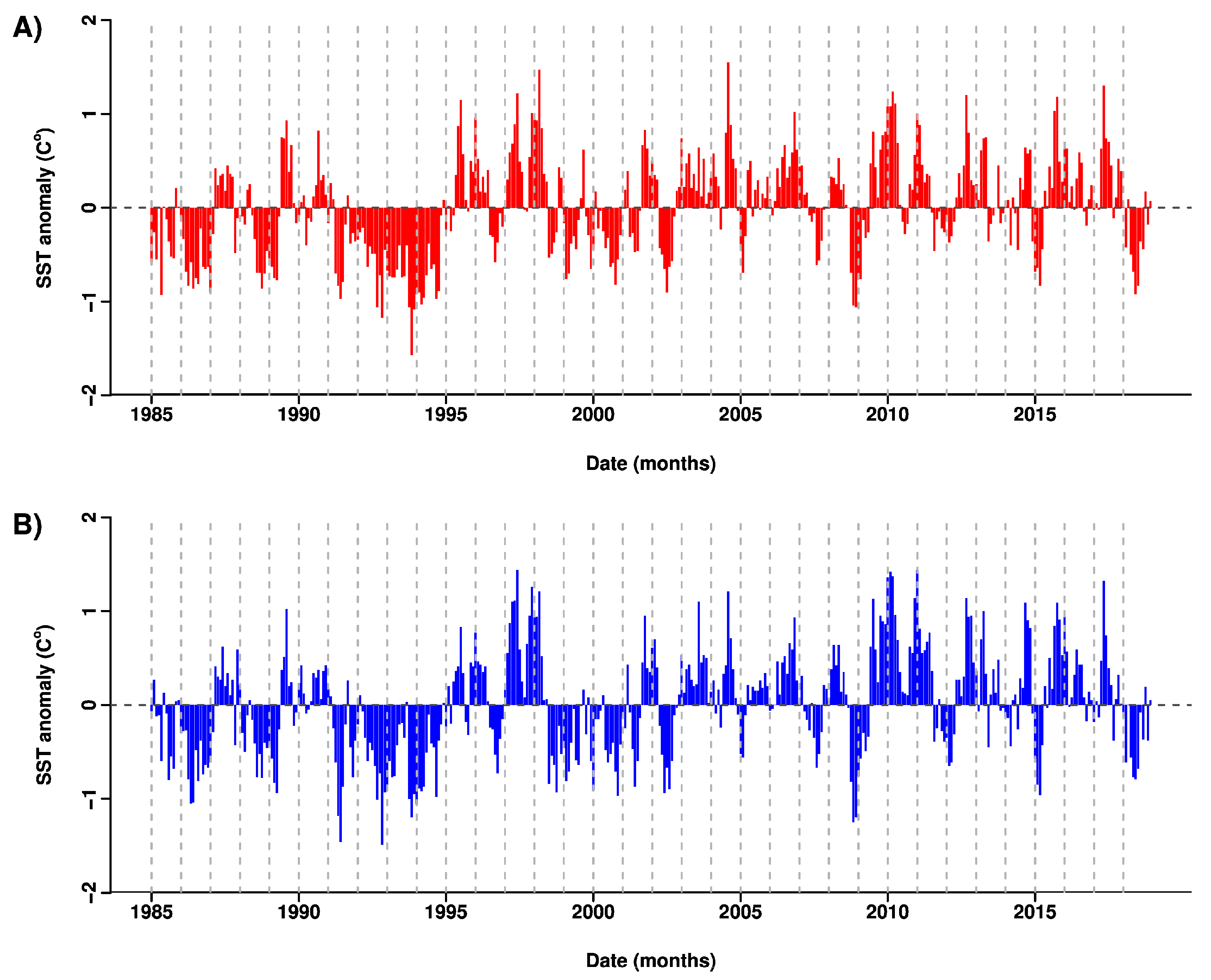
| Site | Estimate | Std. Error | z Value | P |
|---|---|---|---|---|
| El Hierro | ||||
| (Intercept) | −3.497 × 10 | 2.015 × 10 | −1.736 | 0.0826 |
| Year | 1.800 × 10−2 | 1.003 × 10−2 | 1.795 | 0.0726 |
| Effort | 5.481 × 10−3 | 3.842 × 10−4 | 14.265 | < 2 × 10−16 |
| Null deviance | 247.925 | |||
| Residual deviance | 69.953 | |||
| Gran Canaria | ||||
| (Intercept) | −2.006 × 10−2 | 2.437 × 10 | −8.23 | < 2 × 10−16 |
| Year | 1.004 × 10 | 1.213 × 10−2 | 8.28 | < 2 × 10−16 |
| Effort | 2.956 × 10−3 | 2.194 × 10−4 | 13.47 | < 2 × 10−16 |
| Null deviance | 741.221 | |||
| Residual deviance | 71.253 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Espino, F.; Tuya, F.; del Rosario, A.; Bosch, N.E.; Coca, J.; González-Ramos, A.J.; del Rosario, F.; Otero-Ferrer, F.J.; Moreno, Á.C.; Haroun, R. Geographical Range Extension of the Spotfin burrfish, Chilomycterus reticulatus (L. 1758), in the Canary Islands: A Response to Ocean Warming? Diversity 2019, 11, 230. https://doi.org/10.3390/d11120230
Espino F, Tuya F, del Rosario A, Bosch NE, Coca J, González-Ramos AJ, del Rosario F, Otero-Ferrer FJ, Moreno ÁC, Haroun R. Geographical Range Extension of the Spotfin burrfish, Chilomycterus reticulatus (L. 1758), in the Canary Islands: A Response to Ocean Warming? Diversity. 2019; 11(12):230. https://doi.org/10.3390/d11120230
Chicago/Turabian StyleEspino, Fernando, Fernando Tuya, Armando del Rosario, Néstor E. Bosch, Josep Coca, Antonio J. González-Ramos, Francisco del Rosario, Francisco J. Otero-Ferrer, Ángel C. Moreno, and Ricardo Haroun. 2019. "Geographical Range Extension of the Spotfin burrfish, Chilomycterus reticulatus (L. 1758), in the Canary Islands: A Response to Ocean Warming?" Diversity 11, no. 12: 230. https://doi.org/10.3390/d11120230
APA StyleEspino, F., Tuya, F., del Rosario, A., Bosch, N. E., Coca, J., González-Ramos, A. J., del Rosario, F., Otero-Ferrer, F. J., Moreno, Á. C., & Haroun, R. (2019). Geographical Range Extension of the Spotfin burrfish, Chilomycterus reticulatus (L. 1758), in the Canary Islands: A Response to Ocean Warming? Diversity, 11(12), 230. https://doi.org/10.3390/d11120230







