Aquatic Macrophytes are Seasonally Important Dietary Resources for Moose
Abstract
1. Introduction
2. Materials and Methods
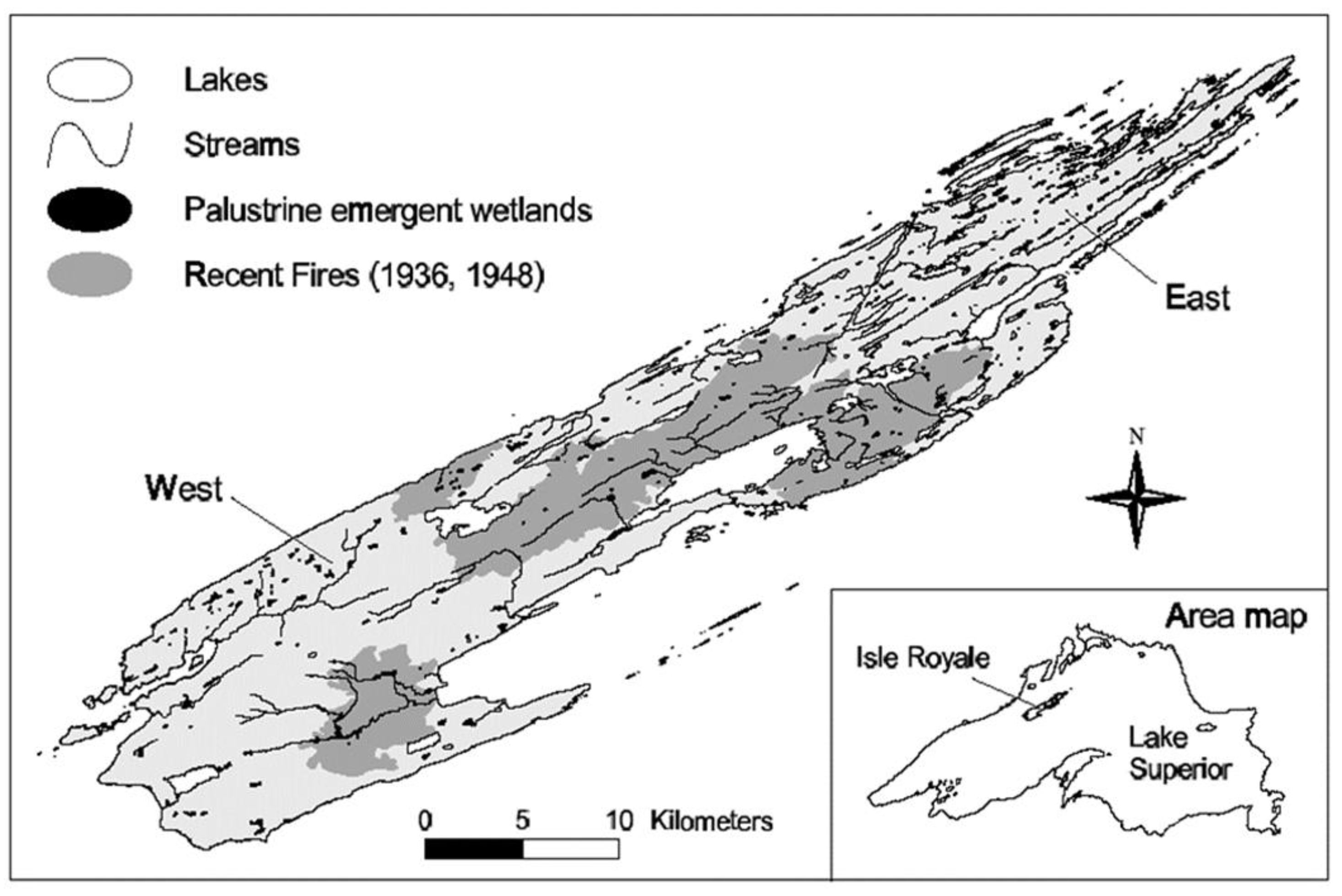
2.1. Study Area
2.2. Sample Collection
2.2.1. Moose Forage
2.2.2. Moose Hooves and Bone Marrow
2.3. Physical and Chemical Sample Pretreatment and Measurement
2.4. Moose Hooves
2.5. Data Analysis
2.5.1. Moose Forage
2.5.2. Moose Hooves and Bone Marrow
2.6. Diet Estimation
3. Results
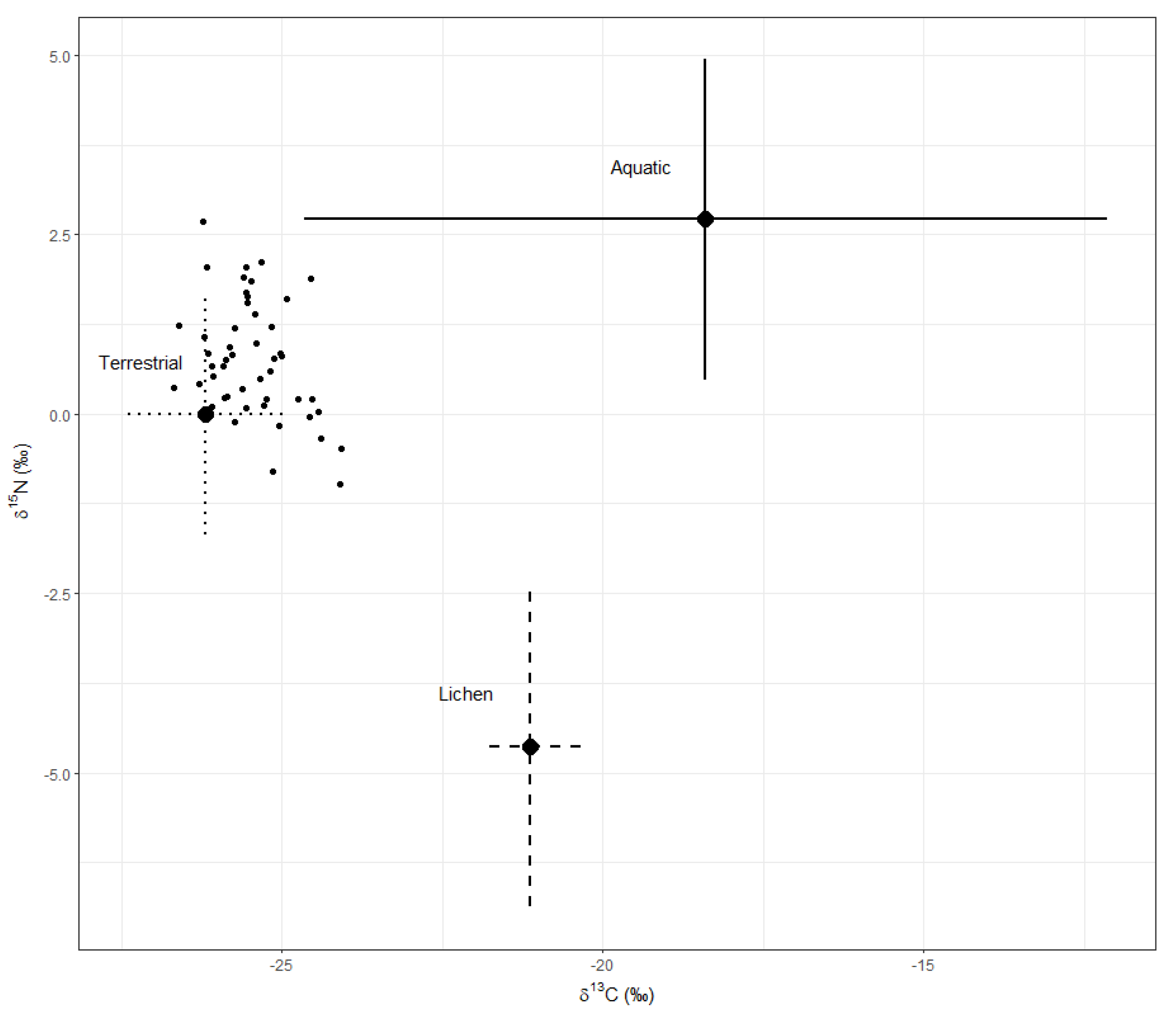
3.1. Moose Forage
3.2. Moose Hooves and Bone Marrow
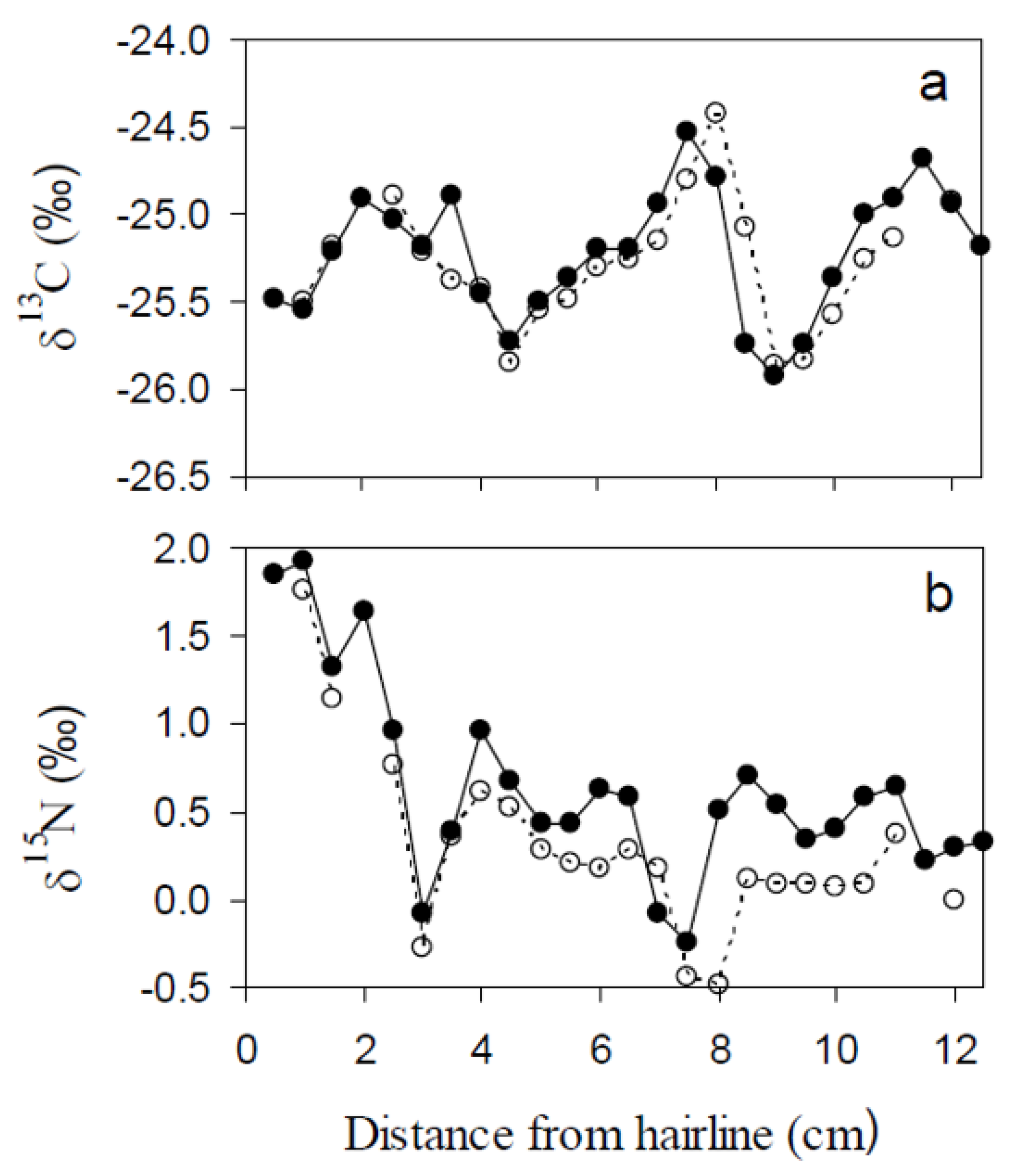
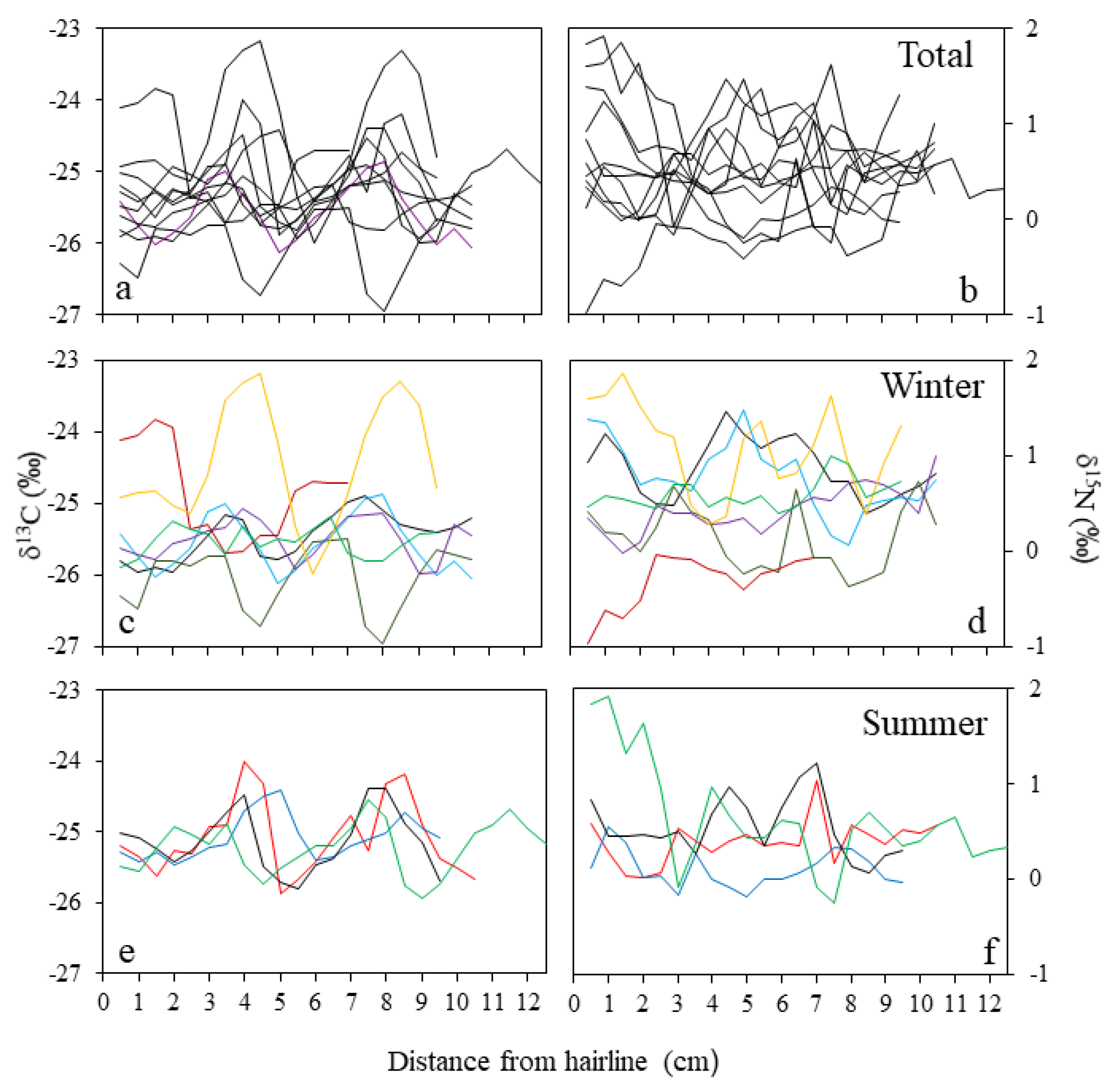
3.2.1. Hairline
3.2.2. Chronology
3.3. Diet Estimation
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Taber, R.D.; Dasmann, R.F. The dynamics of three natural populations of the deer Odocoileus hemionus columbianus. Ecology 1957, 38, 233–246. [Google Scholar] [CrossRef]
- Klein, D.R. Food selection by North American deer and their response to over-utilization of preferred plant species. In Animal Populations in Relation to Their Food Resources; Watson, A., Ed.; British Ecological Society: Oxford, UK, 1970; pp. 25–46. [Google Scholar]
- Eadie, J. Sheep production and pastoral resources. In Animal Populations in Relation to Their Food Resources; Watson, A., Ed.; Blackwell Scientific Publications: Oxford, UK, 1970. [Google Scholar]
- Gibson, R.M.; Guinness, F.E. Differential reproduction among red deer (Cervus elaphus) stags on Rhum. J. Anim. Ecol. 1980, 49, 199–208. [Google Scholar] [CrossRef]
- Elser, J.J.; Fagan, W.F.; Denno, R.F.; Dobberfuhl, D.R.; Folarin, A.; Huberty, A.; Interlandi, S.; Kilham, S.S.; McCauley, E.; Schulz, K.L.; et al. Nutritional constraints in terrestrial and freshwater food webs. Nature 2000, 408, 578–580. [Google Scholar] [CrossRef]
- Guinness, F.E.; Clutton-Brock, T.H.; Albon, S.D. Factors affecting calf mortality in red deer (Cervus elaphus). J. Anim. Ecol. 1978, 47, 817–832. [Google Scholar] [CrossRef]
- Stodart, E.; Myers, K. The effects of different foods on confined populations of wild rabbits, Oryctolagus cuniculus (L.). Csiro Wildl. Res. 1966, 11, 111–124. [Google Scholar] [CrossRef]
- Newton, I.; Kerbes, R.H. Breeding of greylag geese (Anser anser) on the Outer Hebrides, Scotland. J. Anim. Ecol. 1974, 43, 771–783. [Google Scholar] [CrossRef]
- Hunter, M.D.; Ohgushi, T.; Price, P.W. Effects of Resource Distribution on Animal Plant Interactions; Elsevier: San Diego, CA, USA, 2012; ISBN 978-0-12-361955-6. [Google Scholar]
- Jefferies, R.L.; Klein, D.R.; Shaver, G.R. Vertebrate herbivores and northern plant communities: Reciprocal influences and responses. Oikos 1994, 71, 193–206. [Google Scholar] [CrossRef]
- Moen, R.; Pastor, J.; Cohen, Y. A spatially explicit model of moose foraging and energetics. Ecology 1997, 78, 505. [Google Scholar] [CrossRef]
- Mattson, W.J., Jr. Herbivory in relation to plant nitrogen content. Annu. Rev. Ecol. Syst. 1980, 11, 119–161. [Google Scholar] [CrossRef]
- Karns, P.D. Population distribution, density and trends. In Ecology and Management of the North American Moose; Franzmann, A.W., Schwartz, C.C., Eds.; University Press of Colorado: Boulder, CO, USA, 2007; pp. 125–139. [Google Scholar]
- Clifford, A.B.; Witmer, L.M. Case studies in novel narial anatomy: 2. The enigmatic nose of moose (Artiodactyla: Cervidae: Alces alces). J. Zool. 2004, 262, 339–360. [Google Scholar] [CrossRef]
- Schwartz, C.C.; Hubbert, M.E.; Franzmann, A.W. Energy requirements of adult moose for winter maintenance. J. Wildl. Manag. 1988, 52, 26. [Google Scholar] [CrossRef]
- Vucetich, J.A.; Peterson, R.O. The influence of top–down, bottom–up and abiotic factors on the moose (Alces alces) population of Isle Royale. Proc. R. Soc. Lond. B Biol. Sci. 2004, 271, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Peterson, R.O. Wolf Ecology and Prey Relationships on Isle Royale; US Government Printing Office: Washington, DC, USA, 1977.
- McLaren, B.E.; Peterson, R.O. Wolves, moose, and tree rings on Isle Royale. Science 1994, 266, 1555–1558. [Google Scholar] [CrossRef] [PubMed]
- Geist, V. Deer of the World: Their Evolution, Behaviour, and Ecology; Stackpole Books: Mechanicsburg, PA, USA, 1998; ISBN 1-84037-094-7. [Google Scholar]
- Schwartz, C.C.; Renecker, L.A. Nutrition and energetics. In Ecology and Management of the North American Moose; Franzmann, A.W., Schwartz, C.C., Eds.; University Press of Colorado: Boulder, CO, USA, 2007; pp. 141–171. [Google Scholar]
- MacCracken, J.G.; Van Ballenberghe, V.; Peek, J.M. Use of aquatic plants by moose: Sodium hunger or foraging efficiency? Can. J. Zool. 1993, 71, 2345–2351. [Google Scholar] [CrossRef]
- Tischler, K.B. Aquatic Plant Nutritional Quality and Contribution to Moose Diet at Isle Royale National Park. Master’s Thesis, Michigan Technological University, Houghton, MI, USA, 2004. [Google Scholar]
- Fraser, D.; Chavez, E.R.; Palohelmo, J.E. Aquatic feeding by moose: Selection of plant species and feeding areas in relation to plant chemical composition and characteristics of lakes. Can. J. Zool. 1984, 62, 80–87. [Google Scholar] [CrossRef]
- Fryxell, J.M.; Doucet, C.M. Diet choice and the funcional response of beavers. Ecology 1993, 74, 1297–1306. [Google Scholar] [CrossRef]
- Ceacero, F.; Landete-Castillejos, T.; Miranda, M.; García, A.J.; Martínez, A.; Gallego, L. Why do cervids feed on aquatic vegetation? Behav. Process. 2014, 103, 28–34. [Google Scholar] [CrossRef]
- Huber, N.K. Glacial and Postglacial Geologic History of Isle Royale National Park, Michigan; U.S. Geological Survey Professional Paper 754-A; U.S. Government Printing Office: Washington, DC, USA, 1973.
- Peek, J.M. Habitat relationships. In Ecology and Management of the North American Moose; Franzmann, A.W., Schwartz, C.C., Eds.; University Press of Colorado: Boulder, CO, USA, 2007; pp. 351–375. ISBN 1-56098-775-8. [Google Scholar]
- Morris, D.M. Aquatic Habitat Use by North American Moose (Alces alces) and Associated Richness and Biomass of Submersed and Floating-Leaved Aquatic Vegetation in North-Central Minnesota. Master’s Thesis, Lakehead University, Thunder Bay, ON, Canada, 2014. [Google Scholar]
- Kelly, J.F. Stable isotopes of carbon and nitrogen in the study of avian and mammalian trophic ecology. Can. J. Zool. 2000, 78, 1–27. [Google Scholar] [CrossRef]
- Belovsky, G.E.; Jordan, P.A. The time-energy budget of a moose. Theor. Popul. Biol. 1978, 14, 76–104. [Google Scholar] [CrossRef]
- Park, R.; Epstein, S. Carbon isotope fractionation during photosynthesis. Geochim. Cosmochim. Acta 1960, 21, 110–126. [Google Scholar] [CrossRef]
- Bender, M.M. Mass spectrometric studies of carbon 13 variations in corn and other grasses. Radiocarbon 1968, 10, 468–472. [Google Scholar] [CrossRef]
- Bender, M.M. Variations in the 13C/12C ratios of plants in relation to the pathway of photosynthetic carbon dioxide fixation. Phytochemistry 1971, 10, 1239–1244. [Google Scholar] [CrossRef]
- Boutton, T.W.; Arshad, M.A.; Tieszen, L.L. Stable isotope analysis of termite food habits in East African grasslands. Oecologia 1983, 59, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Boutton, T.W.; Smith, B.N.; Harrison, A.T. Carbon isotope ratios and crop analyses of Arphia (Orthoptera: Acrididae) species in southeastern Wyoming grassland. Oecologia 1980, 45, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.; Sherr, E.B. δ13C measurements as indicators of carbon flow in marine and freshwater ecosystems. In Stable Isotopes in Ecological Research; Rundel, P.W., Ehleringer, J.R., Nagy, K.A., Eds.; Springer: New York, NY, USA, 1989; pp. 196–229. [Google Scholar]
- Hobson, K.A. Use of stable-carbon isotope analysis to estimate marine and terrestrial protein content in gull diets. Can. J. Zool. 1987, 65, 1210–1213. [Google Scholar] [CrossRef]
- Tieszen, L.L.; Boutton, T.W. Stable carbon isotopes in terrestrial ecosystem research. In Stable Isotopes in Ecological Research; Rundel, P.W., Ehleringer, J.R., Nagy, K.A., Eds.; Springer: New York, NY, USA, 1989; pp. 167–195. [Google Scholar]
- Högberg, P. Tansley review No. 95 15N natural abundance in soil-plant systems. New Phytol. 1997, 137, 179–203. [Google Scholar]
- Ben-David, M.; Bowyer, R.T.; Duffy, L.K.; Roby, D.D.; Schell, D.M. Social behavior and ecosystem processes: River otter latrines and nutrient dynamics of terrestrial vegetation. Ecology 1998, 79, 2567–2571. [Google Scholar] [CrossRef]
- Miyake, Y.; Wada, E. The abundance ratio of 15N/14N in marine environments. Rec. Oceanogr. Work. Jpn. 1967, 9, 37–53. [Google Scholar]
- Minagawa, M.; Wada, E. Stepwise enrichment of 15N along food chains: Further evidence and the relation between δ15N and animal age. Geochim. Cosmochim. Acta 1984, 48, 1135–1140. [Google Scholar] [CrossRef]
- Hobson, K.A.; Alisauskas, R.T.; Clark, R.G. Stable-nitrogen isotope enrichment in avian tissues due to fasting and nutritional stress: Implications for isotopic analyses of diet. Condor 1993, 95, 388–394. [Google Scholar] [CrossRef]
- Phillips, D.L. Mixing models in analyses of diet using multiple stable isotopes: A critique. Oecologia 2001, 127, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.L.; Koch, P.L. Incorporating concentration dependence in stable isotope mixing models. Oecologia 2002, 130, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Schwarcz, H.P. Some theoretical aspects of isotope paleodiet studies. J. Archaeol. Sci. 1991, 18, 261–275. [Google Scholar] [CrossRef]
- Moore, J.W.; Semmens, B.X. Incorporating uncertainty and prior information into stable isotope mixing models. Ecol. Lett. 2008, 11, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Darimont, C.T.; Reimchen, T.E. Intra-hair stable isotope analysis implies seasonal shift to salmon in gray wolf diet. Can. J. Zool. 2002, 80, 1638–1642. [Google Scholar] [CrossRef]
- Hobson, K.A.; Schell, D.M.; Renouf, D.; Noseworthy, E. Stable carbon and nitrogen isotopic fractionation between diet and tissues of captive seals: Implications for dietary reconstructions involving marine mammals. Can. J. Fish. Aquat. Sci. 1996, 53, 528–533. [Google Scholar] [CrossRef]
- Schoeninger, M.J.; Iwaniec, U.T.; Nash, L.T. Ecological attributes recorded in stable isotope ratios of arboreal prosimian hair. Oecologia 1998, 113, 222–230. [Google Scholar] [CrossRef]
- Hobson, K.A.; McLellan, B.N.; Woods, J.G. Using stable carbon (δ13C) and nitrogen (δ15N) isotopes to infer trophic relationships among black and grizzly bears in the upper Columbia River basin, British Columbia. Can. J. Zool. 2000, 78, 1332–1339. [Google Scholar] [CrossRef]
- Kielland, K. Stable isotope signatures of moose in relation to seasonal forage composition: A hypothesis. Alces 2001, 37, 329–337. [Google Scholar]
- Schoeninger, M.J.; Bada, J.L. Carbon and nitrogen stable isotope variation in primate hair reflects ecological and dietary variation. In Abstracts of Papers of the American Chemical Society; American Chemical Society: Washington, DC, USA, 2001; Volume 221, p. U534. [Google Scholar]
- Severud, W.J.; Belant, J.L.; Windels, S.K.; Bruggink, J.G. Seasonal variation in assimilated diets of American beavers. Am. Midl. Nat. 2013, 169, 30–42. [Google Scholar] [CrossRef]
- Milligan, H.E.; Humphries, M.M. The importance of aquatic vegetation in beaver diets and the seasonal and habitat specificity of aquatic-terrestrial ecosystem linkages in a subarctic environment. Oikos 2010, 119, 1877–1886. [Google Scholar] [CrossRef]
- Zazzo, A.; Harrison, S.M.; Bahar, B.; Moloney, A.P.; Monahan, F.J.; Scrimgeour, C.M.; Schmidt, O. Experimental determination of dietary carbon turnover in bovine hair and hoof. Can. J. Zool. 2007, 85, 1239–1248. [Google Scholar] [CrossRef]
- Phillips, D.L.; Gregg, J.W. Uncertainty in source partitioning using stable isotopes. Oecologia 2001, 127, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Semmens, B.X.; Ward, E.J.; Moore, J.W.; Darimont, C.T. Quantifying inter- and intra-population niche variability using hierarchical Bayesian stable isotope mixing models. PLoS ONE 2009, 4, e6187. [Google Scholar] [CrossRef] [PubMed]
- Linn, R.M. The Spruce-Fir, Maple-Birch Transition in Isle Royale National Park. Ph.D. Thesis, Duke University, Durham, NC, USA, 1957. [Google Scholar]
- Sattler, R.L.; Willoughby, J.R.; Swanson, B.J. Decline of heterozygosity in a large but isolated population: A 45-year examination of moose genetic diversity on Isle Royale. PeerJ 2017, 5, e3584. [Google Scholar] [CrossRef]
- Mech, L.D. The wolves of Isle Royale; U.S. National Park Service Fauna Series, no. 7. U.S. Government Printing Office: Washington, DC, USA, 1966.
- Risenhoover, K.A. Winter Foraging Strategies of Moose in Subarctic and Boreal Forest Habitats. Ph.D. Thesis, Michigan Technological University, Houghton, MI, USA, 1987. [Google Scholar]
- Peek, J.M.; Urich, D.L.; Mackie, R.J. Moose habitat selection and relationships to forest management in northeastern Minnesota. Wildl. Monogr. 1976, 48, 3–65. [Google Scholar]
- Ackerman, T.N. Moose Response to Summer Heat on Isle Royale. Master’s Thesis, Michigan Technological University, Houghton, MI, USA, 1987. [Google Scholar]
- Slavick, A.D.; Janke, R.A. The vascular flora of Isle Royale national park. Mich. Bot. 1987, 26, 91–134. [Google Scholar]
- Wolfe, M.L. Age determination in moose from cemental layers of molar teeth. J. Wildl. Manag. 1969, 33, 428–431. [Google Scholar] [CrossRef]
- Neiland, K.A. Weight of dried marrow as indicator of fat in caribou femurs. J. Wildl. Manag. 1970, 34, 904–907. [Google Scholar] [CrossRef]
- Barnett, B.A. Carbon and Nitrogen Ratios of Caribou Tissues, Vascular Plants, and Lichens from Northern Alaska. Master’s Thesis, University of Alaska, Fairbanks, AK, USA, 1994. [Google Scholar]
- Grue, H.; Jensen, B. Review of the formation of incremental lines in tooth cementum of terrestrial mammals. Dan. Rev. Game Biol. 1979, 11, 3–48. [Google Scholar]
- Guthrie, R.D.; Stoker, S. Paleoecological significance of mummified remains of Pleistocene horses from the North Slope of the Brooks Range, Alaska. Arctic 1990, 43, 267–274. [Google Scholar] [CrossRef]
- Rosing, M.N.; Ben-David, M.; Barry, R.P. Analysis of stable isotope data: AK nearest-neighbors randomization test. J. Wildl. Manag. 1998, 62, 380–388. [Google Scholar] [CrossRef]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar]
- McArdle, B.H.; Anderson, M.J. Fitting multivariate models to community data: A comment on distance-based redundancy analysis. Ecology 2001, 82, 290–297. [Google Scholar] [CrossRef]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002; ISBN 0-521-00976-6. [Google Scholar]
- Parnell, A.C.; Inger, R.; Bearhop, S.; Jackson, A.L. Source partitioning using stable isotopes: Coping with too much variation. PLoS ONE 2010, 5, e9672. [Google Scholar] [CrossRef]
- Sponheimer, M.; Robinson, T.F.; Ayliffe, L.K.; Passey, B.H.; Roeder, B.; Shipley, L.; Lopez, E.; Cerling, T.E.; Dearing, M.D.; Ehleringer, J.R. An experimental study of carbon-isotope fractionation between diet, hair, and feces of mammalian herbivores. Can. J. Zool. 2003, 81, 871–876. [Google Scholar] [CrossRef]
- Jones, R.J.; Ludlow, M.M.; Troughton, J.H.; Blunt, C.G. Changes in the natural carbon isotope ratios of the hair from steers fed diets of C4, C3 and C4 species in sequence. J. Aust. N. Z. Assoc. Adv. Sci. 1981, 12, 85–87. [Google Scholar]
- Cerling, T.E.; Harris, J.M. Carbon isotope fractionation between diet and bioapatite in ungulate mammals and implications for ecological and paleoecological studies. Oecologia 1999, 120, 347–363. [Google Scholar] [CrossRef]
- Sponheimer, M.; Robinson, T.; Ayliffe, L.; Roeder, B.; Hammer, J.; Passey, B.; West, A.; Cerling, T.; Dearing, D.; Ehleringer, J. Nitrogen isotopes in mammalian herbivores: Hair δ15N values from a controlled feeding study. Int. J. Osteoarchaeol. 2003, 13, 80–87. [Google Scholar] [CrossRef]
- Bada, J.L.; Peterson, R.O.; Schimmelmann, A.; Hedges, R.E.M. Moose teeth as monitors of environmental isotopic parameters. Oecologia 1990, 82, 102–106. [Google Scholar] [CrossRef]
- Krefting, L.W. The Ecology of the Isle Royale Moose with Special Reference to the Habitat; Agricultural Experiment Station Technical Bulletin; Minnesota Agricultural Experiment Station: Minneapolis, MN, USA, 1974; pp. 1–75. [Google Scholar]
- DelGiudice, G.D.; Peterson, R.O.; Seal, U.S. Differences in urinary chemistry profiles of moose on Isle Royale during winter. J. Wildl. Dis. 1991, 27, 407–416. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Parikh, G.L.; Forbey, J.S.; Robb, B.; Peterson, R.O.; Vucetich, L.M.; Vucetich, J.A. The influence of plant defensive chemicals, diet composition, and winter severity on the nutritional condition of a free-ranging, generalist herbivore. Oikos 2017, 126, 196–203. [Google Scholar] [CrossRef]
- Peterson, R.O.; Vucetich, J.A. Ecological Studies of Wolves on Isle Royale. Annual Report 2001–2002; Michigan Technological University: Houghton, MI, USA, 2002. [Google Scholar]
- Dunton, K.H.; Schonberg, S.V.; Cooper, L.W. Food web structure of the Alaskan nearshore shelf and estuarine lagoons of the Beaufort Sea. Estuaries Coasts 2012, 35, 416–435. [Google Scholar] [CrossRef]
- Chibowski, P.; Zalewski, A.; Suska-Malawska, M.; Brzeziński, M. Study on geographical differences in American mink diets reveals variations in isotopic composition of potential mink prey. Mamm. Res. 2019, 64, 343–351. [Google Scholar] [CrossRef]
- Hoy, S.R.; Peterson, R.O.; Vucetich, J.A. Ecological Studies of Wolves on Isle Royale; Michigan Technological University: Houghton, MI, USA, 2019; pp. 1–16. [Google Scholar]
- Milligan, H.E.; Pretzlaw, T.D.; Humphries, M.M. Stable isotope differentiation of freshwater and terrestrial vascular plants in two subarctic regions. Ecoscience 2010, 17, 265–275. [Google Scholar] [CrossRef]
- Webb, S.C.; Hedges, R.E.; Simpson, S.J. Diet quality influences the δ13C and δ15N of locusts and their biochemical components. J. Exp. Biol. 1998, 201, 2903–2911. [Google Scholar]
- Oelbermann, K.; Scheu, S. Stable isotope enrichment (δ15N and δ13C) in a generalist predator (Pardosa lugubris, Araneae: Lycosidae): Effects of prey quality. Oecologia 2002, 130, 337–344. [Google Scholar] [CrossRef]
- Gannez, L.Z.; O’Brien, D.M.; Del Rio, C.M. Stable isotopes in animal ecology: Assumptions, caveats, and a call for more laboratory experiments. Ecology 1997, 78, 1271–1276. [Google Scholar] [CrossRef]
- Adams, T.S.; Sterner, R.W. The effect of dietary nitrogen content on trophic level 15N enrichment. Limnol. Oceanogr. 2000, 45, 601–607. [Google Scholar] [CrossRef]
- Boon, P.I.; Bunn, S.E. Variations in the stable isotope composition of aquatic plants and their implications for food web analysis. Aquat. Bot. 1994, 48, 99–108. [Google Scholar] [CrossRef]
- Finlay, J.C.; Kendall, C. Stable isotope tracing of temporal and spatial variability in organic matter sources to freshwater ecosystems. Stable Isot. Ecol. Environ. Sci. 2007, 2, 283–333. [Google Scholar]
- West, A.G.; Ayliffe, L.K.; Cerling, T.E.; Robinson, T.F.; Karren, B.; Dearing, M.D.; Ehleringer, J.R. Short-term diet changes revealed using stable carbon isotopes in horse tail-hair. Funct. Ecol. 2004, 18, 616–624. [Google Scholar] [CrossRef]


| Moose ID | n† | ∆δ13C | δ15N |
|---|---|---|---|
| 3618 | 21 | −25.6 ± 0.4 | 0.8 ± 0.4 |
| 3627 | 16 | −25.8 ± 0.4 | 0.7 ± 0.3 |
| 3628 | 21 | −25.5 ± 0.3 | 0.9 ± 0.3 |
| 3723 | 21 | −26.0 ± 0.4 | 0.1 ± 0.3 |
| 3731 | 21 | −25.5 ± 0.3 | 0.4 ± 0.2 |
| 3734 | 14 | −24.8 ± 0.7 | −0.3 ± 0.3 |
| 3766 | 19 | −24.4 ± 0.8 | 1.1 ± 0.5 |
| 3767 | 21 | −25.1 ± 0.5 | 0.4 ± 0.2 |
| 3774 | 19 | −25.5 ± 0.2 | 0.6 ± 0.2 |
| 3784 | 19 | −25.1 ± 0.3 | 0.1 ± 0.2 |
| 3788 | 16 | −25.1 ± 0.4 | 0.4 ± 0.4 |
| 3808 | 19 | −25.1 ± 0.4 | 0.6 ± 0.3 |
| 3828 | 25 | −25.2 ± 0.4 | 0.6 ± 0.6 |
| Trophic Discrimination | ∆13C1 (3.0‰) | ∆13C2 (1.8‰) | - |
|---|---|---|---|
| Sources | Percent of Diet | |∆−∆| | |
| Summer diet (2-source model) | |||
| Terrestrial leaves | 86.8 ± 3.3 | 72.8 ± 3.6 | 14.0 |
| Aquatic macrophytes | 13.2 ± 3.3 | 27.2 ± 3.6 | |
| Winter diet (2-source model) | |||
| East | |||
| Terrestrial twigs | 99.9 ± 0.01 | 89.2 ± 9.4 | 10.7 |
| Lichens | 0.01 ± 0.5 | 10.8 ± 9.4 | |
| West | |||
| Terrestrial twigs | 96.3 ± 4.9 | 71.8 ± 11.3 | 24.5 |
| Lichens | 3.7 ± 4.9 | 28.2 ± 11.3 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tischler, K.B.; Severud, W.J.; Peterson, R.O.; Bump, J.K. Aquatic Macrophytes are Seasonally Important Dietary Resources for Moose. Diversity 2019, 11, 209. https://doi.org/10.3390/d11110209
Tischler KB, Severud WJ, Peterson RO, Bump JK. Aquatic Macrophytes are Seasonally Important Dietary Resources for Moose. Diversity. 2019; 11(11):209. https://doi.org/10.3390/d11110209
Chicago/Turabian StyleTischler, Keren B., William J. Severud, Rolf O. Peterson, and Joseph K. Bump. 2019. "Aquatic Macrophytes are Seasonally Important Dietary Resources for Moose" Diversity 11, no. 11: 209. https://doi.org/10.3390/d11110209
APA StyleTischler, K. B., Severud, W. J., Peterson, R. O., & Bump, J. K. (2019). Aquatic Macrophytes are Seasonally Important Dietary Resources for Moose. Diversity, 11(11), 209. https://doi.org/10.3390/d11110209






