Bioindication of the Influence of Oil Production on Sphagnum Bogs in the Khanty-Mansiysk Autonomous Okrug–Yugra, Russia
Abstract
:1. Introduction
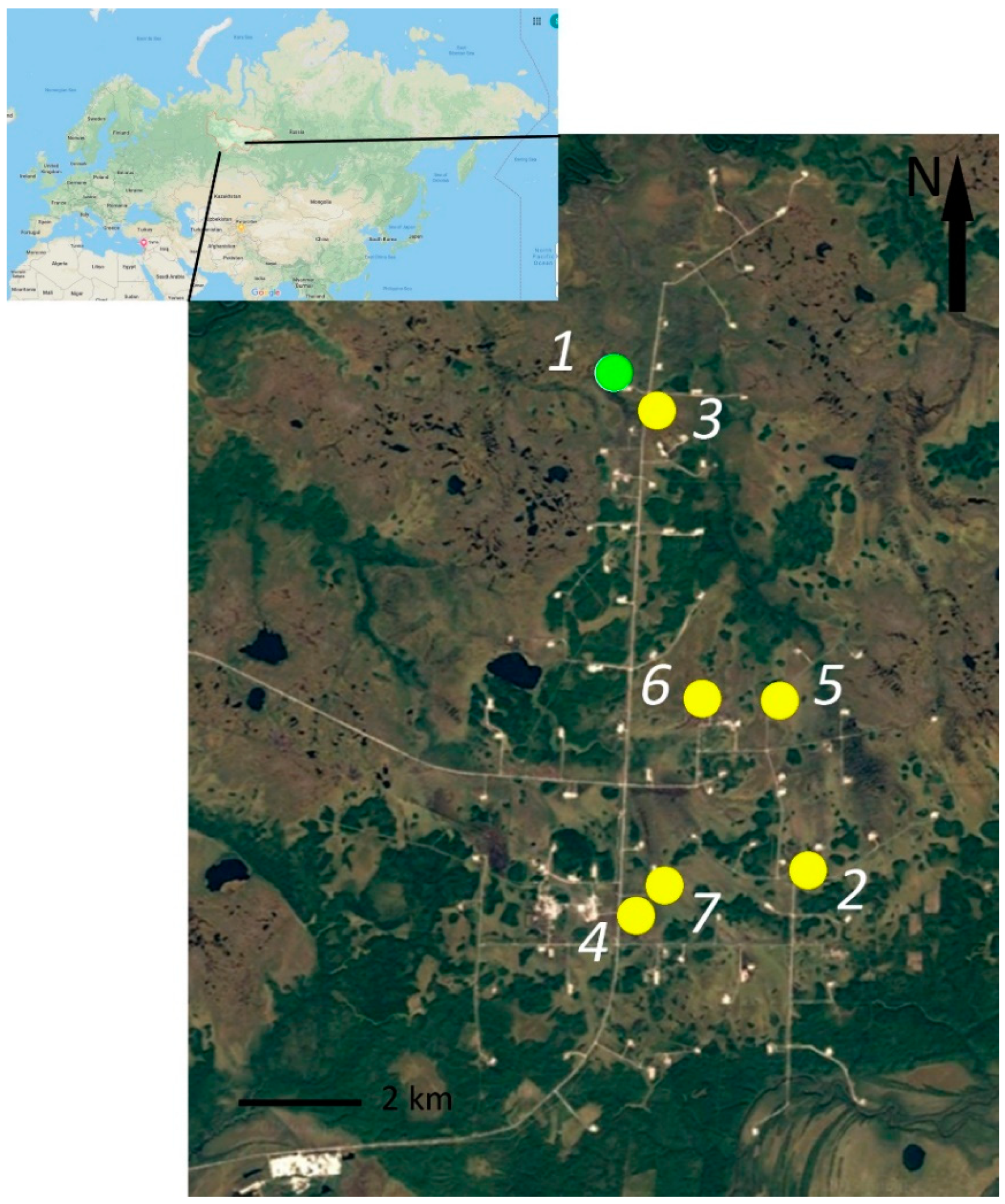
2. Study Site Description
3. Material and Methods
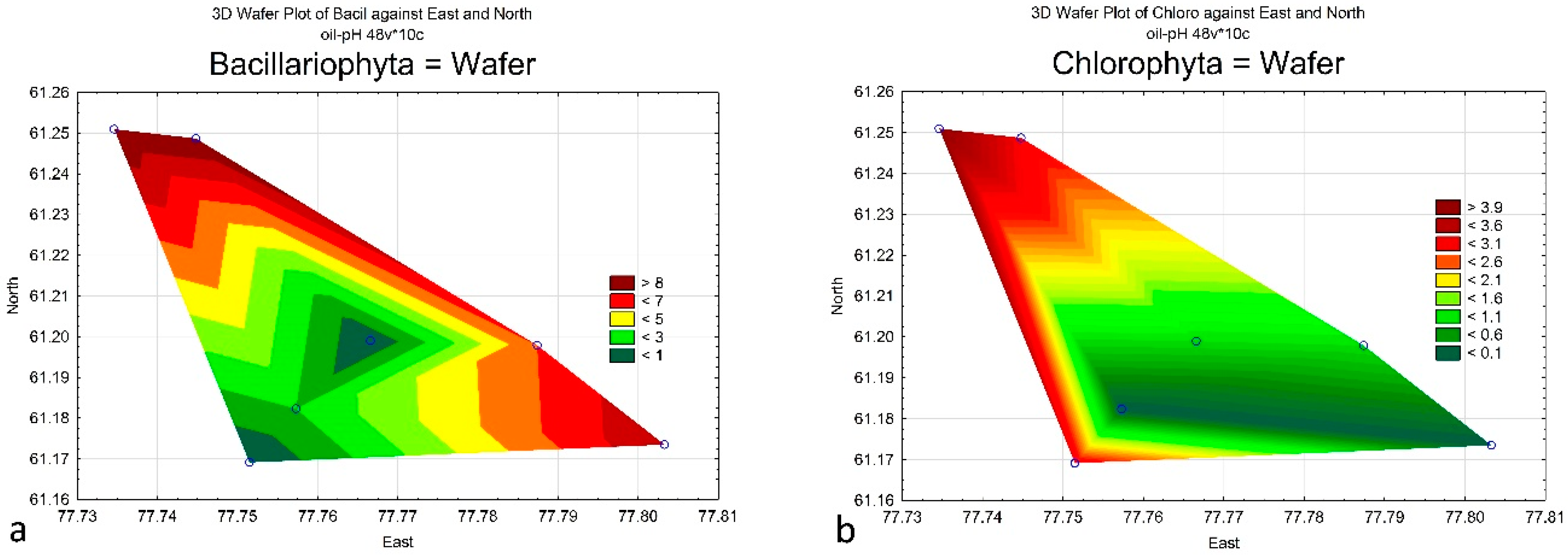
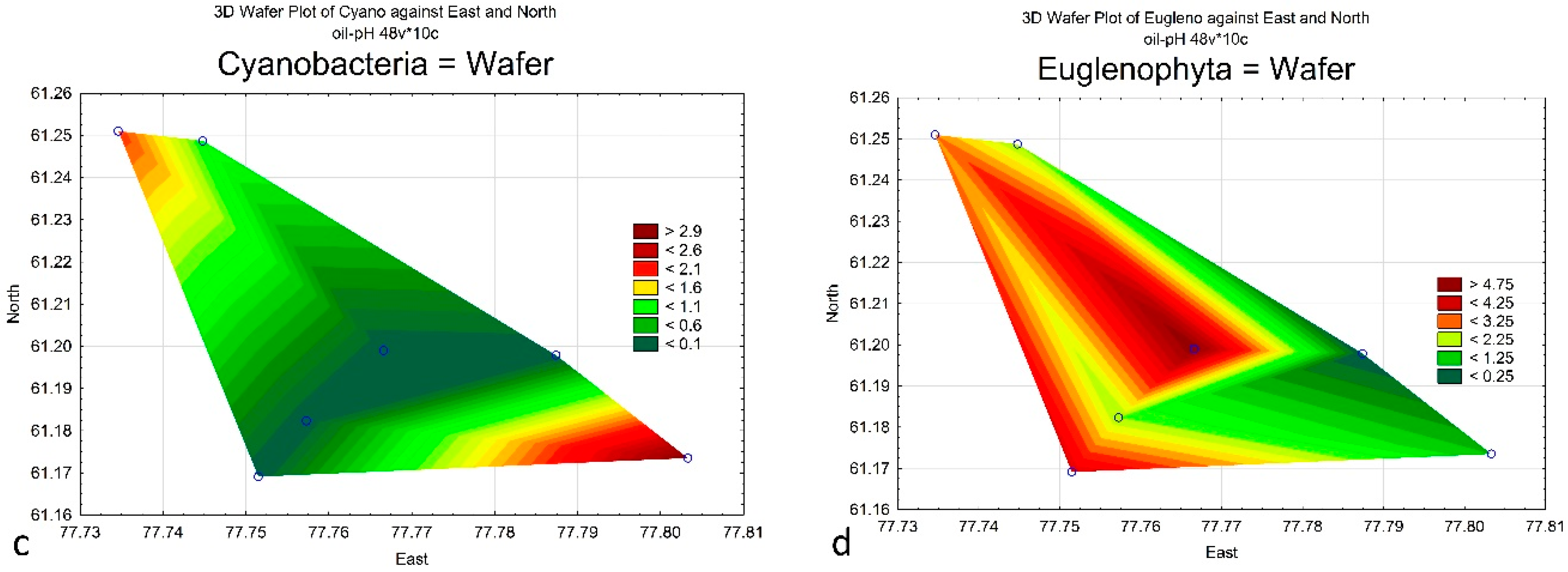
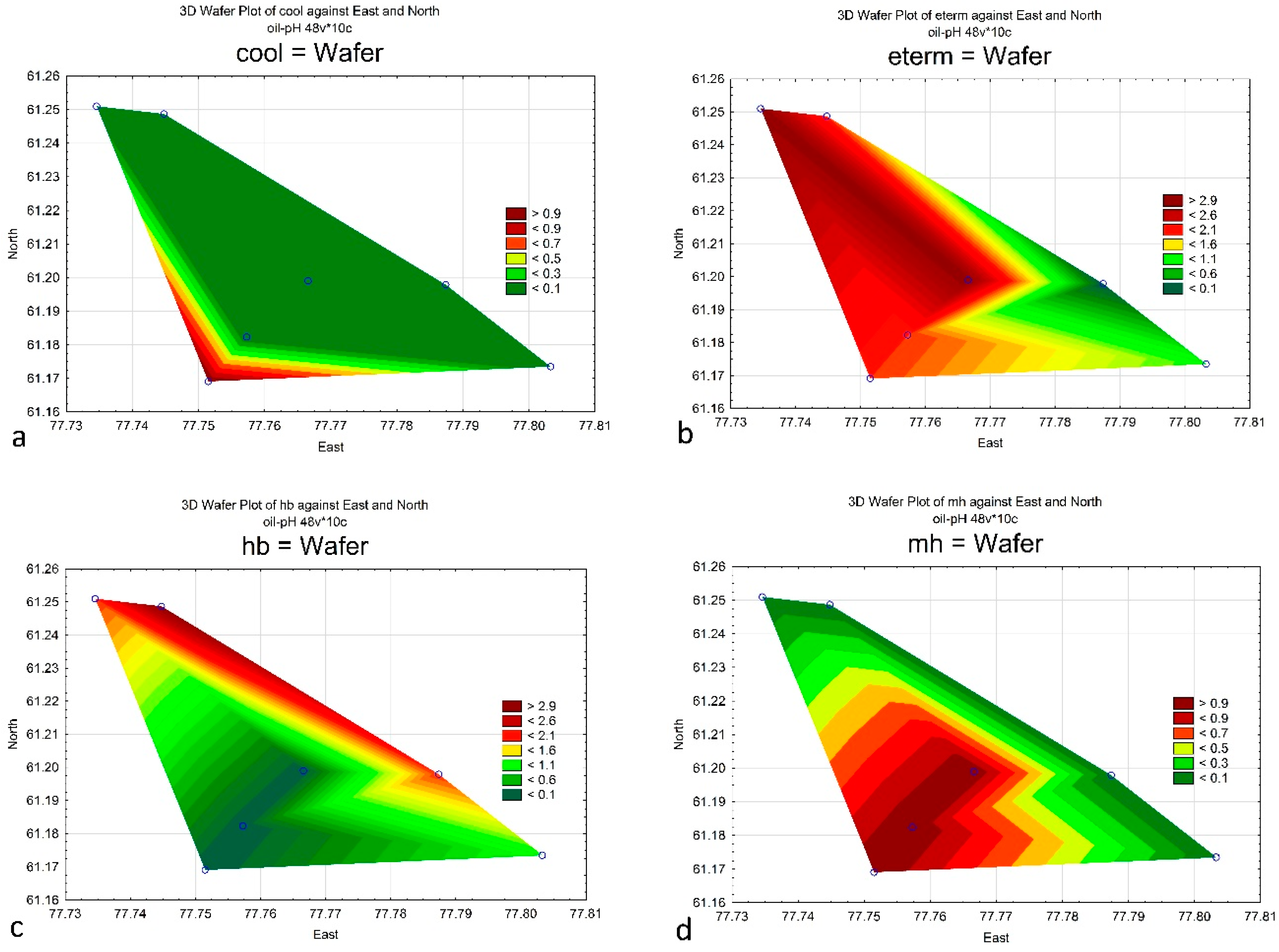
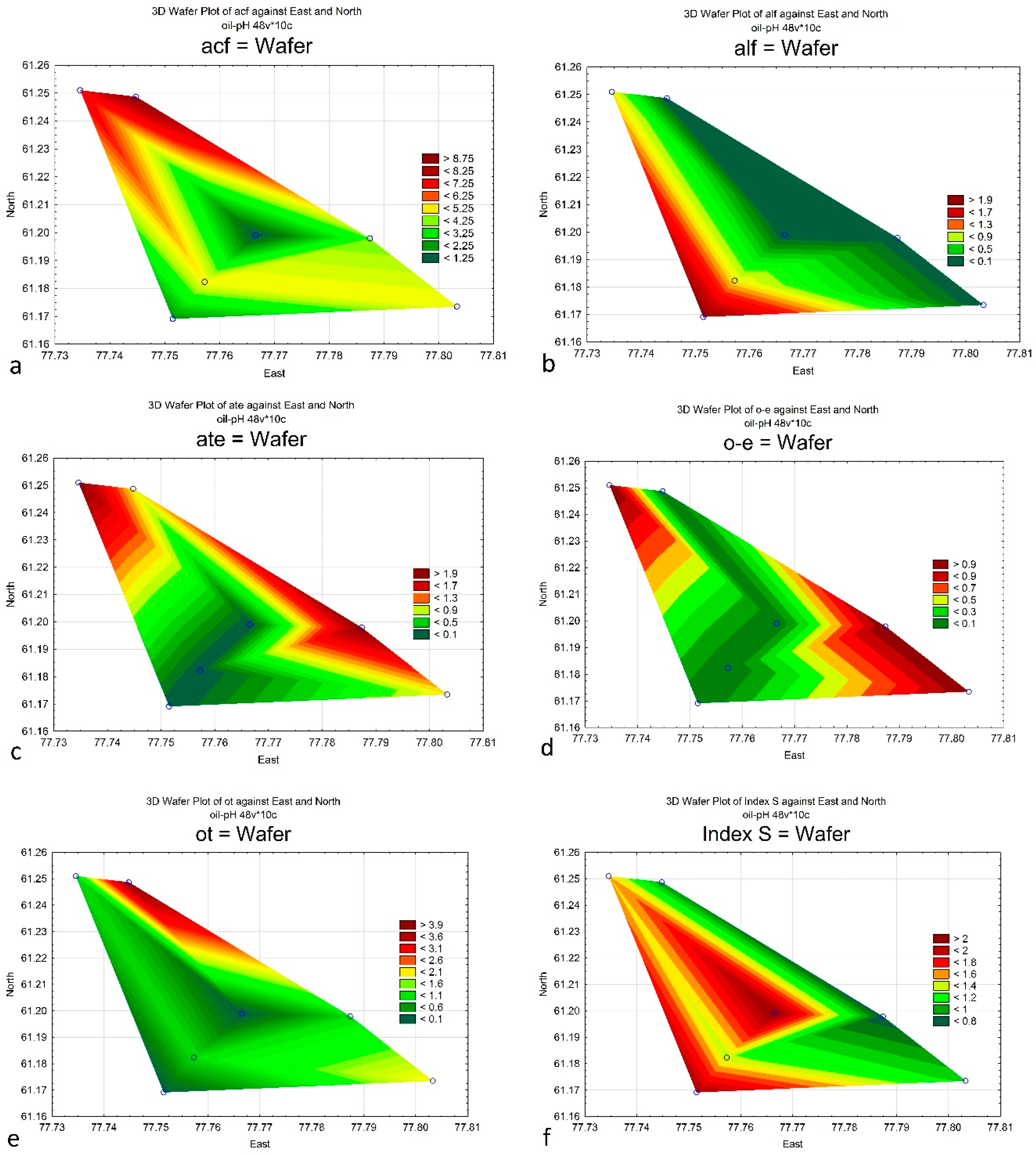
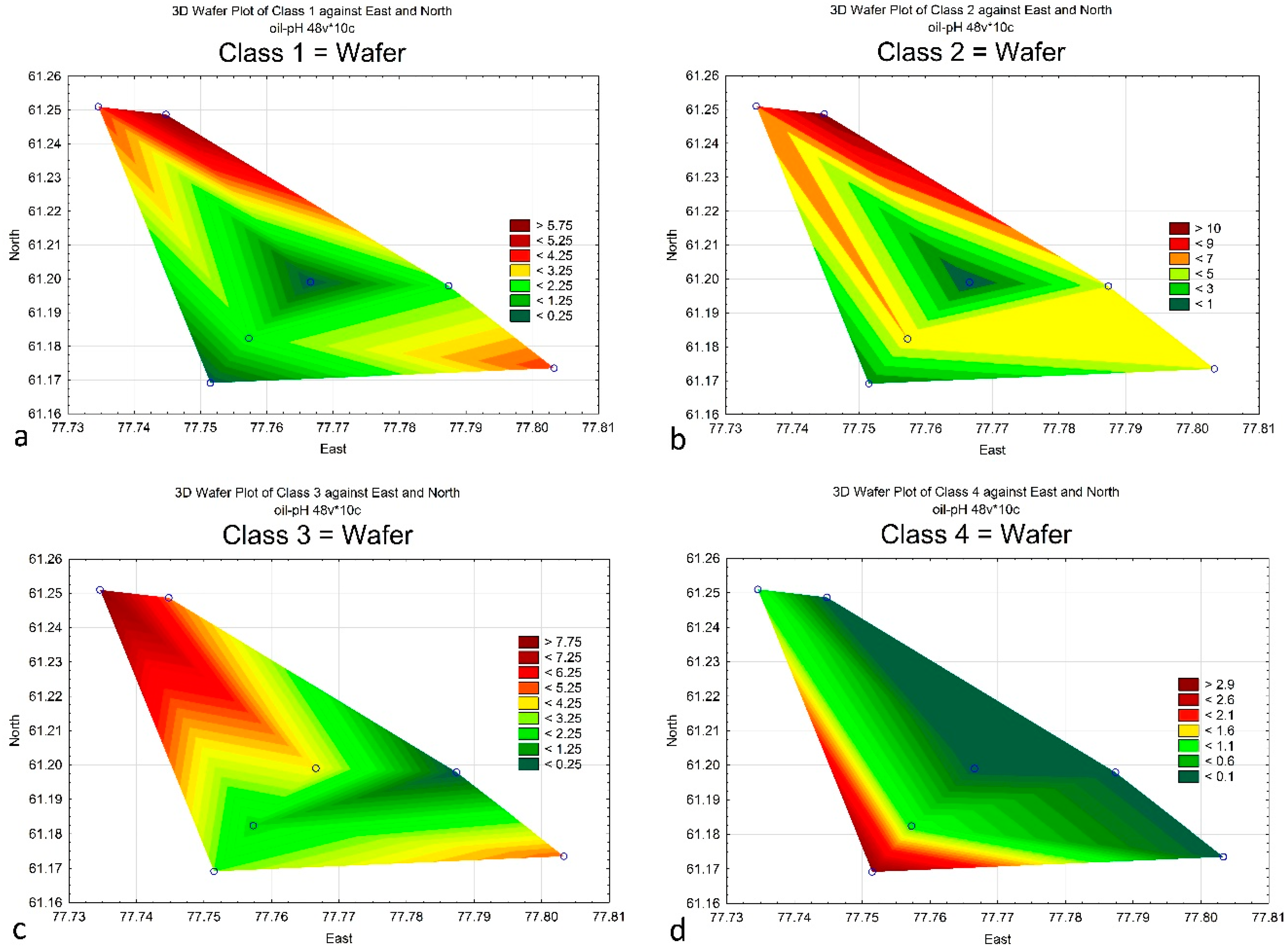
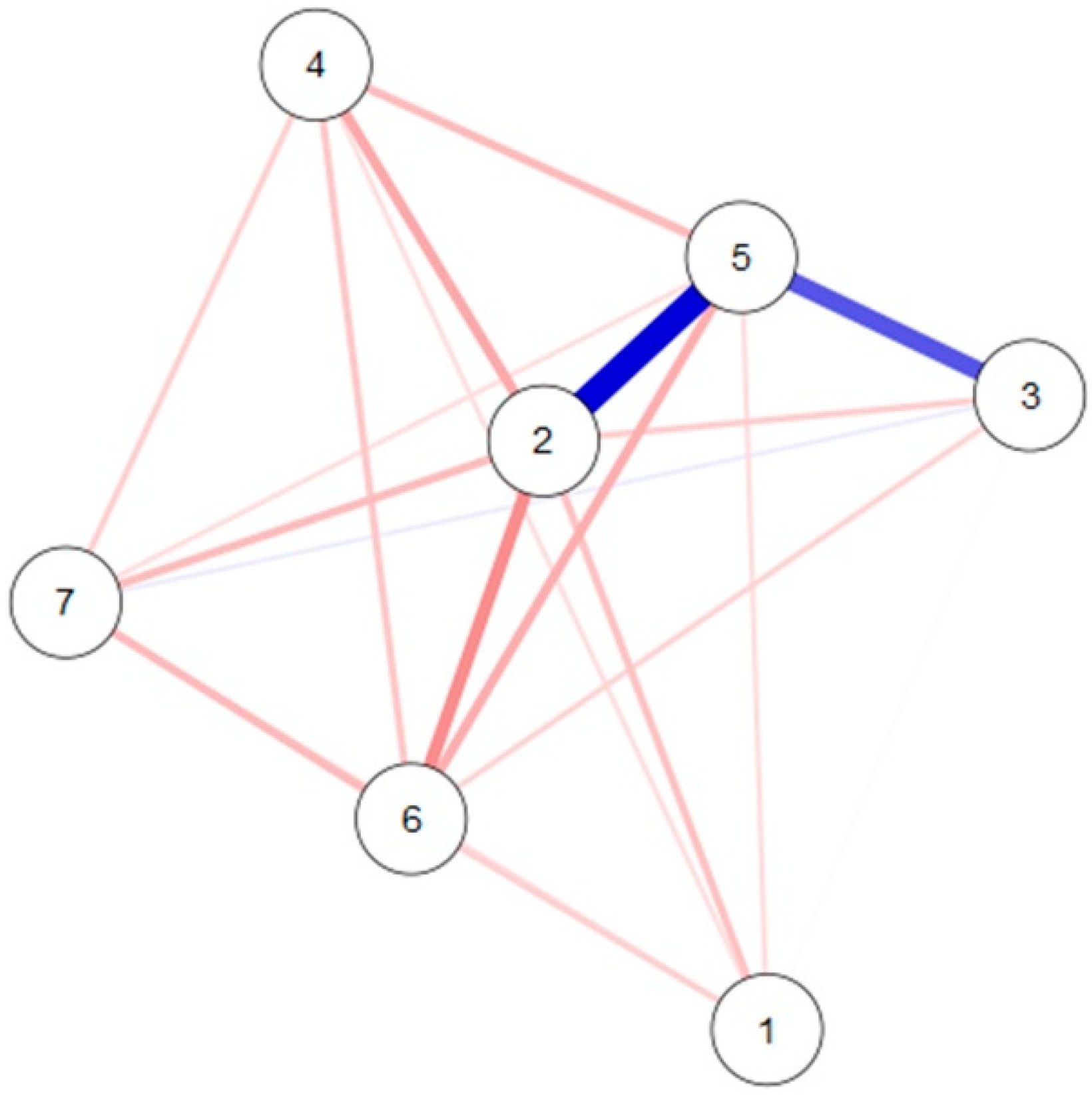
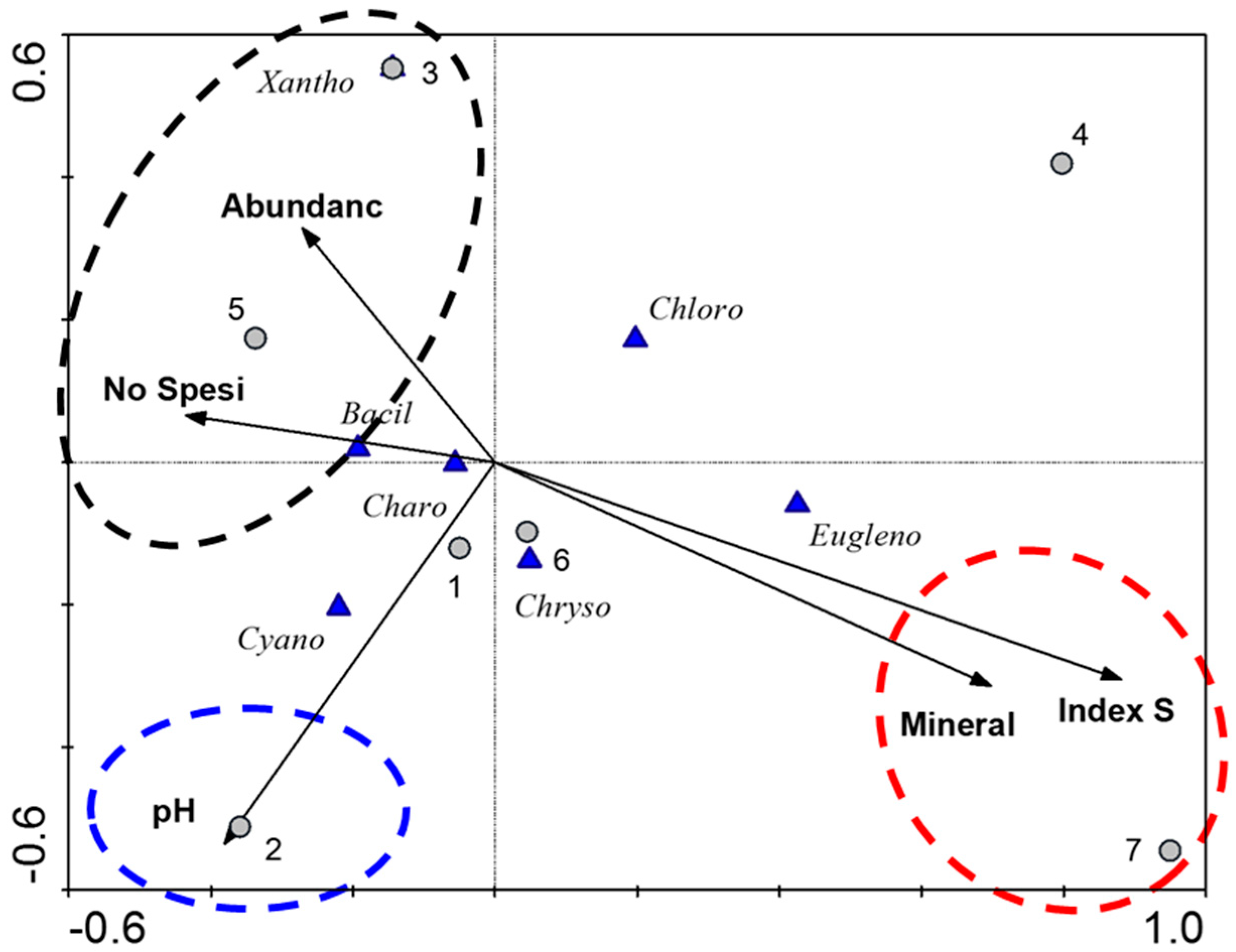
4. Results
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mendelssohn, I.A.; Andersen, G.L.; Baltz, D.M.; Caffey, R.H.; Carman, K.R.; Fleeger, J.W.; Joye, S.B.; Lin, Q.; Maltby, E.; Overton, E.B.; et al. Oil Impacts on Coastal Wetlands: Implications for the Mississippi River Delta Ecosystem after the Deepwater Horizon Oil Spill. BioScience 2012, 62, 562–574. [Google Scholar] [CrossRef]
- EPA. Available online: https://archive.epa.gov/emergencies/content/learning/web/html/freshwat.html (accessed on 29 September 2019).
- Sherrington, M. Biodiversity assessment in the Oil Sands region, northeastern Alberta, Canada. Impact Assess. Proj. Apprais. 2005, 23, 73–81. [Google Scholar] [CrossRef]
- Kazantseva, M.N. The effect of oil extraction on ground cover of West Siberian taiga forests. Contemp. Probl. Ecol. 2011, 4, 582–587. [Google Scholar] [CrossRef]
- Plotnikov, V.V. (Ed.) Ecology of the Khanty-Mansiysk Autonomous Okrug; Soft Design: Tyumen, Russia, 1997; 228p. [Google Scholar]
- Chizhov, B.E. Forest and Oil Khanty-Mansi Autonomous Okrug; Soft Design: Tyumen, Russia, 1998; 144p. [Google Scholar]
- Korkin, S.E.; Isypov, V.A. Channel deformations in the lower reaches of the Vah River. In Proceedings of the Thirty-Third Plenary Interuniversity Coordination Meeting on Erosion, Channel and Estuarine Processes, Nizhnevartovsk, Russia, 2–4 October 2018. [Google Scholar]
- Skorobogatova, O.N.; Usmanov, I.Y. First information about algae of the lakes Vilent and Samotlor (Western Siberia, Khanty-Mansi Autonomous Okrug-Yugra). World Sci. Discov. 2016, 5, 146–151. [Google Scholar] [CrossRef]
- Bellinger, E.G.; Sigee, D.C. Freshwater Algae: Identification and Use as Bioindicators; John Wiley and Sons: Chichester, UK, 2010. [Google Scholar]
- Barinova, S. Essential and practical bioindication methods and systems for the water quality assessment. Int. J. Environ. Sci. Nat. Resour. 2017, 2, 1–11. [Google Scholar] [CrossRef]
- Naumenko, Y.V.; Skorobogatova, O.N. Species of the genus Eunotia Ehr. in phytoplankton of the Vah river (Western Siberia). Turczaninowia 2009, 12, 65–70. [Google Scholar]
- Skorobogatova, O.N.; Galimzyanova, S.T. Floristic-taxonomic review of green algae Chlorophyceae and Conjugatophyceae of Lake Rangetur. In Proceedings of the Culture, Science, Education: Problems and Prospects: Materials of the VI All-Russian Scientific and Practical Conference, Nizhnevartovsk, Russia, 6 February 2017. [Google Scholar]
- Skorobogatova, O.N.; Hydora, O.Y. Structure of algae of bogs (Nizhnevartovsk district, KMAO-Yugra). Proc. Inst. Biol. Intern. Waters RAS 2017, 79, 207–212. [Google Scholar]
- Skorobogatova, O.N.; Naumenko, Y.V. The role of swamps in the formation of phytoplankton of the Vakh River. In Proceedings of the West Siberian Peatlands and the Carbon Cycle: Past and Present, Proceedings of III International field Symposium, Khanty-Mansiysk, Russia, 27 June–5 July 2011; pp. 71–72. [Google Scholar]
- Fazlutdinova, A.I.; Sukhanova, N.V. Composition of diatoms in the zone of influence of oil field complexes. Ecology 2014, 3, 197–203. [Google Scholar]
- Lavrentieva, G.M. Guidelines for the Collection and Processing of Materials in Hydrobiological Studies in Freshwater Bodies; GosNIORH: Leningrad, Russia, 1984; p. 32. [Google Scholar]
- Alekin, O.A.; Semenov, A.D.; Skopintsev, B.A. Manual on the Chemical Analysis of Land Waters; Gidrometeoizdat: Leningrad, Russia, 1973; p. 268. [Google Scholar]
- Guiry, M.D.; Guiry, G.M. AlgaeBase World-wide electronic publication. National University of Ireland, Galway. Available online: http://www.algaebase.org 01.01.2019 (accessed on 17 September 2019).
- Barinova, S. Ecological Mapping in Application to Aquatic Ecosystems BioIndication: Problems and Methods. Int. J. Environ. Sci. Nat. Resour. 2017, 3, 1–7. [Google Scholar] [CrossRef]
- Barinova, S.S.; Bilous, O.P.; Tsarenko, P.M. Algal Indication of Water Bodies in Ukraine: Methods and Prospects; Publishing House of Haifa University: Haifa, Israel, 2019; p. 367. (In Russian) [Google Scholar]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Microcomputer Power Press: Ithaca, NY, USA, 2002; 500p. [Google Scholar]
- Love, J.; Selker, R.; Marsman, M.; Jamil, T.; Dropmann, D.; Verhagen, A.J.; Ly, A.; Gronau, Q.F.; Smira, M.; Epskamp, S.; et al. JASP: Graphical statistical software for common statistical designs. J. Stat. Softw. 2019, 88, 1–17. [Google Scholar] [CrossRef]
- Kireeva, N.A.; Dubovik, I.E.; Yakupova, A.B. Influence of different methods of bioremedia on the algocenosis of oil-contaminated soil. Soil Sci. 2011, 11, 1375–1385. [Google Scholar]
- Pauwels, H.; Talbo, H. Nitrate concentration in wetlands: Assessing the contribution of deeper groundwater from anions. Water Res. 2004, 38, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Julian, P.; Gerber, S.; Bhomia, R.K.; King, J.; Osborne, T.Z.; Wright, A.L.; Powers, M.; Dombrowski, J. Evaluation of nutrient stoichiometric relationships among ecosystem compartments of a subtropical treatment wetland. Do we have “Redfield wetlands”? Ecol. Process. 2019, 8, 20. [Google Scholar] [CrossRef]
- Sotek, Z.; Stasińska, M.; Malinowski, R.; Gamrat, R.; Gałczyńska, M. Birch Bog on Anthropogenically Transformed Raised Bogs. A Case Study from Pomerania (Poland). Water 2019, 11, 1224. [Google Scholar] [CrossRef]
- Volkov, I.V.; Skorobogatova, O.N. Green algae of the swamp massif in the area of the Agan and Novopokursky deposits. In Intercultural dialogue and cooperation between the EU and Russia: Experience in implementing projects of Jean Monnet at the Nizhnevartovsk State University. In Proceedings of the International Scientific and Practical Conference, Nizhnevartovsk, Russia, 15–19 April 2019. [Google Scholar]
- Rochefort, L.; Kooijman, A.M.; Roelofs, J.G. Ecological Restoration of Rich Fens in Europe and North America: From Trial and Error to an Evidence-Based Approach. Biol. Rev. 2014, 90, 182–203. Available online: https://onlinelibrary.wiley.com/doi/full/10.1111/brv.12102 (accessed on 2 October 2019).


| Site | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
| North | 77° 44′ 4.5132″ | 77° 48′ 11.9088″ | 77° 44′ 41.2908″ | 77° 45′ 5.3712″ | 77° 47′ 14.514″ | 77° 45′ 59.9184″ | 77° 45′ 26.1936″ |
| East | 61° 15′ 3.7764″ | 61° 10′ 24.5568″ | 61° 14′ 55.41″ | 61° 10′ 8.7672″ | 61° 11′ 52.3572″ | 61° 11′ 56.3568″ | 61° 10′ 15.8124″ |
| pH | 4.1–4.8 | 5.4 | 4.0–4.5 | 4.6 | 4.3 | 4.2 | 4.1 |
| Mineral oil, mg kg−1 | 255.65 ± 1.50 | 419.40 ± 1.30 | 1203.10 ± 25.00 | 1500.38 ± 21.20 | 1715.47 ± 4.56 | 3308.52 ± 5.33 | 16,893.80 ± 2.54 |
| T °C | 21 | 21 | 22 | 22 | 22 | 23 | 22 |
| Years after pollution | 0 | 20 | 10 | 7 | 10 | 20 | 10 |
| State | Unimpaired | Reclaimed | Reclaimed | Self-healing | Self-healing | - | Self-healing |
| Taxa | 1 | 2 | 3 | 4 | 5 | 6 | 7 | Hab | T | Oxy | pH | Sal | Wat | Sap | S | Tro | Aut-Het |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cyanobacteria | |||||||||||||||||
| Amorphonostoc paludosum (Kützing ex Bornet and Flahault) Elenkin | - | 1 | - | - | - | - | - | P-B, S | - | st | - | - | - | b-o | 1.6 | m | - |
| Anabaena minutissima Lemmermann | - | - | - | 1 | - | - | - | B | - | - | - | - | - | - | - | - | - |
| Anabaena sp. 1 | - | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - |
| Anathece clathrata (West and G.S. West) Komárek, Kastovsky and Jezberová | - | - | - | 1 | - | - | - | P | - | - | - | hl | - | o-a | 1.8 | me | - |
| Aphanizomenon flosaquae Ralfs ex Bornet and Flahault | - | - | - | - | - | - | 1 | P | - | - | - | hl | - | o-a | 1.95 | m | - |
| Aphanizomenon sp. | - | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - |
| Chroococcus turgidus (Kützing) Nägeli | - | - | - | 1 | - | - | - | P-B, S | - | aer | alf | hl | - | x-b | 0.8 | - | - |
| Coelosphaerium dubium Grunow | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Coelosphaerium kuetzingianum Nägeli | - | - | 1 | - | - | - | - | P | - | - | - | i | - | b-o | 1.6 | m | - |
| Cylindrospermum michailovskoense Elenkin | - | - | - | - | - | - | 1 | B | - | - | - | - | - | - | - | me | - |
| Gloeocapsopsis magma (Brébisson) Komárek and Anagnostidis ex Komárek | - | - | - | 1 | - | - | - | S | - | - | ind | i | - | - | - | - | - |
| Leptobasis striatula (F.C. Hy) Elenkin | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Merismopedia tenuissima Lemmermann | 1 | - | - | - | - | - | - | P-B | - | - | - | hl | - | b-a | 2.4 | e | - |
| Microcystis aeruginosa (Kützing) Kützing | - | - | 1 | - | - | - | - | P | - | - | - | hl | - | b | 2.1 | e | - |
| Nodularia spumigena Mertens ex Bornet and Flahault | - | 1 | - | - | - | - | - | B, S | - | - | - | - | - | o-a | 1.8 | - | - |
| Scytonema coactile Montagne ex Bornet and Flahault | - | 1 | - | - | - | - | - | - | - | - | - | - | - | b | 2 | - | - |
| Synechocystis crassa Woronichin | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Ochophyta (Chrysophyta) | |||||||||||||||||
| Chrysococcus rufescens Klebs | 1 | - | - | - | - | - | - | - | - | - | - | hb | - | o-b | 1.4 | - | - |
| Dinobryon cylindricum O.E.Imhof | - | - | 1 | - | - | - | - | P | - | - | - | i | - | o | 1.2 | - | - |
| Dinobryon divergens O.E.Imhof | - | - | 1 | - | - | 1 | - | P | - | st-str | ind | i | - | o-b | 1.45 | - | - |
| Dinobryon pediforme (Lemmermann) Steinecke | - | - | - | - | - | 1 | - | P | - | - | - | - | - | o | 1.2 | - | - |
| Dinobryon sertularia Ehrenberg | 1 | 1 | - | - | - | 1 | - | P | - | - | - | i | - | o | 1.3 | - | - |
| Dinobryon sociale (Ehrenberg) Ehrenberg | - | - | - | - | - | 1 | - | P | - | - | - | i | - | o | 1.2 | - | - |
| Mallomonas sp. | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Bacillariophyta | |||||||||||||||||
| Achnanthhes sp. | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Cyclotella meneghiniana Kützing | 1 | - | - | - | - | - | - | P-B | temp | st | alf | hl | sp | a-o | 2.8 | e | hne |
| Epithemia gibba (Ehrenberg) Kützing | - | 1 | 1 | - | - | - | - | B | temp | - | alb | i | es | x-o | 0.4 | - | - |
| Eunotia arcus Ehrenberg | 1 | - | - | - | - | 1 | - | B | - | st-str | acf | i | - | x-o | 0.5 | ot | ats |
| Eunotia exigua (Brébisson ex Kützing) Rabenhorst | 1 | - | - | - | 1 | - | - | P-B | - | st-str | acf | hb | es | x-o | 0.45 | o-e | ate |
| Eunotia lunaris (Ehrenberg) Grunow var. lunaris | 1 | 1 | 1 | - | 1 | 1 | - | B | - | st | ind | i | - | o | 1 | - | - |
| Eunotia lunaris var. capitata (Grunow) Schönfeldt | - | 1 | - | - | 1 | - | - | B | - | st | ind | i | - | - | - | - | - |
| Eunotia lunaris var. subarcuata (Nägeli ex Kützing) Grunow | - | 1 | - | - | - | - | - | B | - | st | ind | i | - | o | 1 | - | - |
| Eunotia major (W.Smith) Rabenhorst | - | - | 1 | - | - | - | - | B | - | - | acf | hb | - | x-o | 0.4 | - | - |
| Eunotia monodon Ehrenberg | - | - | 1 | - | - | - | - | B | - | st-str | acf | hb | - | x-o | 0.4 | ot | ats |
| Eunotia neocompacta S.Mayama | 1 | - | - | - | 1 | - | - | B | - | - | - | i | - | x-o | 0.5 | - | - |
| Eunotia parallela Ehrenberg | - | 1 | 1 | - | - | - | - | P-B | - | str | acf | i | - | x | 0.3 | ot | ats |
| Eunotia tenella (Grunow) Hustedt | - | - | - | - | 1 | - | - | B | - | str | acf | hb | es | o-x | 0.7 | ot | ats |
| Eunotia sp. | 1 | - | - | - | - | - | - | - | - | - | - | - | - | x-o | 0.4 | - | - |
| Frustulia saxonica Rabenhorst | - | 1 | - | - | - | - | - | B | - | st | acf | hb | es | x | 0.3 | ot | ats |
| Hantzschia amphioxys var. constricta Pantocsek | 1 | - | - | - | - | - | - | B | - | - | ind | i | - | - | - | - | - |
| Pinnularia interrupta W.Smith | - | 1 | - | - | - | - | - | B | - | - | ind | i | - | - | - | - | - |
| Pinnularia major (Kützing) Rabenhorst | - | - | 1 | - | - | - | - | B | temp | st-str | ind | i | - | o-x | 0.6 | me | ate |
| Pinnularia subcapitata W.Gregory | 1 | - | - | - | 1 | - | - | B | - | st-str | ind | i | sp | o-x | 0.6 | o-m | ate |
| Pinnularia sp. | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Tabellaria fenestrata (Lyngbye) Kützing | - | - | 1 | - | - | - | - | P-B | - | st-str | ind | i | es | x | 0.3 | o-m | ats |
| Tabellaria flocculosa (Roth) Kützing | - | - | 1 | - | - | - | - | P-B | eterm | st-str | acf | i | es | o-x | 0.6 | ot | ats |
| Ulnaria ulna (Nitzsch) Compère | - | 1 | - | - | - | - | - | P-B | temp | st-str | ind | i | es | b | 2.25 | o-e | ate |
| Euglenophyta | |||||||||||||||||
| Astasia curvata (G.A.Klebs) G.A.Klebs | - | - | - | 1 | - | - | - | P-B | eterm | st-str | - | - | - | a | 3.4 | - | - |
| Astasia sagittifera Skuja | - | - | - | 1 | - | - | - | P | cool | st-str | - | - | - | a | 3 | - | - |
| Euglena brevis P.Christ | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Euglena pisciformis Klebs | - | - | - | 1 | - | 1 | - | P-B | eterm | st-str | alf | mh | - | a | 3 | - | - |
| Eutreptia lanowii Steuer | - | - | - | - | - | - | 1 | - | - | - | - | mh | - | - | - | - | - |
| Lepocinclis steinii (Lemmermann) Lemmermann | - | - | - | - | - | - | 1 | P | eterm | st | ind | i | - | b | 2.2 | - | - |
| Menoidium tortuosum (Stokes) Lemmermann | - | - | - | - | - | - | 1 | P-B | eterm | st-str | ind | - | - | a | 3 | - | - |
| Phacus oscillans G.A.Klebs | - | - | - | - | - | - | 1 | P-B | - | st-str | ind | i | - | - | - | - | - |
| Strombomonas acuminata var. verrucosa Teodoresco | - | - | 1 | - | - | - | - | P | - | st-str | ind | i | - | b | 2.2 | - | - |
| Trachelomonas oblonga Lemmermann | 1 | - | - | - | - | - | - | P | eterm | st-str | - | i | - | b-a | 2.4 | - | - |
| Trachelomonas planctonica Svirenko | 1 | 1 | - | - | - | - | 1 | P | eterm | st-str | ind | i | - | b | 2.1 | - | - |
| Trachelomonas volvocina (Ehrenberg) Ehrenberg | 1 | - | 1 | - | - | 1 | - | B | eterm | st-str | ind | i | - | b | 2 | - | - |
| Ochrophyta (Xanthophyceae) | |||||||||||||||||
| Tribonema spirotaenia Ettl | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Chlorophyta | |||||||||||||||||
| Coelastrum microporum Nägeli | 1 | - | 1 | - | - | - | - | P-B | - | st-str | ind | i | - | b | 2.3 | - | - |
| Coenococcus planctonicus Korshikov | - | - | 1 | - | 1 | - | - | P | - | - | - | i | - | o-b | 1.5 | - | - |
| Kirchneriella obesa (West) West and G.S. West | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Mucidosphaerium pulchellum (H.C.Wood) C.Bock, Proschold and Krienitz | 1 | - | - | 1 | - | - | - | P-B | - | st-str | ind | i | - | b | 2.3 | - | - |
| Oocystis rhomboidea Fott | 1 | - | - | - | - | - | - | P | - | - | - | - | - | o-a | 1.8 | - | - |
| Sphaerocystis planctonica (Korshikov) Bourrelly | - | - | 1 | - | - | - | - | - | - | - | - | - | - | o | 1 | - | - |
| Tetradesmus obliquus (Turpin) M.J.Wynne | 1 | - | - | 1 | - | - | 1 | P-B | - | st-str | ind | i | - | b | 2.05 | - | - |
| Charophyta | |||||||||||||||||
| Actinotaenium cucurbitinum (Bisset) Teiling | 1 | - | - | - | - | - | - | P-B | - | - | acf | - | - | - | - | o-m | - |
| Actinotaenium globosum (Bulnheim) Kurt Förster ex Compère | - | - | - | 1 | - | - | - | P-B | - | - | acf | - | - | - | - | o-m | - |
| Actinotaenium rufescens (Cleve) Teiling | 1 | 1 | 1 | 1 | - | - | 1 | B | - | - | acf | - | - | - | - | m | - |
| Actinotaenium spinospermum (Joshua) Kouwets and Coesel | - | - | - | - | - | 1 | - | B | - | - | acf | - | - | - | - | m | - |
| Actinotaenium wollei (West & G.S.West) Teiling ex Ruzika and Pouzar | - | - | - | - | 1 | - | - | B | - | - | acf | - | - | - | - | o | - |
| Bambusina borreri (Ralfs) Cleve | - | - | 1 | - | 1 | 1 | - | P-B | - | - | acf | - | - | x-b | 0.9 | o | - |
| Closterium acutum Brébisson | - | - | - | - | - | 1 | - | P-B | - | st-str | ind | - | - | b | 2.05 | m | - |
| Closterium baillyanum (Brébisson ex Ralfs) Brébisson | 1 | - | - | - | - | - | - | B | - | - | ind | - | - | - | - | o-m | - |
| Closterium calosporum var. brasiliense Børgesen | 1 | - | - | - | - | - | - | B | - | - | acf | - | - | - | - | o-m | - |
| Closterium closterioides (Ralfs) A.Louis and Peeters var. closterioides | - | - | 1 | - | - | - | - | B | - | - | acf | - | - | o-x | 0.6 | o-m | - |
| Closterium closterioides var. intermedium (J.Roy and Bisset) Ruzicka | 1 | - | - | - | - | - | - | B | - | - | acf | - | - | o-x | 0.7 | o-m | - |
| Closterium dianae Ehrenberg ex Ralfs | 1 | - | - | - | - | - | - | P-B | - | st-str | acf | - | - | x-b | 0.8 | m | - |
| Closterium striolatum Ehrenberg ex Ralfs | - | 1 | - | - | - | - | - | P-B | - | - | acf | - | - | o | 1.2 | o-m | - |
| Desmidium swartzii C.Agardh ex Ralfs | - | 1 | - | - | - | - | - | B | - | - | ind | i | - | o-x | 0.6 | m | - |
| Euastrum ansatum Ehrenberg ex Ralfs | - | 1 | - | - | - | - | - | P-B | - | - | acf | - | - | x-o | 0.5 | o-m | - |
| Euastrum divergens var. ornatum (O.Borge) Schmidle | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Euastrum dubium var. pseudocambrense Grönblad | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Euastrum lapponicum Schmidle | - | - | 1 | - | - | - | - | P | - | - | ind | hb | - | x-o | 0.5 | m | - |
| Euastrum oblongum Ralfs | - | - | 1 | - | - | - | - | B | - | - | acf | - | - | o-x | 0.6 | m | - |
| Haplotaenium minutum (Ralfs) Bando | - | - | - | - | - | 1 | - | B | - | - | acf | - | - | x-o | 0.5 | m | - |
| Octacanthium bifidum (Brébisson) Compère | - | - | - | - | - | 1 | - | B | - | - | acf | - | - | - | - | o-m | - |
| Spirogyra sp. | - | 1 | - | - | - | - | - | B | - | - | - | - | - | - | - | - | - |
| Staurastrum dilatatum Ehrenberg ex Ralfs | 1 | 1 | 1 | - | 1 | - | - | P | - | - | - | - | - | - | - | - | - |
| Staurastrum gracile Ralfs ex Ralfs | - | - | 1 | - | - | - | - | P-B | - | st | acf | i | - | o | 1.3 | m | - |
| Staurastrum ralfsii var. depressum (J. Roy and Bisset) Coesel and Meesters | 1 | - | - | - | - | - | - | B | - | - | ind | - | - | o | 1.3 | m | - |
| Variable | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
| Division | |||||||
| Bacillariophyta | 9 | 8 | 9 | 0 | 6 | 2 | 0 |
| Charophyta | 10 | 6 | 7 | 2 | 3 | 5 | 1 |
| Chlorophyta | 4 | 0 | 3 | 3 | 1 | 0 | 1 |
| Cyanobacteria | 2 | 3 | 1 | 0 | 0 | 0 | 0 |
| Euglenophyta | 3 | 1 | 2 | 4 | 0 | 2 | 5 |
| Ochophyta (Chrysophyta) | 3 | 1 | 1 | 0 | 0 | 1 | 1 |
| Ochrophyta (Xanthophyceae) | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Abundance, 103 cells L−1 | 40 | 7 | 36 | 1 | 3 | 5 | 1 |
| No. of Species | 31 | 19 | 24 | 9 | 10 | 10 | 8 |
| Substrate | |||||||
| S | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| B | 11 | 10 | 9 | 2 | 6 | 6 | 2 |
| P-B | 8 | 5 | 6 | 6 | 2 | 3 | 3 |
| P | 5 | 3 | 8 | 2 | 2 | 4 | 3 |
| Temperature | |||||||
| cool | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| temp | 1 | 2 | 2 | 0 | 0 | 0 | 0 |
| eterm | 3 | 1 | 2 | 2 | 0 | 2 | 3 |
| Oxygenation | |||||||
| st | 2 | 5 | 2 | 0 | 2 | 1 | 1 |
| st-str | 10 | 2 | 8 | 5 | 2 | 5 | 4 |
| str | 0 | 1 | 1 | 0 | 1 | 0 | 0 |
| aer | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| pH | |||||||
| acf | 7 | 5 | 9 | 2 | 4 | 5 | 1 |
| ind | 10 | 7 | 8 | 3 | 3 | 4 | 5 |
| alf | 1 | 0 | 0 | 2 | 0 | 1 | 0 |
| alb | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| Salinity | |||||||
| hb | 2 | 1 | 3 | 0 | 2 | 0 | 0 |
| i | 12 | 10 | 14 | 3 | 5 | 6 | 4 |
| hl | 2 | 0 | 1 | 2 | 0 | 0 | 1 |
| mh | 0 | 0 | 0 | 1 | 0 | 1 | 1 |
| Watanabe | |||||||
| es | 1 | 3 | 3 | 0 | 2 | 0 | 0 |
| sp | 2 | 0 | 0 | 0 | 1 | 0 | 0 |
| Class of Water quality | |||||||
| Class 1 | 4 | 4 | 6 | 0 | 2 | 2 | 0 |
| Class 2 | 7 | 5 | 11 | 1 | 5 | 6 | 0 |
| Class 3 | 8 | 5 | 5 | 3 | 0 | 2 | 4 |
| Class 4 | 1 | 0 | 0 | 3 | 0 | 1 | 0 |
| Trophic state | |||||||
| ot | 1 | 2 | 4 | 0 | 1 | 1 | 0 |
| o-m | 5 | 2 | 2 | 1 | 1 | 1 | 0 |
| m | 3 | 3 | 5 | 1 | 0 | 3 | 2 |
| me | 0 | 0 | 1 | 1 | 0 | 0 | 1 |
| e | 2 | 0 | 1 | 0 | 0 | 0 | 0 |
| o-e | 1 | 1 | 0 | 0 | 1 | 0 | 0 |
| Nutrition type | |||||||
| ats | 1 | 2 | 4 | 0 | 1 | 1 | 0 |
| ate | 2 | 1 | 1 | 0 | 2 | 0 | 0 |
| hne | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| Index saprobity S | 1.50 | 1.07 | 0.91 | 2.03 | 0.73 | 1.33 | 2.13 |
| Site | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
| 1 | 0.97 | 2.11 | 1.12 | 4.87 | 0.84 | 1.79 | |
| 2 | 2.04 | 1.58 | 17.94 | 2.67 | 2.50 | ||
| 3 | 0.43 | 2.26 | 0.45 | 2.16 | |||
| 4 | 0.00 | 6.06 | 2.08 | ||||
| 5 | 3.81 | 7.26 | |||||
| 6 | 0 | ||||||
| 7 |
| Percent of Unique Species | |
|---|---|
| No of Site | Estimator |
| 1 | 68.0 |
| 2 | 54.6 |
| 3 | 40.6 |
| 4 | 33.2 |
| 5 | 21.2 |
| 6 | 8.2 |
| 7 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skorobogatova, O.; Yumagulova, E.; Storchak, T.; Barinova, S. Bioindication of the Influence of Oil Production on Sphagnum Bogs in the Khanty-Mansiysk Autonomous Okrug–Yugra, Russia. Diversity 2019, 11, 207. https://doi.org/10.3390/d11110207
Skorobogatova O, Yumagulova E, Storchak T, Barinova S. Bioindication of the Influence of Oil Production on Sphagnum Bogs in the Khanty-Mansiysk Autonomous Okrug–Yugra, Russia. Diversity. 2019; 11(11):207. https://doi.org/10.3390/d11110207
Chicago/Turabian StyleSkorobogatova, Olga, Elvira Yumagulova, Tatiana Storchak, and Sophia Barinova. 2019. "Bioindication of the Influence of Oil Production on Sphagnum Bogs in the Khanty-Mansiysk Autonomous Okrug–Yugra, Russia" Diversity 11, no. 11: 207. https://doi.org/10.3390/d11110207
APA StyleSkorobogatova, O., Yumagulova, E., Storchak, T., & Barinova, S. (2019). Bioindication of the Influence of Oil Production on Sphagnum Bogs in the Khanty-Mansiysk Autonomous Okrug–Yugra, Russia. Diversity, 11(11), 207. https://doi.org/10.3390/d11110207






