Comparing Medicinal Uses of Cochlospermaceae throughout Its Geographic Range with Insights from Molecular Phylogenetics
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Farnsworth, N.R.; Soejarto, D.D. Global importance of medicinal plants. In The Conservation of Medicinal Plants; Akerele, O., Heywood, V., Synge, H., Eds.; Cambridge University Press: Cambridge, UK, 1991. [Google Scholar]
- Balick, M.J.; Cox, P.A. Plants, People, and Culture: The Science of Ethnobotany; Scientific American Library: New York, NY, USA, 1996. [Google Scholar]
- Evert, R.F.; Eichhorn, S.E. Raven Biology of Plants; W. H. Freeman: New York, NY, USA, 2012. [Google Scholar]
- Raskin, I.; Ribnicky, D.M.; Komarnytsky, S.; Ilic, N.; Poulev, A.; Borisjuk, N.; Brinker, A.; Moreno, D.A.; Ripoll, C.; Yakoby, N.; et al. Plants and human health in the twenty-first century. Trends Biotechnol. 2002, 20, 522–531. [Google Scholar] [CrossRef]
- Gurib-Fakim, A. Medicinal plants: Traditions of yesterday and drugs of tomorrow. Mol. Asp. Med. 2006, 27, 1–93. [Google Scholar] [CrossRef] [PubMed]
- Benz, B.F.; Cevallos, J.; Santana, F.; Rosales, J. Losing knowledge about plant use in the Sierra de Manantlan Biosphere Reserve, Mexico. Econ. Bot. 2000, 54, 183–191. [Google Scholar] [CrossRef]
- Sutherland, W.J. Parallel extinction risk and global distribution of languages and species. Nature 2003, 423, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Arora, N.K. Biodiversity conservation for sustainable future. Environ. Sustain. 2018, 1, 109–111. [Google Scholar] [CrossRef]
- Glikson, A. The lungs of the Earth: Review of the carbon cycle and mass extinction of species. Energy Procedia 2018, 146, 3–11. [Google Scholar] [CrossRef]
- Saslis-Lagoudakis, C.H.; Klitgaard, B.B.; Forest, F.; Francis, L.; Savolainen, V.; Williamson, E.M.; Hawkins, J.A. The use of phylogeny to interpret cross-cultural patterns in plant use and guide medicinal plant discovery: An example from Pterocarpus (Leguminosae). PLoS ONE 2011, 6, e22275. [Google Scholar] [CrossRef] [PubMed]
- Poppendieck, H.H. A Monograph of the Cochlospermaceae. Bot. Jahrb. 1980, 101, 191–265. [Google Scholar]
- Poppendieck, H.H. Flora Neotropica Monograph, No. 27: Cochlospermaceae; New York Botanical Garden: New York, NY, USA, 1981. [Google Scholar]
- Poppendieck, H.H. Cochlospermaceae. In The Families and Genera of Vascular Plants; Kubitzki, K., Bayer, C., Eds.; Springer: Berlin, Germany, 2003; Volume 5, pp. 71–74. [Google Scholar]
- Morton, J.F. Atlas of Medicinal Plants of Middle America: Bahamas to Yucatan; Charles, C. Thomas Publisher: Springfield, IL, USA, 1981. [Google Scholar]
- Abbiw, D. Useful Plants of Ghana; Intermediate Technology Publications and Royal Botanic Gardens: Kew, UK, 1990. [Google Scholar]
- Sala, A.V. Indian Medicinal Plants: A Compendium of 500 Species; Orient Longman Ltd.: Madras, India, 1994; Volume 2. [Google Scholar]
- Hodgson, W.C. Food Plants of the Sonoran Desert; University of Arizona Press: Tucson, AZ, USA, 2001. [Google Scholar]
- Pullaiah, T. Medicinal plants in Andhra Pradesh, India; Regency Publications: New Delhi, India, 2002. [Google Scholar]
- Yetman, D.A.; Van Devender, T.R. Mayo Ethnobotany; University of California Press: Berkeley, CA, USA, 2002. [Google Scholar]
- Fabricant, D.S.; Farnsworth, N.R. The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect. 2001, 109, 69–75. [Google Scholar] [PubMed]
- Rønsted, N.; Savolainen, V.; Mølgaard, P.; Jäger, A.K. Phylogenetic selection of Narcissus species for drug discovery. Biochem. Syst. Ecol. 2008, 36, 417–422. [Google Scholar] [CrossRef]
- Zhu, F.; Qin, C.; Tao, L.; Liu, X.; Shi, Z.; Ma, X.; Jia, J.; Tan, Y.; Cui, C.; Lin, J.; et al. Clustered patterns of species origins of nature-derived drugs and clues for future bioprospecting. Proc. Natl. Acad. Sci. USA 2011, 108, 12943–12948. [Google Scholar] [CrossRef] [PubMed]
- Rønsted, N.; Symonds, M.R.E.; Birkholm, T.; Christensen, S.; Meerow, A.W.; Molander, M.; Mølgaard, P.; Petersen, G.; Rasmussen, N.; van Staden, J.; et al. Can phylogeny predict chemical diversity and potential medicinal activity of plants? A case study of Amaryllidaceae. BMC Evol. Biol. 2012, 12, 182. [Google Scholar] [CrossRef] [PubMed]
- Saslis-Lagoudakis, C.H.; Savolainen, V.; Williamson, E.M.; Forest, F.; Wagstaff, S.J.; Baral, S.R.; Watson, M.F.; Pendry, C.A.; Hawkins, J.A. Phylogenies reveal predictive power of traditional medicine in bioprospecting. Proc. Natl. Acad. Sci. USA 2012, 109, 15835–15840. [Google Scholar] [CrossRef] [PubMed]
- Hao, D.; Xiao, P. Genomics and evolution in traditional medicinal plants: Road to a healthier life. Evol. Bioinform. 2015, 11, 197–212. [Google Scholar] [CrossRef] [PubMed]
- Yessoufou, K.; Daru, B.; Muasya, A.M. Phylogenetic exploration of commonly used medicinal plants in South Africa. Mol. Ecol. Resour. 2015, 15, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Garnatje, T.; Peñuelas, J.; Vallès, J. Ethnobotany, Phylogeny, and ‘Omics’ for Human Health and Food Security. Trends Plant Sci. 2017, 22, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, C.; Barnes, C.J.; Cornett, C.; Holmfred, E.; Hansen, S.H.; Persson, C.; Antonelli, A.; Rønsted, N. Phylogeny predicts the quantity of antimalarial alkaloids within the iconic yellow Cinchona bark (Rubiaceae: Cinchona calisaya). Front. Plant Sci. 2017, 8, 391. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Morita, A.H.; Kanaya, S.; Altaf-Ul-Amin, M. Metabolite-content-guided prediction of medicinal/edible properties in plants for bioprospecting. Curr. Res. Compliment. Altern. Med. 2018. [CrossRef]
- Terashima, H.; Ichikawa, M. A comparative ethnobotany of the Mbuti and Efe hunter-gatherers in the Ituri forest, Democratic Republic of Congo. Afr. Study Monogr. 2003, 24, 1–168. [Google Scholar]
- Prescott, T.A.K.; Kiapranis, R.; Maciver, S.K. Comparative ethnobotany and in-the-field antibacterial testing of medicinal plants used by the Bulu and inland Kaulong of Papua New Guinea. J. Ethnopharmacol. 2012, 139, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, A.; Langenberger, G.; Sauerborn, J. A comparison of the wild food plant use knowledge of ethnic minorities in Naban River Watershed National Nature Reserve, Yunnan, SW China. J. Ethnobiol. Ethnomed. 2012, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Alexiades, M.N. Selected Guidelines for Ethnobotanical Research: A Field Manual; Botanical Garden: New York, NY, USA, 1996. [Google Scholar]
- Martin, G.J. Ethnobotany: A Method’s Manual; Routledge: Abingdon, UK, 2004. [Google Scholar]
- Johnson-Fulton, S.; Watson, L. Phylogenetic Systematics of Cochlospermaceae (Malvales) Based on Molecular and Morphological Evidence. Syst. Bot. 2017, 42, 271–282. [Google Scholar] [CrossRef]
- Maddison, D.R.; Maddison, W.P. MacClade 4: Analysis of Phylogeny and Character Evolution. Version 4.08a. 2005. Available online: http://macclade.org (accessed on 20 August 2013).
- Martinez, M. Las Plantas Medicinales de Mexico, Quinta ed.; Ediciones Botas: Mexico City, Mexico, 1969. [Google Scholar]
- Esposito-avella, M.; Brown, P.; Tejeira, I.; Buitrago, R.; Barrios, L.; Sanchez, C.; Gupta, M.P.; Cedeño, J. Pharmacological screening of Panamanian medicinal plants. Part 1. Pharm. Biol. 1985, 23, 17–25. [Google Scholar] [CrossRef]
- Salas, T.P.; Peraza, L.S. Lista Anotada de las Plantas Medicinales de Uso Actual en el Estado de Quintana Roo, Mexico; Centro de Investigaciones de Qintana Roo: Chetumal, Mexico, 1993. [Google Scholar]
- Zamora-Martinez, M.C.; Pola, C.N.P. Medicinal plants used in some rural populations of Oaxaca, Puebla and Veracruz, Mexico. J. Ethnopharmacol. 1992, 35, 229–257. [Google Scholar] [CrossRef]
- Stebbins, M. Flowering Trees of Florida; Pineapple Press Inc.: Sarasota, FL, USA, 1999. [Google Scholar]
- Caballero-George, C.; Vanderheyden, P.M.L.; Solis, P.N.; Pieters, L.; Shahat, A.A.; Gupta, M.P.; Vauquelin, G.; Vlietinck, A.J. Biological screening of selected medicinal Panamanian plants by radioligand-binding techniques. Phytomedicine 2001, 8, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Cedano, M.M.; Villasenor, I.L. Usos y nombres communes de lase species de Cochlospermaceae en Mexico. Ethnobiologia 2004, 4, 73–88. [Google Scholar]
- Zuchowski, W. A Guide to Tropical Plants of Costa Rica; Zona Tropical Publication: Ithaca, NY, USA, 2006. [Google Scholar]
- Monroy-Ortíz, C.; España, P.C. Plantas Medicinales Utilizadas en el Estado de Morelos; Universidad Autónoma del Estado de Morelos: Cuernavaca, Morelos, Mexico, 2007. [Google Scholar]
- Lewis, A.C.; Hanson, P.J. Anti-ulcer drugs of plant origin. Prog. Med. Chem. 1991, 28, 201–231. [Google Scholar] [PubMed]
- Salles, A.H.; Reis, G.M.C.I.; Zurlo, M.A. Horto Medicinal do Cerrado; Jardim Botanico de Brasilia: Brasília, Brazil, 1997. [Google Scholar]
- Nunes, W.B.; de Carvalho, S. Evaluation of the mutagenic potential of Cochlospermum regium in Drosophila melanogaster male germ cells. Genet. Mol. Biol. 2003, 26, 545–549. [Google Scholar] [CrossRef]
- Rodrigues, E.; Carlini, E.A. Ritual use of plants with possible action on the central nervous system by the Kraho Indians, Brazil. Phytother. Res. 2005, 19, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Ceschini, L.; Campos, E.G. Cytotoxic effects of Cochlospermum regium (Mart & Schrank) Pilger aqueous root extract on mammalian cells. J. Ethnopharmacol. 2006, 103, 302–305. [Google Scholar] [PubMed]
- Andrade, L.S.; Santos, D.B.; Castro, D.B.; Guillo, L.A.; Chen, L. Absence of antimutagenicity of Cochlospermum regium (Mart. and Schr.) Pilger 1924 by micronucleus test in mice. Braz. J. Biol. 2008, 68, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Solon, S.; Carollo, C.A.; Brandão, L.F.G. Phenolic derivatives and other chemical compounds from Cochlospermum regium. Quim. Nova 2012, 35, 1169–1172. [Google Scholar] [CrossRef]
- Rutter, R.A. Catálogo de Plantas Utiles de la Amazonía Peruana; Instituto Lingüístico de Verano: Yarinacocha, Peru, 1990. [Google Scholar]
- Duke, J.A.; Vasquez, R. Amazonian Ethnobotanical Dictionary; CRC Press: Boca Raton, FL, USA, 1994. [Google Scholar]
- Adam, J.G.; Echard, N.; Lescot, M. Plantes médicinales Hausa de l’Ader (République du Niger). J. Agric. Trop. Bot. Appl. 1972, 19, 259–399. [Google Scholar] [CrossRef]
- Adjanohoun, E.; Ahyi, M.R.A.; Assi, L.A.; Dicko, L.D.; Daouda, H.; Delmas, M.; de Souza, S.; Garba, M.; Guinko, S.; Kayonga, A.; et al. Medecine Traditionnelle et Pharmacopee: Contribution Aux études Ethnobotaniques et Floristiques au Niger; Agence de Coopération Culturelle et Technique: Paris, France, 1980. [Google Scholar]
- Aké Assi, L.; Guinko, S. Plants Used in Traditional Medicine in West Africa; Roche Editions: Basel, Switzerland, 1991. [Google Scholar]
- Etkin, N.L.; Ross, P.J. Recasting malaria, medicine, and meals: A perspective on disease adaptation. In The Anthropology of Medicine: From Culture to Method; Romanucci-Ross, L., Moerman, D.E., Tancredi, L.R., Eds.; Bergin & Garvey: New York, NY, USA, 1991. [Google Scholar]
- Aliyu, R.; Okoye, Z.S.C.; Shier, W.T. The hepatoprotective cytochrome P-450 enzyme inhibitor isolated from the Nigerian medicinal plant Cochlospermum planchonii is a zinc salt. J. Ethnopharmacol. 1995, 48, 89–97. [Google Scholar] [CrossRef]
- Benoit, F.; Valentin, A.; Pélissier, Y.; Marion, C.; Dakuyo, Z.; Mallié, M.; Bastide, J.M. Antimalarial activity in vitro of Cochlospermum tinctorium tubercle extracts. Trans. R. Soc. Trop. Med. Hygiene 1995, 89, 217–218. [Google Scholar] [CrossRef]
- Vonthron-Senecheau, C.; Weniger, D.; Ouattara, M.; Bi, F.T.; Kamenan, A.; Lobstein, A.; Brun, R.; Anton, R. In vitro antiplasmodial activity and cytotoxicity of ethnobotanically selected Ivorian plants. J. Ethnopharmacol. 2003, 87, 221–225. [Google Scholar] [CrossRef]
- Phytofla Laboratory. Les Laboratoires Phytofla. Available online: https://laboratoiresphytofla.com (accessed on 29 July 2012).
- Thiombiano, A.; Schmidt, M.; Dressler, S.; Ouédraogo, A.; Hahn, K.; Zizka, G. Catalogue des Plantes Vasculaires du Burkina Faso; Boissiera 65, Ed.; Conservatoire et Jardin Botaniques de la Ville de Genève: Geneva, Switzerland, 2012. [Google Scholar]
- Dalziel, J.M.; Hutchinson, J. The Useful Plants of West Tropical Africa; Published under the Authority of the Secretary of State for the Colonies by the Crown Agents for the Colonies: London, UK, 1937. [Google Scholar]
- Kerharo, J.; Bouquets, A. Plantes Medicinales et Toxiques de la Côte d’Ivoire-Haute Volta; Vigot Frères: Paris, France, 1950. [Google Scholar]
- Kerharo, J.; Adam, J.G. La Pharmacopee Sénégal aise Traditionnelle: Plantes Medicinales Et Toxiques; Vigot Frères: Paris, France, 1974. [Google Scholar]
- Berhaut, J. Flore illustrée du Sénégal. Tome 2. Ministère Déve-loppement Rural: Direction des Eaux & Forets et Ed; Clairafrique: Dakar, Senegal, 1974. [Google Scholar]
- Burkill, H.M. The Useful Plants of West Tropical Africa, 2nd ed.; Families, A.-D., Ed.; Royal Botanical Gardens: Kew, UK, 1985. [Google Scholar]
- Diniz, M.A.; Silva, O.; Paulo, M.A.; Gomes, E.T. Medicinal uses of plants from Guinea-Bissau. In The Biodiversity of African Plants; van der Maesen, L.J.G., Ed.; Kluwer: Dordrecht, The Netherlands, 1996. [Google Scholar]
- Neuwinger, H.D. African Ethnobotany: Poisons and Drugs: Chemistry, Pharmacology, Toxicology; Chapman & Hall: London, UK, 1996. [Google Scholar]
- Jansen, P.C.M.; Cardon, D. (Eds.) Plant Resources of Tropical Africa (PROTA) 3. Dyes and Tannins; PROTA Foundation: Wageningen, The Netherlands, 2005. [Google Scholar]
- Nergard, C.S.; Diallo, D.; Inngjerdingen, K.; Michaelsen, T.E.; Matsumoto, T.; Kiyohara, H.; Yamada, H.; Paulsen, B.S. Medicinal use of Cochlospermum tinctorium in Mali: Anti-ulcer-, radical scavenging- and immunomodulation activities of polymers in the aqueous extract of the roots. J. Ethnopharmacol. 2005, 96, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Togola, A.; Diallo, D.; Dembele, S.; Barsett, H.; Paulsen, B.S. Ethnopharmacological survey of different uses of seven medicinal plants from Mali, (West Africa) in the regions Doila, Kolokani and Siby. J. Ethnobiol. Ethnomed. 2005, 1, 7. [Google Scholar] [CrossRef] [PubMed]
- Bosch, C.H.; Borus, D.J. (Eds.) Dyes and Tannins of Tropical Africa; Conclusions and Recommendations Based on PROTA 3: ‘Dyes and tannins’; PROTA Foundation: Wageningen, The Netherlands, 2006. [Google Scholar]
- Angola: Country Report to the International Conference and Programme on Plant Genetic Resource. In Proceedings of the International Conference on Plant Genetic Resources, Leipzig, Gemany, 17–23 June 1996; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 1996.
- Presber, W.; Hegenscheid, B.; Hernandez-Alvarez, H.; Herrmann, D.; Brendel, C. Inhibition of the growth of Plasmodium falciparum and Plasmodium berghei in vitro by an extract of Cochlospermum angolense (Welw.). Acta Trop. 1992, 50, 331–338. [Google Scholar] [CrossRef]
- Howes, F.N. Vegetable Gums and Resins; Chronica Botanica: Waltham, MA, USA, 1949. [Google Scholar]
- Jain, N.; Babbar, S.B. Gum katira—A cheap gelling agent for plant tissue culture media. Plant Cell Tissue Organ Cult. 2002, 71, 223–229. [Google Scholar] [CrossRef]
- Panda, H. Hand Book of Ayurvedic Medicines with Formulae, Processes, and Their Uses; National Institute of Industrial Research: Delhi, India, 2004. [Google Scholar]
- Vegi, G.M.N.; Sistla, R.; Srinivasan, P.; Beedu, S.R.; Khar, R.K.; Diwan, P.V. Emulsifying properties of gum kondagogu (Cochlospermum gossypium), a natural biopolymer. J. Sci. Food Agric. 2008, 89, 1271–1276. [Google Scholar] [CrossRef]
- Low, T. Bush Medicine: A Pharmacopoeia of Natural Remedies; Angus & Robertson: Melbourne, Australia, 1990. [Google Scholar]
- Devanesen, D.D. Traditional Aboriginal Medicine Practice in the Northern Territory of Australia. Presented at the International Symposium on Traditional Medicine, Awaji Island, Japan, 11–13 September 2000. [Google Scholar]
- Cook, I.F.; Knox, J.R. Flavonoids from Cochlospermum gillivraei. Phytochemistry 1975, 14, 2510–2511. [Google Scholar] [CrossRef]
- Etkin, N.L. A Hausa herbal pharmacopoeia: Biochemical evaluation of commonly used plant medicines. J. Ethnopharmacol. 1981, 4, 75–98. [Google Scholar] [CrossRef]
- Diallo, B.; Vanhaelen, M. Apocarotenoids from Cochlospermum tinctorium. Phytochemistry 1987, 26, 1491–1492. [Google Scholar] [CrossRef]
- Diallo, B.; Vanhaelen, M.; Kiso, Y.; Hikino, H. Antihepatotoxic actions of Cochlospermum tinctorium rhizomes. J. Ethnopharmacol. 1987, 20, 239–243. [Google Scholar] [CrossRef]
- Diallo, B.; Vanhaelen, M.; Vanhaelen-Fastre, R. Studies on inhibitors of skin-tumor promotion inhibitory effects of triterpenes from Cochlospermum tinctorium on Epstein-Barr virus activation. J. Nat. Prod. 1989, 52, 879–881. [Google Scholar] [CrossRef] [PubMed]
- Diallo, B.; Vanhaelen-Fastre, R.; Vanhaelen, M.; Nkiani-Ibwala, N.Y.; Pelsener-Coremans, J. Antimicrobial activity of two apocarotenoids isolated from Cochlospermum tinctorium rhizome. Fitoterapia 1991, 62, 144–145. [Google Scholar]
- Diallo, B.; Vanhaelen-Fastre, R.; Vanhaelen, M.; Fiegel, C.; Joyeux, M.; Roland, A.; Fleurentin, J. Further studies on the hepatoprotective effects of Cochlospermum tinctorium rhizomes. J. Ethnopharmacol. 1992, 36, 137–142. [Google Scholar] [CrossRef]
- Presber, W.; Hegenscheid, B.; Friedmann-Alvermann, B.; Dörge, S.; Voigt, G.; Hiller, K.; Hils, J.; May, A.; Böthig, B. Antiviral activity of extracts of Cochlospermum angolense (Welw.). Die Pharm. 1987, 42, 707–708. [Google Scholar]
- Presber, W.; Herrmann, D.K.; Hegenscheid, B. The effect of an extract from Cochlospermum angolense (“Burututu”) on Plasmodium berghei in the mouse malaria suppression test. Angew. Parasitol. 1991, 32, 7–9. [Google Scholar] [PubMed]
- Dalvi, R.R.; Séré, A. Protective effect of Cochlospermum tinctorium A. rich extract versus Aflatoxin B induced liver damage in rats. Pharm. Biol. 1988, 26, 117–120. [Google Scholar] [CrossRef]
- Castro, M.S.A.; Siqueira, J.M.; Krieger-Amorin, L.W.; Souza, K.C.B. Efeito analgésico e antiedematogênico do Cochlospermum regium (Mart.) Pilger (‘algodãozinho’). In Proceedings of the XIII Simpósio de Plantas Medicinais do Brasil, Fortaleza, Brazil, 6–9 September 1994. [Google Scholar]
- Castro, M.S.A.; Siqueira, J.M.; Vanderline, F.A. Envolvimento de receptores dopaminérgicos no efeito antinociceptivo de uma flavanona isolada dos rizomas de Cochlospermum regium. In Proceedings of the Anais do XV Simpósio de Plantas Medicinais do Brasil, Águas de Lindóia, Brazil, 15 August 1998. [Google Scholar]
- Oliveira, C.C.; de Siqueira, J.M.; de Souza, K.C.B.; Resende, U.M. Avaliação da atividade bacteriana da raiz de Cochlospermum regium “algodãozinho do campo”. In Proceedings of the XIII Simpósio de Plantas Medicinais do Brasil, Fortaleza, Brazil, 6–9 September 1994. [Google Scholar]
- Oliveira, C.C.; de Siqueira, J.M.; de Souza, K.C.B.; Resende, U.M. Antibacterial activity of rhizomes from Cochlospermum regium preliminary results. Fitotera 1996, 67, 176–177. [Google Scholar]
- Herrera, M.D.; Zarzuelo, A.; Jimenez, J.; Marhuenda, E.; Duarte, J. Effects of flavanoids on rat aortic smooth muscle contractility: Structure-activity relationships. Gener. Pharmacol. 1996, 27, 273–277. [Google Scholar] [CrossRef]
- Toledo, M.I.; Siqueira, J.M.; Araujo, L.C.L.; Oga, S. Acute and subacute toxicity of Cochlospermum regium (Mart. & Schr.) Pilger. Phytother. Res. 2000, 14, 359–361. [Google Scholar] [PubMed]
- Ballin, N.Z.; Traoré, M.; Tinto, H.; Sittie, A.; Mølgaard, P.; Olsen, C.E.; Kharazami, A.; Christensen, S.B. Antiplasmodial compounds from Cochlospermum tinctorium. J. Nat. Prod. 2002, 65, 1325–1327. [Google Scholar] [CrossRef] [PubMed]
- Ajay, M.; Gilani, A.U.; Mustafa, M.R. Effects of flavanoids on vascular smooth muscle of the isolated rat thoracic aorta. Life Sci. 2003, 19, 603–612. [Google Scholar] [CrossRef]
- Sánchez-Salgado, J.C.; Ortiz-Andrade, R.R.; Aguirre-Crespo, F.; Vergara-Galicia, J.; León-Rivera, I.; Montes, S.; Villalobos-Molina, R.; Estrada-Soto, S. Hypoglycemic, vasorelaxant and hepatoprotective effects of Cochlospermum vitifolium (Willd.) Sprengel: A potential agent for the treatment of metabolic syndrome. J. Ethnopharmacol. 2007, 109, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Salgado, J.C.; Castillo-Espana, P.; Ibarra-Barajas, M.; Villalobos-Molina, R.; Estrada-Soto, S. Cochlospermum vitifolium induces vasorelaxant and antihypertensive effects mainly by activation of NO/cGMP signaling pathway. J. Ethnopharmacol. 2010, 130, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Andrade, R.R.; Sánchez-Salgado, J.C.; Navarrete-Vázquez, G.; Webster, S.P.; Binnie, M.; García-Jiménez, S.; León-Rivera, I.; Cigarroa-Vázquez, P.; Villalobos-Molina, R.; Estrada-Soto, S. Antidiabetic and toxicological evaluations of naringenin in normoglycaemic and NIDDM rat models and its implications on extra-pancreatic glucose regulation. Diabetes Obes. Metab. 2008, 10, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Traoré, M.; Diallo, A.; Nikièma, J.B.; Tinto, H.; Dakuyo, Z.P.; Ouédraogo, J.B.; Guissou, I.P.; Guiguemdé, T.R. In vitro and in vivo antiplasmodial activity of ‘Saye’, an herbal Remedy used in Burkina Faso traditional medicine. Phytother. Res. 2008, 22, 550–551. [Google Scholar] [CrossRef] [PubMed]
- Manso, J.A.X.; Figueiredo, F.R.G.; Hanusch, A.L.; Machado, R.C.; Cunha, D.M.C.; da Silva, C.C.; da Cruz, A.D. Bioensaio animal na avaliação da mutagenicidade do decocto de Cochlospermum regium (Cochlospermaceae). In Proceedings of the 2nd Reunião Brasileira de Citogenética, Águas de Lindóia, São Paulo, Brazil, 28–30 August 2011. [Google Scholar]
- Willcox, M. Improved traditional phytomedicines in current use for the clinical treatment of malaria. Planta Med. 2011, 77, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Yerbanga, R.S.; Lucantoni, L.; Lupidi, G.; Dori, G.U.; Tepongning, N.R.; Nikiema, J.B.; Esposito, F.; Habluetzel, A. Antimalarial plant remedies from Burkina Faso: Their potential for prophylactic use. J. Ethnopharmacol. 2012, 140, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Benoit-vical, F.; Valentin, A.; Pelissier, Y.; Mallie, M.; Bastide, J.M.; Bessiere, J.M. In vitro antimalarial activity and cytotoxicity of Cochlospermum tinctorium and Cochlospermum planchonii leaf extracts and essential oils. Planta Med. 1999, 65, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Benoit-Vical, F.; Valentin, A.; Da, B.; Dakuyo, Z.; Descamps, L.; Mallie, M. N’Dribala (Cochlospermum planchonii) versus chloroquine for treatment of uncomplicated Plasmodium falciparum malaria. J. Ehtnopharmacol. 2003, 89, 111–114. [Google Scholar] [CrossRef]
- Deharo, E.; Baelmans, R.; Gimenez, A.; Quenevo, C.; Bourdy, G. In vitro immunomodulatory activity of plants used by the Tacana ethnic group in Bolivia. Phytomedicine 2004, 11, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Devi, V.G.; Rooban, B.N.; Sasikala, V.; Sahasranamam, V.; Abraham, A. Isorhamnetin-3-glucoside alleviates oxidative stress and opacification in selenite cataract in vitro. Toxicol. In Vitro 2010, 24, 1662–1669. [Google Scholar] [CrossRef] [PubMed]
- Muhs, A.; Lyles, J.T.; Parlet, C.P.; Nelson, K.; Kavanaugh, J.S.; Horswill, A.R.; Quave, C.L. Virulence inhibitors from Brazilian Peppertree block quorum sensing and abate dermonecrosis in skin infection models. Sci. Rep. 2017, 7, 42275. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.F.; Tang, H.; Lyles, J.T.; Pineau, R.; Mashwani, Z.R.; Quave, C.L. Antibacterial properties of medicinal plants from Pakistan against multidrug-resistant ESKAPE pathogens. Front. Pharmacol. 2018, 9, 815. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Abdullah, A.A.; Huang, M.; Nishioka, T.; Altaf-Ul-Amin, M.; Kanaya, S. Novel approach to classify plants based on metabolite-content similarity. BioMed Res. Int. 2017, 2017, 5293729. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B.C. Doctrine of Signatures: An explanation of medicinal plant discovery or dissemination of knowledge? Econ. Bot. 2007, 61, 246–255. [Google Scholar] [CrossRef]
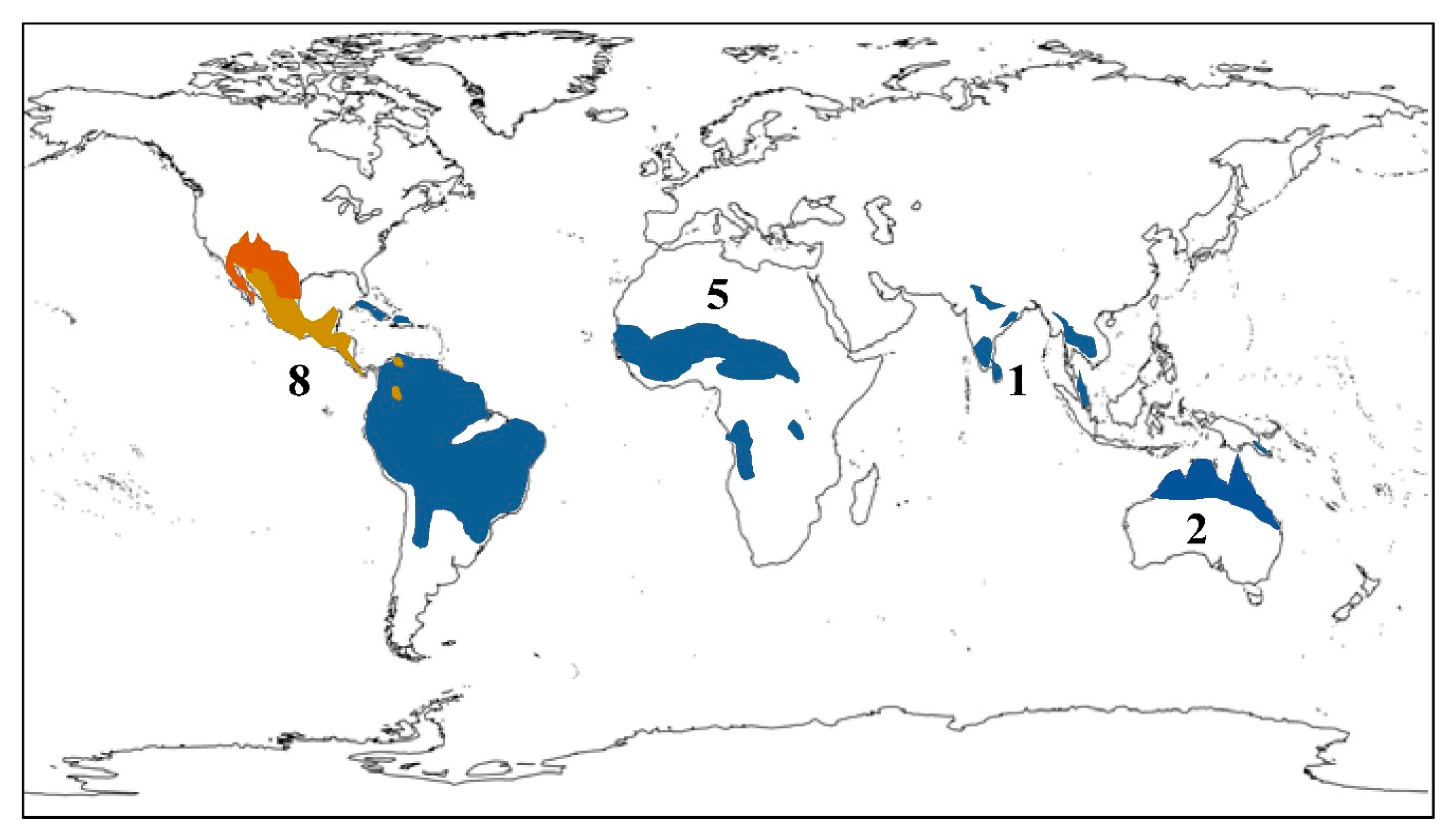

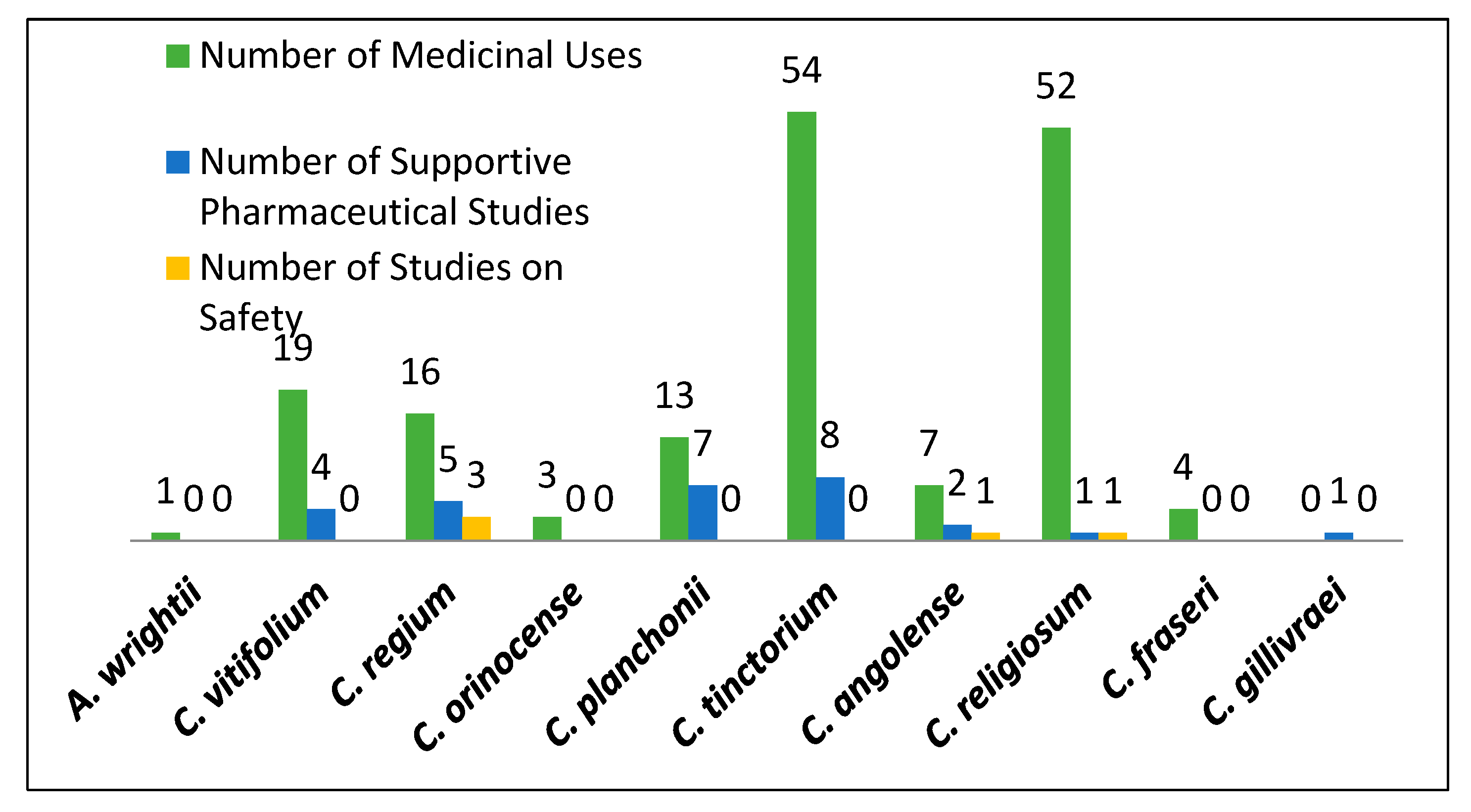
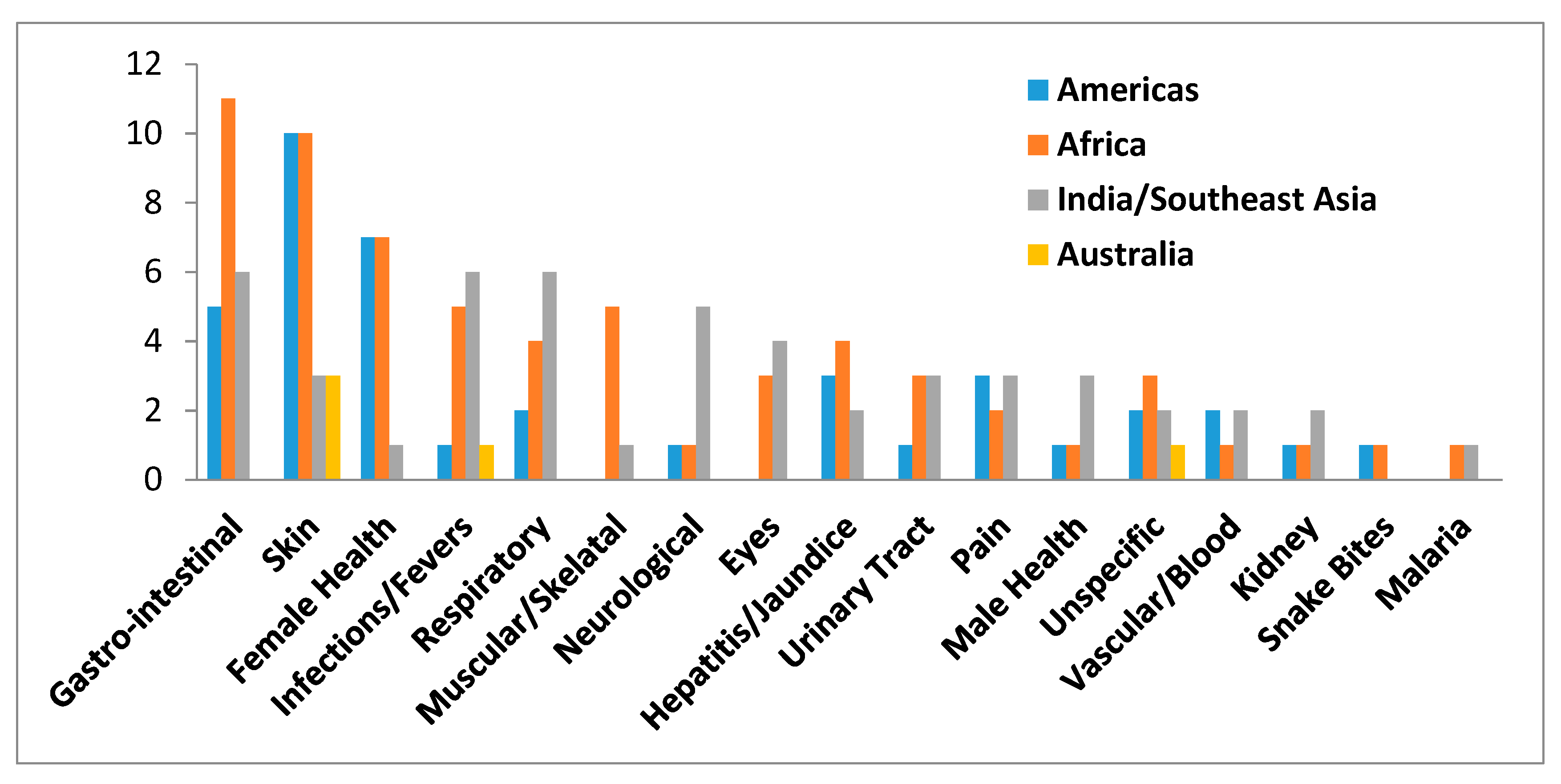
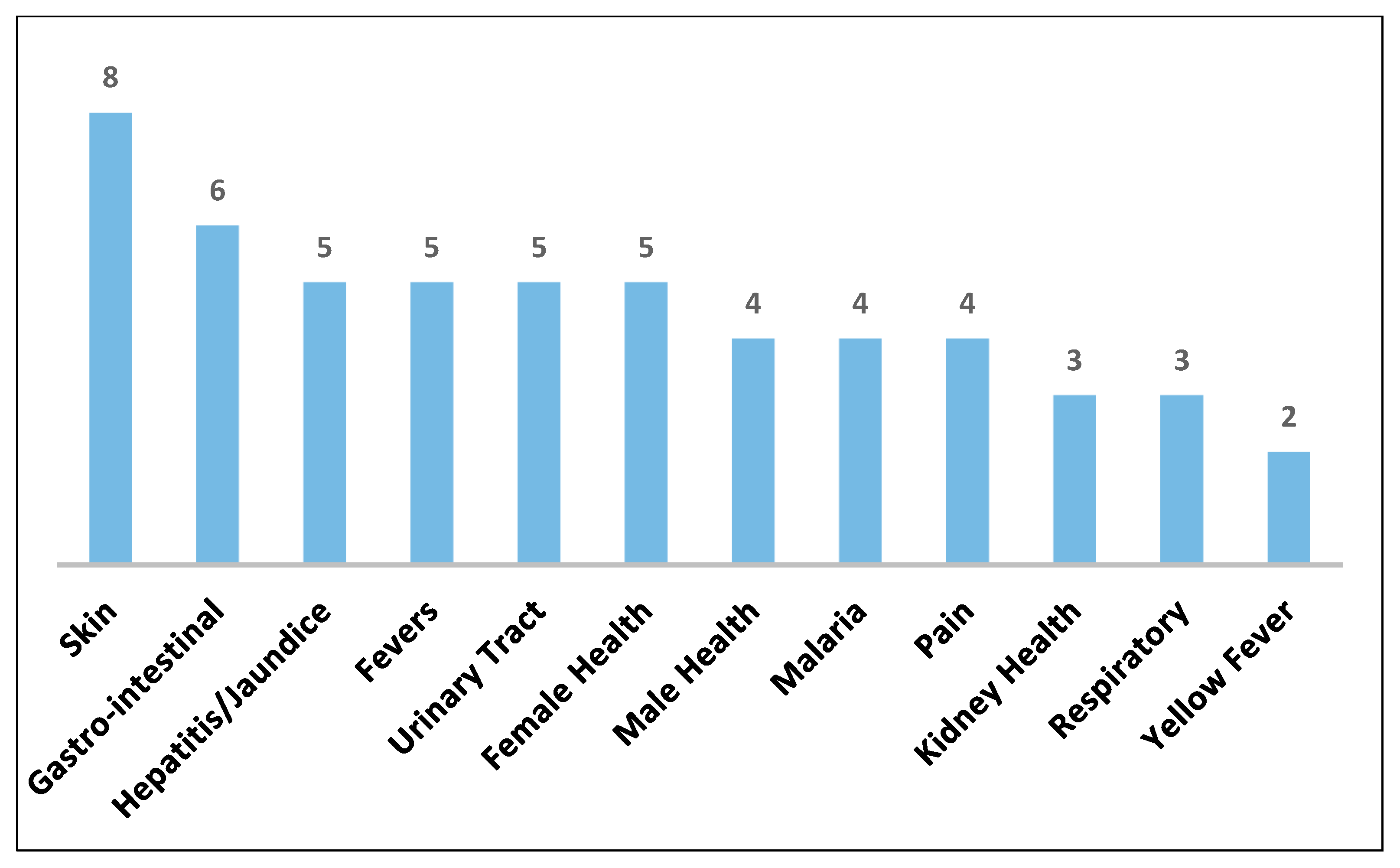
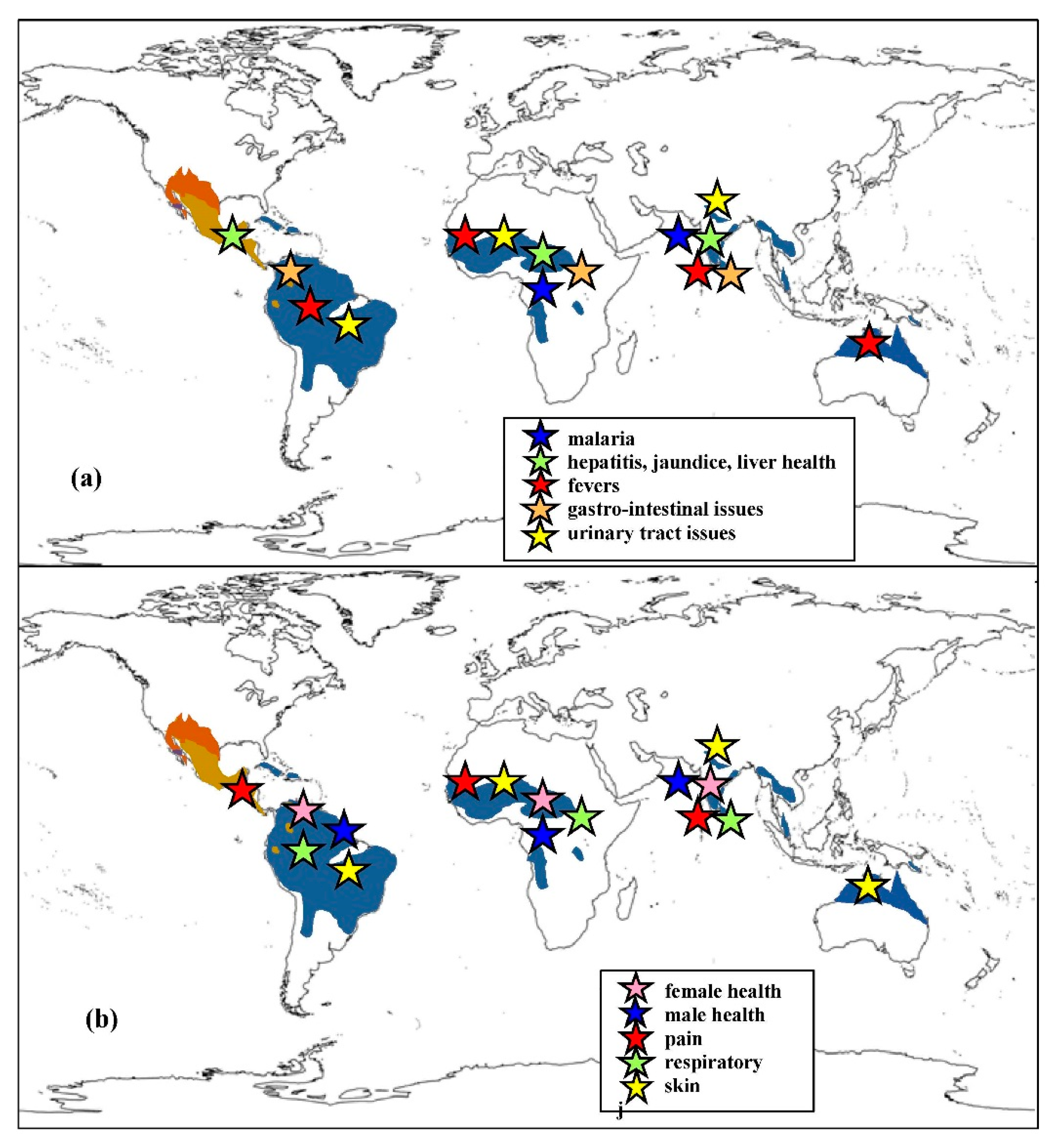

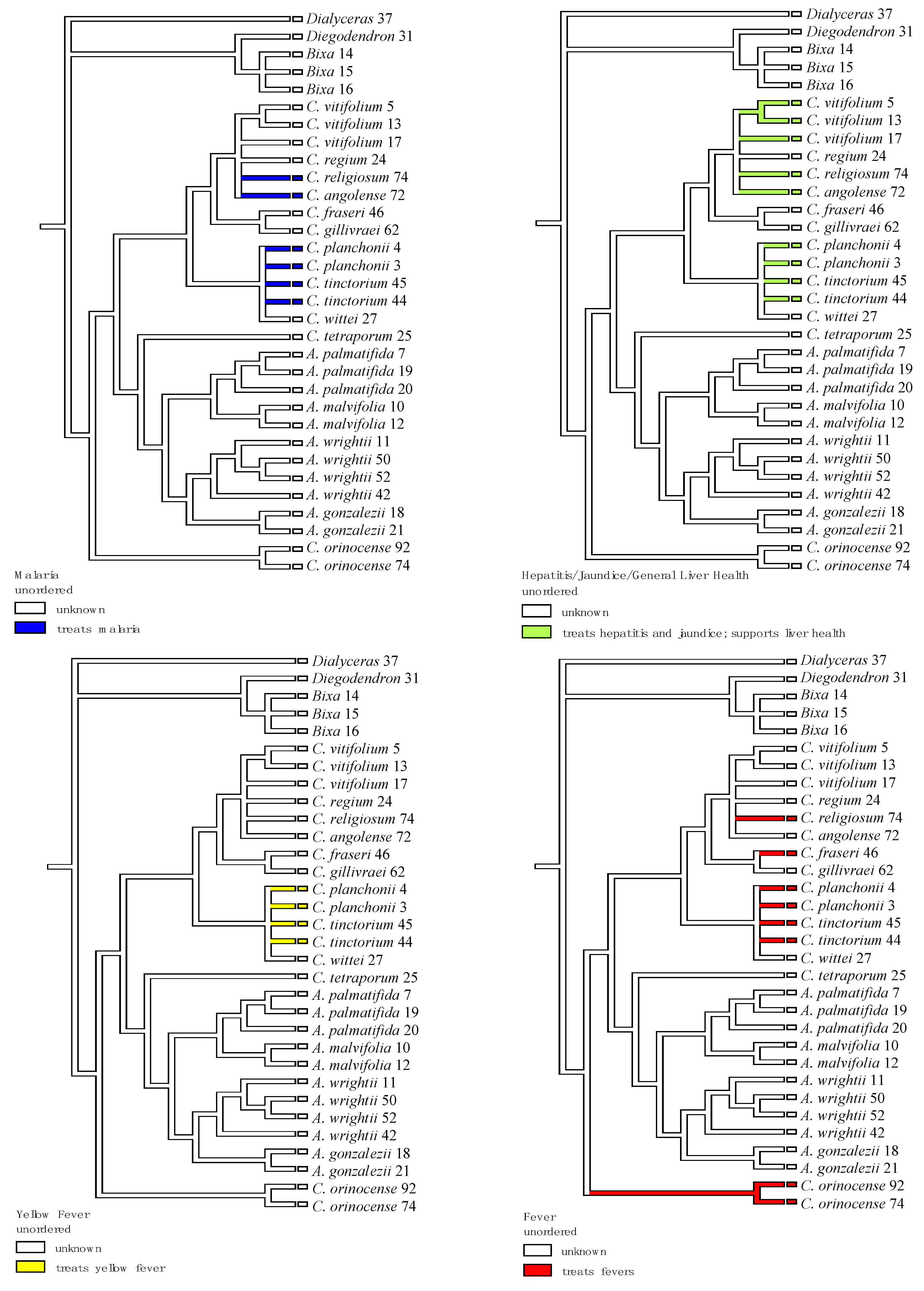
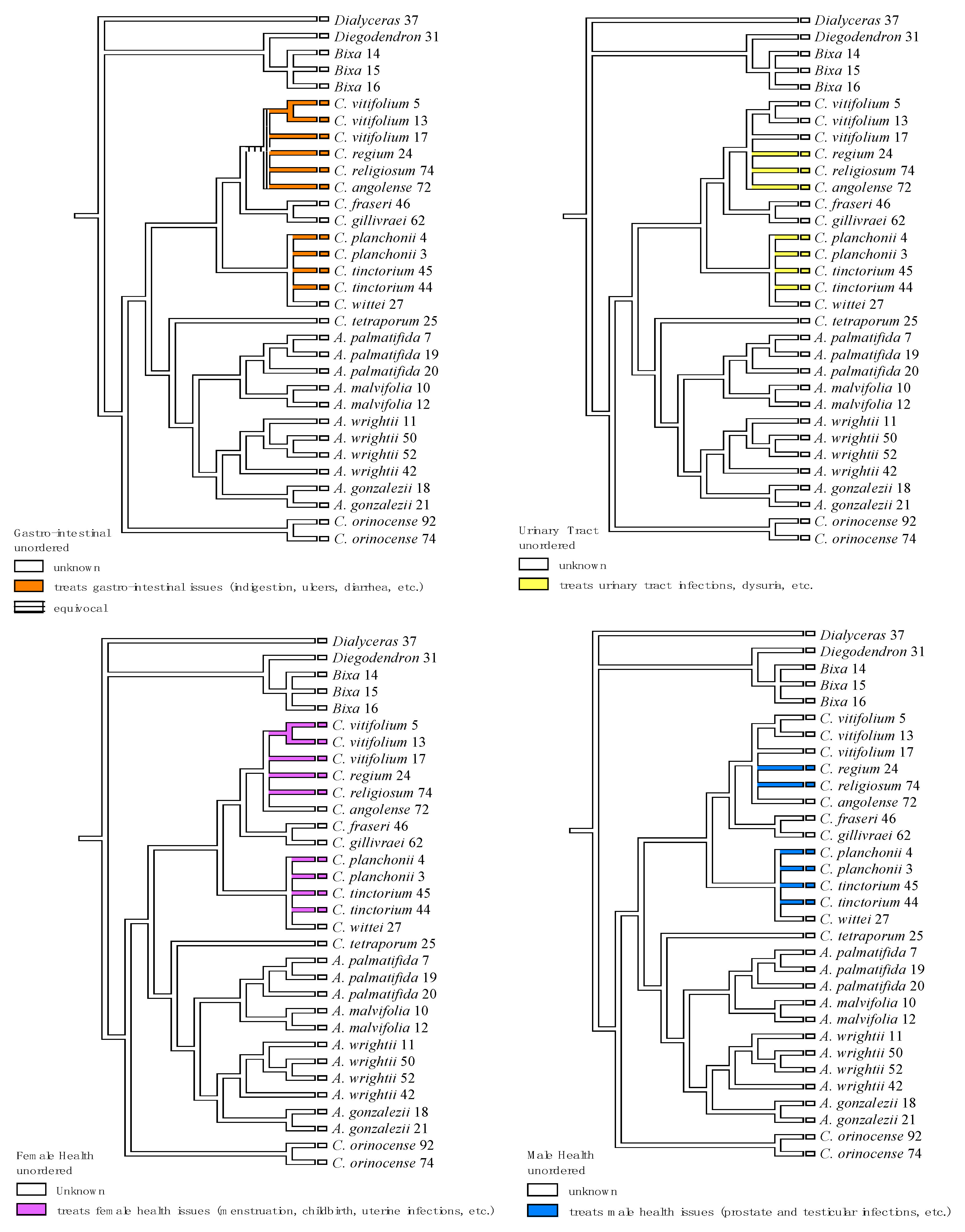
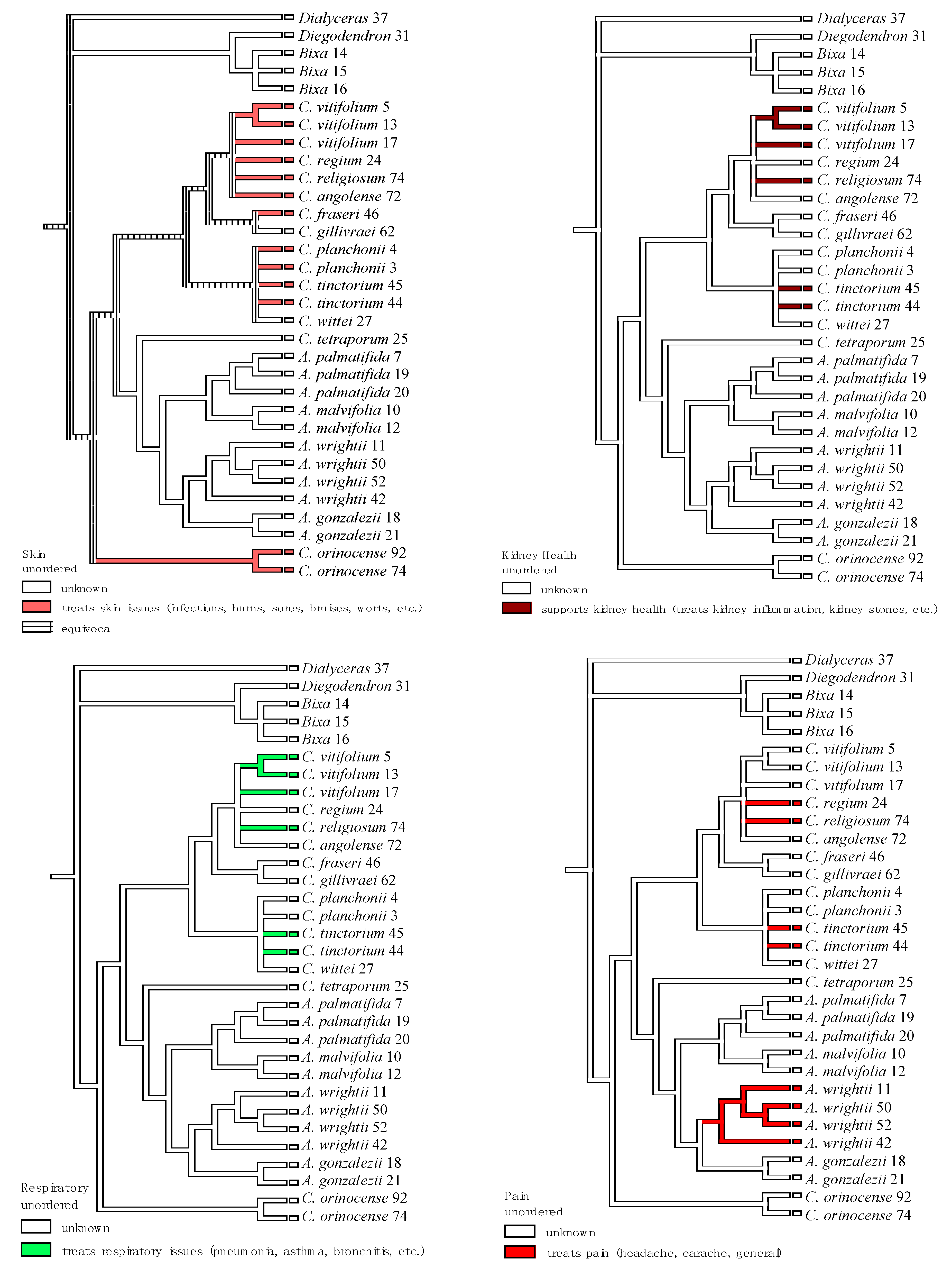
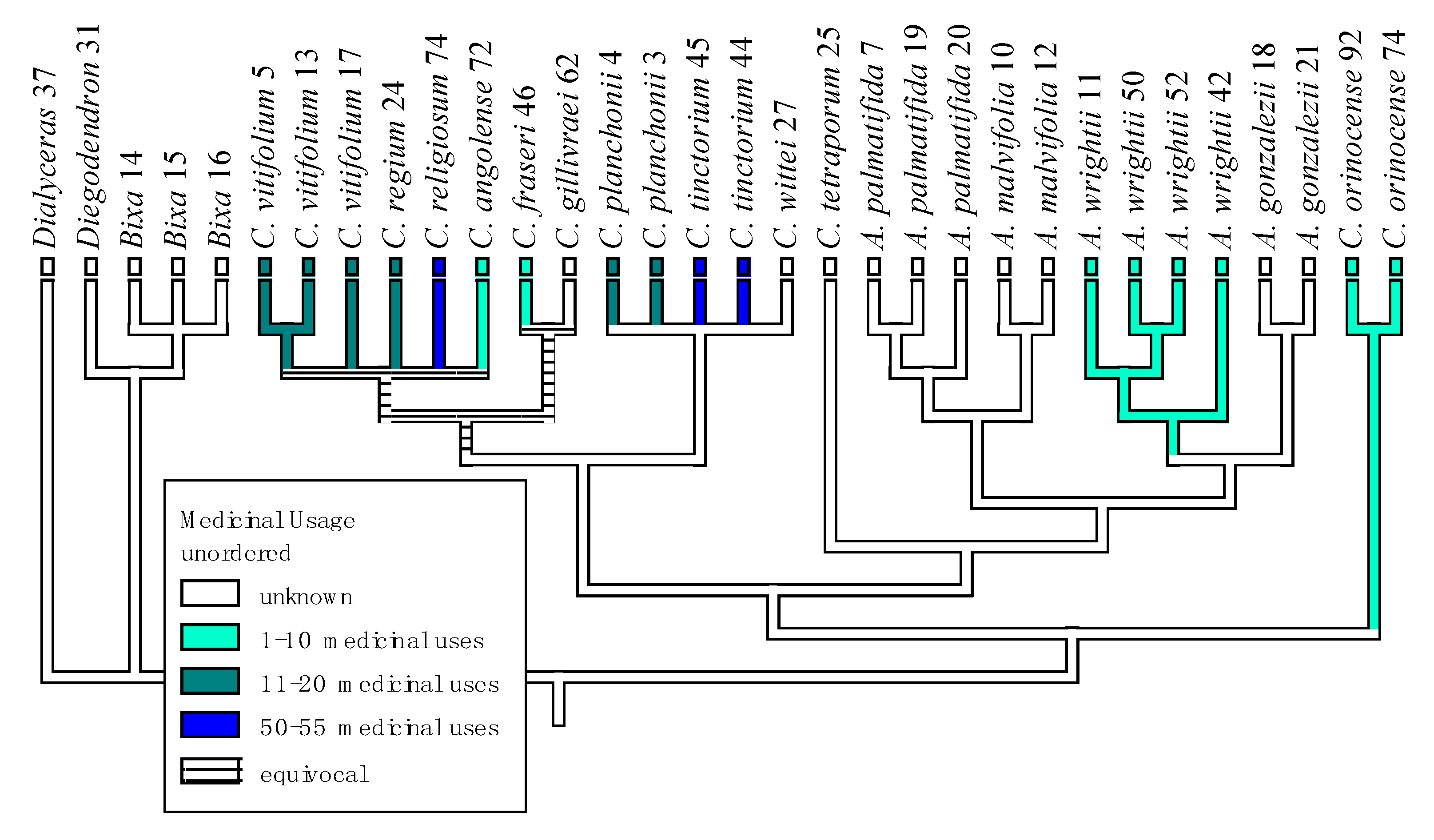
| Plant Species | Geographic Regions | Plant Parts | Medicinal Uses/Ailments Treated |
|---|---|---|---|
| Amoreuxia wrightii A. Gray | Mexico | Leaves | Headache |
| Cochlospermum vitifolium Spreng. | Mexico | Bark | Snakebites, diarrhea, skin rashes, hepatitis, jaundice, liver issues, hypertension, high blood pressure, hyperglycemia, diabetes, asthma, cleanse vagina, veterinary |
| Panama | Bark | Liver issues, kidney issues | |
| Mexico, Guatemala | Wood, leaves | Chest issues, jaundice, liver issues, kidney issues, hasten childbirth | |
| Costa Rica | Wood | Jaundice | |
| Mexico, Cuba | Leaves | Ulcers | |
| Mexico | Flowers | Chest issues, hepatitis | |
| Mexico, Cuba | Root | Stimulate menstruation, abscesses, intestinal issues | |
| Panama | Fruit | Blood circulation | |
| Not specified | Eating disorders, anti-hypertensive | ||
| [11,14,37,38,39,40,41,42,43,44,45] | |||
| Cochlospermum regium Pilg. | Brazil | Roots | Inflammation of the intestine, uterus, and ovaries; skin problems (boils, pimples, dermatitis, and blotchy skin), arthritis and rheumatism, acne, infections of the reproductive and urinary systems |
| Bark | Rheumatoid arthritis, and in a compress to help heal abscesses | ||
| Not specified | Headaches, internal cleansing agent (removing wastes and toxins), heal wounds, cleanse blood, laxative, internal pains, leucorrhea (abnormal vaginal discharge), inflammation/infection of prostate, gastritis, ulcers | ||
| [11,46,47,48,49,50,51,52] | |||
| Cochlospermum orinocense (Kunth) Steud. | Amazon | Bark | Fevers |
| Not specified | Bruises, wounds, promotes formation of scar tissue | ||
| [53,54] | |||
| Cochlospermum planchonii Hook.f. ex Planch. | Burkina Faso | Roots | Malaria, hepatitis, jaundice, liver issues, constipation |
| Ivory Coast | Roots | Fevers, malaria | |
| Nigeria | Roots | Jaundice, fevers, and malaria | |
| Niger | Roots | Jaundice, malaria, fevers, lactation issues | |
| Benin | Leaves | Edema | |
| West Africa | Not specified | Stomach troubles, burns, indigestion, leprosy, jaundice, testicular inflammation, and urethral discharges | |
| [11,55,56,57,58,59,60,61,62,63] | |||
| Cochlospermum tinctorium Perr. | West Africa, including: Ivory Coast, Ghana, Cameroon, Nigeria, Gambia, Guinea-Bissau, Guinea, Togo, and Senegal, | Roots | Jaundice, liver diseases, yellow fever, and malaria |
| West Africa | Not specified | Epilepsy, schistosomiasis, leprosy and other skin infections, burns, edema, urethral discharge, menstrual cramps, testicular inflammation, pneumonia and other lung infections, conjunctivitis, indigestion, stomachache, diarrhea, rickets, intestinal worms, and beriberi, stimulate blood flow in the uterus and pelvic region and aid in childbirth | |
| Mali | Roots | Urinary issues, schistosomiasis, gastrointestinal problems, such as ulcers, stomachaches, constipation, and flatulence, abdominal pains, wounds, hemorrhoids, intestinal worms, schistosomiasis, and hepatitis, snakebites | |
| Leaves | Malaria, ulcers, and flatulence | ||
| Flowers | Constipation | ||
| Senegal | Roots | Rickets, colic, indigestion, ascite, beriberi, hemorrhoids, testicular inflammation, schistosomiasis, syphilis, bronchitis, convulsions, epilepsy, fever, imflammation of lymph nodes, measles, malaria, blepharitis (inflammation of eyelid), gonorrhea, boils, worms | |
| Togo | Root | Painful menstruation, rectal prolapse, jaundice | |
| Cochlospermum tinctorium | Benin | Root | Jaundice, inner hemorrhaging, headache |
| Whole plant | Weakness | ||
| Nigeria | Root | Promote general body health | |
| Root, stem | Veterinary medicine | ||
| Fruit | Snakebites | ||
| Seed (oil) | Leprosy | ||
| Niger | Roots | Jaundice | |
| Ghana | Roots | Bronchial problems (pneumonia, lung inflammation), epileptic convulsions, eye issues (conjunctivitis, trachoma, sore eyes), fevers, hernias, chest pain, leprosy, absence of menstruation, female sterility, hemorrhoids, sprains, burns, bruises, stomachaches, indigestion, inflammation, venereal diseases, snakebites, bleeding diarrhea, rheumatic muscle and back pain | |
| Burkina Faso | Root | Malaria | |
| Ivory Coast | Root | Inflammation of the testes, schistosomiasis, jaundice, fevers, epilepsy, pneumonia, chest pain, bronchial problems (pneumonia), conjunctivitis, indigestion, stomach pain, snakebites, skin diseases | |
| Leaves | Abscesses and boils | ||
| Stems, roots | Urinary and genital pain and disorders, kidney pain, chest pain | ||
| [15,56,63,64,65,66,67,68,69,70,71,72,73,74] | |||
| Cochlospermum angolense Welw. | Angola | Bark, roots | Protect and heal liver, jaundice, hepatitis, prophylaxis and treatment for malaria, stomach, liver, gallbladder, spleen, and urinary tract, and also used externally as a wash for skin problems, such as herpes and open sores |
| [11,75,76] | |||
| Cochlospermum religiosum (L.) Alston | India | Not specified | Dry cough, tuberculosis, bronchitis, laryngitis, colds, sneezing, mucus inflammation in the nose, throat, and sinuses, varicose veins, malaria, weakness, fever, headache, earache, hard of hearing, optic inflammation, and inflammation of the stomach, liver, kidney, and urinary tract. Other uses include expelling phlegm from the lungs, dissolving bladder and kidney stones, removing warts, relieving and moisturizing sore, irritated, and/or congested eyes, and enhancing ejaculation while controlling excessive and/or involuntary emissions, stop or slow bleeding by contracting tissues and/or blood vessels, excessive menstrual bleeding, blood in the urine, bleeding hemorrhoids, lower intestinal bleeding, and vomiting blood. Aphrodisiacs, cerebral tonics (relaxes and soothes anxiety and nervous twitches), astringents, diuretics, general anti-inflammatories, internal cooling agents, and assists with opium addictions |
| Leaves, flowers | Stimulant | ||
| Whole plant | Coughs | ||
| Gum | Pain reliever, sedative, and laxative, and is thought to be thermogenic. It is also used to treat cough, diarrhea, dysentery, pharyngitis, gonorrhoea, syphilis, trachoma, and stomach and urinary tract disorders, protection from heat strokes | ||
| [11,16,18,77,78,79,80] | |||
| Cochlospermum fraseri Planch. | Australia | Root | Treat sores, boils, and cuts |
| Leaves, bark, flowers | Treat fevers | ||
| [81,82] | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson-Fulton, S.B.; Watson, L.E. Comparing Medicinal Uses of Cochlospermaceae throughout Its Geographic Range with Insights from Molecular Phylogenetics. Diversity 2018, 10, 123. https://doi.org/10.3390/d10040123
Johnson-Fulton SB, Watson LE. Comparing Medicinal Uses of Cochlospermaceae throughout Its Geographic Range with Insights from Molecular Phylogenetics. Diversity. 2018; 10(4):123. https://doi.org/10.3390/d10040123
Chicago/Turabian StyleJohnson-Fulton, Susannah B., and Linda E. Watson. 2018. "Comparing Medicinal Uses of Cochlospermaceae throughout Its Geographic Range with Insights from Molecular Phylogenetics" Diversity 10, no. 4: 123. https://doi.org/10.3390/d10040123
APA StyleJohnson-Fulton, S. B., & Watson, L. E. (2018). Comparing Medicinal Uses of Cochlospermaceae throughout Its Geographic Range with Insights from Molecular Phylogenetics. Diversity, 10(4), 123. https://doi.org/10.3390/d10040123





