Estimating Fish Species Richness across Multiple Watersheds
Abstract
1. Introduction
- (1)
- Identify an efficient sampling protocol for accurately estimating species richness at the drainage basin scale by comparing true species richness with species richness estimates produced by different estimators and sampling designs that were derived from sample subsets. I hypothesized that the efficiency of species richness estimation (as indicated by sampling effort required to produce accurate estimates) would be maximized by using a suitable estimator combined with a sampling design that accounted for key spatial factors that influenced species richness within and among drainage basins.
- (2)
- Using methods that were developed under objective 1, compare stream fish species richness among reservations that differed in the degree of disturbance, land use, and other factors. I hypothesized that stream fish species richness would be inversely correlated with anthropogenic development/disturbance within the reservations.
2. Materials and Methods
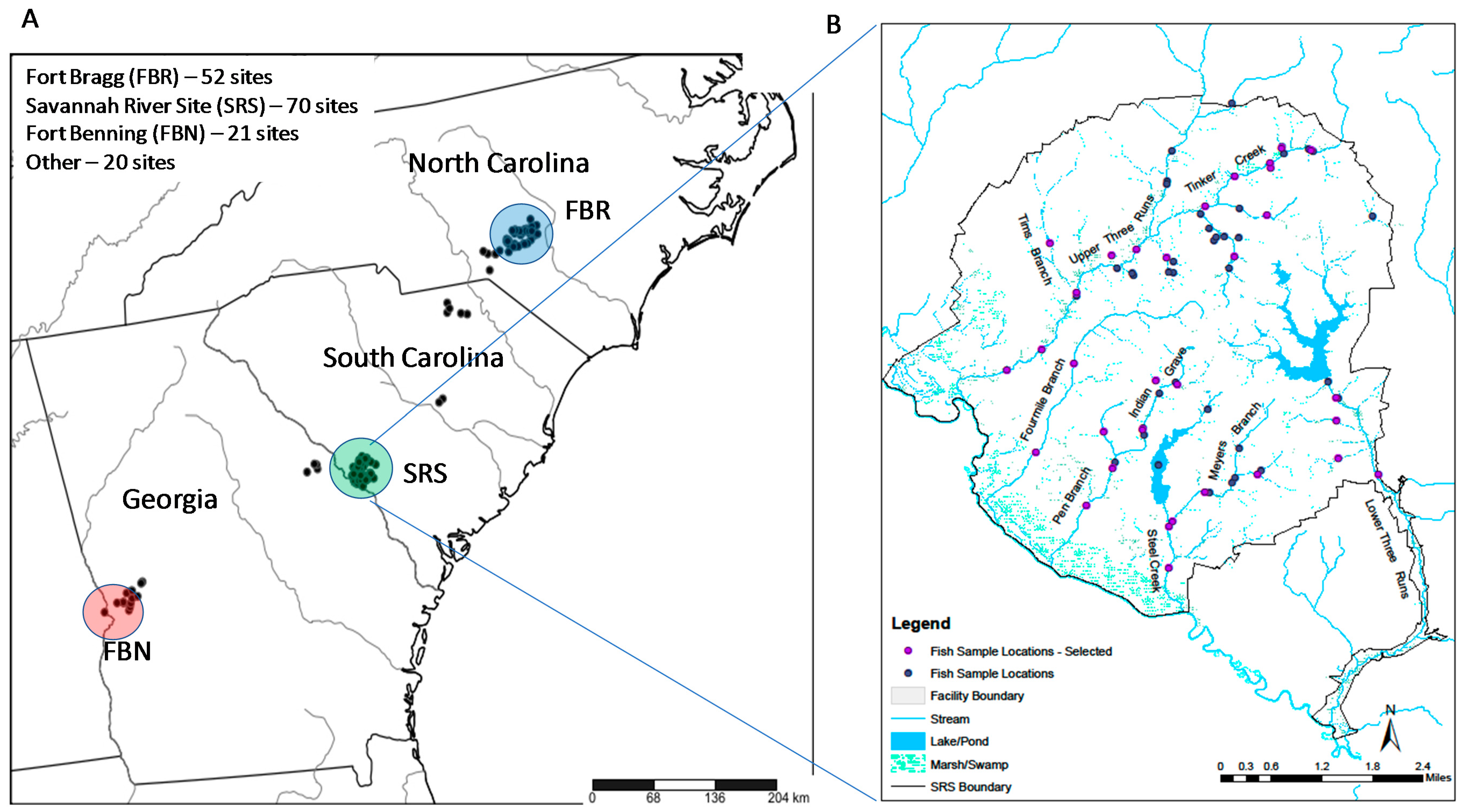
2.1. Study Areas
2.2. Fish Sampling
2.3. Environmental Data
2.4. Data Analysis
2.4.1. Estimating Drainage-Wide Species Richness
2.4.2. Comparing Species Richness Among Reservations
3. Results
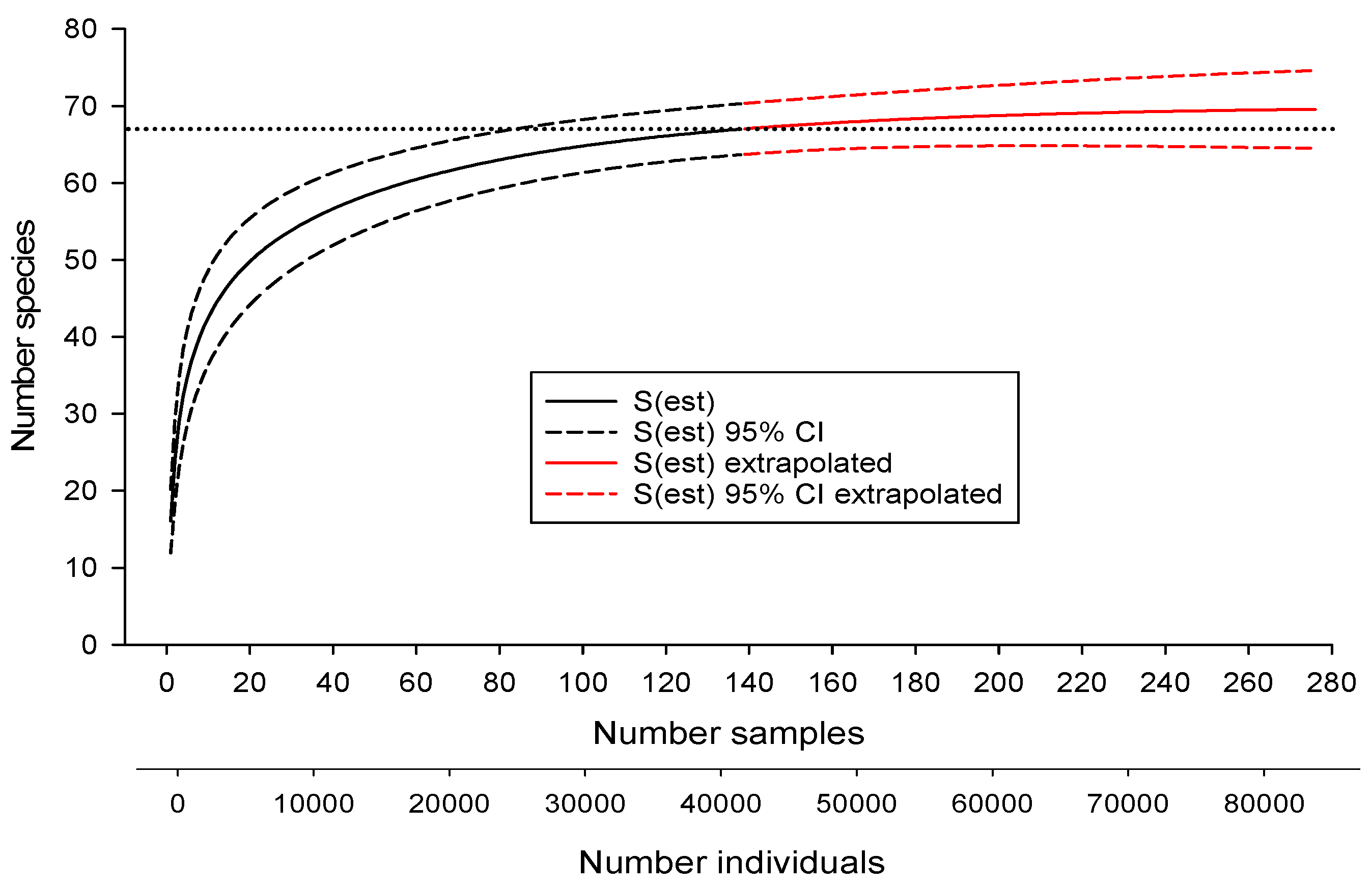
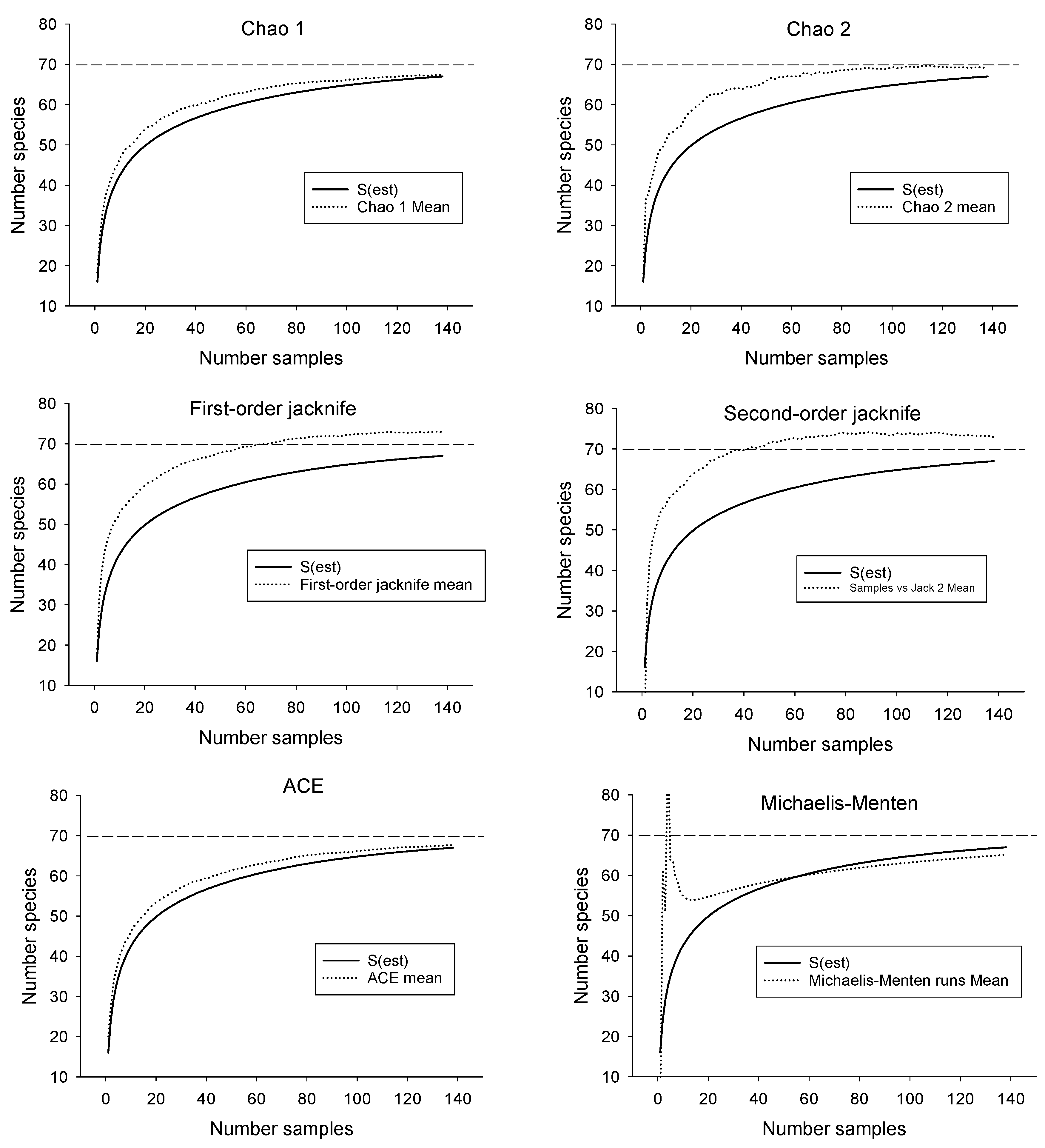
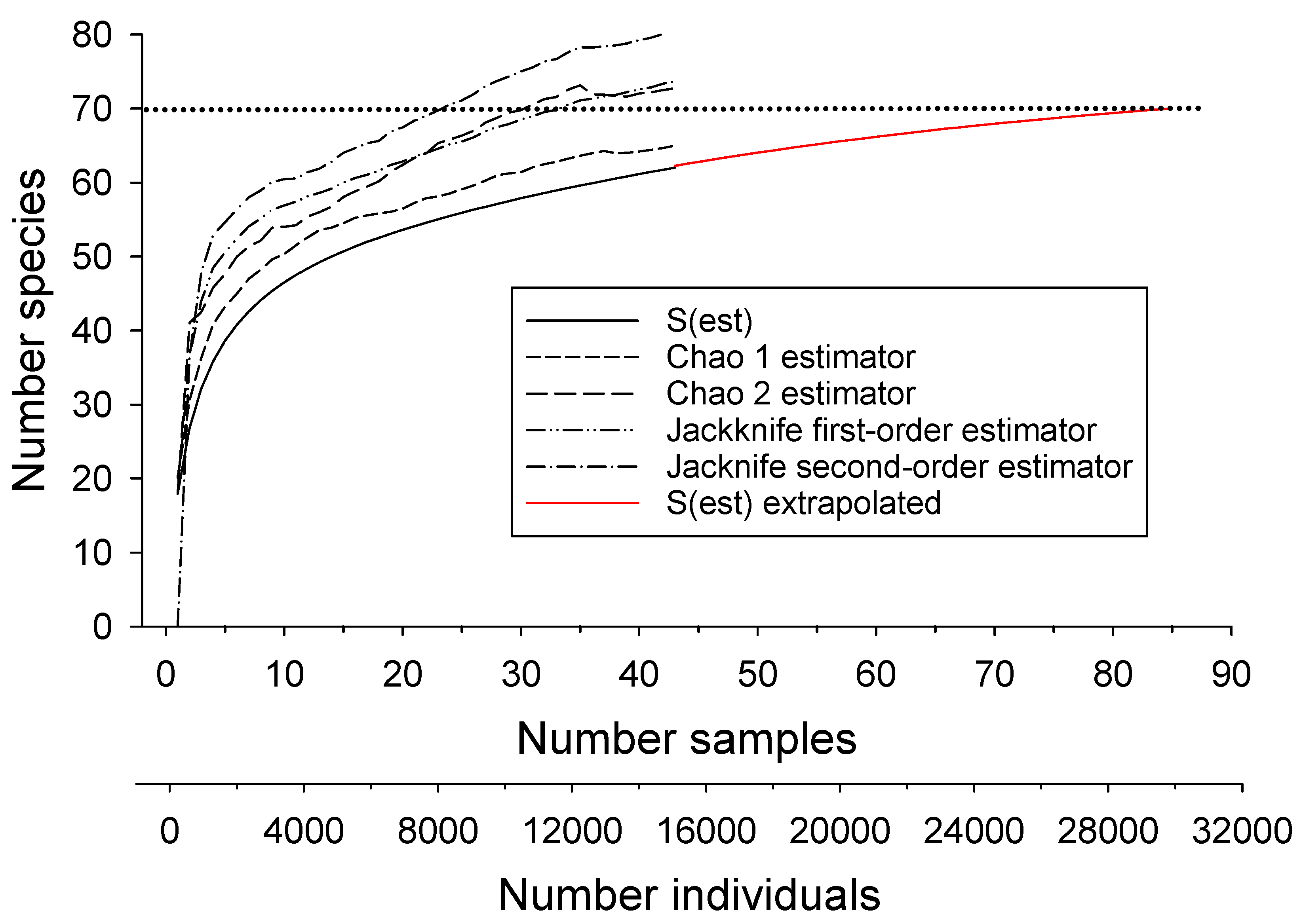
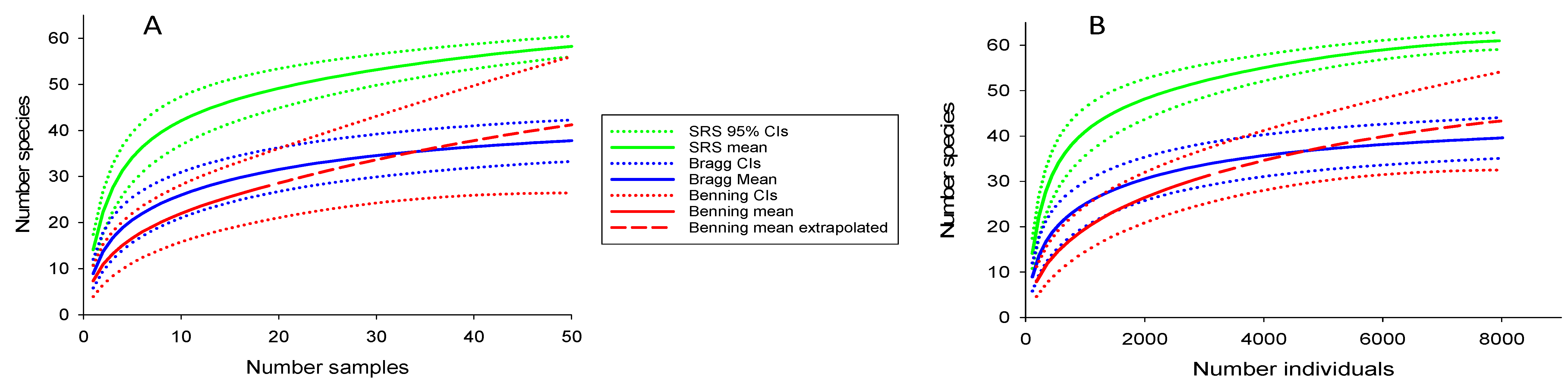
3.1. Estimating Drainage-Wide Species Richness
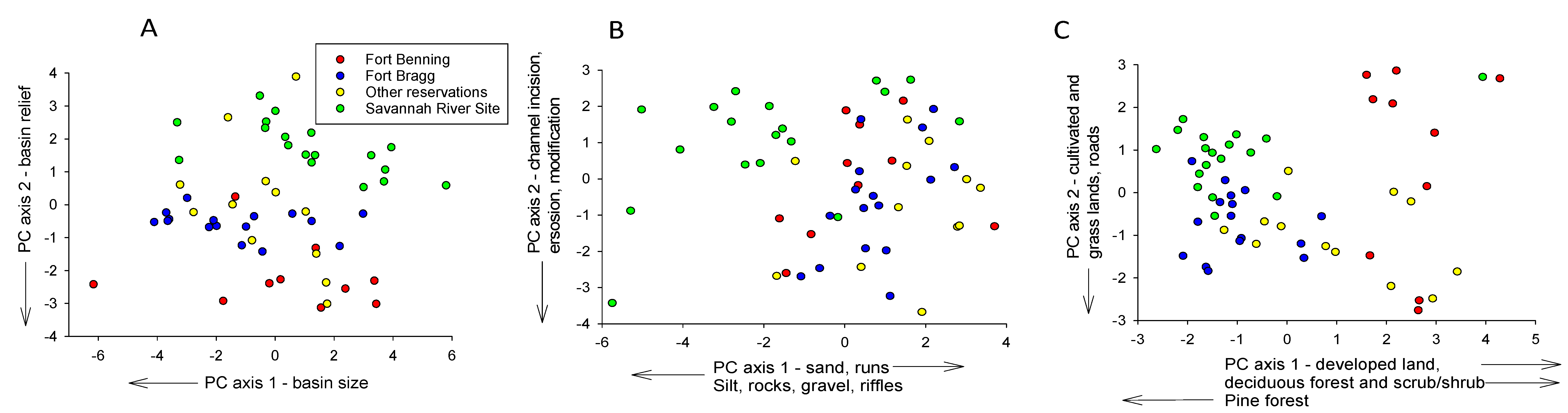
3.2. Comparing Species Richness Among Reservations
4. Discussion
4.1. Estimating Drainage-Wide Species Richness
4.2. Comparing Species Richness Among Reservations
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.; Knowler, D.J.; Lévêque, C.L.; Naiman, R.J.; Prieur-Richard, A.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef] [PubMed]
- Helfman, G.S. Fish Conservation: A Guide to Understanding and Restoring Global Aquatic Biodiversity and Fishery Resources; Island Press: Washington, DC, USA, 2007. [Google Scholar]
- Jelks, H.L.; Walsh, S.J.; Burkhead, N.M.; Contreras-Balderas, S.; Diaz-Pardo, E.; Hendrickson, D.A.; Lyons, J.; Mandrak, N.E.; McCormick, F.; Nelson, J.S.; et al. Conservation status of imperiled North American freshwater and diadromous fishes. Fisheries 2008, 33, 372–407. [Google Scholar] [CrossRef]
- Nichols, J.D.; Conroy, M.J. Estimation of species richness. In Measuring and Monitoring Biodiversity—Standard Methods for Mammals; Wilson, D.E., Cole, J.D., Rudran, R., Foster, M., Eds.; Smithsonian Institution Press: Washington, DC, USA, 1996; pp. 227–234. [Google Scholar]
- Holtrop, A.M.; Cao, Y.; Dolan, C.R. Estimating sampling effort required for characterizing species richness and site-to-site similarity in fish assemblage surveys of wadeable Illinois streams. Trans. Am. Fish. Soc. 2010, 139, 1421–1435. [Google Scholar] [CrossRef]
- Connor, E.F.; McCoy, E.D. The statistics and biology of the species-area relationship. Am. Nat. 1979, 113, 791–833. [Google Scholar] [CrossRef]
- Palmer, M.W.; White, P.S. Scale dependence and the species-area relationship. Am. Nat. 1994, 144, 717–740. [Google Scholar] [CrossRef]
- Scheiner, S.M. Six types of species-area curves. Glob. Ecol. Biogeogr. 2003, 12, 441–447. [Google Scholar] [CrossRef]
- Gotelli, N.; Colwell, R.K. Quantifying biodiversity: Procedures and pitfalls in the measurement and comparison of species richness. Ecol. Lett. 2001, 4, 379–391. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Preston’s ergodic conjecture: The accumulation of species in space and time. In Biodiversity Dynamics: Turnover of Populations, Taxa and Communities; McKinney, M.L., Drake, J., Eds.; Columbia University Press: New York, NY, USA, 1998; pp. 311–348. [Google Scholar]
- Carey, S.; Ostling, A.; Harte, J.; del Moral, R. Impact of curve construction and community dynamics on the species-time relationship. Ecology 2007, 88, 2145–2153. [Google Scholar] [CrossRef] [PubMed]
- Cam, E.; Nichols, J.D.; Hines, J.E.; Sauer, J.R.; Alpizar-Jara, R.; Flather, C.H. Disentangling sampling and ecological explanations underlying species-area relationships. Ecology 2002, 83, 1118–1130. [Google Scholar]
- Colwell, R.K.; Coddington, J.A. Estimating terrestrial biodiversity through extrapolation. Philos. Trans. R. Soc. B 1994, 345, 101–118. [Google Scholar] [CrossRef] [PubMed]
- Holdridge, L.R.; Grenke, W.C.; Hatheway, W.H.; Liang, T.; Tosi, J.A. Forest Environments in Tropical Life Zones; Pergamon Press: Oxford, UK, 1971. [Google Scholar]
- Raaijmakers, J.G.W. Statistical analysis of the Michaelis-Menten equation. Biometrics 1987, 43, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Melo, S.A. A critique of the use of jackknife and related non-parametric techniques to estimate species richness. Community Ecol. 2004, 5, 149–157. [Google Scholar] [CrossRef]
- O’Hara, R.B. Species richness estimators: How many species can dance on the head of a pin? J. Anim. Ecol. 2005, 74, 375–386. [Google Scholar] [CrossRef]
- Gotelli, N.; Colwell, R.K. Estimating species richness. In Frontiers in Measuring Biodiversity; Magurran, A.A., McGill, B.J., Eds.; Oxford University Press: New York, NY, USA, 2011; pp. 39–54. [Google Scholar]
- Rosenzweig, M.L.; Turner, W.R.; Cox, J.G.; Ricketts, T.H. Estimating diversity in unsampled habitats of a biogeographical province. Conserv. Biol. 2003, 17, 864–874. [Google Scholar] [CrossRef]
- Angermeier, P.L.; Smogor, R.A. Estimating number of species and relative abundances in stream-fish communities: Effects of sampling effort and discontinuous spatial distributions. Can. J. Fish. Aquat. Sci. 1995, 52, 936–945. [Google Scholar] [CrossRef]
- Paller, M.H. Relationships among number of fish species sampled, reach length surveyed, and sampling effort in South Carolina coastal plain streams. N. Am. J. Fish. Manag. 1995, 15, 110–120. [Google Scholar] [CrossRef]
- Kennard, M.J.; Pusey, B.J.; Harch, B.D.; Dore, E.; Arthington, A.H. Estimating local stream fish assemblage attributes: Sampling effort and efficiency at two spatial scales. Mar. Freshw. Res. 2006, 57, 635–653. [Google Scholar] [CrossRef]
- Smith, J.M.; Wells, S.P.; Mather, M.E.; Muth, R.M. Fish biodiversity sampling in stream ecosystems: A process for evaluating the appropriate types and amount of gear. Aquat. Conserv. 2014, 24, 338–350. [Google Scholar] [CrossRef]
- Smith, K.L.; Jones, M.L. Watershed-level sampling effort requirements for determining riverine species composition. Can. J. Fish. Aquat. Sci. 2005, 62, 1580–1588. [Google Scholar] [CrossRef]
- Hyde, J.M.; Stewart, P.M.; Miller, J.M. Species richness estimation and rarefaction of fish assemblages in a small watershed. Southeast. Nat. 2014, 13, 208–220. [Google Scholar] [CrossRef]
- Harding, J.S.; Benfield, E.F.; Bolstad, P.V.; Helfman, G.S.; Jones, E.B.D., III. Stream biodiversity: The ghost of land use past. Proc. Natl. Acad. Sci. USA 1998, 95, 14843–14847. [Google Scholar] [CrossRef] [PubMed]
- HydroGeoLogic. Prepared for the Legacy Resource Management Program, Strategic Environmental Research and Development Program, and Environmental Security Technology Certification Program. In Proceedings of the Southeast Region Threatened, Endangered, and At-Risk Species Workshop, Cocoa Beach, FL, USA, 27 February–1 March 2007; HydroGeoLogic, Inc.: Reston, VA, USA, 2007. [Google Scholar]
- Griffith, G.E.; Omernik, J.M.; Comstock, J.A.; Lawrence, S.; Martin, G.; Goddard, A.; Hulcher, V.J.; Foster, T. Ecoregions of Alabama and Georgia, (Color Poster with Map, Descriptive Text, Summary Tables, and Photographs); Map Scale 1:1,700,000; US Geological Survey: Reston, VA, USA, 2001.
- Loehle, C.; Wigley, T.B.; Schilling, E.; Tatum, V.; Beebe, J.; Vance, E.; van Deusen, P.; Weatherford, P. Achieving conservation goals in managed forests of the Southeastern Coastal Plain. Environ. Manag. 2009, 44, 1136–1148. [Google Scholar] [CrossRef] [PubMed]
- Paller, M.H.; Feminella, J.; Kosnicki, E.; Sefick, S.; Miller, J.; Tuberville, T.; Fletcher, D.; Grosse, A.; Harris, B.; Sterrett, S.; et al. Factors Influencing Stream Fish Species Composition and Functional Properties at Multiple Spatial Scales in the Sand Hills of the Southeastern United States. Trans. Am. Fish. Soc. 2016, 145, 545–562. [Google Scholar] [CrossRef]
- Paller, M.H.; Feminella, J.; Kosnicki, E.; Sefick, S.; Miller, J.; Tuberville, T.; Fletcher, D.; Grosse, A.; Harris, B.; Sterrett, S.; et al. Development of Ecological Reference Models and an Assessment Framework for Streams on the Atlantic Coastal Plain; Final Report SERDP Project RC-1694; SRNL-STI-2014-00050; Savannah River National Laboratory: Aiken, SC, USA, 2014.
- SCDHEC (South Carolina Department of Health and Environmental Control). Standard Operating and Quality Control Procedures for Macroinvertebrate Sampling; Technical Report No. 004-98; Bureau of Water, Division of Water Monitoring, Assessment and Protection, Aquatic Biology Section: Columbia, SC, USA, 1998.
- Colwell, R.K.; Mao, C.X.; Chang, J. Interpolating, Extrapolating, and Comparing Incidence-Based Species Accumulation Curves. Ecology 2004, 85, 2717–2727. [Google Scholar] [CrossRef]
- Chazdon, R.L.; Colwell, R.K.; Denslow, J.S.; Guariguata, M.R. Statistical methods for estimating species richness of woody regeneration in primary and secondary rain forests of Northeastern Costa Rica. In Forest Biodiversity Research, Monitoring and Modeling: Conceptual Background and Old World Case Studies; Dallmeier, F., Comiskey, J.A., Eds.; Parthenon Publishing: Paris, France, 1998; pp. 285–309. [Google Scholar]
- Colwell, R.K.; Chao, A.; Gotelli, N.J.; Lin, S.; Mao, C.X.; Chazdon, R.L.; Longino, J.T. Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. J. Plant Ecol. 2012, 5, 3–21. [Google Scholar] [CrossRef]
- Lande, R. Statistics and partitioning of species diversity and similarity among multiple communities. Oikos 1996, 76, 5–13. [Google Scholar] [CrossRef]
- McCune, B.; Grace, J.B. Analysis of Ecological Communities; MjM Software Design: Gleneden Beach, OR, USA, 2002. [Google Scholar]
- Colwell, R.K. EstimateS: Statistical Estimation of Species Richness and Shared Species from Samples, version 9; User’s Guide and Application. 2013. Available online: http://viceroy.eeb.uconn.edu/estimates/EstimateSPages/EstSUsersGuide/EstimateSUsersGuide.htm (accessed on 29 January 2018).
- McCune, B.; Mefford, M.J. PC-ORD. Multivariate Analysis of Ecological Data, version 6; MjM Software: Gleneden Beach, OR, USA, 2011. [Google Scholar]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002. [Google Scholar]
- SYSTAT Software, Inc. SYSTAT 12; SYSTAT Software, Inc.: San Jose, CA, USA, 2007. [Google Scholar]
- Sokal, R.R.; Rohlf, F.J. Biometry the Principles and Practice of Statistics in Biological Research, 3rd ed.; W.H. Freeman and Company: New York, NY, USA, 1995. [Google Scholar]
- Oberdorff, T.; Guégan, J.-F.; Hugueny, B. Global scale patterns of fish species richness in rivers. Ecography 1995, 18, 345–352. [Google Scholar] [CrossRef]
- Griffiths, D. Connectivity and vagility determine spatial gradients and diversification of freshwater fish in North America and Europe. Biol. J. Linn. Soc. 2015, 116, 773–786. [Google Scholar] [CrossRef]
- Tedesco, P.A.; Beauchard, O.; Bigorne, R.; Blanchet, S.; Buisson, L.; Conti, L.; Cornu, J.-F.; Dias, M.S.; Grenouillet, G.; Hugueny, B.; et al. A global database on freshwater fish species occurrence in drainage basins. Sci. Data 2017, 4, 170141. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.L.; Swan, C.M.; Auerbach, D.A.; Campbell Grant, E.H.; Hitt, N.P.; Maloney, K.O.; Patrick, C. Metacommunity theory as a multispecies, multiscale framework for studying the influence of river network structure on riverine communities and ecosystems. J. N. Am. Benthol. Soc. 2011, 30, 310–327. [Google Scholar] [CrossRef]
- Sheldon, A.L. Species diversity and longitudinal succession in stream fishes. Ecology 1968, 49, 193–198. [Google Scholar] [CrossRef]
- Paller, M.H. Relationship between fish assemblage structure and stream order in South Carolina coastal plain streams. Trans. Am. Fish. Soc. 1994, 123, 150–161. [Google Scholar] [CrossRef]
- Súarez, Y.R.; Valério, S.B.; Tondado, K.K.; Florentino, A.C.; Felipe, T.R.A.; Ximenes, L.Q.L.; Lourenço, L.S. Fish species diversity in headwaters streams of Paraguay and Paraná Basins. Braz. Arch. Biol. Technol. 2007, 50, 1678–4324. [Google Scholar] [CrossRef]
- Erős, T.; Schmera, D. Spatio-temporal scaling of biodiversity and the species-time relationship in a stream fish assemblage. Freshw. Biol. 2010, 55, 2391–2400. [Google Scholar] [CrossRef]
- Meador, M.R.; McIntyre, J.P.; Pollock, K.H. Assessing the efficacy of single-pass backpack electrofishing to characterize fish community structure. Trans. Am. Fish. Soc. 2003, 132, 39–46. [Google Scholar] [CrossRef]
- Kimmel, W.G.; Argent, D.G. Efficacy of two-pass electrofishing employing multiple units to assess stream fish species richness. Fish. Res. 2006, 82, 14–18. [Google Scholar] [CrossRef]
- Dos Anjos, M.B.; Zuanon, J. Sampling effort and species richness in small terra firme forest streams of central Amazonia, Brazil. Neotrop. Ichthyol. 2007, 5, 45–52. [Google Scholar] [CrossRef]
- Ebner, B.C.; Thiem, J.D.; Gilligan, D.M.; Lintermans, M.; Wooden, I.J.; Linke, S. Estimating species richness and catch per unit effort from boat electro-fishing in a lowland river in temperate Australia. Austral Ecol. 2008, 33, 891–901. [Google Scholar] [CrossRef]
- Súarez, Y.R.; de Souza, M.M.; Ferreira, F.S.; Pereira, M.J.; da Silva, E.A.; Ximenes, L.Q.L.; de Azevedo, L.G.; Martins, O.C.; Lima Júnior, S.E. Patterns of species richness and composition of fish assemblages in streams of the Ivinhema River basin, Upper Paraná River. Acta Limnol. Bras. 2011, 23, 177–188. [Google Scholar] [CrossRef]
- Glowacki, L.; Penczak, T. Species richness estimators applied to fish in a small tropical river sampled by conventional methods and rotenone. Aquat. Living Resour. 2005, 18, 159–168. [Google Scholar] [CrossRef]
- Chao, A.; Lee, S.M. Estimating the number of classes via sample coverage. J. Am. Stat. Assoc. 1992, 87, 210–217. [Google Scholar] [CrossRef]
- Tonn, W.M.; Magnuson, J.J.; Rask, M.; Toivonen, J. Intercontinental comparison of small-lake fish assemblages: The balance between local and regional processes. Am. Nat. 1990, 136, 345–375. [Google Scholar] [CrossRef]
- Osborne, L.L.; Wiley, M.J. Influence of tributary spatial position on the structure of warmwater fish communities. Can. J. Fish. Aquat. Sci. 1992, 49, 671–681. [Google Scholar] [CrossRef]
- Osborne, L.L.; Kohler, S.L.; Bailey, P.B.; Day, D.M.; Bertrand, W.A.; Wiley, M.J.; Sauer, R. Influence of stream location in a drainage network on the index of biotic integrity. Trans. Am. Fish. Soc. 1992, 121, 635–643. [Google Scholar] [CrossRef]
- Thornbrugh, D.J.; Gido, K.B. Influence of spatial positioning within stream networks on fish assemblage structure in the Kansas River basin, USA. Can. J. Fish. Aquat. Sci. 2009, 67, 143–156. [Google Scholar] [CrossRef]
- Cross, J.N. Distribution of fish in the Virgin River, a tributary of the lower Colorado River. Environ. Biol. Fishes 1985, 12, 13–21. [Google Scholar] [CrossRef]
- Robertson, S.; Parker, M.; Linam, G.; Robertson, C.; Grubb, A. River Studies Report No. 26, Canadian River Bioassessment; Inland Division, Texas Parks and Wildlife: Austin, TX, USA, 2017. [Google Scholar]
- Thomas, D.A.; Hayes, D.B. A comparison of fish community composition of headwater and adventitious streams in a coldwater river system. J. Freshw. Ecol. 2006, 21, 265–275. [Google Scholar] [CrossRef]
- Bettinger, J.; Crane, J.; Bulak, J. Piedmont Stream Survey; Completion Report, 28 February; South Carolina Department of Natural Resources: Columbia, SC, USA, 2006.
- Hossain, M.S.; Das, N.G.; Sarker, S.; Rahaman, M.Z. Fish diversity and habitat relationship with environmental variables at Meghna river estuary, Bangladesh. Egypt. J. Aquat. Res. 2012, 38, 213–226. [Google Scholar] [CrossRef]
- Voelz, N.J.; McArthur, J.V. An exploration of factors influencing lotic insect species richness. Biodivers. Conserv. 2000, 9, 1543–1570. [Google Scholar] [CrossRef]
| Common Name | Scientific Name | Fourmile Branch | Lower Three Runs | Pen Branch | Steel Creek | Upper Three Runs | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1–2 | 3–4 | 1–2 | 3–4 | 1–2 | 3–4 | 1–2 | 3–4 | 1–2 | 3–4 | ||
| American shad | Alosa sapidissima | ✓ | |||||||||
| Blackbanded darter | Percina nigrofasciata | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Brown bullhead | Ameiurus nebulosus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Blackbanded sunfish | Enneacanthus chaetodon | ✓ | ✓ | ||||||||
| Black crappie | Pomoxis nigromaculatus | ✓ | ✓ | ✓ | ✓ | ||||||
| Bowfin | Amia calva | ✓ | ✓ | ✓ | ✓ | ||||||
| Bannerfin shiner | Cyprinella leedsi | ✓ | |||||||||
| Bluegill | Lepomis macrochirus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Bluehead chub | Nocomis leptcephalus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Banded pygmy sunfish | Elassoma zonatum | ✓ | ✓ | ✓ | |||||||
| Banded sunfish | Enneacanthus obesus | ✓ | ✓ | ||||||||
| Brook silverside | Labidesthes sicculus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Blue spotted sunfish | Enneacanthus gloriosus | ✓ | ✓ | ✓ | ✓ | ||||||
| Carp | Cyprinus carpio | ✓ | |||||||||
| Creek chub | Semotilus atromaculatus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||
| Channel catfish | Ictalurus punctatus | ✓ | ✓ | ||||||||
| Creek chubsucker | Erimyzon oblongus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Christmas darter | Etheostoma hopkinsi | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Chain pickerel | Esox niger | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Coastal shiner | Notropis petersoni | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Dollar sunfish | Lepomis marginatus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Dusky shiner | Notropis cummingsae | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| American eel | Anguilla rostrata | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Flat bullhead | Ameiurus platycephalus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Florida gar | Lepisosteus platyrhincus | ✓ | |||||||||
| Flier | Centrarchus macropterus | ✓ | ✓ | ✓ | |||||||
| Green sunfish | Lepomis cyanellus | ✓ | ✓ | ||||||||
| Golden shiner | Notemigonus crysoleucas | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| Golden topminnow | Fundulus chrysotus | ✓ | ✓ | ||||||||
| Ironcolor shiner | Notropis chalybaeus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||
| Lake chubsucker | Erimyzon sucetta | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Lowland shiner | Pteronotropis stonei | ✓ | ✓ | ✓ | ✓ | ✓ | |||||
| Largemouth bass | Micropterus salmoides | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| longnose gar | Lepisosteus osseus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| Lined topminnow | Fundulus lineolatus | ✓ | ✓ | ✓ | ✓ | ✓ | |||||
| Eastern mosquitofish | Gambusia holbrooki | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Eastern mudminnow | Umbra pygmaea | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |||
| Margined madtom | Noturus insignis | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Mud sunfish | Acantharchus pomotis | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||
| Northern hogsucker | Hypentilium nigricans | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| Notchlip redhorse | Moxostoma collapsum | ✓ | ✓ | ✓ | ✓ | ||||||
| Pumpkinseed | Lepomis gibbosus | ✓ | ✓ | ✓ | ✓ | ||||||
| Pugnose minnow | Opsopoeodus emilia | ✓ | |||||||||
| Pirate perch | Aphredoderus sayanus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Redbreast sunfish | Lepomis auritus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Redear sunfish | Lepomis microlophus | ✓ | ✓ | ✓ | ✓ | ✓ | |||||
| Rosyface chub | Hybopsis rubrifrons | ✓ | ✓ | ✓ | |||||||
| Redfin pickerel | Esox americanus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Snail bullhead | Ameiurus brunneus | ✓ | ✓ | ||||||||
| Savannah darter | Etheostoma fricksium | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Speckled madtom | Noturus leptacanthus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Spotted sucker | Minytrema melanops | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||
| Spotted sunfish | Lepomis punctatus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Striped bass | Morone saxatilis | ✓ | |||||||||
| Spottail shiner | Notropis hudsonius | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| Sawcheek darter | Etheostoma serrifer | ✓ | ✓ | ✓ | |||||||
| Tesselated darter | Etheostoma olmstedi | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Taillight shiner | Notropis maculatus | ✓ | |||||||||
| Tadpole madtom | Noturus gyrinus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Turquoise darter | Etheostoma inscriptum | ✓ | |||||||||
| White crappie | Pomoxis annularis | ✓ | |||||||||
| White catfish | Ameiurus catus | ✓ | ✓ | ✓ | |||||||
| Whitefin shiner | Cyprinella nivea | ✓ | ✓ | ||||||||
| Warmouth | Lepomis gulosus | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| Yellow bullhead | Ameiurus natalis | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Yellow perch | Perca flavescens | ✓ | ✓ | ✓ | |||||||
| Yellowfin shiner | Notropis lutipinnis | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Estimator | Estimated Number of Species | SD | Upper 95% CI | Lower 95% CI | Number Samples Required to Reach Estimate of 67 |
|---|---|---|---|---|---|
| All samples (138 from 71 sites)—67 species, 39,087 individuals | |||||
| Chao 1 | 67.3 | 2.9 | 67.0 | 73.0 | 110 |
| Chao 2 | 69.1 | 3.4 | 67.3 | 80.3 | 55 |
| First-order jacknife | 73.0 | 2.7 | 45 | ||
| Second-order jacknife | 73.0 | 1.4 | 37 | ||
| ACE | 67.6 | 0.2 | 108 | ||
| Michaelis-Menton | 65.1 | 67 not reached | |||
| Multiple-pass (4–7) stratified design (32 sites)—60 species, 12,390 individuals | |||||
| Chao 1 | 62.0 | 2.6 | 60.3 | 74.0 | |
| Chao 2 | 70.7 | 8.6 | 62.7 | 102.8 | 29 |
| First-order jacknife | 70.7 | 4.1 | - | - | 30 |
| Second-order jacknife | 77. 3 | 1.7 | - | - | 20 |
| ACE | 63.3 | 0.8 | - | - | 67 not reached |
| Michaelis-Menton | 62.5 | - | - | - | 67 not reached |
| Two-pass stratified design (32 sites)—58 species, 4211 individuals | |||||
| Chao 1 | 61.7 | 4.5 | 58.0 | 79.8 | 67 not reached |
| Chao 2 | 65.9 | 8.3 | 59.2 | 92.6 | 67 not reached |
| First-order jacknife | 67.7 | 4.3 | - | - | 29 |
| Second-order jacknife | 73.4 | 2.6 | - | - | 23 |
| ACE | 62.9 | 1.4 | - | - | 67 not reached |
| Michaelis-Menton | 59.8 | 67 not reached | |||
| Multiple-pass (4–7) stratified design (25 sites) excluding first order streams—60 species, 10,411 individuals | |||||
| Chao 1 | 62.0 | 2.6 | 60.3 | 74.0 | 67 not reached |
| Chao 2 | 70.6 | 8.6 | 62.6 | 102.3 | 24 |
| First-order jacknife | 70.6 | 3.9 | 24 | ||
| Second-order jacknife | 77.2 | 2.1 | 15 | ||
| ACE | 63.1 | 67 not reached | |||
| Michaelis-Menton | 63.6 | 67 not reached | |||
| Variable | Marginal Effect | Conditional Effect | ||
|---|---|---|---|---|
| Lambda | Lambda | F | p | |
| Mean stream width (m) | 0.22 | 0.22 | 13.30 | 0.002 |
| Stream drainage | 0.05 | 0.05 | 3.06 | 0.002 |
| Sample year | 0.07 | 0.05 | 3.14 | 0.002 |
| Strahler stream order | 0.20 | 0.02 | 1.69 | 0.010 |
| Sample reach length (m) | 0.04 | 0.02 | 0.99 | 0.446 |
| Species Richness | Sites (Average) (α1) | Among Sites within Drainages (β1) | Drainages (Average) (α2) | Among Drainages within Reservations (β2) | Reservations (Averages and Total) (α3) | Among Reservations within Sand Hills (β3) | Sand Hills (γ) |
|---|---|---|---|---|---|---|---|
| SRS | |||||||
| No. spp | 14 | 28 | 42 | 25 | 67 | ||
| Percent | 20 | 42 | 38 | 100 | |||
| Fort Bragg | |||||||
| No. spp | 9 | 21 | 30 | 13 | 43 | ||
| Percent | 21 | 49 | 30 | 100 | |||
| Fort Benning | |||||||
| No. spp | 8 | 18 | 26 | 21 | 46 | ||
| Percent | 17 | 38 | 45 | 100 | |||
| Overall average | |||||||
| No. spp | 10 | 22 | 32 | 20 | 52 | 36 | 88 |
| Percent | 12 | 25 | 22 | 41 | 100 |
| Drainage Basin | Stream | Stream Order | Stream Length | Sites | ||
|---|---|---|---|---|---|---|
| Km | % | Number | % | |||
| Fourmile Branch | Fourmile Branch | 3 | 24.0 | 11.5 | 2 ** | 6.3 |
| unmamed tributaries * | 1 | 5.2 | 2.5 | 0 | 0.0 | |
| Lower Three Runs | Lower Three Runs | 4 | 8.2 | 3.9 | 2 | 6.3 |
| unnamed tributaries * | 1-2 | 18.2 | 8.7 | 2 | 6.3 | |
| Pen Branch | Pen Branch | 2 | 24.0 | 11.5 | 4 | 12.5 |
| Indian Grave Branch * | 2 | 4.5 | 2.1 | 1 | 3.1 | |
| unnamed tributaries * | 1 | 2.7 | 1.3 | 1 | 3.1 | |
| Steel Creek | Steel Creek | 3 | 11.0 | 5.3 | 2 | 6.3 |
| Meyers Branch * | 2-3 | 12.6 | 6.0 | 2 | 6.3 | |
| unnamed tributaries * | 1 | 4.0 | 1.9 | 1 | 3.1 | |
| Upper Three Runs | Upper Three Runs | 4 | 28.0 | 13.4 | 5 | 15.6 |
| Tinker Creek * | 3 | 20.8 | 9.9 | 4 | 12.5 | |
| Mill Creek * | 2 | 8.1 | 3.9 | 1 | 3.1 | |
| Reedy Branch * | 2 | 6.1 | 2.9 | 1 | 3.1 | |
| McQueen Branch * | 1-2 | 5.5 | 2.6 | 1 | 3.1 | |
| Tims branch * | 2 | 10.3 | 4.9 | 1 | 3.1 | |
| unnamed tributaries * | 1-2 | 16.2 | 7.7 | 2 | 6.3 | |
| Totals | 209.4 | 100 | 32 | 100 | ||
| Basin Size, Shape, and Relief | Instream Habitat | Basin Land Cover | ||||||
|---|---|---|---|---|---|---|---|---|
| Factors | PC 1 (43%) | PC 2 (22%) | Factors | PC 1 (19%) | PC 2 (12%) | Factors | PC 1 (23%) | PC 2 (14%) |
| Basin area (km2) | −0.94 | Bank height (m) | −0.41 | Developed, low level (%) | 0.60 | |||
| Basin perimeter (km) | −0.94 | Bank angle (degrees) | −0.63 | Developed, medium level (%) | 0.55 | |||
| Stream length (km) | −0.96 | Erosion (0-4) | −0.40 | Cultivated (%) | 0.42 | −0.53 | ||
| Basin length (km) | −0.96 | Bank vegetative cover (%) | 0.59 | Pasture (%) | −0.45 | |||
| Basin shape | 0.83 | Undercut banks (%) c | −0.65 | Grassland/herbaceous (%) | −0.44 | |||
| Stream length (m) | −0.91 | Root masses (%) c | −0.54 | Deciduous trees (%) | 0.68 | |||
| Basin relief ratio | 0.61 | −0.56 | Macrophytes (%) c | Evergreen forest (%) | −0.94 | |||
| Stream gradient (m/km) | −0.59 | Silt/muck substrate (%) | −0.60 | Mixed forest (%) | 0.48 | |||
| Basin relief (m) | −0.57 | −0.60 | Woody debris (%) | −0.48 | Shrub/scrub (%) | 0.70 | ||
| Sand substrate (%) c | 0.80 | Palustrine (%) | 0.49 | |||||
| Gravel substrate (%) c | −0.70 | Unpaved roads (km) | −0.58 | |||||
| Rock substrate (%) c | −0.55 | Road crossings (km) | −0.40 | |||||
| Proportion riffles | −0.60 | |||||||
| Proportion runs | 0.73 | |||||||
| Proportion shallow pools | −0.50 | |||||||
| Depth (m) | 0.60 | |||||||
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paller, M.H. Estimating Fish Species Richness across Multiple Watersheds. Diversity 2018, 10, 42. https://doi.org/10.3390/d10020042
Paller MH. Estimating Fish Species Richness across Multiple Watersheds. Diversity. 2018; 10(2):42. https://doi.org/10.3390/d10020042
Chicago/Turabian StylePaller, Michael H. 2018. "Estimating Fish Species Richness across Multiple Watersheds" Diversity 10, no. 2: 42. https://doi.org/10.3390/d10020042
APA StylePaller, M. H. (2018). Estimating Fish Species Richness across Multiple Watersheds. Diversity, 10(2), 42. https://doi.org/10.3390/d10020042




