1. Introduction
Seasonal and annual variation in climate affect the temporal and spatial patterns of precipitation and temperature, on both local and global scales, of many coastal regions around the world [
1]. Climate change is considered to be a threat to individuals, species, and ecosystems, because it affects their structure and functioning, as well as the interactions of ecosystems, such as biological, geochemical, and hydrological systems. Climate change also affects the welfare of human society [
2,
3,
4].
Drexler et al. [
5] demonstrated that short-term climatic disturbances, such as El Niño/South Oscillation (ENSO) and the Pacific Decadal Oscillation (DPO) influence important coastal processes. Disrupting the normal intensity, frequency, and duration of precipitation, storms and hurricanes, droughts, hails, and frosts. With repeated drought cycles, these disturbances can potentially affect the structure and function of mangrove forests and upstream ecosystems [
6,
7]. This situation was observed in 2008 when the cold phase of ENSO, called LA NIÑA, was recorded, and the warm phase of this phenomenon, called EL NIÑO, then occurred in 2009. Thus, rapid environmental changes have prompted increased interest in understanding current and future threats to biodiversity and their consequences for ecosystem health and services [
8]. Growing evidence is demonstrating that climate change has altered the phenological synchrony of various organisms, but the underlying mechanisms are unclear, particularly in plants [
9,
10]. Other authors have indicated that the phenological patterns are closely related to climate [
11]. As such, climate change is expected to affect growth patterns in the phenological stage and during the flowering and fruiting of plants, including mangroves. Wang’ondu et al. [
12] reported that the plasticity of the reproductive phenology of
Rhizophora mucronata and
Sonneratia alba in Kenya is related to certain variations in the climate and environment.
Plants respond to imbalances caused by environmental variations, offsetting this plasticity through mechanisms that change the biochemical capacity for assimilation of resources, such as changes in biomass and the rate of tissue loss. This adaptation allows plants to grow in a broad range of levels of reserves. However, the plasticity or the suitability of the species to its environment is limited, and substantial changes in the balance of resources can cause changes in the composition of the species, with a greater impact on the phenology of the plants [
10,
13]. Consequently, studying phenology is an excellent tool for examining climate change [
14,
15]. Conversely, resistance is the capacity to withstand change, which is the ability to not be perturbed or affected by a climate extreme; resilience is the capacity to recover after perturbation, i.e., the ability to recover after being affected by a climate extreme [
16,
17]. Changes in rainfall, hydroperiod, and the physicochemical properties of pore water and sediments as consequences of climatic variations affect the functioning and modify the structures of mangrove communities such as photosynthesis, biomass, composition, richness, and productivity [
5,
18,
19,
20,
21].
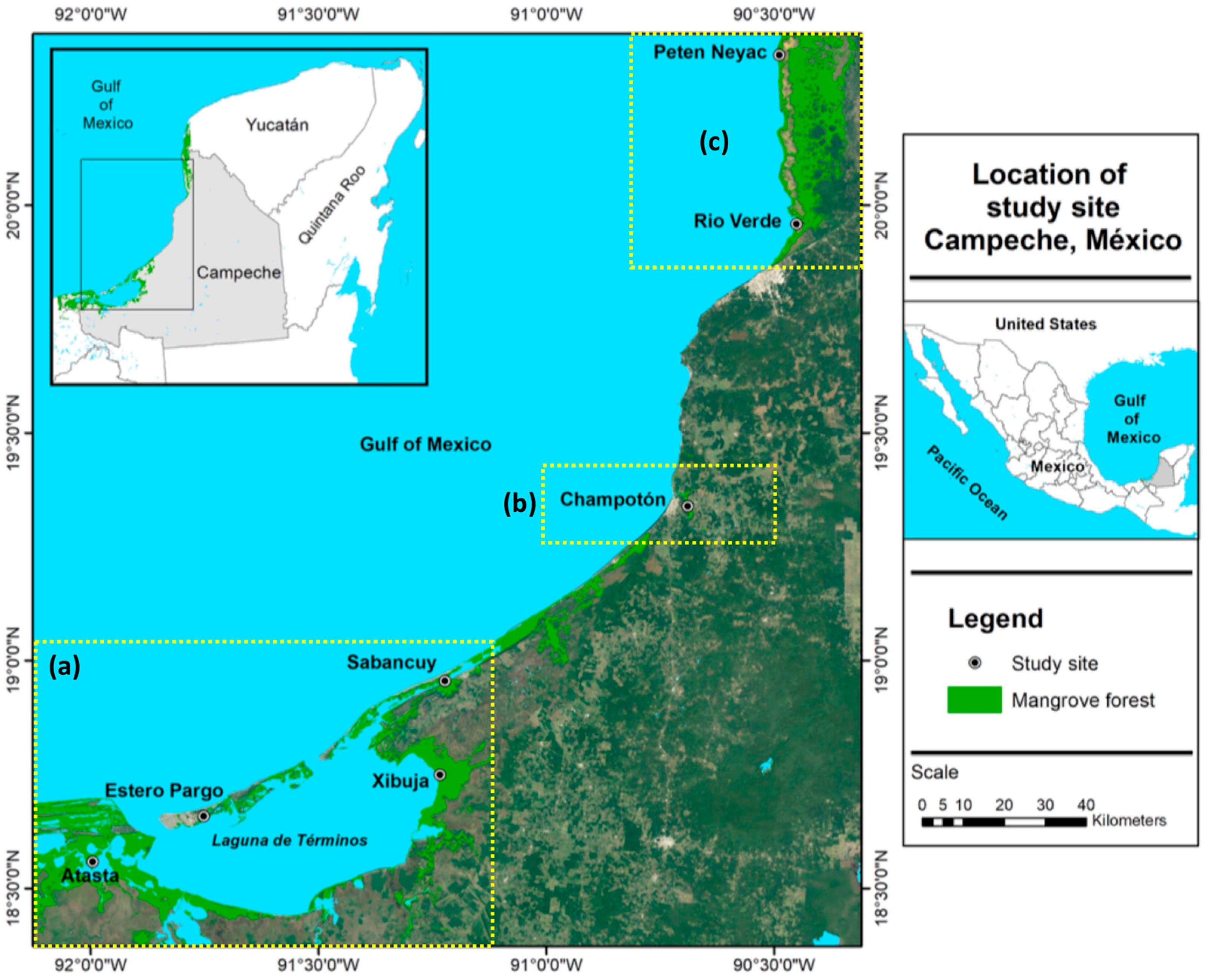
The distribution of Mexican mangroves varies along the coast of México. The extensive continental platform and high rainfall in the Gulf of Mexico promote the development of vast coastal wetlands. This is particularly obvious in the Yucatan Peninsula, which has the most extensive mangrove coverage in the state of Campeche [
22,
23]. However, climate variation in the state of Campeche may alter the periods of reduction or an excess of fresh water, generating a combination of extreme conditions, including salinization of soils, sustained waterlogging, and decreases in oxygen and pH [
24]. From 2006 to 2010, significant changes in the chemical conditions of the pore water and the production of total litterfall and hypocotyls occurred as a result of the variations in the seasonal precipitation in monospecific forests of
Rhizophora mangle L., located in Laguna de Terminos, Campeche, México [
18]. The mangrove forests located along the coast of Campeche exhibited drought conditions in 2009 compared to in 2010. During this period, seasonal changes in rainfall were also apparent [
25]. There is a study which reported on the annual reduction in rainfall in the Maya region of México (which included the states of Quintana Roo, Campeche, Chiapas, Tabasco, and Yucatan), Belize, Guatemala, Honduras, and El Salvador during the 21st century [
26]. These authors predicted a decrease in rainfall between 10% and 22% during the rainy season, and 48% during the dry season. This severe change in the regime of rainfall in 2009 caused a hydrologic change along the coast of Campeche, in the south–southwest region. As a result, salinity increased and the redox potential in the pore water decreased. Laguna de Terminos and the Rio Champoton showed anoxic conditions, whereas the Los Petenes Biosphere Reserve showed hypoxic conditions, which were considered remarkably marginal conditions [
25]. The hydrological variations that occurred in 2009 also caused significant changes in the physiological indicators studied in populations of mangroves along the coast of Campeche.
The objective of this study was to evaluate the variations in the hydrochemical characteristics of the pore water, including salinity and the redox potential, and precipitation and its effects in seven R. mangle (L.) dominated mangrove forests along the coast of Campeche during 2009, 2010, 2014, 2015, and 2016. We asked the following questions: (1) How does the precipitation vary, on a yearly and seasonal basis, along the coast of Campeche, during the study period? (2) Are there significant differences among years and seasons in the variation of the salinity and the redox potential of the groundwater? (3) What are the responses to this variation in terms of the productivity (total litterfall) and the reproductive phenology (flower and hypocotyl production) with the environmental factors in the different R. mangle forests between the three areas? (4) Is R. mangle resilient to seasonal and yearly variations in salinity and redox potential?
4. Discussion
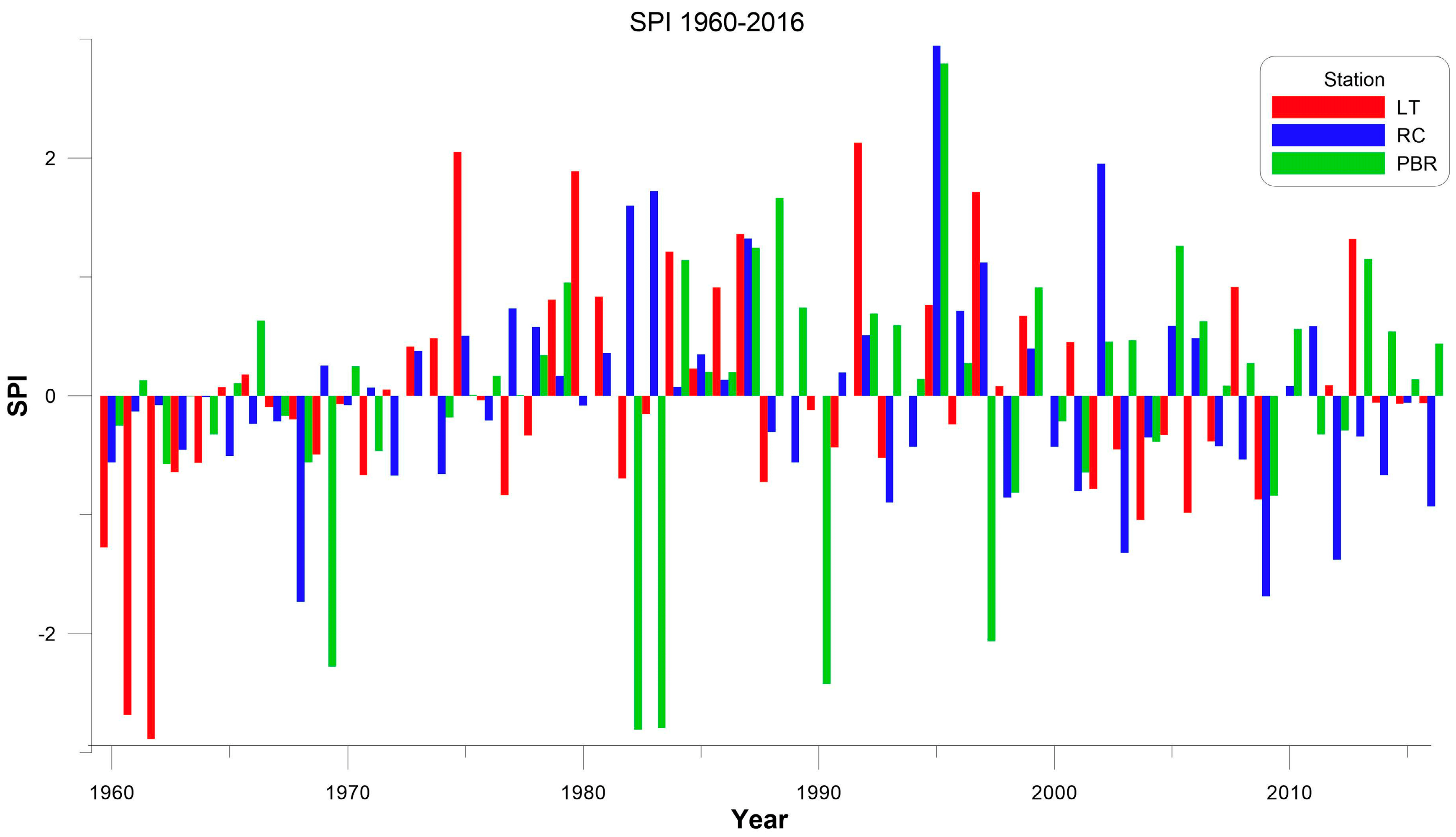
The severe drought conditions registered in 2009 (SPI = −1.50) and 2015 (SPI = −1.16) were caused by a precipitation deficit. The northern area (PBR and RC) registered the most severe drought with 64.4% less precipitation compared to 2010, which was the wettest year. Since the 1950s in Mexico, the second most extreme drought year was 2009 [
39]. In contrast, 2010 was the wettest year. In addition, 2011 and 2012 showed severe drought conditions in the Mexican territory. Consequently, 64 of the 174 natural protected areas in Mexico were at risk due to extreme drought [
40,
41,
42,
43]. These results may be related to the impacts of ENSO and DPO, climatic phenomena that disrupt the normal intensity, frequency, and duration of storms and hurricanes, droughts, hail, and frosts [
44]. These events affect biodiversity, the ecosystem services, and the general wellbeing of human society [
3]. Mangrove communities are particularly vulnerable to changes in precipitation because the hydroperiod and the physicochemical conditions of the soil and pore water are directly modified by the climatic variables [
5,
36]. A small change in the precipitation dynamics could cause a response in the entire biota, including the composition species richness, and the productivity of mangroves [
45]. According to Field [
10], changes in precipitation may cause differences in the distribution and the area covered by mangrove ecosystems, affecting the unusually diverse regeneration processes of these ecosystems. Pore water salinity gradients caused by precipitation changes, the inundation regime, and the microtopography, among other hydroperiod factors, may act as regulating determinants of physiologic processes in mangrove communities, such as tree photosynthesis and growth [
21].
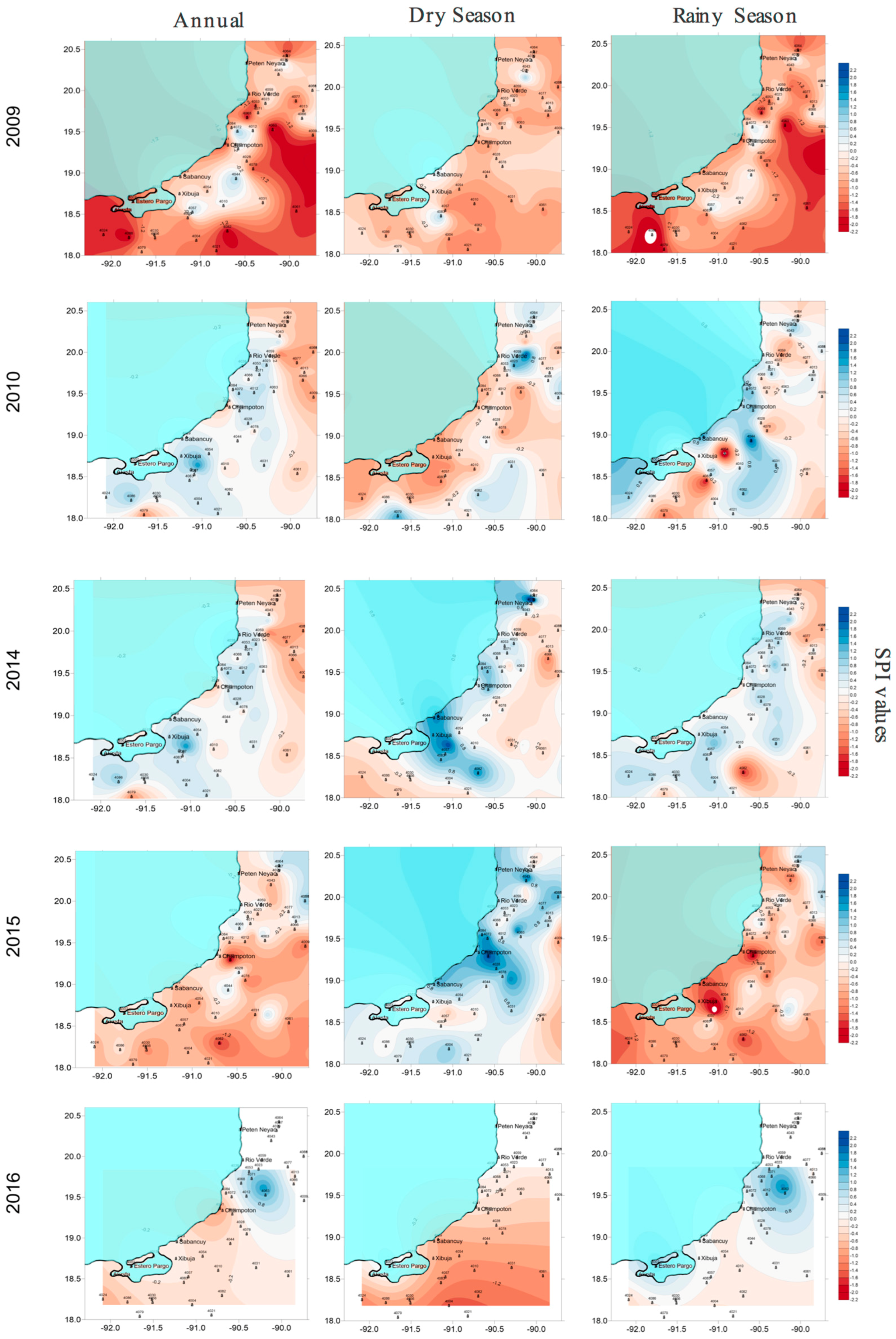
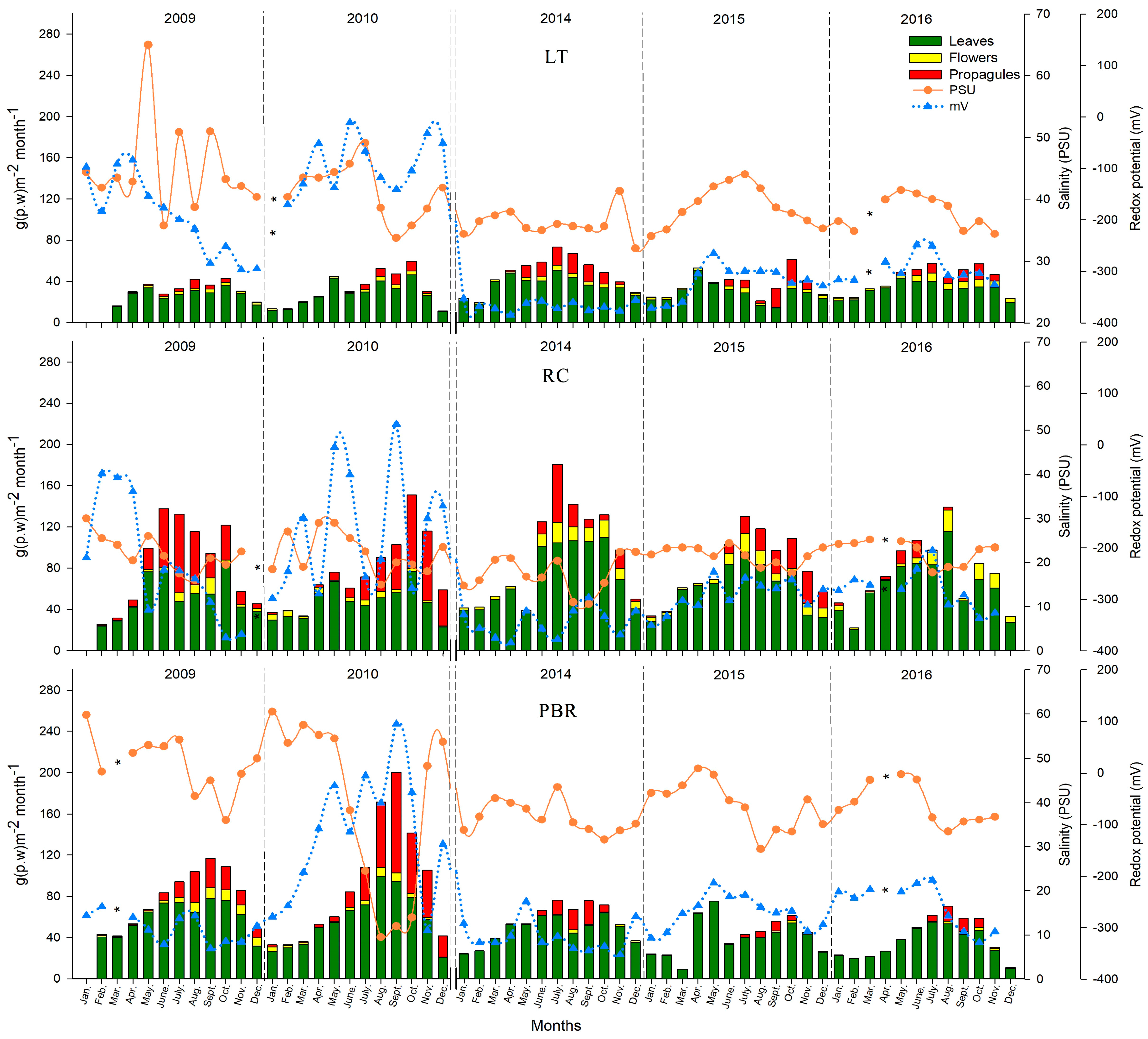
Along the coast of Campeche, the salinity and redox potential of the pore water were heterogeneous. Salinity increased during the drought years in 2009 and 2015, creating mesohaline–hypersaline and hypoxic to anoxic conditions. In contrast, during the wet years of 2010 and 2014, oxic conditions were registered. During the drought in 2009 and 2015, salinity and precipitation had an inverse correlation with each other, whereas salinity and the redox potential among the sites showed an inverse relationship only during 2009. Similarly, Saravanakumar et al. [
46] reported a high inverse correlation between oxygen in the pore water and salinity and temperature in mangroves along the Indian coast. When precipitation decreases, the entry of fresh surface water and interstitial water also decrease, and soil salinity increases, altering the average salinity between the seasons [
47]. By reducing the input of fresh water in wetlands, the dissolved oxygen decreases in the pore water and increases their demand for bacterial respiration processes and the degradation of the organic matter, among other biogeochemical processes [
48]. Variations in the dissolved oxygen concentrations in the pore water depend on the salinity concentration and temperature, establishing an inverse correlation between these parameters [
46,
49].
During the rainy seasons of 2014 to 2016, all three mangrove areas (LT, RC, and PBR), demonstrated mesohaline conditions. However, PBR in the north showed the highest salinity (52.93 ± 7.1 PSU) and anoxic conditions (−331.8 ± 32.0 mV). Meanwhile, in LT in the south, a shift in the seasons from 2014 to 2016 was detected, with the most rainfall occurring in the dry season and drought occurring during the rainy season. In LT, anoxic conditions of less than −300 mV were recorded during the 12 months of 2014, where the southwest site (Atasta), had the most stressful conditions for
R. mangle (>50 PSU, −313.3 ± 25.2 mV) during the rainy season. Along the coast of Campeche, the periods of reduction or the excess of fresh water generated a combination of extreme conditions, including salinization of soils, sustained waterlogging, oxygen, and decreases in pH [
24,
50]. The southern region (LT) has many rivers compared to the northern region (PBR), where the rivers are scarce and the soils are karstic. The circulation of the water is also limited, increasing the temperature and evaporation [
51]. CONAGUA [
25] reported extreme drought during the first nine months of 2009, including the basins of the Palizada and Candelaria rivers, which provide water to the wetlands of LT. The effect of climate variability reduces the flow in the rivers and increases the variability of the hydrological regime which involves episodes of drought and changes in the river system [
52]. CONAGUA [
53] also reported severe drought in south and southeast Mexico during the period of 2014 to 2016.
In the Gulf of Mexico, climate change is impacting the increase in the surface temperature of the ocean and the rising sea level, changing the rainfall regime and the discharge pattern of fresh water, changing the frequency and intensity of tropical storms, and increasing the inland ambient temperature [
54]. Méndez and Magaña [
55] mentioned that Mexico is extremely vulnerable to the negative impacts of significant rainfall deficits (drought) due to its geographic location, with scarce water availability to the north and with drought more significantly impacting the south. Conversely, several studies have reported on drought scenarios in the Yucatan peninsula, with high severity indices in the central and southern areas, and severe in the northwestern region [
28,
56].
Litterfall Production and Phenological Reproduction
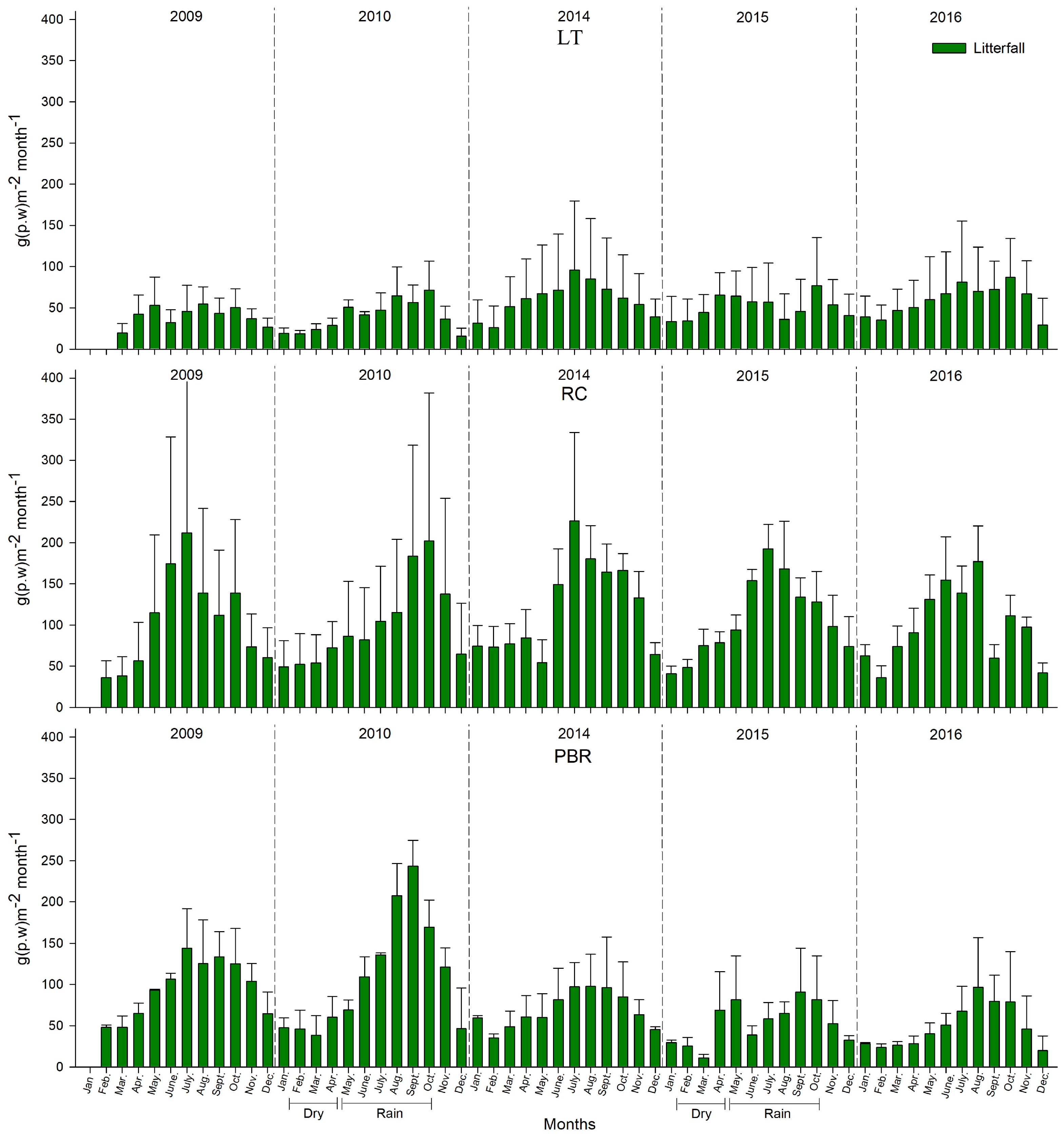
From 2009 to 2010 and 2014 to 2016, significant decreases in litterfall production were found of 14.2 ± 10.5% and 17.5 ± 9.5%, respectively. Litterfall production was significantly lowest in RBP at 29.3 ± 19.3%, which corresponds to a precipitation deficit (22.1%) and hypersaline and anoxic conditions, which are stressful for
R. mangle. A greater impact was registered in the PBR mangroves, where leaf and propagule production decreased by 32.3% and 75%, respectively, from 2014 to 2016 during the rainy season, and 49% from 2010 to 2016 during the dry season (
p < 0.05). However, although hypoxic–anoxic and mesohaline conditions in the pore water were registered in LT from 2014 to 2016, the litter production declined by only 1.7%, the leaf production was unchanged, and the flowering and production of propagules recovered in 2015 to 2016, in comparison to 2009–2010. Several authors indicated that changes in the precipitation pattern affect the productivity decline and survival of the propagules in the mangrove forests [
10,
57,
58,
59]. Gilman et al. [
60] stated that an increase in salinity conditions can cause a severe reduction in the mangrove coverage due to the conversion of upper tidal zones into hypersaline flats. This behaviour reflects differences in the degree of tolerance to drought, saline stress, and prolonged anoxia. The mangroves in LT exhibited more adaptive plasticity than those in PBR. The mangroves of PBR are less tolerant to drought stress, increased salinity, and poorer pore water conditions, meaning that they are more dependent on surface runoff and the contribution of fresh water flow via interstitial means. These mangroves are also influenced by the tide, because the tide helps to dilute the salts throughout a large portion of the year, and by the fresh water that receives surface runoff and groundwater during the rainy season. The mangroves in PBR are also dependent on rain to contribute nutrients during leaching, mainly consisting of phosphates which are critical for the production of photosynthetic tissue.
Rhizophora mangle is especially vulnerable to a PSU value below 30 because this species neither has desalination glands, nor accumulates salts in vacuoles, and it requires oxygen availability and frequent flooding [
48,
61]. For this species, the elimination of salts requires a high energy investment. When pore water salinity becomes extremely high, above 90 PSU, and the redox potential becomes lower than −230 mV, individual mangrove trees suffer detrimental effects, such as the inhibition of photosynthesis and enzyme processes, and, consequently, a reduction in the carbon dioxide (CO
2) assimilation rate [
21,
48]. Also, the transpiration and exclusion of salts may be affected as a result of an excess of salts inside mangrove plants [
61].
Along the coast of Campeche, the ecophysiological response of
R. mangle varies according to the environmental factors, which is reflected in the differences in the salinity and redox potential between sites (
p < 0.05), between years (
p < 0.001), and between the seasons (
p < 0.05). These factors are significantly correlated with the production of
R. mangle. Drought reduces the ecological functions and services of many estuaries and mangroves, inhibiting the production of hypocotyls of
R. mangle to exponentially increase the salinity [
47,
62].
Some authors estimated that the variation in the flooding period modifies the physiological process, productivity, and phenology of mangroves [
18,
63]. Sharma et al. [
64] mentioned that the environmental factors change the ecophysiological responses, such as litterfall production, propagules, leaves, and flowers. Saenger [
48] reported that the plants show an enzymatic and photosynthetic inhibition under anoxic (less than −320 mV) and hypersaline conditions. Some plants modify their morphology, physiology, and reproduction to different environmental conditions through variation in genetic expression [
65,
66]. Adaptive phenotypic plasticity is the ability of a particular genotype to express different phenotypes depending on variation in the environment by altering their morphology and physiology [
65,
67]. Mainly, the morphophysiological adaptations of the mangrove forests depend on the restrictions on the gas exchange, flood levels, temperature, electrochemical characteristics, nutrients, and salinity [
18].
5. Conclusions
Significant variation in precipitation was observed in the periods of 2009 to 2010 and 2014 to 2016, associated with changes in the salinity concentration and the redox potential of the pore water, which caused stress in the litterfall production and phenology of
R. mangle along the coast of the state of Campeche, Mexico. The forests in LT and RC showed the most significant adaptive plasticity (resilience). This behaviour reflects differences in the degree of tolerance to drought, saline stress, and prolonged anoxia. The ecophysiological response of
R. mangle was diverse among the three mangrove areas in the state of Campeche (LT, RC, and PBR). RC had the lowest propagule production (65%) during the rainy season. An extreme impact on the forests of PBR was observed in the period of 2014 to 2016, decreasing litterfall by 29.31% and lowering flowering rates, with a more significant effect on the phenological characteristics during the rainy season in terms of the leaves and propagules (32.3% and 75%, respectively). In addition, a 49% decrease in the production of leaves was observed from 2010 to 2016 during the dry season. The survival of mangroves depends on their ability to adapt to these ecosystems given ongoing climate change and the random effects of anthropogenic activities. Finally, we concluded that the ecophysiological response of
R. mangle is a reflection of the intense droughts that occurred in 2009–2010 and 2014–2016 [
68].
The salinity was the determining factor in the production of flowers during the rainy and dry seasons, and precipitation was the determining factor in the production of propagules during the rainy season. Likewise, for the generation of leaves, salinity and redox potential were the determining factors. The results of this study suggest a low phenotypic plasticity of R. mangle.