Amphibian Declines Are Not Uniquely High amongst the Vertebrates: Trend Determination and the British Perspective
Abstract
:1. Introduction
2. Methods
3. Results
3.1. Taxonomic Group Comparisons
| Taxon group | Number of species (IUCN 2008) | Total number of publications | Number of “decline” publications (%) |
|---|---|---|---|
| Amphibians | 6,347 | 504 | 153 (30) |
| Reptiles | 8,734 | 749 | 42 (6) |
| Mammals | 5,488 | 1,298 | 24 (2) |
| Birds | 9,990 | 2,793 | 161 (6) |
| Taxon group | % of species evaluated | Number of threatened species | Number of threatened species as: | |
|---|---|---|---|---|
| % of species | % of evaluated species | |||
| Amphibians | 98.6 | 1905 | 30 | 30 |
| Reptiles | 15.9 | 423 | 5 | 31 |
| Mammals | 100.0 | 1141 | 21 | 21 |
| Birds | 100.0 | 1222 | 12 | 12 |
| Taxon group | Number of native species | Number of declining and/or strictly protected species (%) | Number of extinctions (as resident, breeding) since 1950 |
|---|---|---|---|
| Amphibians | 7 | 3 (43) | 1 |
| Reptiles | 7 | 3 (43) | 0 |
| Mammals | 59 | 14 (24) | 2 |
| Birds | 223 | 88 (39) | 1 |
3.2. The British Situation: Seeking a Baseline
| Year [reference] | ||||
|---|---|---|---|---|
| 1869 [23] | 1893 [24] | 1901–1903 [25,26] | 1948 [27] | |
| Resolution | Mostly national, some county | Mostly national, some county | Regional and vice-county | Vice-county |
| R. temporaria | Common in Britain & Ireland | |||
| R. lessonae | Present in East Anglia | |||
| B. bufo | Absent from Ireland | Widespread in Britain | ||
| B. calamita | Hampshire, London, East Anglia & Solway | Surrey, Suffolk & south-west Ireland | Dorset, Cheshire, Lancashire | |
| T. vulgaris | Widespread & common | Commoner than T. cristatus, and occurs in Ireland | ||
| T. helveticus | Edinburgh, Dorset, Isle of Wight, Somerset | Devon & Hereford | Widespread in Britain, absent in Ireland | |
| T. cristatus | Widespread & common | |||
| L. vivipara | Common in Britain (including Scotland) & Ireland | |||
| L. agilis | Dorset | Surrey, Hants, Cheshire, Lancashire | ||
| A. fragilis | Widespread, including Scotland | Absent from Ireland | ||
| N. natrix | Common in England, maybe Scotland, not in Ireland | Common in south. Only in south of Scotland | Rare in northern England, absent from Scotland | |
| V. berus | In Britain, not Ireland | Common in Scotland | ||
| C. austriaca | Dumfries (!) Hants & Dorset | Surrey & Berkshire | ||
| D. coriacea | Cornwall, Dorset & North Sea coasts | |||
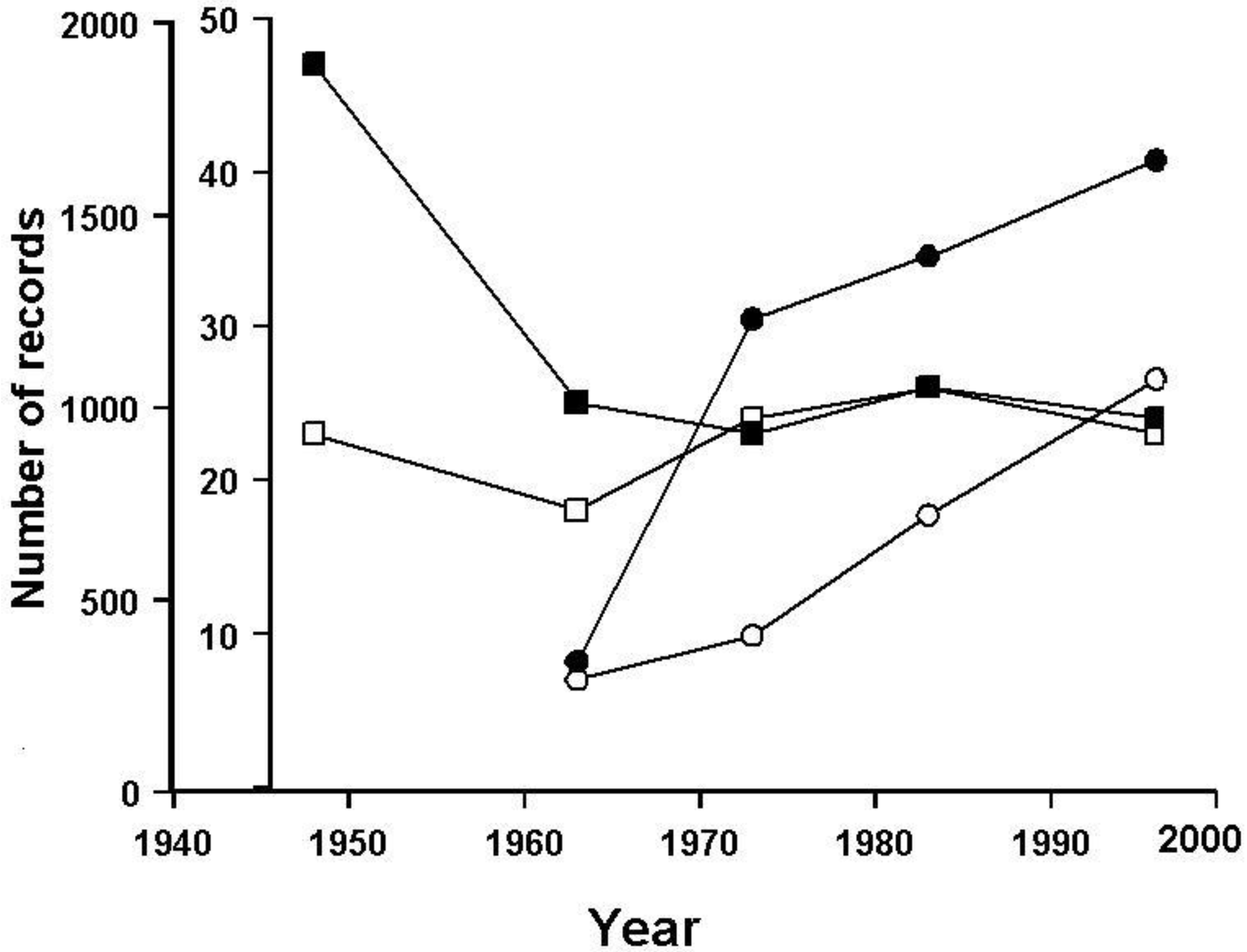
3.3. Population Trends in Britain
| Species | Timespan | Change | Evidence type | References |
|---|---|---|---|---|
| R. temporaria | 1940–1970 | Major decline | Questionnaire | [32] |
| 1970–1980 | Small decline | Questionnaire | [35] | |
| 1980–1990 | Medium increase | Questionnaire | [36] | |
| R. lessonae | 1990–2000 | Extinction | Fieldwork | [22] |
| B. bufo | 1940–1970 | Major decline | Questionnaire | [32] |
| 1970–1980 | Small decline | Questionnaire | [35] | |
| 1980–1990 | Small decline | Questionnaire | [36] | |
| 1985–2000 | Medium decline | Questionnaire | [37] | |
| 1985–2003 | Major declines | Road casualties | [38] | |
| B. calamita | 1900–1970 | Major decline | Fieldwork/ | [33] |
| 1970–1999 | No change | literature/ | [ 39] | |
| questionnaire | ||||
| Fieldwork | ||||
| T. vulgaris | 1970–1980 | Medium decline | Questionnaire | [35] |
| 1980–1990 | No change | Questionnaire | [36] | |
| T. helveticus | 1970–1980 | Small decline | Questionnaire | [35] |
| 1980–1990 | Small decline | Questionnaire | [36] | |
| T. cristatus | 1950–1970 | Strong decline | Questionnaire | [40] |
| 1970–1980 | Strong decline | Questionnaire | [35] | |
| 1980–1990 | Medium decline | Questionnaire | [36] | |
| L. vivipara | 1970–1980 | Medium decline | Questionnaire | [35] |
| 1980–1990 | Medium decline | Questionnaire | [36] | |
| L. agilis | 1970–1987 | Strong decline | Fieldwork | [41] |
| 1800–2000 | Strong decline | Habitat loss | [42,43,44,45] | |
| A. fragilis | 1970–1980 | Medium decline | Questionnaire | [35] |
| 1980–1990 | Medium decline | Questionnaire | [36] | |
| 1980–2003 | No change | Questionnaire/ fieldwork | [46] | |
| N. natrix | 1970–1980 | Major decline | Questionnaire | [35] |
| 1980–1990 | Medium decline | Questionnaire | [36] | |
| V. berus | 1970–1980 | Small decline | Questionnaire | [36] |
| 1980–1990 | Medium decline | Questionnaire | [36] | |
| 1980–2003 | Major decline | Questionnaire/ fieldwork | [46] | |
| C. austriaca | 1970–1987 | Minor decline | Fieldwork | [47] |
| 1970-1979 | 1980-1989 | 1990-1999 | 2000-2009 | |
|---|---|---|---|---|
| B. calamita | ||||
| Total sites | 42 | 44 | 47 | 49 |
| Extinctions | 0 | 4 | 2 | 6* |
| Translocations | 0 | 6 | 5 | 8 |
| L. agilis | ||||
| Total sites | 89 | 93 | 110 | 120 |
| Extinctions | 22 | 2 | 3 | 1 |
| Translocations | 7 | 6 | 20 | 11 |
| Country | Start date | Coverage (no. species) | Coverage (geographical) | Measurement |
|---|---|---|---|---|
| Netherlands | 1997 amphibians 1994 reptiles | 16 amphibians7 reptiles | 320 areas >500 areas | Presence and abundance |
| Britain (NARRS) | 2007 amphibians 2007 reptiles | 5 amphibians 6 reptiles | 400 ponds 400 1 km2 | Presence |
3.4. Reasons for Amphibian and Reptile Declines
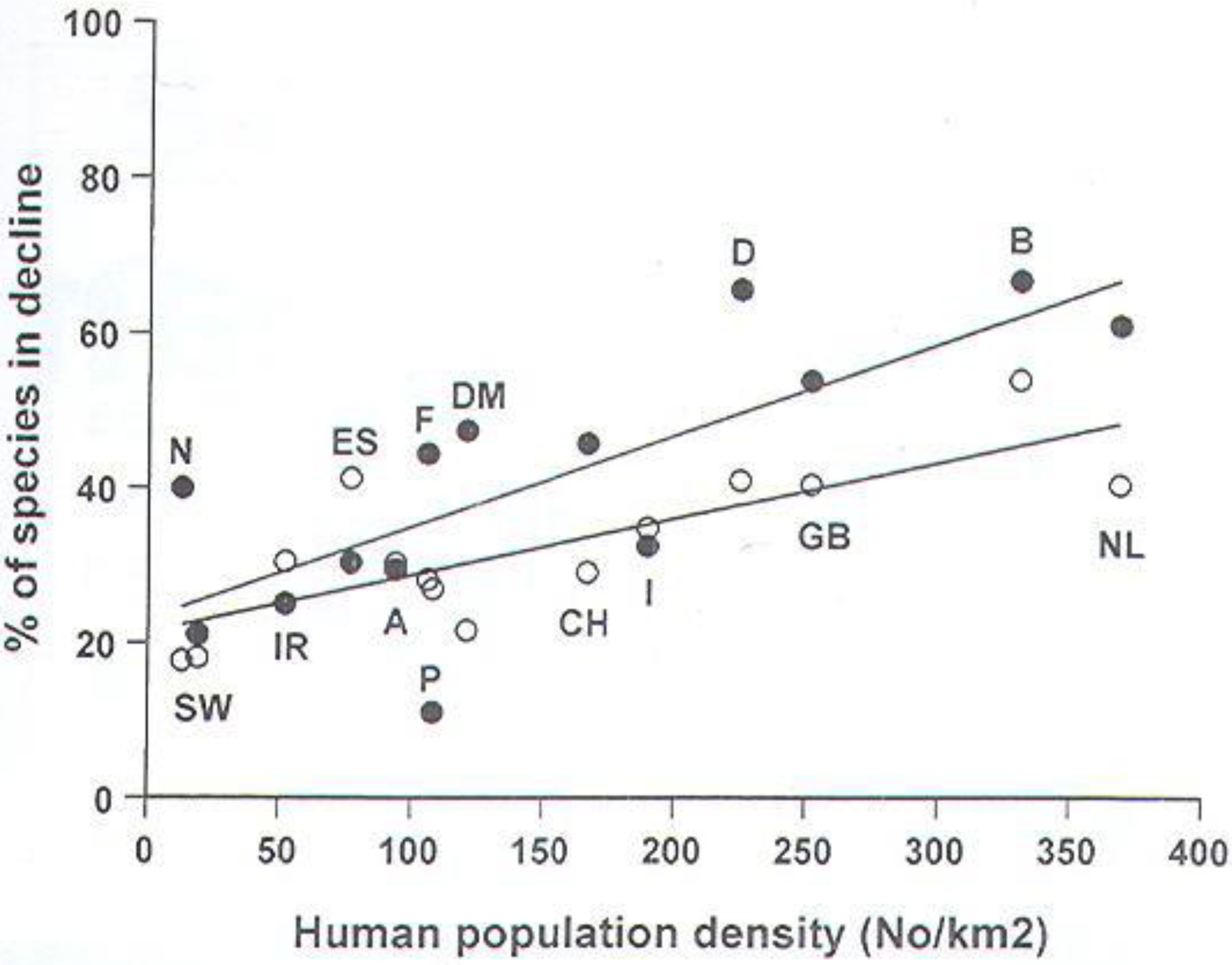
4. Discussion
4.1. Amphibian and Other Declines
| Taxonomic group | Habitat loss | Invasives | Harvesting | Accidents | Persecution |
|---|---|---|---|---|---|
| Amphibians | 77(92) | 40(48) | 22(26) | 3(4) | 0 |
| Threatened | 17(89) | 5(26) | 6(32) | 1(1) | 0 |
| Reptiles | 98(71) | 22(16) | 36(26) | 28(20) | 35(25) |
| Threatened | 22(81) | 9(33) | 16(60) | 4[15) | 4(15) |
| Taxonomic group | Pollution/ climate change | Native population dynamics | Intrinsic factors | Disturbance | |
| Amphibians | 62(75) | 11(13) | 26(31) | 5(6) | |
| Threatened | 10(53) | 3(16) | 12(63) | 4(21) | |
| Reptiles | 34(24) | 8(6) | 22(16) | 16(13) | |
| Threatened | 10(37) | 2(7) | 10(37) | 8(30) |
| Species | Factor | Likely impact | References |
|---|---|---|---|
| B. bufo | (1) Road mortality | Minor/moderate | [37,38,48] |
| (2) Changes in habitat management | Moderate | [37] | |
| (3) Climate change reducing female fecundity and survival | Minor/moderate | [58] | |
| B. calamita | (1) Pond acidification | Minor | [59] |
| (2) Competition from B. bufo after habitat change | Severe | [57] | |
| (3) Inbreeding in isolated populations | Minor | [60] | |
| (4) Chytrid infection | Uncertain | Unpublished | |
| R. lessonae | Changes in habitat management and water abstraction levels | Severe | [22] |
| V. berus | Habitat fragmentation and inbreeding effects | Moderate | [46] |
| L. agilis | Competition with introduced lizards (Podarcis muralis) | Minor | Unpublished |
4.2. Trend Detection and the British Experience
4.3. Research Needs and Future Prospects
References and Notes
- Wake, D.B. Declining amphibian populations. Science 1991, 253, 860. [Google Scholar] [PubMed]
- Stuart, S.N.; Chanson, J.S.; Cox, N.A.; Young, B.E.; Rodrigues, A.S.L.; Fischmann, D.L.; Waller, R.W. Status and trends of amphibian declines and extinctions worldwide. Science 2004, 306, 1783–1786. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.P.; Storfer, A. Global amphibian declines: sorting the hypotheses. Divers. Distrib. 2003, 9, 89–98. [Google Scholar] [CrossRef]
- Beebee, T.J.C.; Griffiths, R.A. The amphibian decline crisis: a watershed for conservation biology? Biol. Conserv. 2005, 125, 271–285. [Google Scholar] [CrossRef]
- Pounds, J.A.; Bustamante, M.R.; Coloma, L.A.; Consuegra, J.A.; Fogden, M.P.L.; Foster, P.N.; La Marca, E.; Masters, K.L.; Merino-Viteri, A.; Puschendorf, R.; Ron, S.R.; Sánchez-Azofeifa, G.A.; Still, C.J.; Young, B.E. Widespread amphibian extinctions from epidemic disease driven by global warming. Nature 2006, 439, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Infectious disease and amphibian population declines. Divers. Distrib. 2003, 9, 141–150. [Google Scholar] [CrossRef]
- Hopkins, W.A. Amphibians as models for studying environmental change. Ilar. J. 2007, 48, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, R.A.; Pavajeau, L. Captive breeding, reintroduction, and the conservation of amphibians. Conserv. Biol. 2008, 22, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Beebee, T.J.C. Amphibian decline? Nature 1992, 355, 120. [Google Scholar] [CrossRef]
- McKinney, M.L. High rates of extinction and threat in poorly studied taxa. Conserv. Biol. 1999, 13, 1273–1281. [Google Scholar] [CrossRef]
- Pechmann, J.H.K.; Scott, D.E.; Semlitsch, R.D.; Caldwell, J.P.; Vitt, L.J.; Gibbons, J.W. Declining amphibian populations: the problem of separating human impacts from natural fluctuations. Science 1991, 253, 892–895. [Google Scholar] [CrossRef] [PubMed]
- Available online: www.IUCN.org (accessed 21 September, 2009).
- Temple, H.J.; Cox, N.A. European Red List of Amphibians; Cambridge Publications: Cambridge, UK, 2009. [Google Scholar]
- Cox, N.A.; Temple, H.J. European Red List of Reptiles; Cambridge Publications: Cambridge, UK, 2009. [Google Scholar]
- Collins, J.P.; Halliday, T.R. Forecasting changes in amphibian biodiversity: aiming at a moving target. Phil. Trans. Roy. Soc. B–Biol. Sci. 2005, 360, 309–314. [Google Scholar]
- Gibbons, J.W.; Scott, D.E.; Ryan, T.J.; Buhlmann, K.A.; Tuberville, T.D.; Metts, B.S.; Greene, J.L.; Mills, T.; Leiden, Y.; Poppy, S.; Winne, C.T. The global decline of reptiles, déjà vu amphibians. BioScience 2000, 50, 653–666. [Google Scholar] [CrossRef]
- Schipper, J.; Chanson, J.S.; Chiozza, F.; Cox, N.A.; Hoffmann, M.; Katariya, V.; Lamoreux, J.; Rodrigues, A.S.L.; Stuart, S.N.; Temple, H.J.; et al. The status of the world’s land and marine mammals: diversity, threat and knowledge. Science 2008, 322, 225–230. [Google Scholar] [CrossRef] [PubMed]
- ekercioğlu, C.; Daily, G.C.; Ehrlich, P.R. Ecosystem consequences of bird declines. Proc. Nat. Acad. Sci. USA 2004, 101, 18042–18047. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.; Yalden, D.W. Mammals of the British Isles: Handbook, 4th ed.; The Mammal Society: Southampton, UK, 2008. [Google Scholar]
- Mayer, W.; Bischoff, W. Beitrage zur taxonomischen revision der gattung Lacerta (Reptilia: Laceridae).1. Zootoca, Omanosaura, Timon and Tiera als eigenstandige gattungen. Salamandra 1996, 32, 163–170. [Google Scholar]
- Frost, D.R.; Grant, T.; Faivovich, J.; Bain, R.H.; Haas, A.; Haddad, C.F.B.; Desa, R.O.; Channing, A.; Wilkinson, M.; Donnellan, S.C.; Raxworthy, C.J.; Campbell, J.A.; Blotto, B.L.; Moler, P.; Derwes, R.C.; Nussbaum, R.A.; Lynch, J.D.; Green, D.M.; Wheeler, W.C. The amphibian tree of life. Bull. Am. Mus. Nat. Hist. 2006, 297, 1–370. [Google Scholar] [CrossRef]
- Beebee, T.J.C.; Buckley, J.; Evans, I.; Foster, J.P.; Gent, A.H.; Gleed-Owen, C.P.; Kelly, G.; Rowe, G.; Snell, C.; Wycherley, J.T.; Zeisset, E. Neglected native or undesirable alien? Resolution of a conservation dilemma concerning the pool frog. Rana lessonae. Biodivers. Conser. 2005, 14, 1607–1626. [Google Scholar] [CrossRef]
- Bell, T. A History of British Reptiles, 2nd ed.; J. Van Voorst: London, UK, 1869. [Google Scholar]
- Cooke, M.C. Our Reptiles and Batrachians; W.H. Allen & Co.: London, UK, 1893. [Google Scholar]
- Leighton, G.R. The Life-History of British Serpents; Blackwood & Sons: London, UK, 1901. [Google Scholar]
- Leighton, G.R. British Lizards; Blackwood & Sons: London, UK, 1903. [Google Scholar]
- Taylor, R.H.R. The distribution of reptiles and amphibia in the British Isles, with notes on species recently introduced. Brit. J. Herpetol. 1948, 1, 1–38. [Google Scholar]
- Taylor, R.H.R. The distribution of amphibians and reptiles in England and Wales, Scotland and Ireland and the Channel Islands: a revised survey. Brit. J. Herpetol. 1963, 3, 95–115. [Google Scholar]
- Arnold, H.R. Provisional Atlas of the Amphibians and Reptiles of the British Isles; Biological Records Centre: Abbots Ripton, UK, 1973. [Google Scholar]
- Arnold, H.R. Distribution Maps of the Amphibians and Reptiles of the British Isles; Biological Records Centre: Abbots Ripton, UK, 1983. [Google Scholar]
- Arnold, H.R. Atlas of Amphibians and Reptiles in Britain; ITE Research Publication 10; HMSO: London, UK, 1996.
- Cooke, A.S. Indications of recent changes of status in the British Isles of the frog (Rana temporaria) and the toad (Bufo bufo). J. Zoo. (London) 1972, 167, 161–178. [Google Scholar] [CrossRef]
- Beebee, T.J.C. The natterjack toad (Bufo calamita) in the British Isles: a study of past and present status. Brit. J. Herpetol. 1976, 5, 515–521. [Google Scholar]
- Prestt, I.; Cooke, A.S.; Corbett, K.F. British Amphibians and Reptiles. In The Changing Flora and Fauna of Britain; Hawksworth, D.L., Ed.; Academic Press: London, UK, Systematics Association Special Volume No. 6.
- Cooke, A.S.; Scorgie, H.R.A. The Status of the Commoner Amphibians and Reptiles in Britain; Nature Conservancy Council: Peterborough, UK, 1983; Focus on Nature Conservation 3. [Google Scholar]
- Hilton-Brown, D.; Oldham, R.S. The Atatus of the Widespread Amphibians and Reptiles in Britain, 1990, and Changes during the 1980’s; Nature Conservancy Council: Peterborough, UK, 1991; Nature Conservancy Council Contract Survey 131. [Google Scholar]
- Carrier, J-A.; Beebee, T.J.C. Recent, substantial and unexplained declines of the common toad Bufo bufo in lowland England. Biol. Conserv. 2003, 111, 395–399. [Google Scholar] [CrossRef]
- Cooke, A.S.; Sparks, T.H. Population declines of common toads (Bufo bufo): the contribution of road traffic and monitoring value of casualty counts. Brit. Herpetol. Soc. Bull. 2004, 88, 13–26. [Google Scholar]
- Buckley, J.; Beebee, T.J.C. Monitoring the conservation status of an endangered amphibian: the natterjack toad Bufo calamita in Britain. Anim. Conserv. 2004, 7, 221–228. [Google Scholar] [CrossRef]
- Beebee, T.J.C. Changes in status of the great crested newt Triturus cristatus in the British Isles. Brit. J. Herpetol. 1975, 5, 481–490. [Google Scholar]
- Corbett, K.F. Distribution and status of the sand lizard, Lacerta agilis agilis, in Britain. Mertensiella 1988, 1, 92–99. [Google Scholar]
- Moore, N.W. The heaths of Dorset and their conservation. J. Ecol. 1962, 50, 369–391. [Google Scholar] [CrossRef]
- Jackson, H.C. The decline of the sand lizard, Lacerta agilis L. population on the sand dunes of the Merseyside coast, England. Biol. Conserv. 1979, 15, 177–193. [Google Scholar]
- Webb, N. Changes on the heathlands of Dorset, England between 1978 and 1987. Biol. Conserv. 1990, 51, 273–286. [Google Scholar] [CrossRef]
- Rose, R.J.; Webb, N.R.; Clarke, R.T.; Traynor, C.H. Changes on the heathland in Dorset, England, between 1987 and 1996. Biol. Conserv. 2000, 93, 117–125. [Google Scholar] [CrossRef]
- Baker, J.; Suckling, J.; Carey, R. Status of the Adder Vipera Berus and Slow-Worm Anguis Fragilis in England; English Nature: Peterborough, UK, 2004; English Nature Research report 546. [Google Scholar]
- Braithwaite, A.C.; Buckley, J.; Corbett, K.F.; Edgar, P.W.; Haslewood, E.S.; Haslewood, G.A.D.; Langton, T.E.S.; Whitaker, W.J. The distribution in England of the smooth snake (Coronella austriaca Laurenti). Herpetol. J. 1989, 1, 370–376. [Google Scholar]
- Young, S.L.; Beebee, T.J.C. An Investigation of Recent Declines in the Common Toad Bufo Bufo; English Nature: Peterborough, UK, 2004; English Nature Research Report 584. [Google Scholar]
- Reading, C.J. A proposed standard method for surveying reptiles on dry lowland heath. J. Appl. Ecol. 1997, 34, 1057–1069. [Google Scholar] [CrossRef]
- Goverse, E.; Smit, G.; van der Meij, T. 10 Years of Amphibian Monitoring in the Netherlands: Preliminary Results. In Proceedings of the 14th European Congress of Herpetology, Porto, Portugal; 2007. [Google Scholar]
- Zuiderwijk, A.; Janssen, I. Results of 14 Years Reptile Monitoring in the Netherlands. In Proceedings of the 6th World Congress of Herpetology, Manaus, Brazil; 2008. [Google Scholar]
- Weir, L.A.; Royle, J.A.; Nanjappa, P.; Jung, R.E. Modeling anuran detection and site occupancy on North American amphibian monitoring program (NAAMP) routes in Maryland. J. Herpetol. 2005, 39, 627–639. [Google Scholar]
- Available online: http://www.narrs.org.uk (accessed 21 September, 2009).
- MacKenzie, D.I.; Nichols, J.D.; Lachman, G.B.; Droege, S.; Royle, J.A.; Langtim, C.A. Estimating site occupancy rates when detection probabilities are less than one. Ecology 2002, 83, 2248–2255. [Google Scholar] [CrossRef]
- Bosch, J.; Carrascal, L.M.; Durán, L.; Walker, S.; Fisher, M.C. Climate change and outbreaks of amphibian chytridiomycosis in a montane area of central Spain: is there a link? Proc. Roy. Soc. B–Biol. Sci. 2007, 274, 253–260. [Google Scholar]
- Houlahan, J.E.; Findlay, C.S.; Schmidt, B.R.; Meyer, A.H.; Kuzmin, S.L. Quantitative evidence for global amphibian population declines. Nature 2000, 404, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Beebee, T.J.C. Environmental change as a cause of natterjack toad (Bufo calamita) declines in Britain. Biol. Conserv. 1977, 11, 87–102. [Google Scholar] [CrossRef]
- Reading, C.J. Linking global warming to amphibian declines through its effects on female body condition and survivorship. Oecologia 2007, 151, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Beebee, T.J.C.; Flower, R.J.; Stevenson, A.C.; Patrick, S.T.; Appleby, P.G.; Fletcher, C.; Marsh, C.; Natkanski, J.; Rippey, B.; Battarbee, R.W. Decline of the natterjack toad (Bufo calamita) in Britain: Palaeoecological, documentary and experimental evidence for breeding site acidification. Biol. Conserv. 1990, 53, 1–20. [Google Scholar] [CrossRef]
- Rowe, G.; Beebee, T.J.C. Population on the verge of a mutational meltdown? Fitness costs of genetic load for an amphibian in the wild. Evolution 2003, 57, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Beebee, T.J.C. British wildlife and human numbers: the ultimate conservation issue? Brit. Wildlife 2001, 13, 1–8. [Google Scholar]
- Kotterat, M.; Freyhof, J. Handbook of European Freshwater Fishes; IUCN: Gland, Swiss, 2007. [Google Scholar]
- Hall, R.J.; Henry, P.F.P. Assessing the effects of pesticides on amphibians and reptiles: status and needs. Herpetol. J. 1992, 2, 65–71. [Google Scholar]
- Lawler, J.J.; White, D.; Sifneos, J.C.; Master, L.L. Rare species and the use of indicator groups for conservation planning. Conserv. Biol. 2003, 17, 875–882. [Google Scholar] [CrossRef]
- Dudgeon, D.; Arthrington, A.H.; Gessner, M.O.; Kawabata, Z.I.; Knowler, J.D.; Leveque, C.; Naiman, R.J.; Prieur-Richard, A.H.; Soto, D.; Stiassny, M.L.J.; Sullivan, C.A. Freshwater biodiversity: importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar]
- Davidson, A.D.; Hamilton, M.J.; Boyer, A.G.; Brown, J.H.; Ceballos, G. Multiple ecological pathways to extinction in mammals. Proc. Nat. Acad. Sci. USA 2009, 106, 10702–10705. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.A.; Telfer, M.G.; Roy, D.B.; Preston, C.D.; Greenwood, J.J.D.; Aher, J.; Fox, R.; Clarke, R.T.; Lawton, J.H. Comparative losses of British butterflies, birds and plants and the global extinction crisis. Science 2004, 303, 1879–1881. [Google Scholar] [CrossRef] [PubMed]
- Gent, T.; Gibson, S. Herpetofauna Workers’ Manual; Joint Nature Conservation Committee: Peterborough, UK, 1998.
- Pellet, J.; Schmidt, B.R. Monitoring distributions using call surveys: estimating site occupancy, detection probabilities and inferring absence. Biol. Conser. 2005, 123, 27–35. [Google Scholar] [CrossRef]
- Kéry, M.; Schmidt, B.R. Imperfect detection and its consequences for monitoring for conservation. Community Ecol. 2008, 9, 207–216. [Google Scholar] [CrossRef] [Green Version]
- Lips, K.R.; Brem, F.; Brenes, R.; Reeve, J.D.; Alford, R.A.; Voyles, J.; Carey, C.; Livo, L.; Pessier, A.P.; Collins, J.P. Emerging infectious disease and the loss of biodiversity in a neotropical amphibian community. Proc. Nat. Acad. Sci. USA 2006, 103, 3165–3170. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.J.; Lips, K.R.; Eichholz, M.W. Decline and extirpation of an endangered Panamanian stream frog population (Craugastor punctariolus) due to an outbreak of cytridiomycosis. Biol. Conserv. 2008, 141, 1636–1647. [Google Scholar] [CrossRef]
- Bosch, J.; Carrascal, L.M.; Durán, L.; Walker, S.; Fisher, M.C. Climate change and outbreaks of amphibian chytridiomycosis in a montane area of central Spain: is there a link? Proc. Roy. Soc. B–Biol. Sci. 2006, 274, 253–260. [Google Scholar]
- Rodder, D.; Veith, M.; Loetters, S. Environmental gradients explaining the prevalence and intensity of infection with the amphibian chytrid fungus: the host’s perspective. Anim. Conserv. 2008, 11, 513–517. [Google Scholar] [CrossRef]
- Holdich, D.; Sibley, P.; Peay, S. The white-clawed crayfish—a decade on. Brit. Wildlife 2004, 16, 153–164. [Google Scholar]
- Rushton, S.P.; Lurz, P.W.W.; Gurnell, J.; Nettleton, P.; Bruemmer, C.; Shirley, M.D.F.; Sainsbury, A.W. Disease threats posed by alien species: the role of a poxvirus in the decline of the native red squirrel in Britain. Epidemiol. Infect. 2006, 134, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Denton, J.S.; Hitchings, S.P.; Beebee, T.J.C.; Gent, A. A recovery program for the natterjack toad (Bufo calamita) in Britain. Conserv. Biol. 1997, 11, 1329–1338. [Google Scholar] [CrossRef]
- Corbett, K.F.; Moulton, N.R. The Sand Lizard Species Recovery Programme Project (1994-1997); English Nature: Peterborough, UK, 1994; English Nature Research reports No. 288. [Google Scholar]
- Baker, J.M.R.; Halliday, T.R. Amphibian colonization of new ponds in an agricultural landscape. Herpetol. J. 1999, 9, 1–8. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Beebee, T.J.C.; Wilkinson, J.W.; Buckley, J. Amphibian Declines Are Not Uniquely High amongst the Vertebrates: Trend Determination and the British Perspective. Diversity 2009, 1, 67-88. https://doi.org/10.3390/d1010067
Beebee TJC, Wilkinson JW, Buckley J. Amphibian Declines Are Not Uniquely High amongst the Vertebrates: Trend Determination and the British Perspective. Diversity. 2009; 1(1):67-88. https://doi.org/10.3390/d1010067
Chicago/Turabian StyleBeebee, Trevor J. C., John W. Wilkinson, and John Buckley. 2009. "Amphibian Declines Are Not Uniquely High amongst the Vertebrates: Trend Determination and the British Perspective" Diversity 1, no. 1: 67-88. https://doi.org/10.3390/d1010067




