Crystal Structures of Two 4-Alkyl-8-hydroxyquinolines
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis of 4-Methyl- (1) and 4-ethyl-8-hydroxyquinoline (2)
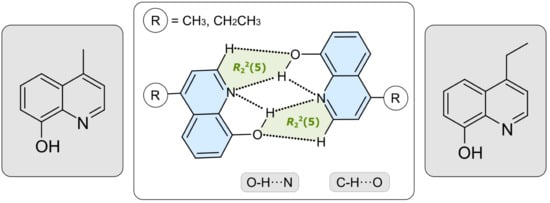
2.2. Structural Elucidation
3. Materials and Methods
3.1. Synthesis and Crystallization of 4-Alkyl-8-hydroxyquinolines 1 and 2
3.2. X-Ray Structure Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rohini; Kamaldeep, P.; Luxami, V. 8-Hydroxyquinoline Fluorophore for Sensing of Metal Ions and Anions. Chem. Rec. 2020, 20, 1430–1473. [Google Scholar] [CrossRef]
- Phillips, J.P. The Reactions of 8-Quinolinol. Chem. Rev. 1956, 56, 271–297. [Google Scholar] [CrossRef]
- Zborowski, K.K.; Solá, M.; Poater, J.; Proniewicz, L.M. Aromatic properties of 8-hydroxyquinoline and its metal complexes. Cent. Eur. J. Chem. 2013, 11, 655–663. [Google Scholar] [CrossRef]
- Hollingshead, R.G.W. Oxine and Its Derivatives; Butterworths Scientific Publications: London, UK, 1956. [Google Scholar]
- Rao, T.P.; Gladis, J.M. Oxines as Preconcentration Agents in Inorganic Trace Analysis. Rev. Anal. Chem. 2001, 20, 145–159. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, T.; Lv, G.; Chao, X.; Yang, X. Extraction and Utilization of Valuable Elements from Bauxite and Bauxite Residue: A Review. Bull. Environ. Contam. Toxicol. 2022, 109, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Melegari, M.; Neri, M.; Falco, A.; Tegoni, M.; Maffini, M.; Fornari, F.; Mucchino, C.; Artizzu, F.; Serpe, A.; Marchiò, L. Tailoring the Use of 8-Hydroxyquinolines for the Facile Separation of Iron, Dysprosium and Neodymium. ChemSusChem 2024, 17, e202400286. [Google Scholar] [CrossRef]
- Kekesi, T. Gallium extraction from synthetic Bayer liquors using Kelex 100-kerosene, the effect of loading and stripping conditions on selectivity. Hydrometallurgy 2007, 88, 170–179. [Google Scholar] [CrossRef]
- Berdimurodov, E.; Verma, C.; Berdimuradov, K.; Quraishi, M.A.; Kholikov, A.; Akbarov, K.; Umirov, N.; Borikhonov, B. 8–Hydroxyquinoline is key to the development of corrosion inhibitors: An advanced review. Inorg. Chem. Commun. 2022, 144, 109839. [Google Scholar] [CrossRef]
- Lewinska, G.; Sanetra, J.; Marszalek, K.W. Application of quinoline derivatives in third-generation photovoltaics. J. Mater. Sci. Mater. Electron. 2021, 32, 18451–18465. [Google Scholar] [CrossRef]
- Hamada, Y.; Sano, T.; Fujita, M.; Fujii, T.; Nishio, Y.; Shibata, K. Organic Electroluminescent Devices with 8-Hydroxyquinoline Derivative-Metal Complexes as an Emitter. Jpn. J. Appl. Phys. 1993, 32, L514. [Google Scholar] [CrossRef]
- Chen, C.H.; Shi, C. Metal chelates as emitting materials for organic electroluminescence. Coord. Chem. Rev. 1998, 171, 161–174. [Google Scholar] [CrossRef]
- Wu, S.; Li, S.; Li, C.-Z.; Shi, M.; Chen, H. Chemical modification of AlQ3 to a potential electron acceptor for solution-processed organic solar cells. Tetrahedron Lett. 2016, 57, 2797–2799. [Google Scholar] [CrossRef]
- Geffert, C.; Kuschel, M.; Mazik, M. Molecular Recognition of N-Acetylneuraminic Acid by Acyclic Pyridinium- and Quinolinium-Based Receptors in Aqueous Media: Recognition through Combination of Cationic and Neutral Recognition Sites. J. Org. Chem. 2013, 78, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Mazik, M.; Geffert, C. 8-Hydroxyquinoline as a building block for artificial receptors: Binding preferences in the recognition of glycopyranosides. Org. Biomol. Chem. 2011, 9, 2319–2326. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, M.; Fiege, M.; Osetska, O. 8-Hydroxyquinolines in metallosupramolecular chemistry. Coord. Chem. Rev. 2008, 252, 812–824. [Google Scholar] [CrossRef]
- Prachayasittikul, V.; Prachayasittikul, S.; Ruchirawar, S.; Prachayasittikul, V. 8-Hydroxyquinolines: A review of their metal chelating properties and medicinal applications. Drug Des. Dev. Ther. 2013, 7, 1157–1178. [Google Scholar] [CrossRef]
- Song, Y.; Xu, H.; Chen, W.; Zhan, P.; Liu, X. 8-Hydroxyquinoline: A privileged structure with a broad-ranging pharmacological potential. MedChemCommun 2015, 6, 61–74. [Google Scholar] [CrossRef]
- Gupta, R.; Luxami, V.; Paul, K. Insights of 8-hydroxyquinolines: A novel target in medicinal chemistry. Bioorg. Chem. 2021, 108, 104633. [Google Scholar] [CrossRef]
- Gasparin, C.B.; Pilger, D.A. 8-Hydroxyquinoline, Derivatives and Metal-Complexes: A Review of Antileukemia Activities. ChemistrySelect 2023, 8, e202204219. [Google Scholar] [CrossRef]
- Savić-Gajić, I.M.; Savić, I.M. Drug design strategies with metal-hydroxyquinoline complexes. Expert Opin. Drug Discov. 2019, 15, 383–390. [Google Scholar] [CrossRef]
- Schulze, M.; Löwe, R.; Pollex, R.; Mazik, M. Structure–extractability relationships for substituted 8-hydroxyquinolines: Solvent extraction of indium ions from acidic aqueous media. Monatsh. Chem. 2019, 150, 983–990. [Google Scholar] [CrossRef]
- Fuhrmann, B.; Meier, E.; Mazik, M. Crystal structure of bis(μ2-5-nonanoylquinolin-8-olato)bis[aquadichloridoindium(III)]. Acta Cryst. 2024, E80, 1020–1023. [Google Scholar] [CrossRef]
- Huang, D.-F.; Chow, T.J.; Wu, C.-Y.; Sun, S.-S.; Tsai, S.-H.; Wen, Y.-S.; Polosan, S.; Tsuboi, T. The Preparation of (8-Hydroxyquinolinato)Bis(2-Phenylpyridyl)Iridium Complexes and Their Photophysical Properties. J. Chin. Chem. Soc. 2013, 55, 439–448. [Google Scholar] [CrossRef]
- Nagata, Y.; Chujo, Y. Synthesis of Methyl-Substituted Main-Chain-Type Organoboron Quinolate Polymers and Their Emission Color Tuning. Macromolecules 2008, 41, 2809–2813. [Google Scholar] [CrossRef]
- Deaton, J.C.; Place, D.W.; Brown, C.T.; Rajeswaran, M.; Kondakova, M.E. The blue aluminum and gallium chelates for OLEDs. Inorg. Chim. Acta 2008, 361, 1020–1035. [Google Scholar] [CrossRef]
- El Safadi, M.; Wilson, K.A.; Strudwicke, I.J.; O’Mara, M.L.; Bhadbhade, M.; Rawling, T.; McDonagh, A.M. Amphetamine-like Deferiprone and Clioquinol Derivatives as Iron Chelating Agents. Molecules 2024, 29, 4213. [Google Scholar] [CrossRef]
- Phillips, J.P.; Elbinger, R.L.; Merritt, L.L. Preparation of Some Substituted 8-Hydroxy- and 8-Methoxyquinolines. J. Am. Chem. Soc. 1949, 71, 3986–3988. [Google Scholar] [CrossRef]
- Matsugi, M.; Tabusa, F.; Minamikawa, J. Doebner–Miller synthesis in a two-phase system: Practical preparation of quinolines. Tetrahedron Lett. 2000, 41, 8523–8525. [Google Scholar] [CrossRef]
- Steiner, T.; Desiraju, G.R. The Weak Hydrogen Bond in Chemistry and Structural Biology; Oxford University Press: New York, NY, USA, 1999. [Google Scholar]
- Desiraju, G.R. C–H⋯O and other weak hydrogen bonds. From crystal engineering to virtual screening. Chem. Commun. 2005, 2995–3001. [Google Scholar] [CrossRef]
- Mazik, M.; Bläser, D.; Boese, R. Intermolecular CH⋯N/CH⋯O hydrogen bonds in the crystal structures of α,β-unsaturated ketones carrying a terminal pyridine subunit. Tetrahedron 2001, 57, 5791–5797. [Google Scholar] [CrossRef]
- Mazik, M.; Hartmann, A.; Jones, P.G. Hydrogen and Halogen Bonding in the Crystal Structure of a 1,3,5-Substituted 2,4,6-Triethylbenzene Consisting of Three Phenanthroline Units. Eur. J. Org. Chem. 2010, 2010, 458–463. [Google Scholar] [CrossRef]
- Castellano, R.K. Progress Toward Understanding the Nature and Function of C-H⋯O Interactions. Curr. Org. Chem. 2004, 8, 845–865. [Google Scholar] [CrossRef]
- Steiner, T. Unrolling the hydrogen bond properties of C–H···O interactions. Chem. Commun. 1997, 727–734. [Google Scholar] [CrossRef]
- Etter, M.C.; MacDonald, J.C.; Bernstein, J. Graph set Analysis of Hydrogen Bond Patterns in Organic Crystals. Acta Crystallogr. Sect. B 1990, 46, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, J.; Davies, R.E.; Shimoni, L.; Chang, N.L. Patterns in Hydrogen Bonding: Functionality and Graph Set Analysis in Crystals. Angew. Chem. Int. Ed. 1995, 34, 1555–1573. [Google Scholar] [CrossRef]
- Dance, I. π-π Interactions: Theory and Scope. In Encyclopedia of Supramolecular Chemistry; Atwood, J.L., Steed, J.M., Eds.; Dekker: New York, NY, USA, 2004; pp. 1076–1092. [Google Scholar]
- James, S.L. π-π Stacking as a Crystal Engineering Tool. In Encyclopedia of Supramolecular Chemistry; Atwood, J.L., Steed, J.M., Eds.; Dekker: New York, NY, USA, 2004; pp. 1093–1099. [Google Scholar]
- X-AREA, version 2.7; Software Package for Data Collection and Data Evaluation; Stoe & Cie: Darmstadt, Germany, 2002.
- X-RED, version 2.3.3; Software Program for Data Reduction and Absorption Correction; Stoe & Cie: Darmstadt, Germany, 2002.
- Sheldrick, G.M. SHELXT—Integrated Space Group and Crystal Structure Determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- XSTEP-32, Revision 1.07e; Stoe & Cie: Darmstadt, Germany, 2000.
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Farrugia, L.J. WinGX and ORTEP for Windows: An Update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Castaňeda, R.; Antal, S.A.; Draguta, S.; Timofeevaa, T.V.; Khrustalev, V.N. Crystal structure of 8-hydroxyquinoline: A new monoclinic polymorph. Acta Cryst. 2014, E70, o924–o925. [Google Scholar] [CrossRef]





| Atoms | Distance (Å) | Angle (°) | Slippage (Å) | |
|---|---|---|---|---|
| D-H∙∙∙A π∙∙∙π | D∙∙∙A Cg∙∙∙Cg | H∙∙∙A | D-H∙∙∙A | |
| 1 | ||||
| O(1)-H(10)···N(1) | 2.735(2) | 2.25(2) | 114(2) | |
| O(1)-H(10)···N(1) | 2.869(3) | 2.17(2) | 135(2) | |
| C(1)-H(1)···O(1) | 3.002(3) | 2.45 | 117 | |
| Cg(A)···Cg(A) a | 3.556(3) | 1.063 | ||
| Cg(B)···Cg(B) a | 3.631(2) | 1.234 | ||
| Cg(A)···Cg(B) a | 3.719(3) | 1.504 | ||
| 2 | ||||
| O(1)-H(10)···N(1) | 2.740(2) | 2.27(2) | 114(2) | |
| O(1)-H(10)···N(1) | 2.859(2) | 2.18(2) | 134(2) | |
| C(1)-H(1)···O(1) | 3.033(2) | 2.53 | 113 | |
| Cg(A)···Cg(A) a | 3.662(2) | 1.167 | ||
| Cg(B)···Cg(B) a | 3.728(2) | 1.345 | ||
| Cg(A)···Cg(B) a | 3.753(2) | 1.491 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Braun, S.; Schwarzer, A.; Mazik, M. Crystal Structures of Two 4-Alkyl-8-hydroxyquinolines. Molbank 2026, 2026, M2176. https://doi.org/10.3390/M2176
Braun S, Schwarzer A, Mazik M. Crystal Structures of Two 4-Alkyl-8-hydroxyquinolines. Molbank. 2026; 2026(3):M2176. https://doi.org/10.3390/M2176
Chicago/Turabian StyleBraun, Sara, Anke Schwarzer, and Monika Mazik. 2026. "Crystal Structures of Two 4-Alkyl-8-hydroxyquinolines" Molbank 2026, no. 3: M2176. https://doi.org/10.3390/M2176
APA StyleBraun, S., Schwarzer, A., & Mazik, M. (2026). Crystal Structures of Two 4-Alkyl-8-hydroxyquinolines. Molbank, 2026(3), M2176. https://doi.org/10.3390/M2176