(E)-5-(3-Oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)benzo[d]oxazol-2(3H)-one
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Synthesis of (E)-5-(3-Oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)benzo[d]oxazol-2(3H)-one (3)
3.2.1. Base-Catalyzed Aldol Condensation
3.2.2. Acid-Catalyzed Aldol Condensation
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jasim, H.A.; Nahar, L.; Jasim, M.A.; Moore, S.A.; Ritchie, K.J.; Sarker, S.D. Chalcones: Synthetic Chemistry Follows Where Nature Leads. Biomolecules 2021, 11, 1203. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, D.K.; Bharti, S.K.; Asati, V. Anti-cancer chalcones: Structural and molecular target perspectives. Eur. J. Med. Chem. 2015, 98, 69–114. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, C.; Zhang, W.; Sheng, C.; Zhang, W.; Xing, C.; Miao, Z. Chalcone: A Privileged Structure in Medicinal Chemistry. Chem. Rev. 2017, 117, 7762–7810. [Google Scholar] [CrossRef] [PubMed]
- Constantinescu, T.; Mihis, A.G. Two Important Anticancer Mechanisms of Natural and Synthetic Chalcones. Int. J. Mol. Sci. 2022, 23, 11595. [Google Scholar] [CrossRef] [PubMed]
- Bilginer, S.; Gul, H.I.; Erdal, F.S.; Sakagami, H.; Gulcin, I. New halogenated chalcones with cytotoxic and carbonic anhydrase inhibitory properties: 6-(3-Halogenated phenyl-2-propen-1-oyl)-2(3H)-benzoxazolones. Arch. Pharm. 2020, 353, e1900384. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, Y.; Momekov, G.; Petrov, O.; Karaivanova, M.; Kalcheva, V. Cytotoxic Mannich bases of 6-(3-aryl-2-propenoyl)-2(3H)-benzoxazolones. Eur. J. Med. Chem. 2007, 42, 1382–1387. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, D.K.; Bharti, S.K.; Asati, V. Chalcone Derivatives: Anti-inflammatory Potential and Molecular Targets Perspectives. Curr. Top. Med. Chem. 2017, 17, 3146–3169. [Google Scholar] [CrossRef] [PubMed]
- Lagu, S.B.; Yejella, R.P.; Bhandare, R.R.; Shaik, A.B. Design, Synthesis, and Antibacterial and Antifungal Activities of Novel Trifluoromethyl and Trifluoromethoxy Substituted Chalcone Derivatives. Pharmaceuticals 2020, 13, 375. [Google Scholar] [CrossRef] [PubMed]
- Henry, E.J.; Bird, S.J.; Gowland, P.; Collins, M.; Cassella, J.P. Ferrocenyl chalcone derivatives as possible antimicrobial agents. J. Antibiot. 2020, 73, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Okolo, E.N.; Ugwu, D.I.; Ezema, B.E.; Ndefo, J.C.; Eze, F.U.; Ezema, C.G.; Ezugwu, J.A.; Ujam, O.T. New chalcone derivatives as potential antimicrobial and antioxidant agent. Sci. Rep. 2021, 11, 21781. [Google Scholar] [CrossRef] [PubMed]
- Kudličková, Z.; Michalková, R.; Salayová, A.; Ksiažek, M.; Vilková, M.; Bekešová, S.; Mojžiš, J. Design, Synthesis, and Evaluation of Novel Indole Hybrid Chalcones and Their Antiproliferative and Antioxidant Activity. Molecules 2023, 28, 6583. [Google Scholar] [CrossRef] [PubMed]
- Pati, H.N.; Das, U.; Sharma, R.K.; Dimmock, J.R. Cytotoxic Thiol Alkylators. Mini-Rev. Med. Chem. 2007, 7, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Petrov, O.I.; Ivanova, Y.B.; Gerova, M.S.; Momekov, G.T. Synthesis and Cytotoxicity of New Mannich Bases of 6-[3-(3,4,5-Trimetoxyphenyl)-2-propenoyl]-2(3H)-Benzoxazolone. Lett. Drug Des. Discov. 2020, 17, 512–517. [Google Scholar] [CrossRef]
- Ivanova, Y.; Momekov, G.; Petrov, O. Synthesis of novel substituted 1,3-diarylpropenone derivatives and their in vitro cytotoxic activity. Lett. Drug Des. Discov. 2009, 6, 353–357. [Google Scholar] [CrossRef]
- Ivanova, Y.B.; Gerova, M.S.; Momekov, G.T.; Petrov, O.I. Synthetic chalcones of 2(3H)-benzothiazolone with potential cytotoxic activity. C. R. Acad. Bulg. Sci. 2007, 60, 641–644. [Google Scholar]
- Ivanova, Y.B.; Momekov, G.T.; Petrov, O.I. New heterocyclic chalcones. Part 6. Synthesis and cytotoxic activities of 5- or 6-(3-aryl-2-propenoyl)-2(3H)-benzoxazolones. Heterocycl. Commun. 2013, 19, 23–28. [Google Scholar] [CrossRef]
- Blum, G.; Gazit, A.; Levitzki, A. Development of New Insulin-like Growth Factor-1 Receptor Kinase Inhibitors Using Catechol Mimics. J. Biol. Chem. 2003, 278, 40442–40454. [Google Scholar] [CrossRef] [PubMed]
- Petrov, O.; Ivanova, Y.; Gerova, M. SOCl2/EtOH: Catalytic system for synthesis of chalcones. Catal. Commun. 2008, 9, 315–316. [Google Scholar] [CrossRef]
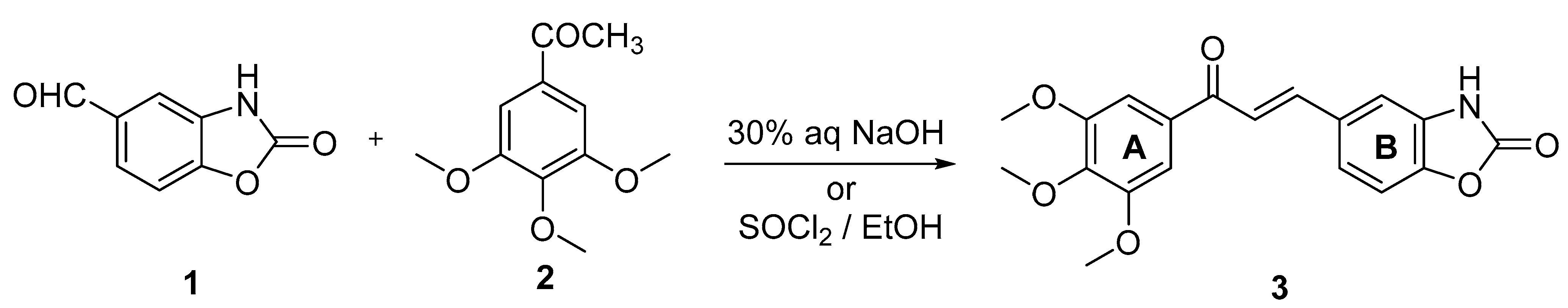
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivanova, Y.B.; Svetoslavov, F.E.; Petrov, O.I. (E)-5-(3-Oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)benzo[d]oxazol-2(3H)-one. Molbank 2024, 2024, M1866. https://doi.org/10.3390/M1866
Ivanova YB, Svetoslavov FE, Petrov OI. (E)-5-(3-Oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)benzo[d]oxazol-2(3H)-one. Molbank. 2024; 2024(3):M1866. https://doi.org/10.3390/M1866
Chicago/Turabian StyleIvanova, Yordanka B., Filip E. Svetoslavov, and Ognyan I. Petrov. 2024. "(E)-5-(3-Oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)benzo[d]oxazol-2(3H)-one" Molbank 2024, no. 3: M1866. https://doi.org/10.3390/M1866
APA StyleIvanova, Y. B., Svetoslavov, F. E., & Petrov, O. I. (2024). (E)-5-(3-Oxo-3-(3,4,5-trimethoxyphenyl)prop-1-en-1-yl)benzo[d]oxazol-2(3H)-one. Molbank, 2024(3), M1866. https://doi.org/10.3390/M1866






