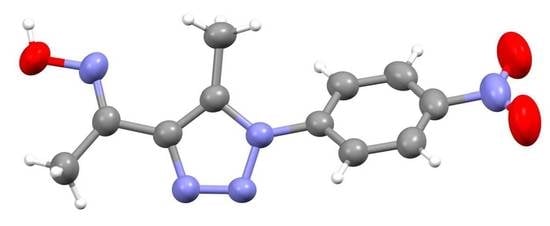
(E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethan-1-one Oxime
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis
2.2. NMR Spectroscopy
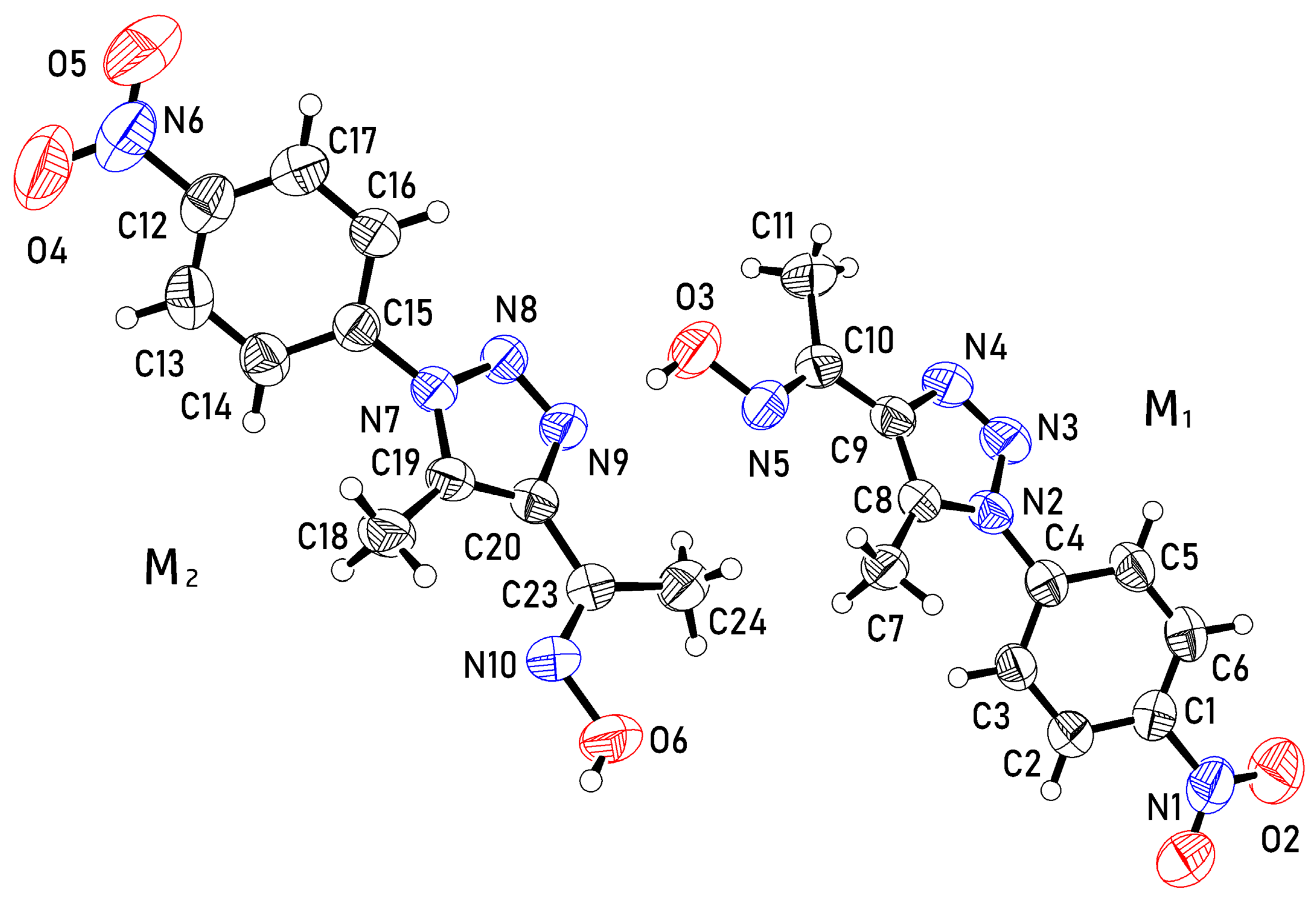
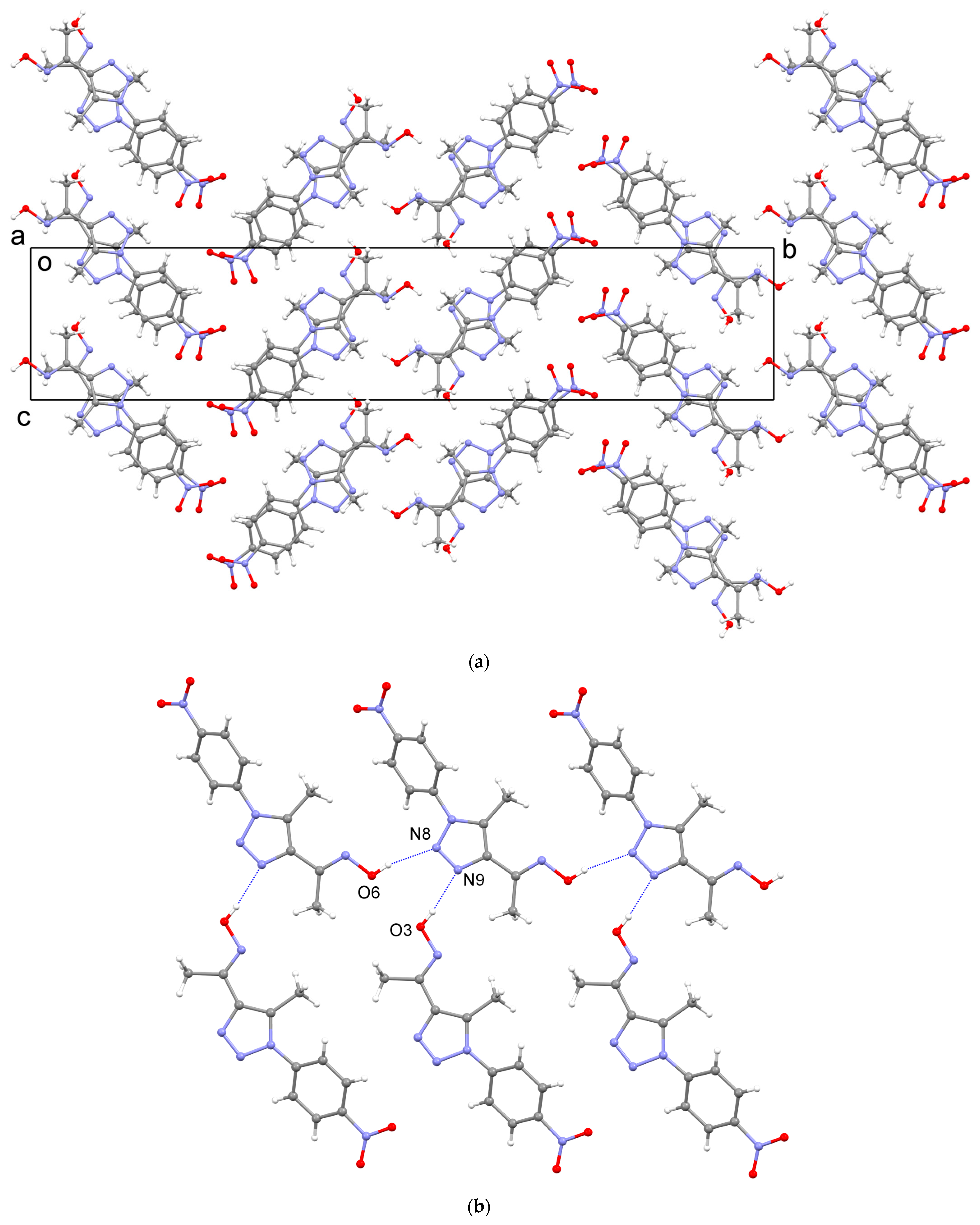
2.3. X-ray Crystal Structure
3. Materials and Methods
3.1. General
3.2. Synthesis of 2
3.3. Crystal Structure Determination
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Zohreh Mirjafary, Z.; Abdoli, M.; Saeidian, H.; Kakanejadifard, A.; Farniad, S.M.F. Review of the synthesis of acyclic and cyclic oxime ethers. RSC Adv. 2016, 6, 17740–17758. [Google Scholar] [CrossRef]
- Rykaczewski, K.A.; Wearing, E.R.; Blackmun, D.E.; Schindler, C.S. Reactivity of oximes for diverse methodologies and synthetic applications. Nat. Synth. 2022, 1, 24–36. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Plotnikov, M.B.; Khlebnikov, A.I.; Plotnikova, T.M.; Quinn, M.T. Oximes: Novel therapeutics with anticancer and anti-Inflammatory potential. Biomolecules 2021, 11, 777. [Google Scholar] [CrossRef] [PubMed]
- Walton, J.C. Functionalised oximes: Emergent precursors for carbon-, nitrogen- and oxygen-centred radicals. Molecules 2016, 21, 63. [Google Scholar] [CrossRef]
- Dhuguru, J.; Zviagin, E.; Skouta, R. FDA-Approved oximes and their significance in medicinal chemistry. Pharmaceuticals 2022, 15, 66. [Google Scholar] [CrossRef]
- Canario, C.; Silvestre, S.; Falcao, A.; Alves, G. Steroidal oximes: Useful compounds with antitumor activities. Curr. Med. Chem. 2018, 25, 660–686. [Google Scholar] [CrossRef]
- Sorensen, M.; Neilson, E.H.J.; Moller, B.L. Oximes: Unrecognized chameleons in general and specialized plant metabolism. Mol. Plant 2018, 11, 95–117. [Google Scholar] [CrossRef]
- Surowiak, A.K.; Lochyński, S.; Strub, D.J. Unsubstituted oximes as potential therapeutic agents. Symmetry 2020, 12, 575. [Google Scholar] [CrossRef]
- Westhuyzen, R.; Mabhula, A.; Njaria, P.M.; Müller, R.; Muhunga, D.N.; Taylor, D.; Lawrence, N.; Njoroge, M.; Brunschwig, C.; Moosa, A.; et al. Benzoheterocyclic oxime carbamates active against mycobacterium tuberculosis: Synthesis, structure–activity relationship, metabolism, and biology triaging. J. Med. Chem. 2021, 64, 9444–19457. [Google Scholar] [CrossRef]
- Liu, C.; Tang, X.; Zhang, W.; Li, G.; Chen, Y.; Guo, A.; Hu, C. 6-bromoindirubin-3′-oxime suppresses LPS-induced inflammation via inhibition of the TLR4/NF-κB and TLR4/MAPK signaling pathways. Inflammation 2019, 42, 2192–2204. [Google Scholar] [CrossRef]
- Hwang, T.L.; Wang, W.H.; Wang, T.Y.; Yu, H.P.; Hsieh, P.W. Synthesis and pharmacological characterization of 2-aminobenzaldehyde oxime analogs as dual inhibitors of neutrophil elastase and proteinase 3. Bioorg. Med. Chem. 2015, 23, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Zeferino-Diaz, R.; Olivera-Castillo, L.; Davalos, A.; Grant, G.; Kantun-Moreno, N.; Rodriguez-Canul, R.; Bernes, S.; SandovalRamirez, J.; Fernandez-Herrera, M.A. 22-Oxocholestane oximes as potential anti-inflammatory drug candidates. Eur. J. Med. Chem. 2019, 168, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Xu, N.; Klamer, G.; Ko, K.H.; Khoo, M.; Ma, D.; Moore, J.; O’Brien, T.A.; Dolnikov, A. Small-molecule inhibitor of glycogen synthase kinase 3β 6-bromoindirubin-3-oxime inhibits hematopoietic regeneration in stem cell recipient mice. Stem. Cells Dev. 2015, 24, 724–736. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Sha, S.; Wang, K.; Zhang, Y.H.; Liu, Y.D.; Ju, G.D.; Wang, B.; Zhu, H.L. Discovery of chromeno [4,3-c]pyrazol-4(2H)-one containing carbonyl or oxime derivatives as potential, selective inhibitors PI3Kα. Chem. Pharm. Bull. 2016, 64, 1576–1581. [Google Scholar] [CrossRef]
- Begum, J.; Skamnaki, V.T.; Moffatt, C.; Bischler, N.; Sarrou, J.; Skaltsounis, A.L.; Leonidas, D.D.; Oikonomakos, N.G.; Hayes, J.M. An evaluation of indirubin analogues as phosphorylase kinase inhibitors. J. Mol. Graph. Model. 2015, 61, 231–242. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Khlebnikov, A.I.; Potapov, A.S.; Kovrizhina, A.R.; Matveevskaya, V.V.; Belyanin, M.L.; Atochin, D.N.; Zanoza, S.O.; Gaidarzhy, N.M.; Lyakhov, S.A.; et al. Synthesis, biological evaluation, and molecular modeling of 11H-indeno [1,2-b]quinoxalin-11-one derivatives and tryptanthrin-6-oxime as c-Jun N-terminal kinase inhibitors. Eur. J. Med. Chem. 2019, 161, 179–191. [Google Scholar] [CrossRef]
- Kerru, N.; Gummidi, L.; Maddila, S.; Gangu, K.K.; Jonnalagadda, S.B. A Review on recent advances in nitrogen-containing molecules and their biological applications. Molecules 2020, 25, 1909. [Google Scholar] [CrossRef]
- Bozorov, K.; Zhao, J.; Aisa, H.A. 1,2,3-Triazole-containing hybrids as leads in medicinal chemistry: A recent overview. Bioorg. Med. Chem. 2019, 27, 3511–3531. [Google Scholar] [CrossRef]
- Mohamed, H.A.; Bekheit, M.S.; Ewies, E.F.; Awad, H.M.; Betz, R.; Hosten, E.C.; Abdel-Wahab, B.F. Design of new hybrids indole/phthalimide/oxadiazole-1,2,3 triazole agents and their anticancer properties. J. Mol. Struct. 2023, 1274, 134415. [Google Scholar] [CrossRef]
- Matin, M.M.; Matin, P.; Rahman, M.R.; Ben Hadda, T.; Almalki, F.A.; Mahmud, S.; Ghoneim, M.M.; Alruwaily, M.; Alshehri, S. Triazoles and their derivatives: Chemistry, synthesis, and therapeutic applications. Front. Mol. Biosci. 2022, 25, 864286. [Google Scholar] [CrossRef]
- Abdulghani, S.M.; Al-Rawi, M.S.; Tomma, J.H. Synthesis of new 1,2,4-triazole derivatives with expected biological activities. Chem. Methodol. 2022, 6, 59–66. [Google Scholar] [CrossRef]
- Aouad, M.R.; Khan, D.J.O.; Said, M.A.; Al-Kaff, N.S.; Rezki, N.; Ali, A.A.; Bouqellah, N.; Hagar, M. Novel 1,2,3-triazole derivatives as potential inhibitors against COVID-19 main protease: Synthesis, characterization, molecular docking and DFT studies. ChemistrySelect 2021, 6, 3468–3486. [Google Scholar] [CrossRef]
- Chandrashekhar, M.; Nayak, V.L.; Ramakrishna, S.; Mallavadhani, U.V. Novel triazole hybrids of myrrhanone C, a natural polypodane triterpene: Synthesis, cytotoxic activity and cell based studies. Eur. J. Med. Chem. 2016, 114, 293–307. [Google Scholar] [CrossRef]
- Madasu, C.; Karri, S.; Sangaraju, R.; Sistla, R.; Uppuluri, M.V. Synthesis and biological evaluation of some novel 1,2,3-triazole hybrids of myrrhanone B isolated from Commiphora mukul gum resin: Identification of potent antiproliferative leads active against prostate cancer cells (PC-3). Eur. J. Med. Chem. 2020, 188, 111974. [Google Scholar] [CrossRef]
- Bakherad, M.; Ali Keivanloo, A.; Amin, A.A.; Farkhondeh, A. Synthesis of 1,2,3-triazole-linked benzimidazole through a copper-catalyzed click reaction. Heterocycl. Commun. 2019, 25, 122–129. [Google Scholar] [CrossRef]
- Agalave, S.G.; Maujan, S.R.; Pore, V.S. Click chemistry: 1,2,3-triazoles as pharmacophores. Chem. Asian J. 2011, 6, 2696–2718. [Google Scholar] [CrossRef]
- Pokhodylo, N.T.; Matiychuk, V.S.; Obushak, N.B. Synthesis of 1H-1,2,3-triazole derivatives by the cyclization of aryl azides with 2-benzothiazolylacetonone, 1,3-benzo-thiazol-2-ylacetonitrile, and (4-aryl-1,3-thiazol-2-yl)acetonitriles. Chem. Heterocycl. Comp. 2009, 45, 483–488. [Google Scholar] [CrossRef]
- Lu, H.; Li, Z. Synthesis of 1,2,3-triazolyl-based ketoximes using calcium carbide as an acetylene source. Eur. J. Org. Chem. 2020, 2020, 845–851. [Google Scholar] [CrossRef]
- Ashok, D.; Reddy, M.R.; Dharavath, R.; Ramakrishna, K.; Nagaraju, N.; Sarasija, M. Microwave-assisted synthesis of some new 1,2,3-triazole derivatives and their antimicrobial activity. J. Chem. Sci. 2020, 132, 47. [Google Scholar] [CrossRef]
- Kariuki, B.M.; Abdel-Wahab, B.F.; Mohamed, H.A.; Bekheit, M.S.; El-Hiti, G.A. Synthesis and characterization of novel 2-(1,2,3-triazol-4-yl)-4,5-dihydro-1H-pyrazol-1-yl)thiazoles and 2-(4,5-dihydro-1H-pyrazol-1-yl)-4-(1H-1,2,3-triazol-4-yl)thiazoles. Molecules 2022, 27, 8904. [Google Scholar] [CrossRef]
- Kariuki, B.M.; Abdel-Wahab, B.F.; Bekheit, M.S.; El-Hiti, G.A. (Z)-2-(1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethylidene)-N-phenylhydrazine-1-carbothioamide. Molbank 2022, 2022, M1462. [Google Scholar] [CrossRef]
- Kariuki, B.M.; Abdel-Wahab, B.F.; Mohamed, H.A.; El-Hiti, G.A. (E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)-3-(naphthalen-2-yl)prop-2-en-1-one. Molbank 2022, 2022, M1464. [Google Scholar] [CrossRef]
- Mohamed, H.A.; Khidre, R.E.; Kariuki, B.M.; El-Hiti, G.A. Synthesis of novel heterocycles using 1,2,3-triazole-4-carbohydrazides as precursors. J. Heterocycl. Chem. 2020, 57, 1055–1062. [Google Scholar] [CrossRef]
- Gökce, H.; Şen, F.; Sert, Y.; Abdel-Wahab, B.F.; Kariuki, B.M.; El-Hiti, G.A. Quantum computational investigation of (E)-1-(4-methoxyphenyl)-5-methyl-N’-(3-phenoxybenzylidene)-1H-1,2,3-triazole-4-carbohydrazide. Molecules 2022, 27, 2193. [Google Scholar] [CrossRef]
- Abdel-Wahab, B.F.; Mohamed, H.A.; Farahat, A.A.; Kariuki, B.M.; El-Hiti, G.A. Reactivity of 4-bromoacetyl-1,2,3-triazoles towards amines and phenols: Synthesis and antimicrobial activity of novel heterocycles. Heterocycles 2022, 104, 1601–1613. [Google Scholar] [CrossRef]
- Kamalraj, V.R.; Senthil, S.; Kannan, P. One-pot synthesis and the fluorescent behavior of 4-acetyl-5-methyl-1,2,3-triazole regioisomers. J. Mol. Struct. 2008, 892, 210–215. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdel-Wahab, B.F.; Farahat, A.A.; Kariuki, B.M.; El-Hiti, G.A. (E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethan-1-one Oxime. Molbank 2023, 2023, M1593. https://doi.org/10.3390/M1593
Abdel-Wahab BF, Farahat AA, Kariuki BM, El-Hiti GA. (E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethan-1-one Oxime. Molbank. 2023; 2023(1):M1593. https://doi.org/10.3390/M1593
Chicago/Turabian StyleAbdel-Wahab, Bakr F., Abdelbasset A. Farahat, Benson M. Kariuki, and Gamal A. El-Hiti. 2023. "(E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethan-1-one Oxime" Molbank 2023, no. 1: M1593. https://doi.org/10.3390/M1593
APA StyleAbdel-Wahab, B. F., Farahat, A. A., Kariuki, B. M., & El-Hiti, G. A. (2023). (E)-1-(5-Methyl-1-(4-nitrophenyl)-1H-1,2,3-triazol-4-yl)ethan-1-one Oxime. Molbank, 2023(1), M1593. https://doi.org/10.3390/M1593