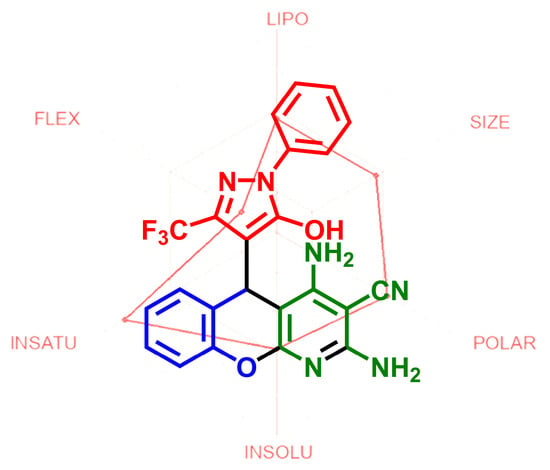
2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile
Abstract
1. Introduction
2. Results and Discussion
2.1. Multicomponent Synthesis of 2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile 4
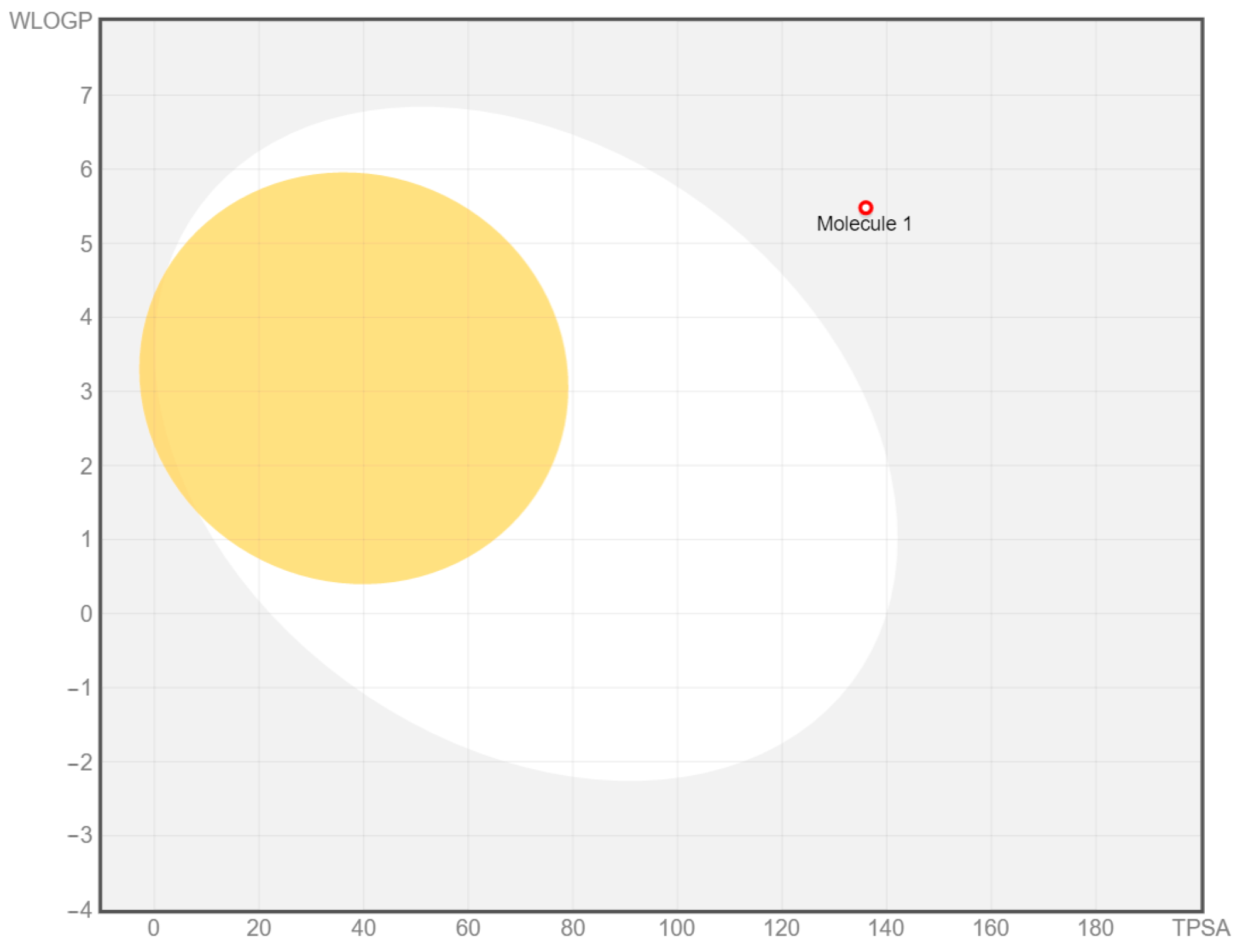
2.2. ADME Prediction
| Parameter | Value |
|---|---|
| Fraction Csp3 | 0.09 |
| Num. rotatable bonds | 3 |
| Topological polar surface area | 136.00 Å2 |
| Consensus Log Po/w (Lipophilicity) | 3.55 |
| Log S (ESOL) [29] | −5.72 |
| Water solubility | 8.88 × 10−4 mg/mL; 1.91 × 10−6 mol/L |
| Class | Moderately soluble |
| Gastrointestinal absorption | Low |
| BBB permeant | No |
| P-gp substrate | No |
| Log Kp (skin permeation) | −6.09 cm/s |
| Bioavailability score | 0.55 |
3. Materials and Methods
3.1. General Methods
3.2. Multicomponent Synthesis of 2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile 4
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pantaleão, S.Q.; Fernandes, P.O.; Gonçalves, J.E.; Maltarollo, V.G.; Honorio, K.M. Recent Advances in the Prediction of Pharmacokinetics Properties in Drug Design Studies: A Review. ChemMedChem 2022, 17, e202100542. [Google Scholar] [CrossRef] [PubMed]
- Hay, M.; Thomas, D.W.; Craighead, J.L.; Economides, C.; Rosenthal, J. Clinical development success rates for investigational drugs. Nat. Biotechnol. 2014, 32, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xing, J.; Xu, Y.; Zhou, N.; Peng, J.; Xiong, Z.; Liu, X.; Luo, X.; Luo, C.; Chen, K.; et al. In silico ADME/T modelling for rational drug design. Q. Rev. Biophys. 2015, 48, 488–515. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Dietrich, J. Privileged scaffolds in lead generation. Expert Opin. Drug Discov. 2015, 10, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Elinson, M.N.; Ryzhkova, Y.E.; Ryzhkov, F.V. Multicomponent design of chromeno[2,3-b]pyridine systems. Russ. Chem. Rev. 2021, 90, 94–115. [Google Scholar] [CrossRef]
- Makino, H.; Saijo, T.; Ashida, Y.; Kuriki, H.; Maki, Y. Mechanism of Action of an Antiallergic Agent, Amlexanox (AA-673), in Inhibiting Histamine Release from Mast Cells. Int. Arch. Allergy Immunol. 1987, 82, 66–71. [Google Scholar] [CrossRef]
- Akyol-Salman, İ.; Leçe-Sertöz, D.; Baykal, O. Topical Pranoprofen 0.1% Is As Effective Anti-Inflammatory and Analgesic Agent as Diclofenac Sodium 0.1% After Strabismus Surgery. J. Ocul. Pharmacol. Ther. 2007, 23, 280–283. [Google Scholar] [CrossRef]
- Penning, T.D.; Talley, J.J.; Bertenshaw, S.R.; Carter, J.S.; Collins, P.W.; Docter, S.; Graneto, M.J.; Lee, L.F.; Malecha, J.W.; Miyashiro, J.M.; et al. Synthesis and biological evaluation of the 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors: Identification of 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benze nesulfonamide (SC-58635, celecoxib). J. Med. Chem. 1997, 40, 1347–1365. [Google Scholar] [CrossRef]
- Maxwell, B.D. The radiolabeled syntheses of JV 485, a herbicide candidate for winter wheat. J. Label. Compd. Radiopharm. 2000, 43, 645–654. [Google Scholar] [CrossRef]
- Elinson, M.N.; Vereshchagin, A.N.; Anisina, Y.E.; Fakhrutdinov, A.N.; Goloveshkin, A.S.; Egorov, M.P. A facile and efficient multicomponent approach to 5-[5-hydroxy-3-(trifluoromethyl)-1H-pyrazol-4-yl]-5H-chromeno[2,3-b]pyridines. J. Fluor. Chem. 2018, 213, 31–36. [Google Scholar] [CrossRef]
- Ryzhkova, Y.E.; Elinson, M.N.; Maslov, O.I.; Fakhrutdinov, A.N. Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids in DMSO. Molecules 2021, 26, 6839. [Google Scholar] [CrossRef] [PubMed]
- Ryzhkova, Y.E.; Maslov, O.I.; Elinson, M.N. Dimethyl 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonate. Molbank 2022, 2022, M1308. [Google Scholar] [CrossRef]
- Ryzhkova, Y.E.; Ryzhkov, F.V.; Maslov, O.I.; Elinson, M.N. 2,4-Diamino-5-(nitromethyl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile. Molbank 2022, 2022, M1365. [Google Scholar] [CrossRef]
- Domling, A.; Wang, W.; Wang, K. Chemistry and biology of multicomponent reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef] [PubMed]
- Ryzhkova, Y.E.; Ryzhkov, F.V.; Elinson, M.N.; Maslov, O.I.; Fakhrutdinov, A.N. One-Pot Solvent-Involved Synthesis of 5-O-Substituted 5H-Chromeno[2,3-b]pyridines. Molecules 2023, 28, 64. [Google Scholar] [CrossRef]
- Elinson, M.N.; Vereshchagin, A.N.; Anisina, Y.E.; Fakhrutdinov, A.N.; Goloveshkin, A.S.; Egorov, M.P. Pot-, Atom- and Step-Economic (PASE) Multicomponent approach to the 5-(Dialkylphosphonate)-Substituted 2,4-Diamino-5H-chromeno-[2,3-b]pyridine scaffold. Eur. J. Org. Chem. 2019, 2019, 4171–4178. [Google Scholar] [CrossRef]
- Ryzhkov, F.V.; Ryzhkova, Y.E.; Elinson, M.N.; Vorobyev, S.V.; Fakhrutdinov, A.N.; Vereshchagin, A.N.; Egorov, M.P. Catalyst-Solvent System for PASE Approach to Hydroxyquinolinone-Substituted Chromeno[2,3-b]pyridines Its Quantum Chemical Study and Investigation of Reaction Mechanism. Molecules 2020, 25, 2573. [Google Scholar] [CrossRef]
- Patai, S.; Israeli, Y. 411. The kinetics and mechanisms of carbonyl–methylene condensations. Part VII. The reaction of malononitrile with aromatic aldehydes in ethanol. J. Chem. Soc. 1960, 2025–2030. [Google Scholar] [CrossRef]
- Prediction of ADME Parameters, Pharmacokinetic Properties, Druglike Nature and Medicinal Chemistry Friendliness of One or Multiple Small Molecules to Support Drug Discovery. Available online: http://www.swissadme.ch (accessed on 31 October 2022).
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- Ritchie, T.J.; Ertl, P.; Lewis, R. The graphical representation of ADME-related molecule properties for medicinal chemists. Drug Discov. Today 2011, 16, 65–72. [Google Scholar] [CrossRef]
- Lovering, F.; Bikker, J.; Humblet, C. Escape from Flatland: Increasing Saturation as an Approach to Improving Clinical Success. J. Med. Chem. 2009, 52, 6752–6756. [Google Scholar] [CrossRef] [PubMed]
- Daina, A.; Zoete, V. A BOILED-Egg To Predict Gastrointestinal Absorption and Brain Penetration of Small Molecules. ChemMedChem 2016, 11, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Ambudkar, S.; Kimchi-Sarfaty, C.; Sauna, Z.; Gottesman, M.M. P-glycoprotein: From genomics to mechanism. Oncogene 2003, 22, 7468–7485. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Ghose, A.K.; Viswanadhan, V.N.; Wendoloski, J.J. A Knowledge-Based Approach in Designing Combinatorial or Medicinal Chemistry Libraries for Drug Discovery. 1. A Qualitative and Quantitative Characterization of Known Drug Databases. J. Comb. Chem. 1999, 1, 55–68. [Google Scholar] [CrossRef]
- Veber, D.F.; Johnson, S.R.; Cheng, H.-Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef]
- Muegge, I.; Heald, S.L.; Brittelli, D. Simple selection criteria for drug-like chemical matter. J. Med. Chem. 2001, 44, 12, 1841–1846. [Google Scholar] [CrossRef]
- Delaney, J.S. ESOL: Estimating aqueous solubility directly from molecular structure. J. Chem. Inf. Comput. Sci. 2004, 44, 1000–1005. [Google Scholar] [CrossRef]
- Mittelbach, M. An improved and facile synthesis of 2-amino-1,1,3-tricyanopropene. Monatsh. Chem. 1985, 116, 689–691. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, S.; Zhang, K.; Chen, J.; Deng, H.; Shao, M.; Zhang, H.; Cao, W. An efficient and highly stereoselective synthesis of novel trifluoromethylated trans-dihydrofuro[2,3-c]pyrazoles using arsonium ylides. Tetrahedron 2012, 68, 2121–2127. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryzhkova, Y.E.; Ryzhkov, F.V.; Maslov, O.I.; Elinson, M.N. 2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile. Molbank 2023, 2023, M1541. https://doi.org/10.3390/M1541
Ryzhkova YE, Ryzhkov FV, Maslov OI, Elinson MN. 2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile. Molbank. 2023; 2023(1):M1541. https://doi.org/10.3390/M1541
Chicago/Turabian StyleRyzhkova, Yuliya E., Fedor V. Ryzhkov, Oleg I. Maslov, and Michail N. Elinson. 2023. "2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile" Molbank 2023, no. 1: M1541. https://doi.org/10.3390/M1541
APA StyleRyzhkova, Y. E., Ryzhkov, F. V., Maslov, O. I., & Elinson, M. N. (2023). 2,4-Diamino-5-(5-hydroxy-1-phenyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitrile. Molbank, 2023(1), M1541. https://doi.org/10.3390/M1541