Hybrid Pyridine Bis-Anthracene-Imidazolium Salt: NMR Studies on Zn-Acetate Complexation
Abstract
1. Introduction
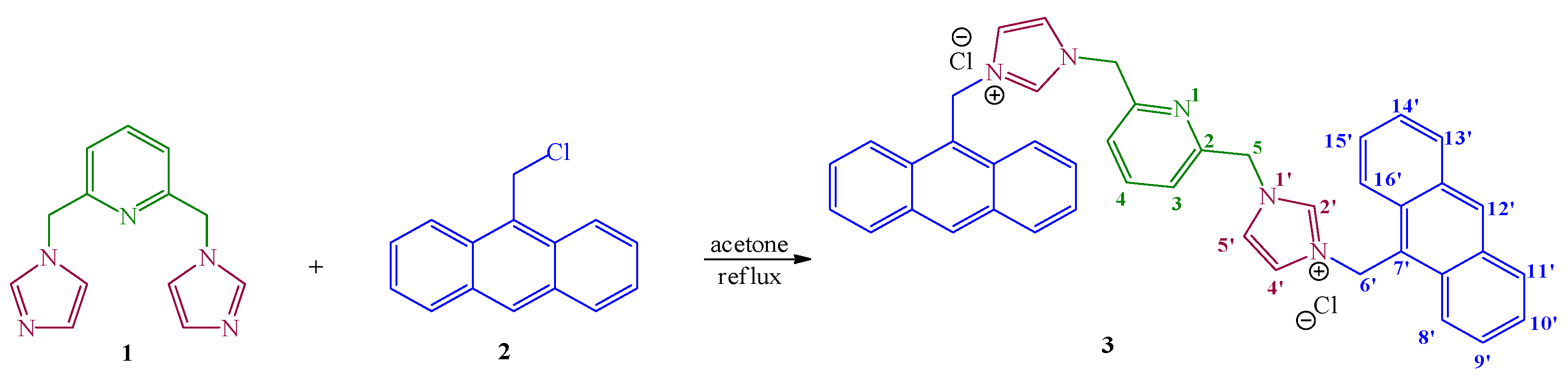
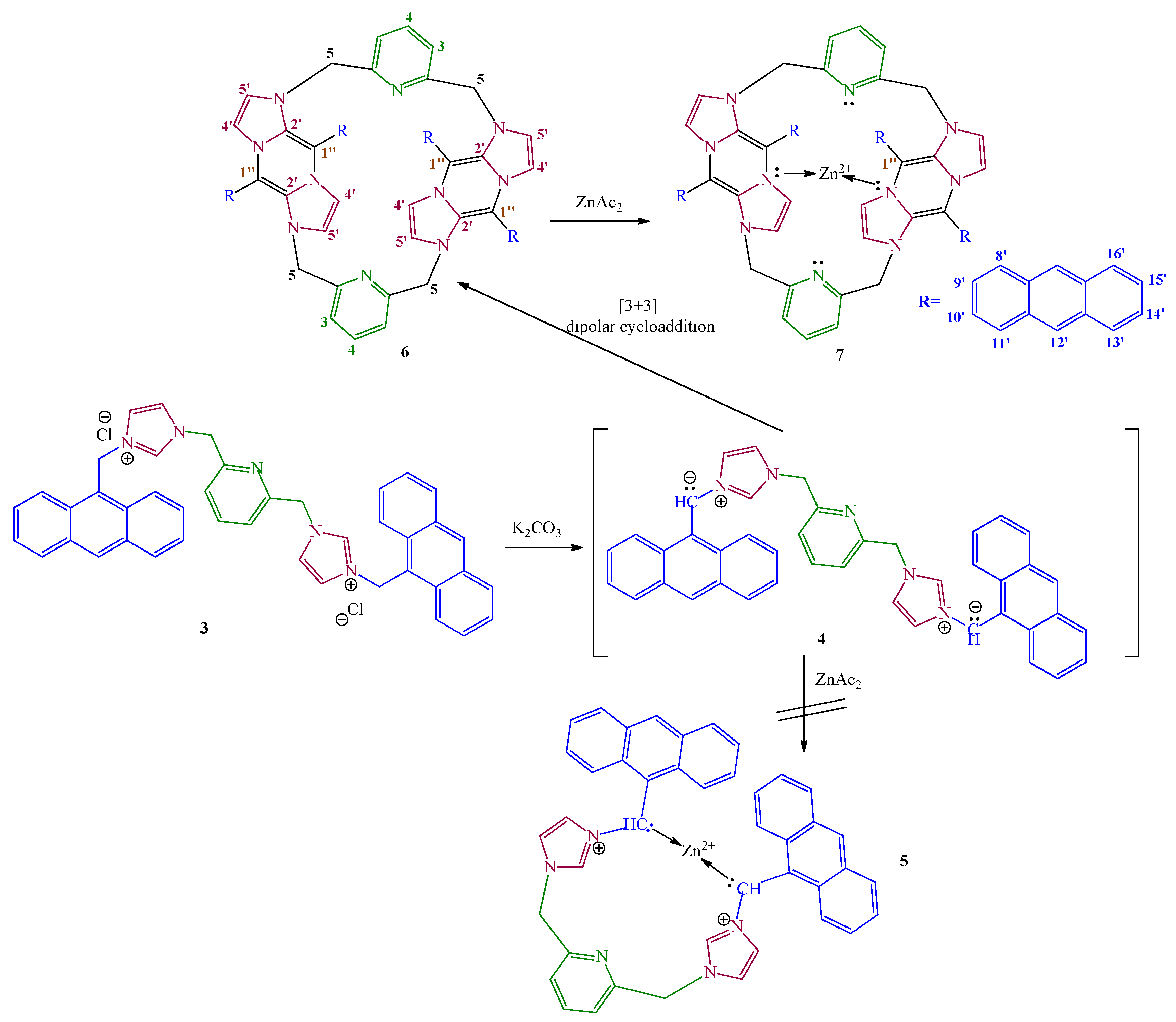
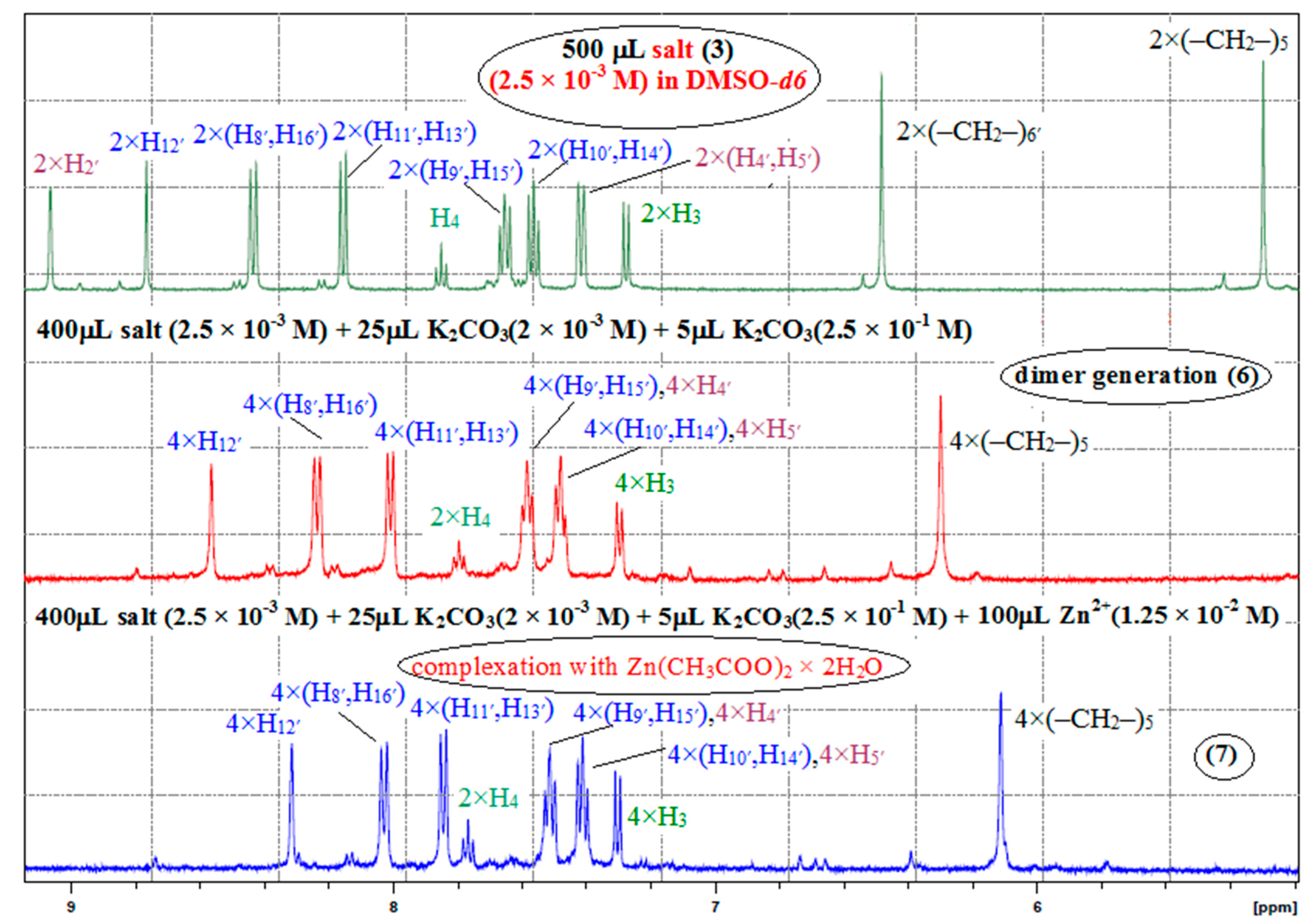
2. Results and Discussion
3. Materials and Methods
3.1. Instrumentation
3.2. General Procedure for Synthesis of Hybrid Quaternary Salt 3
3.3. General Procedure for NMR Studies
3.3.1. Dimer Generation 6
3.3.2. Dimer Complex with Zn2+ 7
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tolpygin, I.E.; Revinskii, Y.V.; Starikov, A.G.; Dubonosov, A.D.; Bren, V.A.; Minkin, V.I. Effective pH sensors based on 1-(anthracen-9-ylmethyl)-1H-benzimidazol-2-amine. Chem. Heterocycl. Compd. 2012, 47, 1230–1236. [Google Scholar] [CrossRef]
- Ciobanu, C.I.; Antoci, V.; Mantu, D.; Mangalagiu, I.I. One Pot Synthesis of Cyclophane with Imidazolium Skeleton. An Improved Method. Rev. Chim. 2015, 66, 497–498. [Google Scholar]
- Quang, D.T.; Kim, J.S. Calixarene-Derived Fluorescent Probes. Chem. Rev. 2007, 107, 3780–3799. [Google Scholar]
- Ghosh, K.; Sen, T. Anthracene coupled adenine for the selective sensing of copper ions. Beilstein J. Org. Chem. 2010, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sabater, P.; Zapata, F.; Caballero, A.; Fernández, I.; Ramirez de Arellano, C.; Molina, P. 2,4,5-Trimethylimidazolium Scaffold for Anion Recognition Receptors Acting Through Charge-Assisted Aliphatic and Aromatic C–H Interactions. J. Org. Chem. 2016, 81, 3790–3798. [Google Scholar] [CrossRef]
- Lungu, C.N.; Bratanovici, B.I.; Grigore, M.M.; Antoci, V.; Mangalagiu, I.I. Hybrid imidazole-pyridine derivatives: An approach to novel anticancer DNA intercalators. Curr. Med. Chem. 2020, 27, 154–169. [Google Scholar] [CrossRef]
- Antoci, V.; Cucu, D.; Zbancioc, G.; Moldoveanu, C.; Mangalagiu, V.; Amăriucăi-Mantu, D.; Aricu, A.; Mangalagiu, I.I. Bis-(imidazole/benzimidazole)-pyridine derivatives: Synthesis, structure and antimycobacterial activity. Future Med. Chem. 2020, 12, 207–222. [Google Scholar] [CrossRef]
- Mangalagiu, I.I.; Amăriucăi-Mantu, D.; Antoci, V.; Zbancioc, G.; Moldoveanu, C.; Cucu, D.; Dănac, R.; Mangalagiu, V. Obtaining class of anthracene-imidazole compounds with anti-tuberculosis effect by performing N-alkylation imidazole and benzimidazole with 9-chloromethyl-anthracene, and subjecting intermediates to quaternization/Procedeu pentru obţinerea unei noi clase de compuşi antracen-imidazolici cu activitate antituberculoasă. Patent RO (State Office for Inventions and Trademarks-OSIM) no. RO134192-A0/2020; Derwent Primary Accession Number: 2020-605892, 30 June 2020. [Google Scholar]
- Cucu, D.; Mangalagiu, V. Pyridine-Imidazolium Salts: Oxidatively Cleavage of N-C Bond via Nitration. Molbank 2019, 2019, M1095. [Google Scholar] [CrossRef]
- Antoci, V.; Humelnicu, I.; Vasilache, V.; Mantu, D. Synthesis, Structure and Biological Activity of Some Hybrid Benzimidazole/Quinoline Derivatives. Rev. Chim. 2016, 67, 1713–1716. [Google Scholar]
- Mantu, D.; Antoci, V.; Moldoveanu, C.; Zbancioc, G.; Mangalagiu, I.I. Hybrid imidazole (benzimidazole)/pyridine(quinoline) derivatives and evaluation of their anticancer and antimycobacterial activity. J. Enz. Inhib. Med. Chem. 2016, 31 (Suppl. S2), 96–103. [Google Scholar] [CrossRef]
- Antoci, V.; Moldoveanu, C.; Danac, R.; Mangalagiu, V.; Zbancioc, G. Huisgen [3+2] Dipolar Cycloadditions of Phthalazinium Ylides to Activated Symmetric and Non-Symmetric Alkynes. Molecules 2020, 25, 4416. [Google Scholar] [CrossRef] [PubMed]
- Cucu, D.; Mangalagiu, V.; Amăriucăi-Mantu, D.; Antoci, V.; Mangalagiu, I.I. Imidazolium ylides: Cycloaddition versus hydrolysis. Studia UBB Chem. 2019, 64, 59–66. [Google Scholar] [CrossRef]
- Moldoveanu, C.; Amăriucăi-Mantu, D.; Mangalagiu, V.; Antoci, V.; Maftei, D.; Mangalagiu, I.I.; Zbancioc, G. Microwave Assisted Reactions of Fluorescent Pyrrolodiazine Building Blocks. Molecules 2019, 24, 3760. [Google Scholar] [CrossRef] [PubMed]
- Popovici, L.; Amarandi, R.M.; Mangalagiu, I.I.; Mangalagiu, V.; Danac, R. Synthesis, molecular modelling and anticancer evaluation of new pyrrolo[1,2-b]pyridazine and pyrrolo[2,1-a]phthalazine derivatives. J. Enz. Inhib. Med. Chem. 2019, 34, 230–243. [Google Scholar] [CrossRef] [PubMed]
- Moldoveanu, C.; Zbancioc, G.; Mantu, D.; Maftei, D.; Mangalagiu, I.I. The Cycloaddition of the Benzimidazolium Ylides with Alkynes: New Mechanistic Insights. PLoS ONE 2016, 11, e0156129. [Google Scholar] [CrossRef]
- Al Matarneh, C.M.; Mangalagiu, I.I.; Shova, S.; Danac, R. Synthesis, structure, antimycobacterial and anticancer evaluation of new pyrrolo-phenanthroline derivatives. J. Enz. Inhib. Med. Chem. 2016, 31, 470–480. [Google Scholar] [CrossRef]
- Al Matarneh, C.M.; Apostu, M.O.; Mangalagiu, I.I.; Danac, R. Reactions of ethyl cyanoformate with cycloimmonium salts: A direct pathway to fused or substituted azaheterocycles. Tetrahedron 2016, 72, 4230–4238. [Google Scholar] [CrossRef]
- Antoci, V.; Mantu, D.; Cozma, D.G.; Usru, C.; Mangalagiu, I.I. Hybrid anticancer 1,2-diazine derivatives with multiple mechanism of action. Part 3 [4,5]. Med. Hypotheses 2014, 82, 11–15. [Google Scholar] [CrossRef]
- Mantu, D.; Maftei, D.; Iurea, D.; Ursu, C.; Bejan, V. Synthesis, structure, and in vitro anticancer activity of new polycyclic 1,2-diazines. Med. Chem. Res. 2014, 23, 2909–2915. [Google Scholar] [CrossRef]
- Tucaliuc, R.; Cotea, V.; Niculaua, M.; Tuchilus, C.; Mantu, D.; Mangalagiu, I.I. New pyridazine–fluorine derivatives: Synthesis, chemistry and biological activity. Part II. Eur. J. Med. Chem. 2013, 67, 367–372. [Google Scholar] [CrossRef]
- Mantu, D.; Maftei, D.; Iurea, D.; Bejan, V. Crystal Structure of Ethyl 5,10-dioxo-5,10-dihydrobenzo[f]pyridazino[6,1-a]isoindole-11-carboxylate. Rev. Chim. 2012, 63, 1239–1242. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amăriucăi-Mantu, D.; Mangalagiu, V.; Ciobanu, C.-I.; Antoci, V. Hybrid Pyridine Bis-Anthracene-Imidazolium Salt: NMR Studies on Zn-Acetate Complexation. Molbank 2021, 2021, M1280. https://doi.org/10.3390/M1280
Amăriucăi-Mantu D, Mangalagiu V, Ciobanu C-I, Antoci V. Hybrid Pyridine Bis-Anthracene-Imidazolium Salt: NMR Studies on Zn-Acetate Complexation. Molbank. 2021; 2021(3):M1280. https://doi.org/10.3390/M1280
Chicago/Turabian StyleAmăriucăi-Mantu, Dorina, Violeta Mangalagiu, Catalina-Ionica Ciobanu, and Vasilichia Antoci. 2021. "Hybrid Pyridine Bis-Anthracene-Imidazolium Salt: NMR Studies on Zn-Acetate Complexation" Molbank 2021, no. 3: M1280. https://doi.org/10.3390/M1280
APA StyleAmăriucăi-Mantu, D., Mangalagiu, V., Ciobanu, C.-I., & Antoci, V. (2021). Hybrid Pyridine Bis-Anthracene-Imidazolium Salt: NMR Studies on Zn-Acetate Complexation. Molbank, 2021(3), M1280. https://doi.org/10.3390/M1280





