(E)-5-Benzyl-7-(3,4-dimethoxybenzylidene)-3-(3,4-dimethoxyphenyl)-2-phenyl-3,3a,4,5,6,7-hexahydro-2H-pyrazolo[4,3c] Pyridine
Abstract
1. Introduction
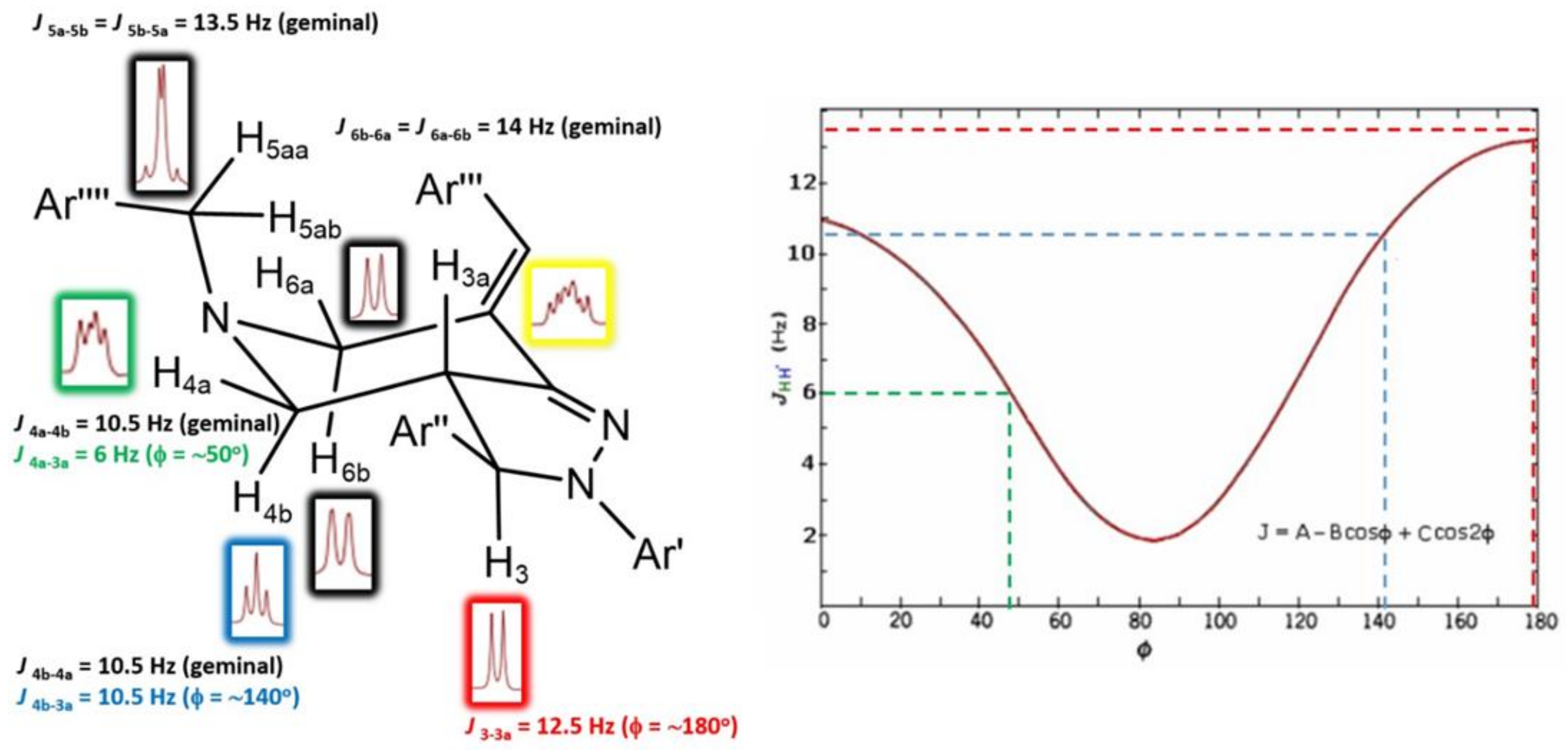
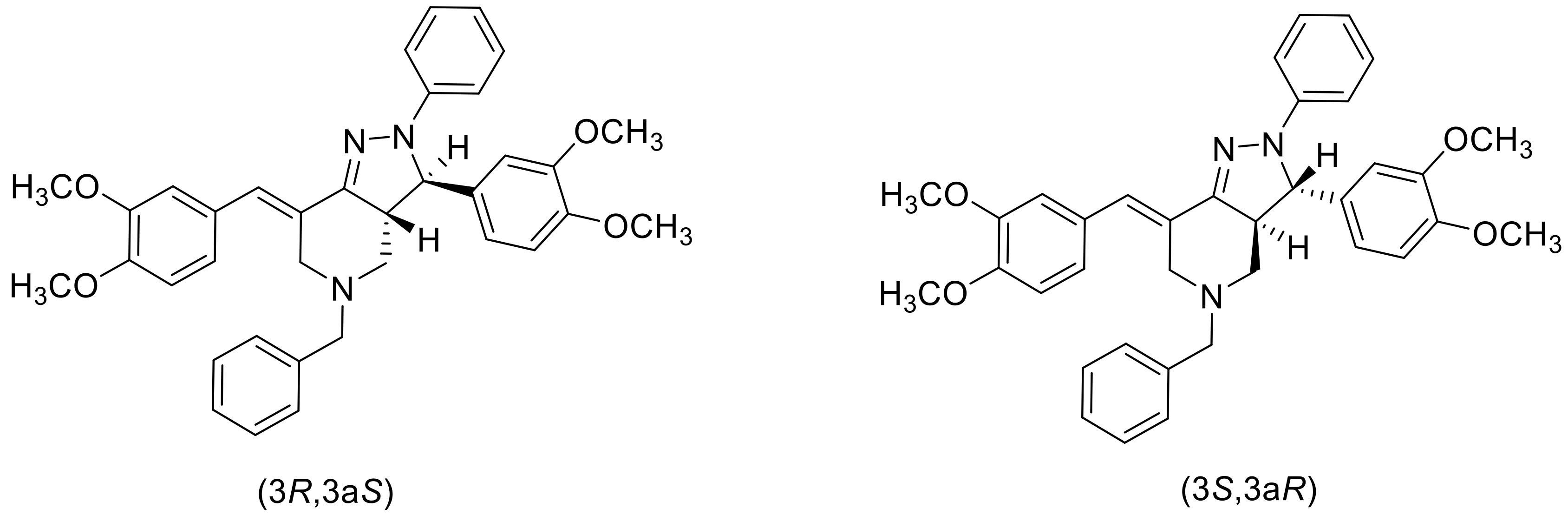
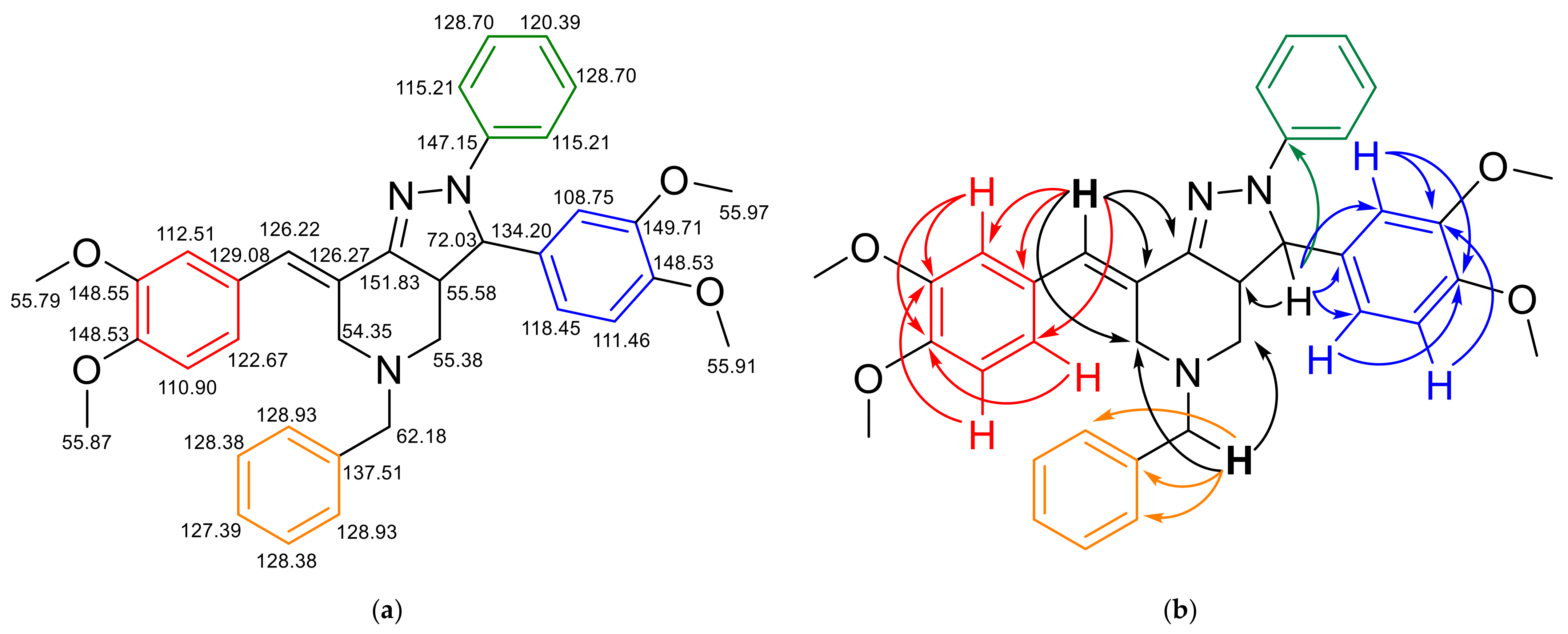
2. Results and Discussions
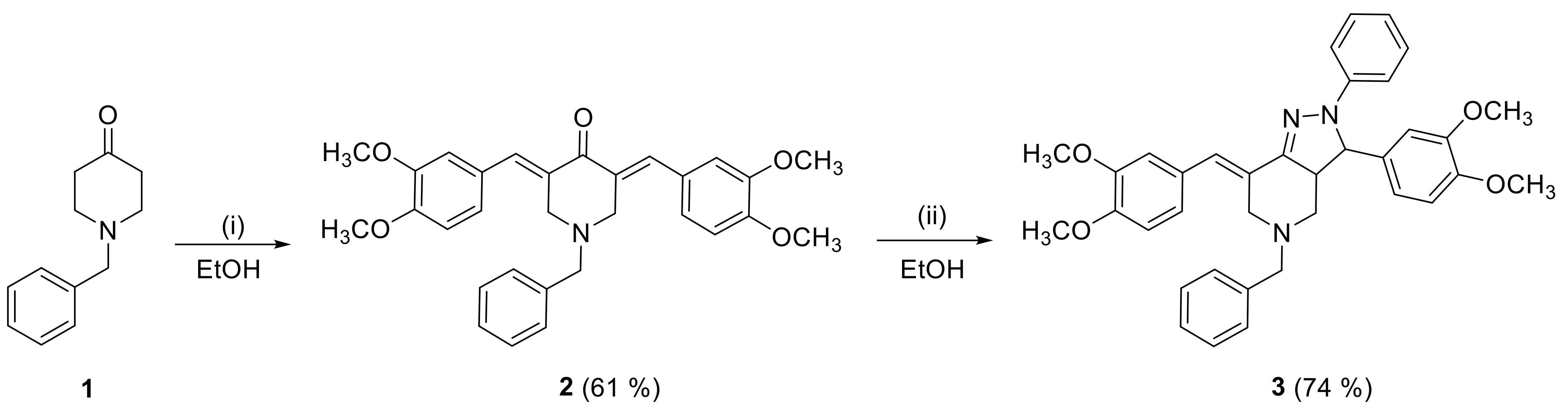
2.1. Synthesis
2.2. Test for Antioxidant Activity
3. Materials and Methods
3.1. Materials
3.2. Instrumentations
3.3. Methods
3.3.1. Synthesis of Compound 2
3.3.2. Synthesis of Compound 3
3.3.3. Test for Antioxidant Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sánchez, V.P.J.; Bautista, N.P.K.; Serrano, N.C.; Rincon, M.Y.; Garg, N.J. Potential Role of Antioxidants as Adjunctive Therapy in Chagas Disease. Oxidative Med. Cell. Longev. 2020, 1, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.; Stozhko, N.; Vidrevich, M. Antioxidants: Terminology, Methods and Future Considerations. Antioxidants 2019, 8, 297. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Norhaizan, M.E.; Liew, W.P.P.; Rahman, H.S. Antioxidant and oxidative stress: A mutual interplay in age-related diseases. Front. Pharmacol. 2018, 9, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, M.; Andruniów, T.; Sroka, Z. Flavones’ and flavonols’ antiradical structure-activity relationship—A quantum chemical study. Antioxidants 2020, 9, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Jasril, J.; Ikhtiarudin, I.; Hasti, S.; Reza, A.I.; Frimayanti, N. Microwave-assisted synthesis, in silico studies and in vivo evaluation for the antidiabetic activity of new brominated pyrazoline analogs. TJPS 2019, 43, 83–89. [Google Scholar]
- Ikhtiarudin, I.; Jasril, J.; Frimayanti, N. Microwave-assisted synthesis and in vivo antidiabetic activity of 5-(2-bromophenyl)-3-(naphthalen-1-yl)-4,5-dihydro-1H-pyrazole. In Proceedings of the Science and Mathematics International Conference (SMIC 2018), Jakarta, Indonesia, 2–4 November 2018; pp. 35–40. [Google Scholar]
- Zamri, A.; Teruna, H.Y.; Wulansari, S.; Herfindo, N.; Frimayanti, N.; Ikhtiarudin, I. 3-(3,4-Dimethoxyphenyl)-5-(2-fluorophenyl)-1-phenyl-4,5-dihydro-1H-pyrazole. Molbank 2019, 2019, M1088. [Google Scholar] [CrossRef]
- Jasril, J.; Teruna, H.Y.; Frimayanti, N.; Hasti, S.; Ikhtiarudin, I. Design, molecular docking study, synthesis and in vivo evaluation of some bromonaphthyl pyrazolines as new anti-inflammatory agents. In Proceedings of the Science and Mathematics International Conference (SMIC 2018), Jakarta, Indonesia, 2–4 November 2018; pp. 41–47. [Google Scholar]
- Jasril, J.; Ikhtiarudin, I.; Zamri, A.; Teruna, H.Y.; Frimayanti, N. New fluorinated chalcone and pyrazoline analogs: Synthesis, docking, and molecular dynamic studies as anticancer agents. TJPS 2017, 41, 93–98. [Google Scholar]
- Jasril, J.; Frimayanti, N.; Ikhtiarudin, I. In silico studies of fluorinated chalcone and pyrazoline analogues as inhibitors for cervical cancer. AIP Conf. Proc. 2020, 2242, 040008. [Google Scholar] [CrossRef]
- Jasril, J.; Nurulita, Y.; Desviana, L.; Ikhtiarudin, I.; Frimayanti, N. Microwave-assisted synthesis of a fluorinated pyrazoline, in silico study and in vitro cytotoxic evaluation against HeLa cell line. AIP Conf. Proc. 2021, 2331, 040014. [Google Scholar] [CrossRef]
- Jasril, J.; Teruna, H.Y.; Aisyah, A.; Nurlaili, N.; Hendra, R. Microwave assisted synthesis and evaluation of toxicity and antioxidant activity of pyrazoline derivatives. Indones. J. Chem. 2019, 19, 583–591. [Google Scholar] [CrossRef]
- Kumar, A.; Varadaraj, B.G.; Singla, R.K. Synthesis and evaluation of antioxidant activity of novel 3,5-disubstituted-2-pyrazolines. Bull. Fac. Pharm. Cairo Univ. 2013, 51, 167–173. [Google Scholar] [CrossRef][Green Version]
- Nagargoje, A.A.; Akolkar, S.V.; Siddiqui, M.M.; Bagade, A.V.; Kodam, K.M.; Sangshetti, J.N.; Damale, M.G.; Shingate, B.B. Synthesis and evaluation of pyrazole-incorporated monocarbonyl curcumin analogues as antiproliferative and antioxidant agents. J. Chin. Chem. Soc. 2019, 66, 1–8. [Google Scholar] [CrossRef]
- Zhou, D.Y.; Zhang, K.; Conney, A.H.; Ding, N.; Cui, X.X.; Wang, H.; Verano, M.; Zhao, S.Q.; Fan, Y.X.; Zheng, X.; et al. Synthesis and evaluation of curcumin-related compounds containing benzyl piperidone for their effects on human cancer cells. Chem. Pharm. Bull. 2013, 61, 1149–1155. [Google Scholar] [CrossRef]
- Perrin, C.L.; Chang, K.L. The complete mechanism of an aldol condensation. J. Org. Chem. 2016, 81, 5631–5635. [Google Scholar] [CrossRef] [PubMed]
- Kaka, K.N.; Taher, S.G.; Hamad, W.M.; Ibrahim, A.H. Synthesis of novel series of pyrazoline, and study their kinetics and reaction mechanism. ARO Sci. J. Koya Univ. 2019, 7, 5–13. [Google Scholar] [CrossRef]
- Ikhtiarudin, I.; Agistia, N.; Frimayanti, N.; Harlianti, T.; Jasril, J. Microwave-assisted synthesis of 1-(4-hydroxyphenyl)-3-(4-methoxyphenyl)prop-2-en-1-one and its activities as an antioxidant, sunscreen, and antibacterial. J. Kim. Sains Dan Apl. 2020, 23, 51–60. [Google Scholar] [CrossRef]
- Jasril, J.; Nurulita, Y.; Afriana, N.; Ikhtiarudin, I.; Frimayanti, N. Microwave-assisted synthesis and antioxidant activity of a hydrazone, (E)-1-(4-methoxybenzylidene)-2-phenylhydrazine. AIP Conf. Proc. 2021, 2331, 040018. [Google Scholar] [CrossRef]
- Jasril, J.; Ikhtiarudin, I.; Nurulita, Y.; Nurisma. Microwave-assisted synthesis and antioxidant activity of an imine, (E)-1-(3-bromobenzylidene)-2-phenylhydrazine. AIP Conf. Proc. 2020, 2242, 040041. [Google Scholar] [CrossRef]
| Atomic Numbering | 1H-NMR δH (ppm), J (Hz) | 13C-NMR, HSQC | COSY | Important HMBC |
|---|---|---|---|---|
| 1 | - | - | - | - |
| 2 | - | - | - | - |
| 3 | 4.53 (d, 1H), J = 12.5 Hz | 72.03 | 3a | 4, 2′′, 6′′, 1′′, 1′ |
| 3a | 3.34 (m, 1H) | 55.58 | 3, 4a, 4b | 4, 3, 1”, 7a |
| 4 | 4a = 3.24 (dd, 1H), J = 11 Hz, 6 Hz | 55.38 | 4b, 3a | 6, 4, 5a, 7a |
| 4b = 2.50 (t, 1H), J = 10.5 Hz | 4a, 3a | |||
| 5 | - | - | - | - |
| 5a | 5aa = 3.69 (d, 1H), J = 13.5 Hz | 62.18 | 5ab | 6, 4, 2′′′′, 6′′′′, 1′′′′ |
| 5ab = 3.66 (d, 1H), J = 13.0 Hz | 5ab | |||
| 6 | 6a = 4.09 (d, 1H), J = 14 Hz | 54.35 | 6b | 4, 7b, 7a |
| 6b = 3.16 (dd, 1H), J = 14 Hz, 3 Hz | 6a | |||
| 7 | - | 126.27 | - | - |
| 7a | - | 151.83 | - | - |
| 7b | 7.29 (s, 1H) | 126.22 | - | 6, 2′′′, 6′′′, 7, 1′′′, 7a |
| 3’’-OCH3 | 3.83 (s, 3H) | 55.97 | 3′′ | |
| 4’’-OCH3 | 3.89 (s, 3H) | 55.91 | 4′′ | |
| 3’’’-OCH3 | 3.81 (s, 3H) | 55.79 | 3′′′ | |
| 4’’’-OCH3 | 3.88 (s, 3H) | 55.87 | 4′′′ | |
| 1’ | - | 147.15 | - | - |
| 2’, 6’ | 7.10 (d, 2H), J = 8 Hz | 115.21 (2C) | 3′, 5′ | |
| 3’, 5’ | 7.17 (t, 2H), J = 8.5 Hz | 128.70 (2C) | 2′, 6′, 4′ | |
| 4′ | 6.85 (t, 1H), J = 8 Hz | 120.39 | 3′, 5′ | |
| 1” | - | 134.20 | - | - |
| 2” | 6.95 (d, 1H), J = 2 Hz | 108.75 | - | 3, 6′′, 1′′, 4′′, 3′′ |
| 3” | - | 149.71 | - | - |
| 4” | - | 148.53 | - | - |
| 5” | 6.86 (d, 1H), J = 8 Hz | 111.46 | 6” | 3′′ |
| 6” | 6.91 (dd, 1H), J = 8 Hz, 2 Hz | 118.45 | 5” | 4′′ |
| 1′’’ | - | 129.08 | - | - |
| 2’’’ | 6.77 (s, 1H) | 112.51 | - | 6′′′, 7b, 4′′′, 3′′′ |
| 3’’’ | - | 148.55 | - | - |
| 4’’’ | - | 148.53 | - | - |
| 5’’’ | 6.83 (s, 2H) | 110.90 | 6′′′ | 3′′′ |
| 6’’’ | 6.83 (s, 2H) | 122.67 | 5′′′ | 4′′′ |
| 1’’’’ | - | 137.51 | - | - |
| 2’’’’, 6′’’’ | 7.27–7.22 (m, 5H) | 128.93 (2C) | 3′′′′, 5′′′′ | |
| 3’’’’, 5’’’’ | 7.27–7.22 (m, 5H) | 128.38 (2C) | 2′′′′, 6′′′′, 4′′′′ | |
| 4’’’’ | 7.27–7.22 (m, 5H) | 127.39 (2C) | 3′′′′, 5′′′′ |
| Concentration (µg/mL) | Ln Concentration | % Inhibition | IC50 (µg/mL) |
|---|---|---|---|
| 1000 | 6.908 | 82.06 ± 0.78 | 194.06 ± 7.88 |
| 500 | 6.215 | 78.73 ± 0.34 | |
| 250 | 5.521 | 55.68 ± 0.94 | |
| 125 | 4.828 | 34.80 ± 3.68 | |
| 62.5 | 4.135 | 23.63 ± 1.48 | |
| 31.25 | 3.442 | 12.84 ± 3.41 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mora, E.; Zamri, A.; Teruna, H.Y.; Frimayanti, N.; Ikhtiarudin, I.; Herfindo, N.; Rindiana, E.N. (E)-5-Benzyl-7-(3,4-dimethoxybenzylidene)-3-(3,4-dimethoxyphenyl)-2-phenyl-3,3a,4,5,6,7-hexahydro-2H-pyrazolo[4,3c] Pyridine. Molbank 2021, 2021, M1240. https://doi.org/10.3390/M1240
Mora E, Zamri A, Teruna HY, Frimayanti N, Ikhtiarudin I, Herfindo N, Rindiana EN. (E)-5-Benzyl-7-(3,4-dimethoxybenzylidene)-3-(3,4-dimethoxyphenyl)-2-phenyl-3,3a,4,5,6,7-hexahydro-2H-pyrazolo[4,3c] Pyridine. Molbank. 2021; 2021(3):M1240. https://doi.org/10.3390/M1240
Chicago/Turabian StyleMora, Enda, Adel Zamri, Hilwan Y. Teruna, Neni Frimayanti, Ihsan Ikhtiarudin, Noval Herfindo, and Elsa Natia Rindiana. 2021. "(E)-5-Benzyl-7-(3,4-dimethoxybenzylidene)-3-(3,4-dimethoxyphenyl)-2-phenyl-3,3a,4,5,6,7-hexahydro-2H-pyrazolo[4,3c] Pyridine" Molbank 2021, no. 3: M1240. https://doi.org/10.3390/M1240
APA StyleMora, E., Zamri, A., Teruna, H. Y., Frimayanti, N., Ikhtiarudin, I., Herfindo, N., & Rindiana, E. N. (2021). (E)-5-Benzyl-7-(3,4-dimethoxybenzylidene)-3-(3,4-dimethoxyphenyl)-2-phenyl-3,3a,4,5,6,7-hexahydro-2H-pyrazolo[4,3c] Pyridine. Molbank, 2021(3), M1240. https://doi.org/10.3390/M1240









