5-((8-Hydroxyquinolin-5-yl)diazenyl)-3-methyl-1H-pyrazole-4-carboxylic Acid
Abstract
1. Introduction
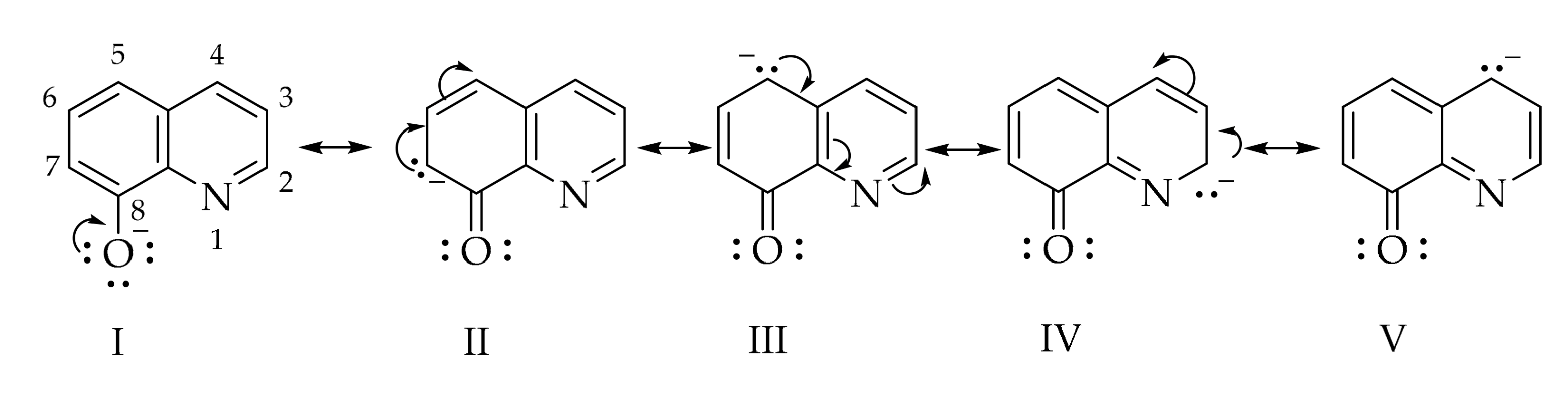
2. Results and Discussion
Experimental NMR Data for Compound (3) Explanation
3. Materials and Methods
4. Experimental
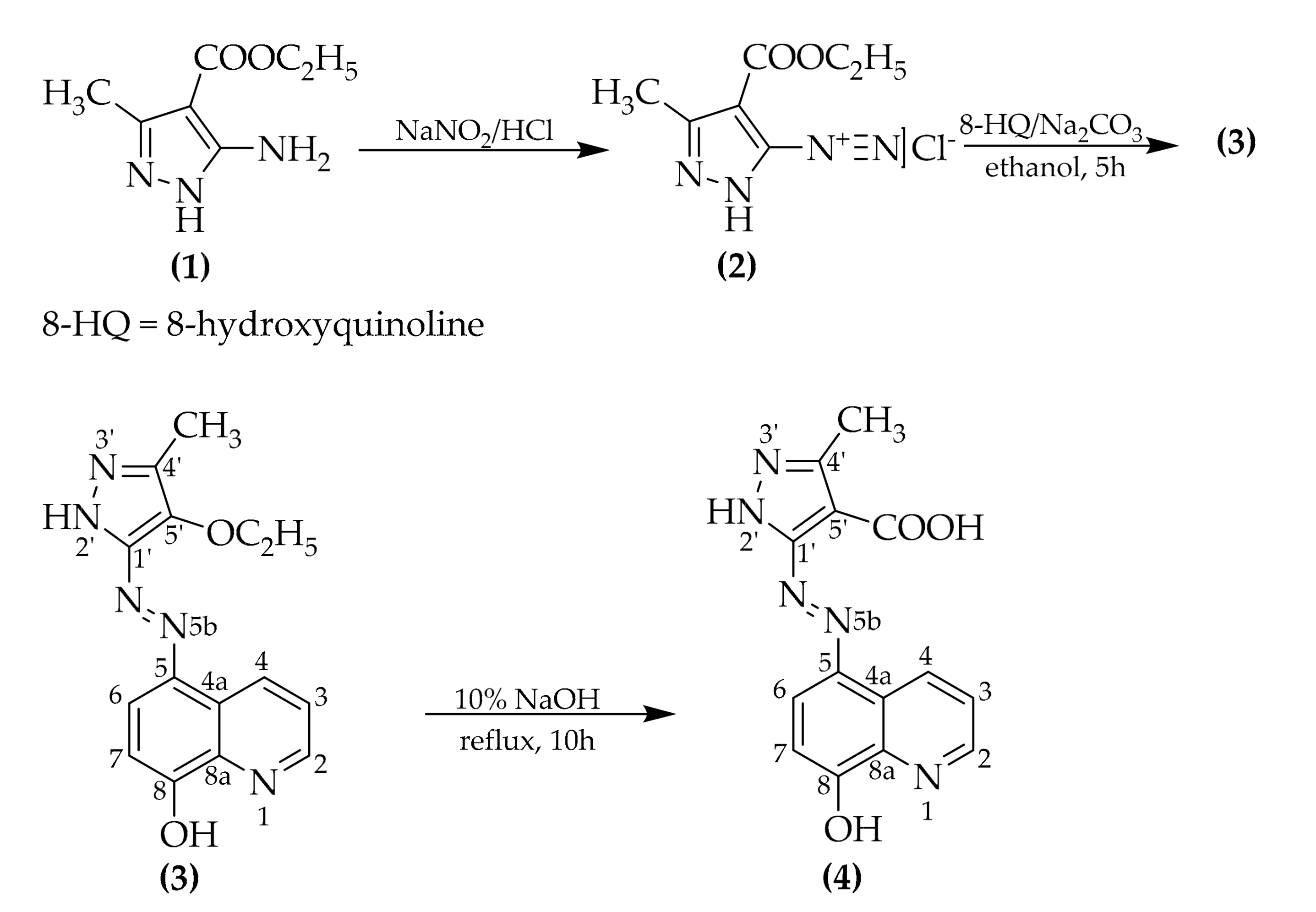
4.1. Synthesis of Ethyl 5-((8-Hydroxyquinolin-5-Yl)Diazenyl)-3-Methyl-1h-Pyrazole-4-Carboxylate (3)
4.2. Synthesis of 5-((8-Hydroxyquinolin-5-Yl)Diazenyl)-3-Methyl-1h-Pyrazole-4-Carboxylic Acid (4)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hunger, K.; Mischke, P.; Rieper, W.; Raue, R.; Kunde, K.; Engel, A. Azo Dyes. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar] [CrossRef]
- Clark, M. Azoic dyeing. In Handbook of Textile and Industrial Dyeing; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 1, pp. 604–605. [Google Scholar]
- Leulescu, M.; Rotaru, A.; Pălărie, I.; Moanţă, A.; Cioateră, N.; Popescu, M.; Morîntale, E.; Bubulică, M.V.; Florian, G.; Hărăbor, A.; et al. Tartrazine: Physical, thermal and biophysical properties of the most widely employed synthetic yellow food-colouring azo dye. J. Therm. Anal. Calorim. 2018, 134, 209–231. [Google Scholar] [CrossRef]
- Devika, B.G.; Doreswamy, B.H.; Tandon, H.C. Corrosion behaviour of metal complexes of antipyrine based azo dye ligand for soft-cast steel in 1 M hydrochloric acid. J. King Saud Univ. Sci. 2020, 32, 881–890. [Google Scholar] [CrossRef]
- Elmaaty, T.A.; Sofan, M.; Elsisi, H.; Kosbar, T.; Negm, E.; Hirogaki, K.; Tabata, I.; Hori, T. Optimization of an eco-friendly dyeing process in both laboratory scale and pilot scale supercritical carbon dioxide unit for polypropylene fabrics with special new disperse dyes. J. CO2 Util. 2019, 33, 365–371. [Google Scholar] [CrossRef]
- Deng, Y.; Jiang, H.; Ye, D.; Zhou, R.; Li, H.; Tang, B.; Jin, M.; Li, N.; Guo, Y.; Zhou, G. Synthesis and application of an alkylated pyrazole-based azo dye for electrofluidic display. J. Soc. Inf. Disp. 2018, 26, 369–375. [Google Scholar] [CrossRef]
- Adiguzel, R.; Turan, N.; Buldurun, K.; Korkoca, H. Spectral, Thermal and Antimicrobial Properties of Novel Mixed Ligand-Metal Complexes Derived from Saccharinate Complexes and Azo Dye Ligand. Int. J. Pharmacol. 2018, 14, 9–19. [Google Scholar] [CrossRef]
- Bartwal, G.; Aggarwala, K.; Khurana, J.M. An Ampyrone based azo dye as pH-responsive and chemo-reversible colorimetric fluorescent probe for Al3+ in semi-aqueous medium: Implication towards logic gate analysis. New J. Chem. 2018, 42, 2224–2231. [Google Scholar] [CrossRef]
- Tang, C.W.; VanSlyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- Al-Busafi, S.N.; O Suliman, F.E.; Al-Alawi, Z.R. 8-Hydroxyquinoline and its Derivatives. Res. Rev. J. Chem. 2014, 3, 1–10. [Google Scholar]
- Yet, L. 4.01—Pyrazoles. In Comprehensive Heterocyclic Chemistry III; Elsevier Science: Amsterdam, The Netherlands, 2008; Volume 4, pp. 1–141. [Google Scholar] [CrossRef]
- Roos, G.; Roos, C. Chapter 7—Functional Classes II, Reactions. In Organic Chemistry Concepts: An EFL Approach; Academic Press: Cambridge, MA, USA, 2015; pp. 103–149. [Google Scholar] [CrossRef]
- Godovikova, T.I.; Rakitin, O.A.; Khmel’nitskii, L.I. Diazotisation of Weakly Basic Aromatic and Heterocyclic Amines in Strongly Acid Media. Russ. Chem. Rev. 1983, 52, 440–445. [Google Scholar] [CrossRef]
- Butler, R.N. Diazotization of heterocyclic primary amines. Chem. Rev. 1975, 75, 241–257. [Google Scholar] [CrossRef]
- Parekh, V.J.; Rathod, V.K.; Pandit, A.B. 2.10—Substrate Hydrolysis: Methods, Mechanism, and Industrial Applications of Substrate Hydrolysis. In Comprehensive Biotechnology, 2nd ed.; Pergamon: Oxford, UK, 2011; pp. 103–118. [Google Scholar] [CrossRef]
- Saadeh, H.A.; Sweidan, K.A.; Mubarak, M.S. Recent Advances in the Synthesis and Biological Activity of 8-Hydroxyquinolines. Molecules 2020, 25, 4321. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.P. The Reactions Of 8-Quinolinol. Chem. Rev. 1956, 56, 271–297. [Google Scholar] [CrossRef]
- Yazdanbakhsh, M.R.; Mahmoodi, N.O.; Dabiry, S. Preparation and characterization of diazenyl quinolin-8-ol with trifluoromethyl substituents. Mendeleev Commun. 2006, 16, 192–194. [Google Scholar] [CrossRef]
- Beyer, H.; Wolter, G. Über Thiazole, XXIX. Mitteil: Über die Kondensationsprodukte von Thiosemicarbazid mit α-Chloracetessigester und eine neuartige Ringverengung des 2-Amino-5- methyl-6-carbäthoxy − 1.3.4-thiodiazins zum 3-Methyl-4- carbäthoxy-5-amino-pyrazol. Chem. Ber. 1956, 89, 1652–1658. [Google Scholar] [CrossRef]
| HSQC 1H-13C | HMBC 1H-13C | HMBC 1H-15N | COSY 1H-1H | NOESY 1H-1H |
|---|---|---|---|---|
| 9.45→133.3 (4-H) (4-C) | 4.47→164.8 (-CH2-) (-C = O) | 8.97→298.8 (2-H) (1-N) | 9.45→7.47 (4-H) (3-H) | 7.47→9.45 (3-H) (4-H) |
| 8.97→149.6 (2-H) (2-C) | 8.49→160.6 (6-H) (8-C) | 7.52→298.8 (7-H) (1-N) | 8.97→7.47 (2-H) (3-H) | 7.47→8.97 (3-H) (2-H) |
| 8.49→118.9 (6-H) (6-C) | 7.52→160.6 (7-H) (8-C) | 8.56→7.52 (6-H) (7-H) | 7.52→8.49 (7-H) (6-H) | |
| 7.52→113.0 (7-H) (7-C) | 9.45→149.6 (4-H) (2-C) | 4.47→1.32 (-CH2-CH3) (-CH2-CH3) | ||
| 7.47→123.6 (3-H) (3-C) | 7.47→149.6 (3-H) (2-C) | |||
| 8.49→140.8 (6-H) (8a-C) | ||||
| 7.52→140.8 (7-H) (8a-C) | ||||
| 9.45→139.7 (4-H) (4a-C) | ||||
| 8.97→139.7 (2-H) (4a-C) | ||||
| 7.52→139.7 (7-H) (4a-C) | ||||
| 8.97→133.3 (2-H) (4-C) | ||||
| 8.49→133.3 (6-H) (4-C) | ||||
| 8.97→128.9 (2-H) (5-C) | ||||
| 8.49→128.9 (6-H) (5-C) | ||||
| 7.47→128.9 (3-H) (5-C) | ||||
| 2.73→159.4 (-CH3) (4′-C) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burcă, I.; Badea, V.; Deleanu, C.; Bercean, V.-N. 5-((8-Hydroxyquinolin-5-yl)diazenyl)-3-methyl-1H-pyrazole-4-carboxylic Acid. Molbank 2021, 2021, M1238. https://doi.org/10.3390/M1238
Burcă I, Badea V, Deleanu C, Bercean V-N. 5-((8-Hydroxyquinolin-5-yl)diazenyl)-3-methyl-1H-pyrazole-4-carboxylic Acid. Molbank. 2021; 2021(2):M1238. https://doi.org/10.3390/M1238
Chicago/Turabian StyleBurcă, Ion, Valentin Badea, Calin Deleanu, and Vasile-Nicolae Bercean. 2021. "5-((8-Hydroxyquinolin-5-yl)diazenyl)-3-methyl-1H-pyrazole-4-carboxylic Acid" Molbank 2021, no. 2: M1238. https://doi.org/10.3390/M1238
APA StyleBurcă, I., Badea, V., Deleanu, C., & Bercean, V.-N. (2021). 5-((8-Hydroxyquinolin-5-yl)diazenyl)-3-methyl-1H-pyrazole-4-carboxylic Acid. Molbank, 2021(2), M1238. https://doi.org/10.3390/M1238






