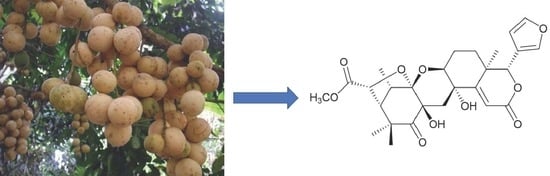
Kokosanolide D: A New Tetranortriterpenoid from Fruit Peels of Lansium domesticum Corr. cv Kokossan
Abstract
:1. Introduction
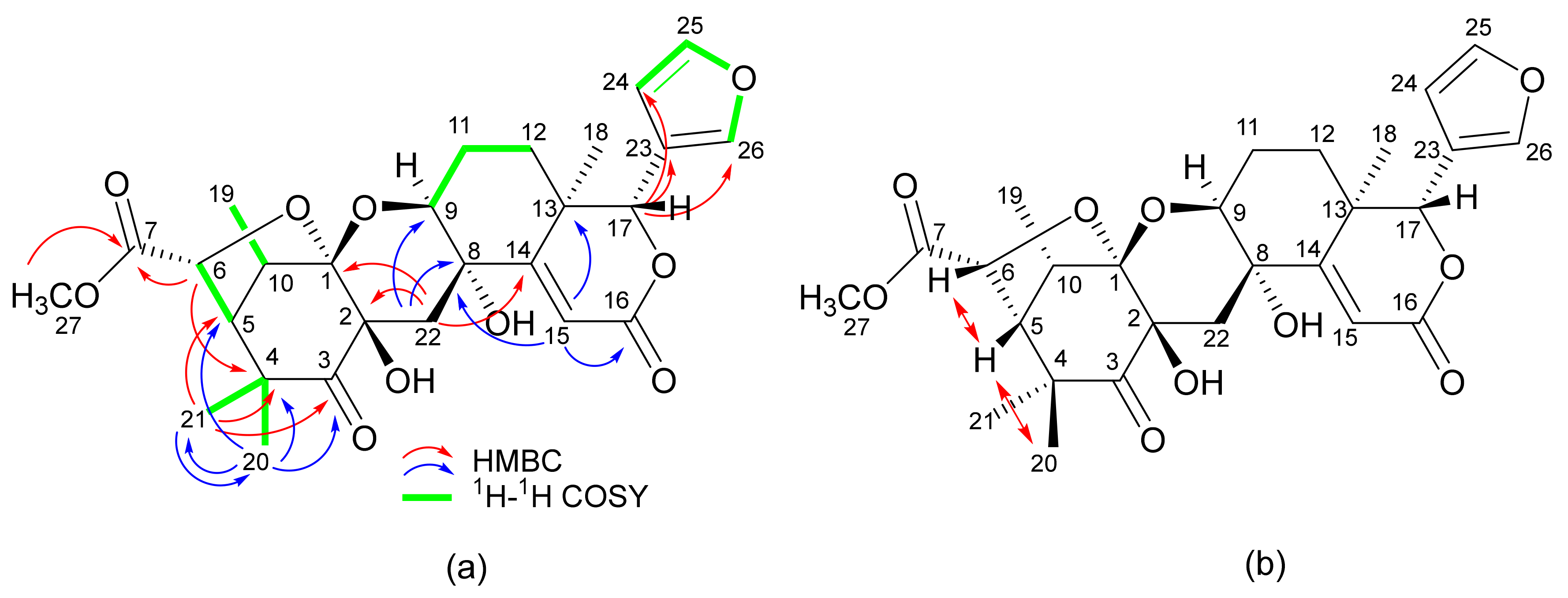
2. Results
Extraction and Isolation
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kiang, A.K.; Tan, E.L.; Lim, F.Y.; Habaguchi, K.; Nakanishi, K.; Fachan, L.; Ourisson, G. Lansic acid, a bicyclic triterpene. Tetrahedron Lett. 1967, 37, 3571–3574. [Google Scholar] [CrossRef]
- Ghani, U. Chapter four—Terpenoids and steroids. In Alpha-Glucosidase Inhibitors, Clinically Promising Candidates for Anti-Diabetic Drug Discovery; Elsevier: Amsterdam, The Netherlands, 2020; pp. 101–117. [Google Scholar]
- Techavuthiporn, C. Langsat—Lansium domesticum. In Exotic Fruits Reference Guides; Rodrigues, S., de Brito, E., Silva, E., Eds.; Academic Press: London, UK, 2018; pp. 279–283. [Google Scholar]
- Dong, S.H.; Zhang, C.R.; Dong, L.; Wu, Y.; Yue, J.M. Onoceranoid-type triterpenoids from Lansium domesticum. J. Nat. Prod. 2011, 74, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Mayanti, T.; Supratman, U.; Mukhtar, M.R.; Awang, K.; Ng, S.W. Kokosanolide from the seed of Lansium domesticum Corr. Acta Crystallogr. 2009, E65, o750. [Google Scholar]
- Nishizawa, M.; Nishide, H.; Hayashi, Y.; Kosela, S. The structure of lansioside A: A novel triterpene glycoside with amino-sugar from Lansium domesticum. Tetrahedron Lett. 1982, 23, 1349–1350. [Google Scholar] [CrossRef]
- Nishizawa, M.; Nishide, H.; Kosela, S.; Hayashi, Y. Structure of lansiosides: Biologically active new triterpene glycosides from Lansium domesticum. J. Org. Chem. 1983, 48, 4462–4466. [Google Scholar] [CrossRef]
- Tanaka, T.; Ishibashi, M.; Fujimoto, H.; Okuyama, E.; Koyano, T.; Kowiyhayakorn, T.; Hayashi, M.; Komiyama, K. New onoceranoid constituents from Lansium domesticum. J. Nat. Prod. 2002, 65, 1709–1711. [Google Scholar] [CrossRef] [PubMed]
- Habaguchi, K.; Watanabe, M.; Nakadaira, Y.; Nakanishi, K.; Kaing, A.K.; Lim, F.L. The full structures of lansic acid and its minor congener, an unsymmetric onoceradienedione. Tetrahedron Lett. 1986, 34, 3731–3734. [Google Scholar] [CrossRef]
- Zulfikar; Putri, N.K.; Fajriah, S.; Yusuf, M.; Maharani, R.; Al Anshori, J.; Supratman, U.; Mayanti, T. 3-Hydroxy-8,14-secogammacera-7,14-dien-21-one: A new onoceranoid triterpenes from Lansium domesticum Corr. cv kokossan. Molbank 2020, 2020, M1157. [Google Scholar] [CrossRef]
- Mayanti, T.; Sianturi, J.; Harneti, D.; Darwati; Supratman, U.; Rosli, M.M.; Fun, H.K. 9,19-Cyclolanost-24-en-3-one,21,23-epoxy-21,22-dihydroxy (21R, 22S, 23S) from the leaves of Lansium domesticum Corr cv Kokossan. Molbank 2015, 2015, M880. [Google Scholar] [CrossRef]
- Mayanti, T.; Tjokronegoro, R.; Supratman, U.; Mukhtar, M.R.; Awang, K.; Hadi, A.H.A. Antifeedant Triterpenoids from the Seeds and Bark of Lansium domesticum cv Kokossan (Meliaceae). Molecules 2011, 16, 2785–2795. [Google Scholar] [CrossRef] [PubMed]
- Fadhilah, K.; Wahyuona, S.; Astuti, P. A bioactive compound isolated from duku (Lansium domesticum Corr) fruit peels exhibits cytotoxicity against T47D cell line. F1000Research 2020, 9, 3. [Google Scholar] [CrossRef]
- Ragasa, C.Y.; Labrador, P.; Rideout, J.A. Antimicrobial terpenoid from Lansium domesticum. Philipp. Agric. Sci. 2006, 89, 101–105. [Google Scholar]
- Matsumoto, T.; Kitagawa, T.; Teo, S.; Anai, Y.; Ikeda, R.; Imahori, D.; Ahmad, H.S.; Watanabe, T. Structures and antimutagenic effects of onoceranoid-type triterpenoids from the leaves of Lansium domesticum. J. Nat. Prod. 2018, 81, 2187–2194. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Kitagawa, T.; Ohta, T.; Yoshida, T.; Imahori, D.; Teo, S.; Ahmad, H.S.; Watanabe, T. Structures of triterpenoids from the leaves of Lansium domesticum. J. Nat. Med. 2019, 73, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Saewan, N.; Sutherland, J.D.; Chantrapromma, K. Antimalarial tetranortriterpenoids from the seed of Lansium domesticum Corr. Phytrochemistry 2006, 67, 2288–2293. [Google Scholar] [CrossRef] [PubMed]
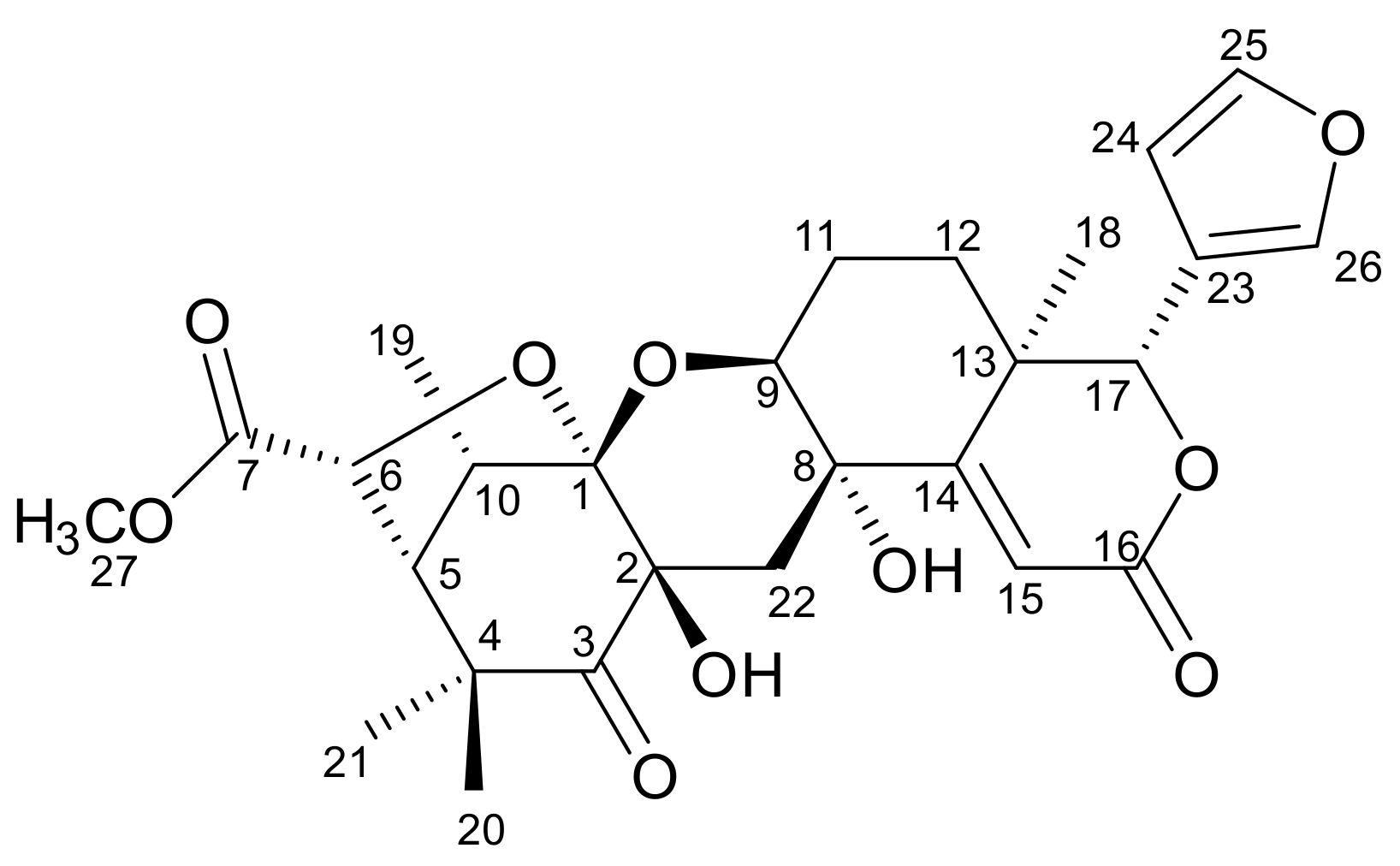
| Position | δC | δH (ΣH, mult., J (Hz)) | HMBC |
|---|---|---|---|
| 1 | 107.0 | - | - |
| 2 | 77.1 | - | - |
| 3 | 209.7 | - | - |
| 4 | 47.8 | - | - |
| 5 | 55.9 | 2.30 (1H, d, 4) | 1, 3 |
| 6 | 76.7 | 4.82 (1H, d, 3.5) | 7, 4 |
| 7 | 172.0 | - | - |
| 8 | 67.8 | - | - |
| 9 | 73.4 | 4.16 (1H, d, 2.5) | 8, 12, 14 |
| 10 | 36.5 | 3.29 (1H, d, 7) | 4, 5, 6, 19 |
| 11 | 20.9 | 1.75 (1H, t, 3.5) 2.18 (1H, tt, 6) | 9, 13 |
| 12 | 26.6 | 1.22 (1H, t, 13) 1.71 (1H, dd, 5) | 13 |
| 13 | 38.4 | - | - |
| 14 | 167.5 | - | - |
| 15 | 117.4 | 6.46 (1H, s) | 8, 13, 16 |
| 16 | 165.7 | - | - |
| 17 | 81.8 | 5.15 (1H, s) | 12, 13, 14, 18, 23, 24, 26 |
| 18 | 19.7 | 1.26 (3H, s) | 12, 13, 14, 17 |
| 19 | 11.8 | 1.16 (3H, d, 6.5) | 1, 5, 10 |
| 20 | 23.1 | 0.98 (3H, s) | 3, 4, 5, 21 |
| 21 | 29.6 | 1.37 (3H, s) | 3, 4, 5, 20 |
| 22 | 34.4 | 2.40 (1H, d, 15) 3.02 (1H, d, 15) | 1, 2, 8, 9, 14 |
| 23 | 119.7 | - | - |
| 24 | 110.2 | 6.43 (1H, s) | 23, 25 |
| 25 | 142.8 | 7.39 (1H, s) | 23, 24 |
| 26 | 141.5 | 7.48 (1H, s) | 23, 24 |
| 27 | 52.5 | 3.67 (3H, s) | 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fauzi, F.M.; Meilanie, S.R.; Zulfikar; Farabi, K.; Herlina, T.; Al Anshori, J.; Mayanti, T. Kokosanolide D: A New Tetranortriterpenoid from Fruit Peels of Lansium domesticum Corr. cv Kokossan. Molbank 2021, 2021, M1232. https://doi.org/10.3390/M1232
Fauzi FM, Meilanie SR, Zulfikar, Farabi K, Herlina T, Al Anshori J, Mayanti T. Kokosanolide D: A New Tetranortriterpenoid from Fruit Peels of Lansium domesticum Corr. cv Kokossan. Molbank. 2021; 2021(2):M1232. https://doi.org/10.3390/M1232
Chicago/Turabian StyleFauzi, Fawwaz M., Sylvia R. Meilanie, Zulfikar, Kindi Farabi, Tati Herlina, Jamaludin Al Anshori, and Tri Mayanti. 2021. "Kokosanolide D: A New Tetranortriterpenoid from Fruit Peels of Lansium domesticum Corr. cv Kokossan" Molbank 2021, no. 2: M1232. https://doi.org/10.3390/M1232
APA StyleFauzi, F. M., Meilanie, S. R., Zulfikar, Farabi, K., Herlina, T., Al Anshori, J., & Mayanti, T. (2021). Kokosanolide D: A New Tetranortriterpenoid from Fruit Peels of Lansium domesticum Corr. cv Kokossan. Molbank, 2021(2), M1232. https://doi.org/10.3390/M1232