(E)-1-(3,4-Dimethoxyphenyl)-2-methyl-3-phenylprop-2-en-1-one: A P-Type Acid-Stable Photochromic α-Methylchalcone
Abstract
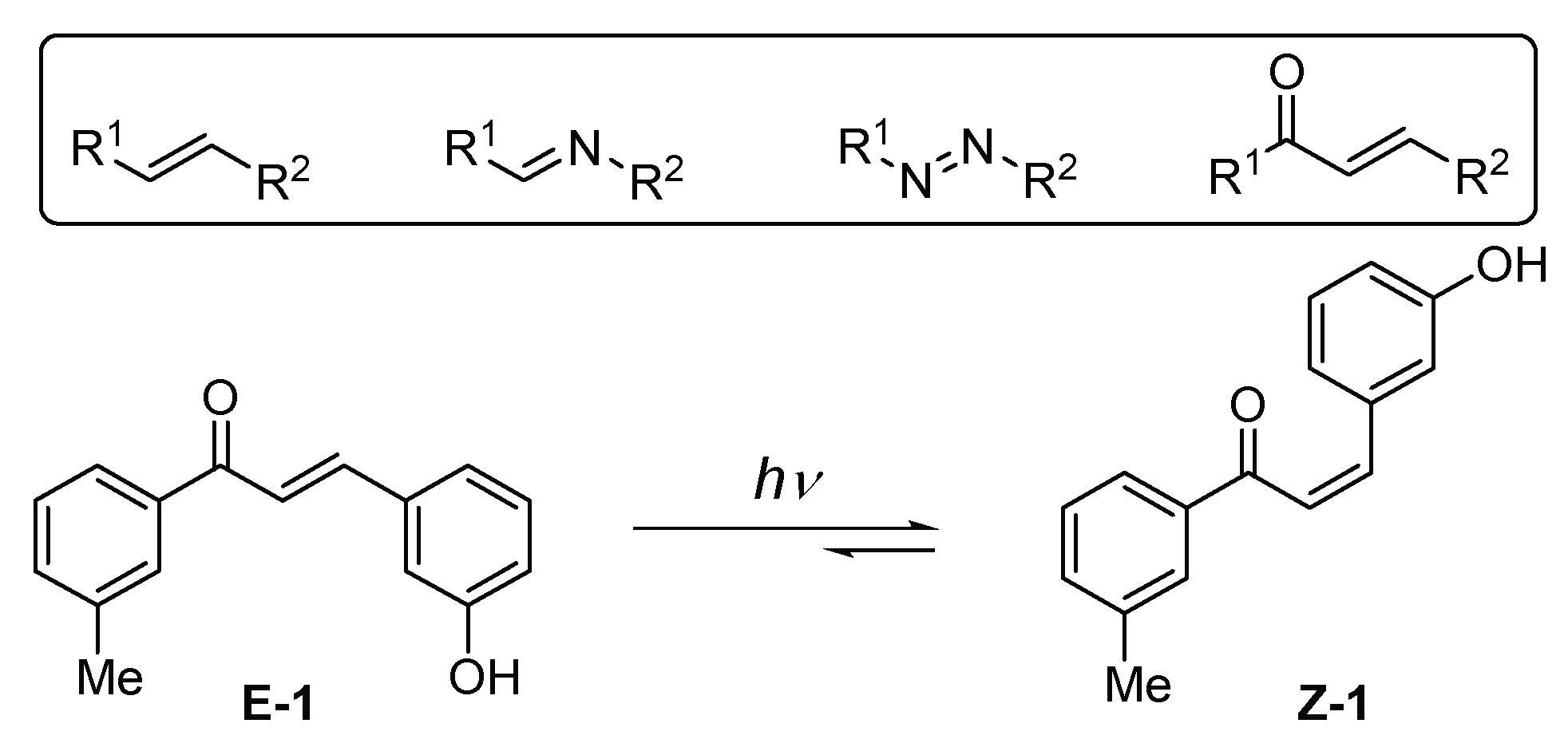
1. Introduction
2. Results
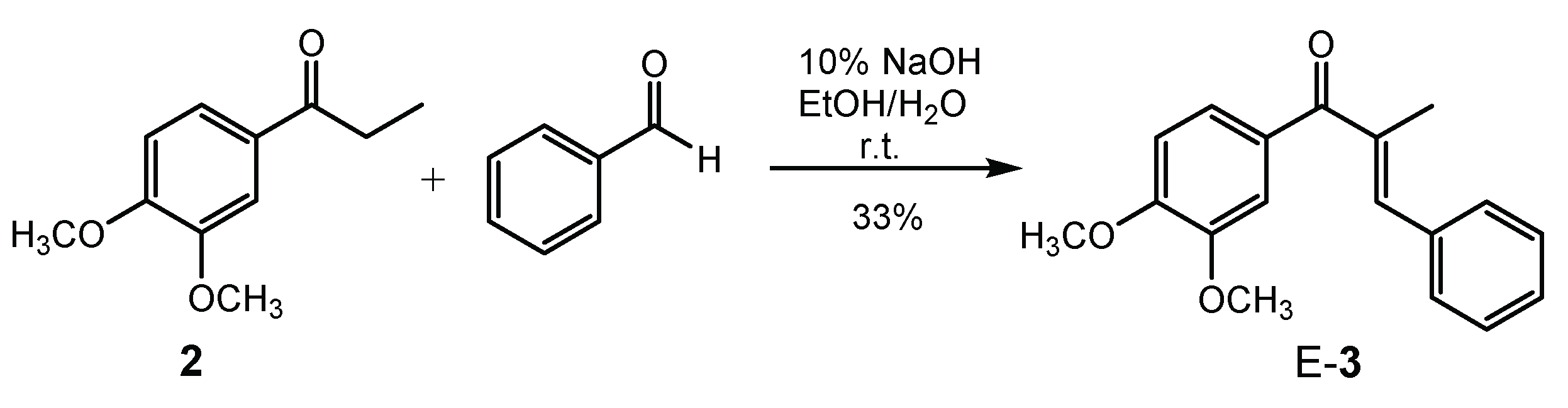
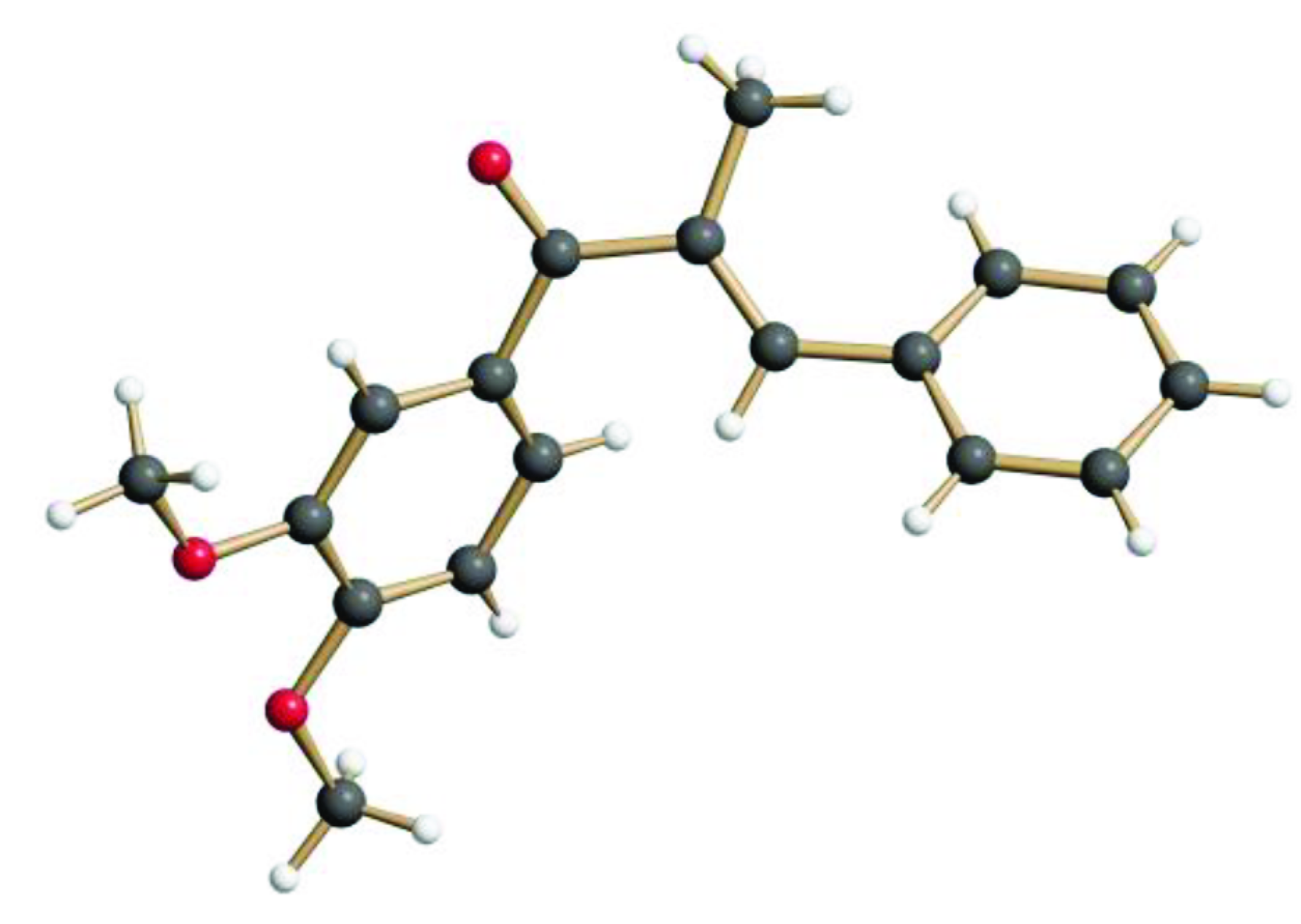
2.1. Synthesis and Structure of the α-Methyl Chalcone 2
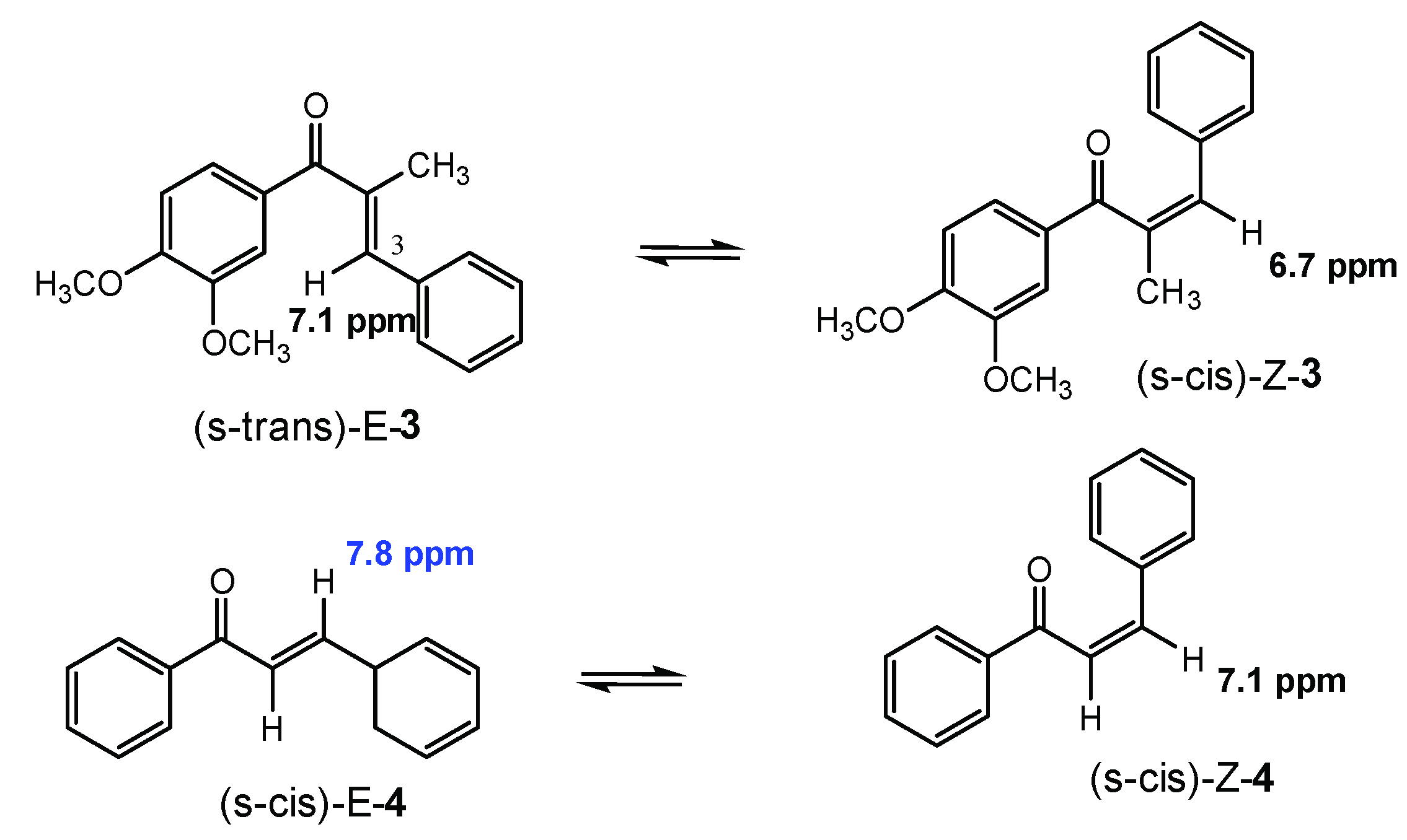
2.2. E/Z-Photoswitching, PSS, and PSS-Stability of the α-Methyl Chalcone 3
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bouas-Laurent, H.; Castellan, A.; Desvergne, J.P. From Anthracene Photo-Dimerization to Jaw Photochromic Materials and Photocrowns. Pure Appl. Chem. 1980, 52, 2633–2648. [Google Scholar]
- Bouas-Laurent, H.; Dürr, H. Organic Photochromism (IUPAC Technical Report). Pure Appl. Chem. 2001, 73, 639–665. [Google Scholar]
- Feringa, B.L.; van Delden, R.A.; Koumura, N.; Geertsema, E.M. Chiroptical Molecular Switches. Chem. Rev. 2000, 100, 1789–1816. [Google Scholar] [PubMed]
- Hull, K.; Morstein, J.; Trauner, D. In Vivo Photopharmacology. Chem. Rev. 2018, 118, 10710–10747. [Google Scholar] [CrossRef] [PubMed]
- Lerch, M.M.; Hansen, M.J.; van Dam, G.M.; Szymanski, W.; Feringa, B.L. Emerging Targets in Photopharmacology. Angew. Chem. Int. Ed. 2016, 55, 10978–10999. [Google Scholar] [CrossRef] [PubMed]
- Irie, M. Diarylethenes for Memories and Switches. Chem. Rev. 2000, 100, 1685–1716. [Google Scholar] [CrossRef] [PubMed]
- Bleger, D.; Hecht, S. Visible-Light-Activated Molecular Switches. Angew. Chem. Int. Ed. 2015, 54, 11338–11349. [Google Scholar] [CrossRef] [PubMed]
- Aubry, J.M.; Pierlot, C.; Rigaudy, J.; Schmidt, R. Reversible binding of oxygen to aromatic compounds. Acc. Chem. Res. 2003, 36, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Sakata, Y.; Fukushima, S.; Akine, S.; Setsune, J. Solvent-dependent dual-mode photochromism between T-and P-types in a dipyrrinone derivative. Chem. Commun. 2016, 52, 1278–1281. [Google Scholar] [CrossRef] [PubMed]
- Kobatake, S.; Terakawa, Y. Acid-induced photochromic system switching of diarylethene derivatives between P- and T-types. Chem. Commun. 2007, 43, 1698–1700. [Google Scholar] [CrossRef] [PubMed]
- Hadjoudis, E.; Mavridis, I.M. Photochromism and thermochromism of Schiff bases in the solid state: Structural aspects. Chem. Soc. Rev. 2004, 33, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.; You, J.; Kim, J.; Huh, J.; Kim, E. Photochromic fluorescence switching from diarylethenes and its applications. J. Photochem. Photobiol. C 2009, 10, 111–129. [Google Scholar] [CrossRef]
- Iwata, S.; Nishino, T.; Inoue, H.; Nagata, N.; Satomi, Y.; Nishino, W.; Shibata, S. Antitumorigenic Activities of Chalcones (II). Photo-isomerization of Chalcones and the Correlation with Their Biological Activities. Biol. Pharm. Bull. 1997, 20, 1266–1270. [Google Scholar] [CrossRef] [PubMed]
- Ducki, S.; Forrest, R.; Ducki, S.; Forrest, R.; Hadfield, J.A.; Kendall, A.; Lawrence, N.J.; McGown, A.T.; Rennison, D. Potent Antimitotic and Cell Growth Inibitory Properties of Substituted Chalcones. Bioorg. Med. Chem. Lett. 1998, 8, 1051–1056. [Google Scholar] [CrossRef]
- Öngel, B. Novel Photoswitches Based on Phthalimide-Based Azobenzenes and Chalcones: Synthesis and Photophysical Study of Reversible E/Z Isomerization. Ph.D. Thesis, University of Cologne, Cologne, Germany, 2020. [Google Scholar]
- Griesbeck, A.G.; Öngel, B.; Brüllingen, E.; Renner, M. New Photochromic α-Methylchalcones are Highly Photostable, Even under Singlet Oxygen Conditions: Breaking the α-Methyl Michael-System Reactivity by Reversible Peroxybiradical Formation. Molecules 2021, 26, 642. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Nakamura, M.; Abdussalam Salhin, M.A.; Ikeda, T.; Kamada, K.; Ando, H.; Shibutani, Y.; Kimura, K. Synthesis and Photochromism of Spirobenzopyran Derivatives Bearing an Oxymethylcrown Ether Moiety: Metal Ion-Induced Switching between Positive and Negative Photochromisms. J. Org. Chem. 2001, 66, 1533–1537. [Google Scholar] [CrossRef] [PubMed]
- Megerle, U.; Lechner, R.; König, B.; Riedle, E. Laboratory apparatus for the accurate, facile and rapid determination of visible light photoreaction quantum yields. Photochem. Photobiol. Sci. 2010, 9, 1400–1406. [Google Scholar] [CrossRef] [PubMed]
- Data from Crystal Structure Analysis are Deposited at the Cambridge Crystallographic Data Centre (CCDC) with the Deposition Number CCDC 2081479. Available online: https://www.ccdc.cam.ac.uk/ (accessed on 2 June 2021).
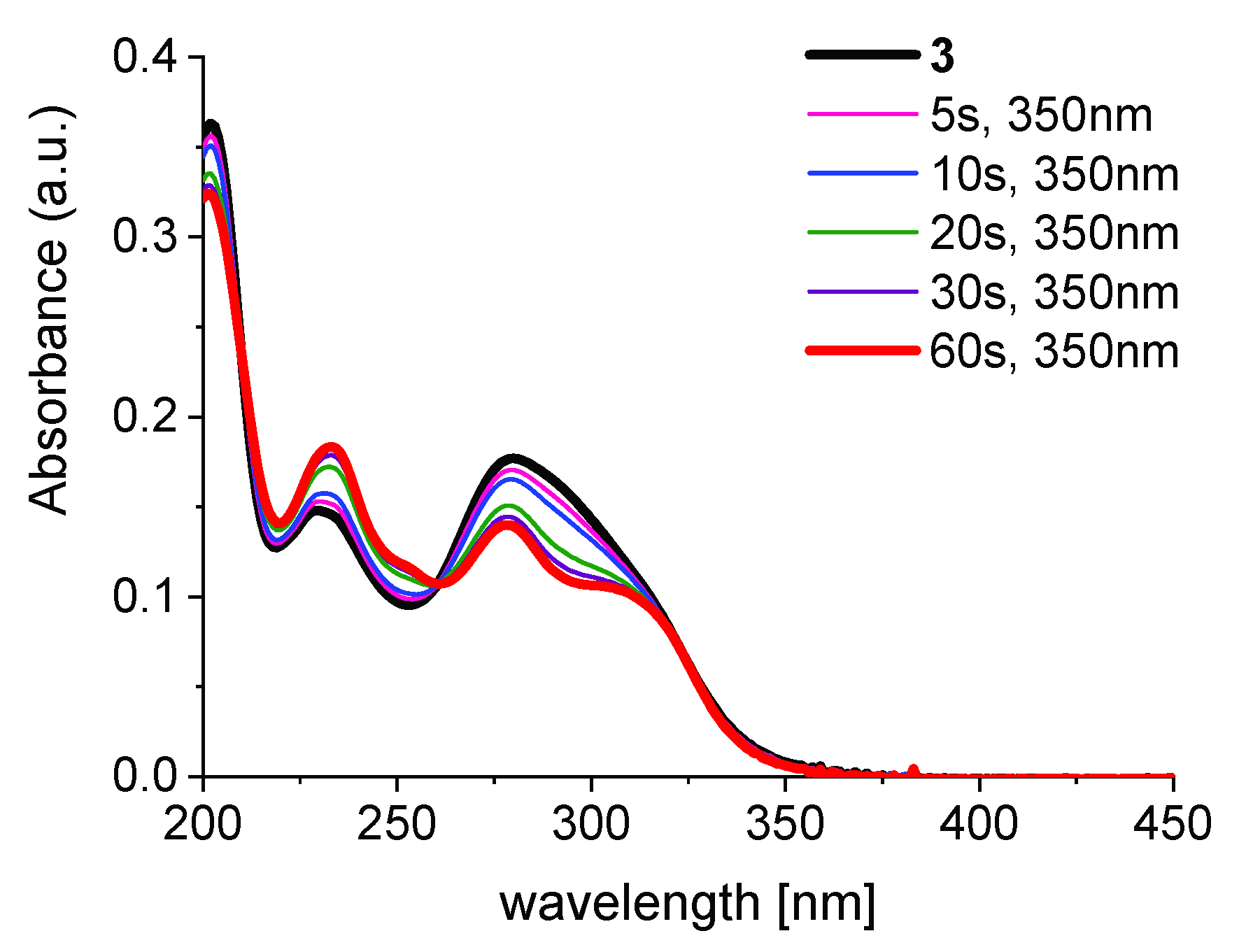
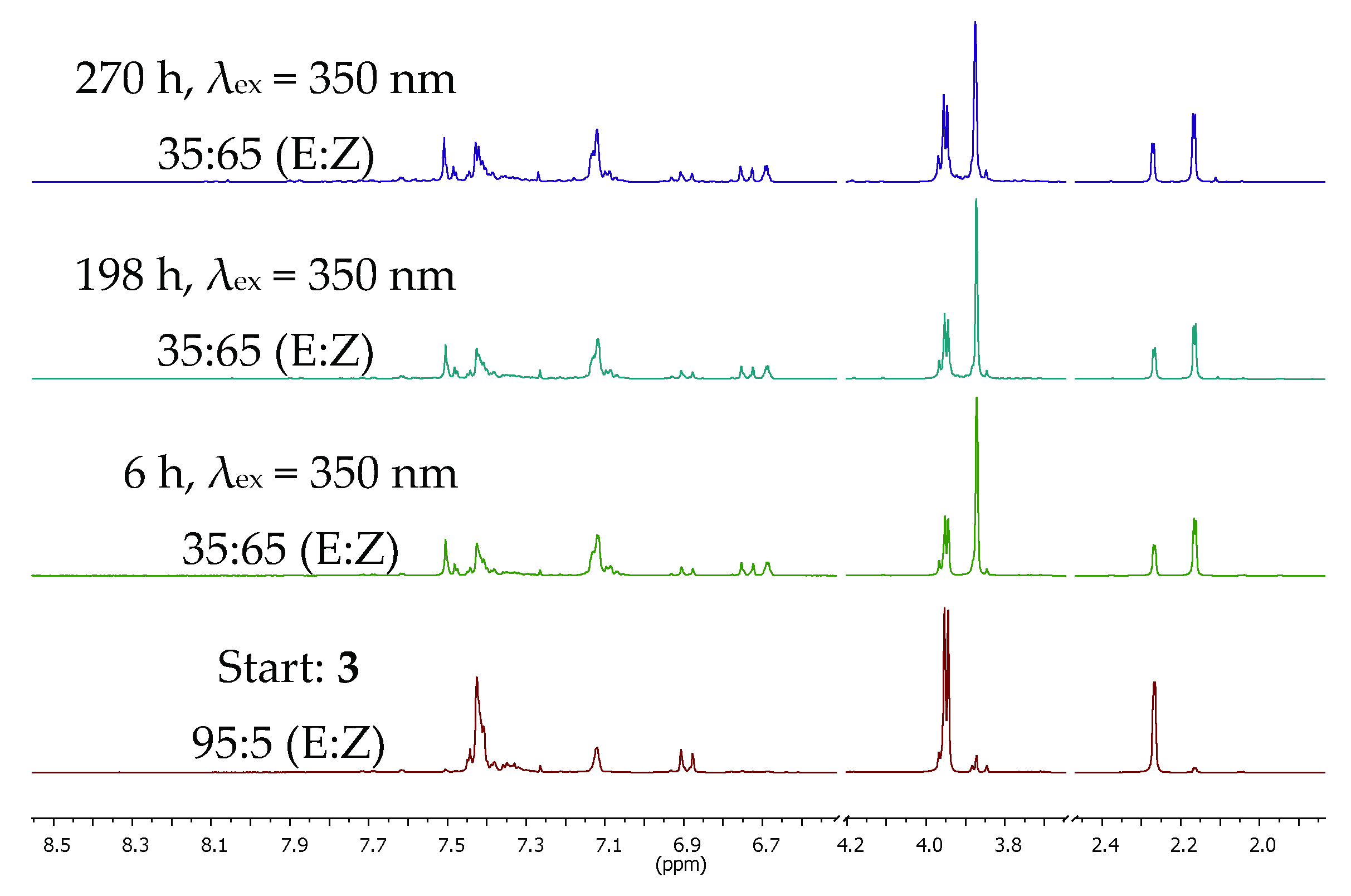
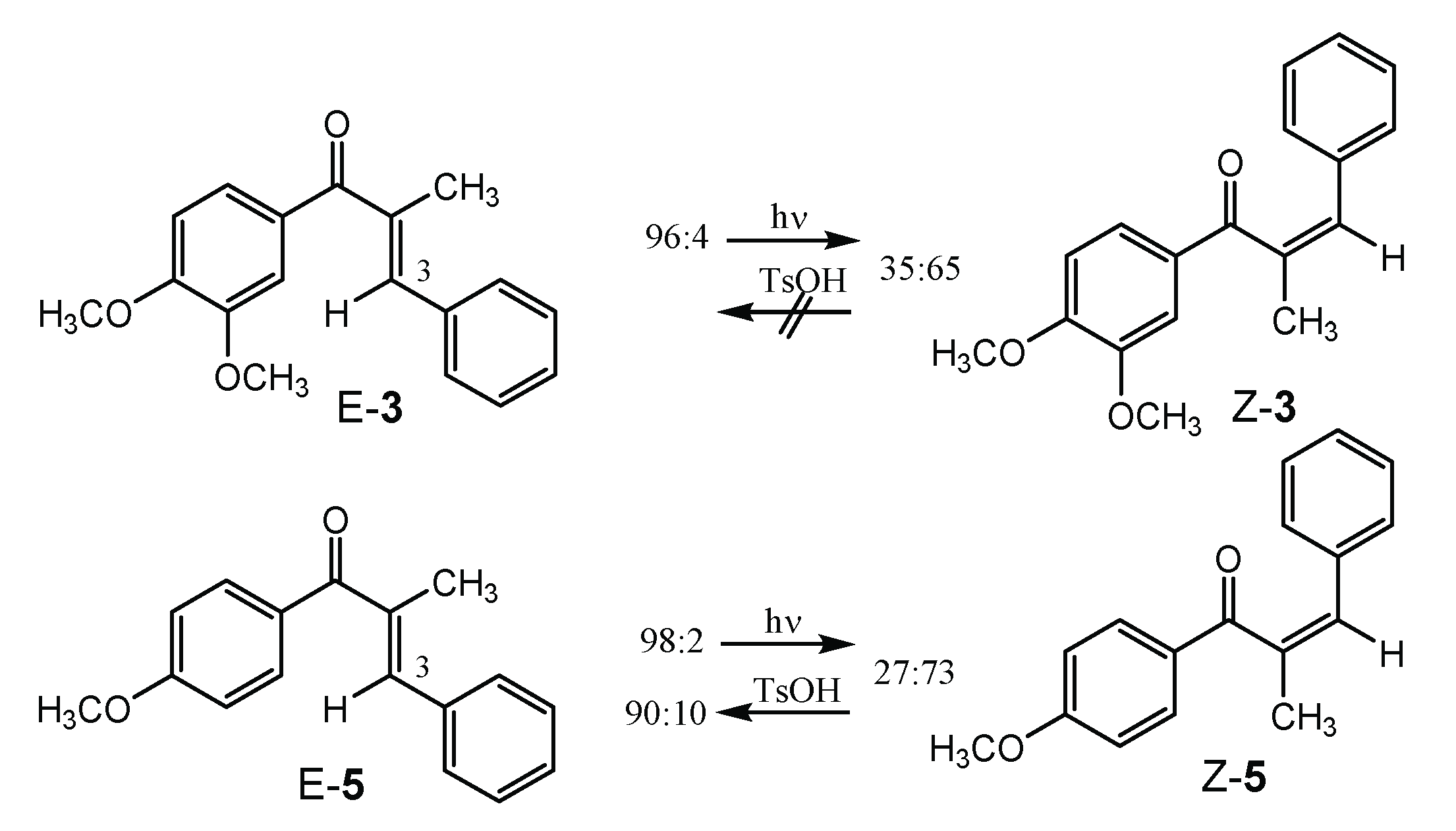
| Excitation Wavelength λex [nm] | PSS [E/Z Ratio, %] | ΦE→Z 1 | ΦZ→E 1 |
|---|---|---|---|
| 254 | 46:54 | - | - |
| 300 | 40:60 | - | - |
| 350 | 35:65 | 0.18 | 0.64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Öngel, B.; Neudörfl, J.M.; Griesbeck, A.G. (E)-1-(3,4-Dimethoxyphenyl)-2-methyl-3-phenylprop-2-en-1-one: A P-Type Acid-Stable Photochromic α-Methylchalcone. Molbank 2021, 2021, M1226. https://doi.org/10.3390/M1226
Öngel B, Neudörfl JM, Griesbeck AG. (E)-1-(3,4-Dimethoxyphenyl)-2-methyl-3-phenylprop-2-en-1-one: A P-Type Acid-Stable Photochromic α-Methylchalcone. Molbank. 2021; 2021(2):M1226. https://doi.org/10.3390/M1226
Chicago/Turabian StyleÖngel, Banu, Jörg M. Neudörfl, and Axel G. Griesbeck. 2021. "(E)-1-(3,4-Dimethoxyphenyl)-2-methyl-3-phenylprop-2-en-1-one: A P-Type Acid-Stable Photochromic α-Methylchalcone" Molbank 2021, no. 2: M1226. https://doi.org/10.3390/M1226
APA StyleÖngel, B., Neudörfl, J. M., & Griesbeck, A. G. (2021). (E)-1-(3,4-Dimethoxyphenyl)-2-methyl-3-phenylprop-2-en-1-one: A P-Type Acid-Stable Photochromic α-Methylchalcone. Molbank, 2021(2), M1226. https://doi.org/10.3390/M1226







