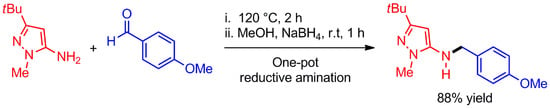
3-(tert-Butyl)-N-(4-methoxybenzyl)-1-methyl-1H-pyrazol-5-amine
Abstract
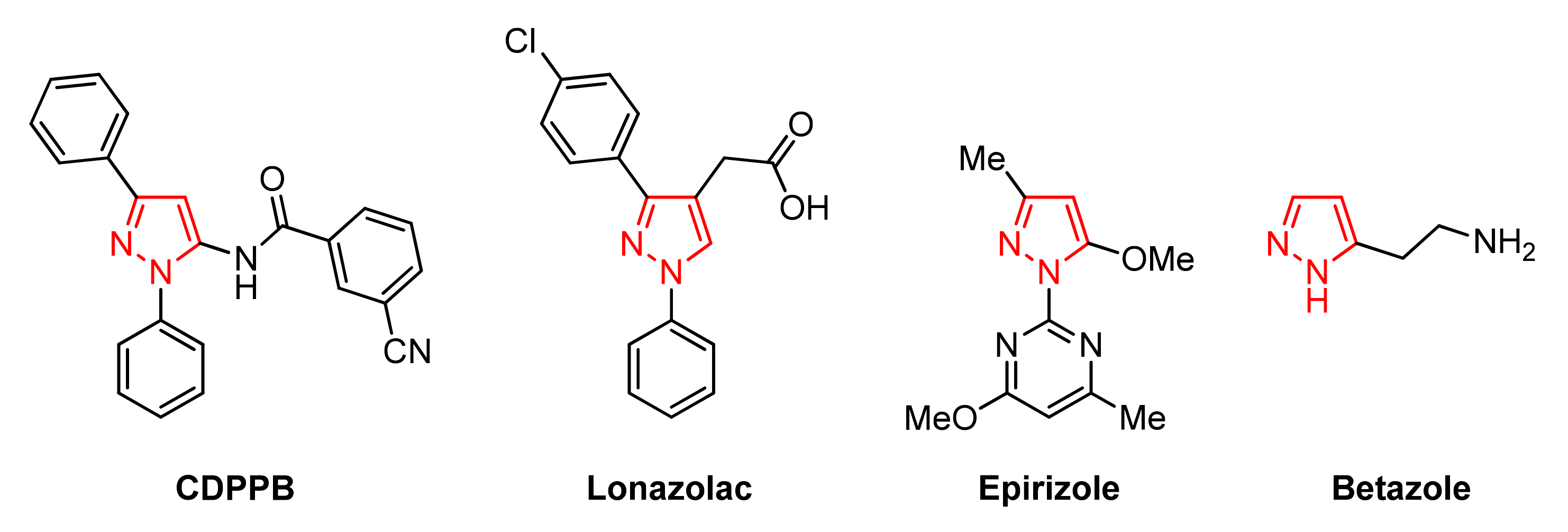
1. Introduction
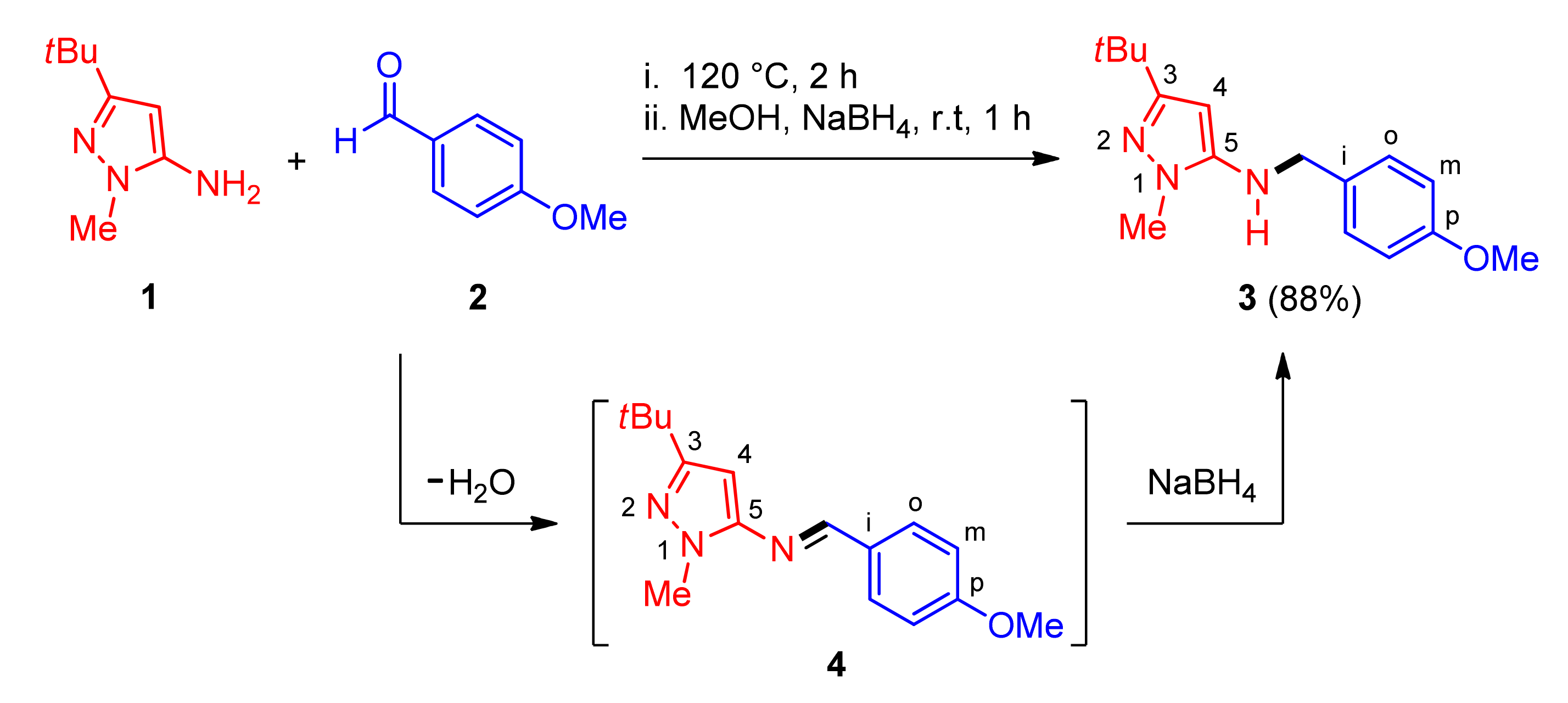
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Synthesis of 3-(tert-Butyl)-N-(4-methoxybenzyl)-1-methyl-1H-pyrazol-5-amine (3)
3.3. Synthesis of (E)-3-(tert-butyl)-N-(4-methoxybenzylidene)-1-methyl-1H-pyrazol-5-amine (4)
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Roughley, S.D.; Jordan, A.M. The medicinal chemist’s toolbox: An analysis of reactions used in the pursuit of drug candidates. J. Med. Chem. 2011, 54, 3451–3479. [Google Scholar] [CrossRef]
- Afanasyev, O.I.; Kuchuk, E.; Usanov, D.L.; Chusov, D. Reductive amination in the synthesis of pharmaceuticals. Chem. Rev. 2019, 119, 11857–11911. [Google Scholar] [CrossRef]
- Kourounakis, A.P.; Xanthopoulos, D.; Tzara, A. Morpholine as a privileged structure: A review on the medicinal chemistry and pharmacological activity of morpholine containing bioactive molecules. Med. Res. Rev. 2020, 40, 709–752. [Google Scholar] [CrossRef] [PubMed]
- Trowbridge, A.; Walton, S.M.; Gaunt, M.J. New strategies for the transition-metal catalyzed synthesis of aliphatic amines. Chem. Rev. 2020, 120, 2613–2692. [Google Scholar] [CrossRef]
- Castillo, J.-C.; Orrego-Hernández, J.; Portilla, J. Cs2CO3-Promoted direct N-alkylation: Highly chemoselective synthesis of N-alkylated benzylamines and anilines. Eur. J. Org. Chem. 2016, 2016, 3824–3835. [Google Scholar] [CrossRef]
- Hahn, G.; Kunnas, P.; de Jonge, N.; Kempe, R. General synthesis of primary amines via reductive amination employing a reusable nickel catalyst. Nat. Catal. 2019, 2, 71–77. [Google Scholar] [CrossRef]
- Cabrero-Antonino, J.R.; Adam, R.; Papa, V.; Beller, M. Homogeneous and heterogeneous catalytic reduction of amides and related compounds using molecular hydrogen. Nat. Commun. 2020, 11, 3893. [Google Scholar] [CrossRef]
- Kumar, V.; Kaur, K.; Gupta, G.M.; Sharma, K.S. Pyrazole containing natural products: Synthetic preview and biological significance. Eur. J. Med. Chem. 2013, 69, 735–753. [Google Scholar] [CrossRef]
- Insuasty, B.; Ramírez, J.; Becerra, D.; Echeverry, C.; Quiroga, J.; Abonia, R.; Robledo, S.M.; Vélez, I.D.; Upegui, Y.; Muñoz, J.A.; et al. An efficient synthesis of new caffeine-based chalcones, pyrazolines and pyrazolo [3,4-b][1,4]diazepines as potential antimalarial, antitrypanosomal and antileishmanial agents. Eur. J. Med. Chem. 2015, 93, 401–413. [Google Scholar] [CrossRef]
- Khan, M.F.; Alam, M.M.; Verma, G.; Akhtar, W.; Akhter, M.; Shaquiquzzaman, M. The therapeutic voyage of pyrazole and its analogs: A review. Eur. J. Med. Chem. 2016, 120, 170–201. [Google Scholar] [CrossRef]
- Ansari, A.; Ali, A.; Asif, M.; Shamsuzzaman, S. Review: Biologically active pyrazole derivatives. New J. Chem. 2017, 41, 16–41. [Google Scholar] [CrossRef]
- Karrouchi, K.; Radi, S.; Ramli, Y.; Taoufik, J.; Mabkhot, Y.N.; Al-aizari, F.A.; Ansar, M. Synthesis and pharmacological activities of pyrazole derivatives: A review. Molecules 2018, 23, 134. [Google Scholar] [CrossRef] [PubMed]
- Bennani, F.E.; Doudach, L.; Cherrah, Y.; Ramli, Y.; Karrouchi, K.; Ansar, M.; Faouzi, M.E.A. Overview of recent developments of pyrazole derivatives as an anticancer agent in different cell line. Bioorg. Chem. 2020, 97, 103470. [Google Scholar] [CrossRef] [PubMed]
- Kleizienė, N.; Arbačiauskienė, E.; Holzer, W.; Šačkus, A. (2E)-3-(3-Methoxy-1-phenyl-1H-pyrazol-4-yl)-2-propenal. Molbank 2009, 2009, M644. [Google Scholar] [CrossRef]
- Kleizienė, N.; Arbačiauskienė, E.; Holzer, W.; Šačkus, A. 4-Bromo-3-methoxy-1-phenyl-1H-pyrazole. Molbank 2009, 2009, M639. [Google Scholar] [CrossRef]
- Chen, B.; Zhu, C.; Tang, Y.; Ma, S. Copper-mediated pyrazole synthesis from 2,3-allenoates or 2-alkynoates, amines and nitriles. Chem. Commun. 2014, 50, 7677–7679. [Google Scholar] [CrossRef] [PubMed]
- Insuasty, D.; Castillo, J.; Becerra, D.; Rojas, H.; Abonia, R. Synthesis of biologically active molecules through multicomponent reactions. Molecules 2020, 25, 505. [Google Scholar] [CrossRef]
- Tigreros, A.; Portilla, J. Recent progress in chemosensors based on pyrazole derivatives. RSC Adv. 2020, 10, 19693–19712. [Google Scholar] [CrossRef]
- Pearce, A.J.; Harkins, R.P.; Reiner, B.R.; Wotal, A.C.; Dunscomb, R.J.; Tonks, I.A. Multicomponent pyrazole synthesis from alkynes, nitriles, and titanium imido complexes via oxidatively induced N–N bond coupling. J. Am. Chem. Soc. 2020, 142, 4390–4399. [Google Scholar] [CrossRef]
- Acosta, P.; Becerra, D.; Goudedranche, S.; Quiroga, J.; Constantieux, T.; Bonne, D.; Rodriguez, R. Exploiting the reactivity of 1,2-ketoamides: Enantioselective synthesis of functionalized pyrrolidines and pyrrolo-1,4-benzodiazepine-2,5-diones. Synlett 2015, 26, 1591–1595. [Google Scholar] [CrossRef]
- Bianchini, G.; Ribelles, P.; Becerra, D.; Ramos, M.T.; Menéndez, J.C. Efficient synthesis of 2-acylquinolines based on an aza-vinylogous Povarov reaction. Org. Chem. Front. 2016, 3, 412–422. [Google Scholar] [CrossRef]
- Moreno-Fuquen, R.; Arango-Daraviña, K.; Becerra, D.; Castillo, J.-C.; Kennedy, A.R.; Macías, M.A. Catalyst- and solvent-free synthesis of 2-fluoro-N-(3-methylsulfanyl-1H-1,2,4-triazol-5-yl)benzamide through a microwave-assisted Fries rearrangement: X-ray structural and theoretical studies. Acta Cryst. 2019, C75, 359–371. [Google Scholar] [CrossRef]
- Abonia, R.; Castillo, J.; Insuasty, B.; Quiroga, J.; Nogueras, M.; Cobo, J. An efficient synthesis of 7-(arylmethyl)-3-tert-butyl-1-phenyl-6,7-dihydro-1H,4H-pyrazolo [3,4-d][1,3]oxazines. Eur. J. Org. Chem. 2010, 2010, 6454–6463. [Google Scholar] [CrossRef]
- Castillo, J.-C.; Quiroga, J.; Abonia, R.; Rodriguez, J.; Coquerel, Y. The aryne aza-Diels–Alder reaction: Flexible syntheses of isoquinolines. Org. Lett. 2015, 17, 3374–3377. [Google Scholar] [CrossRef] [PubMed]
- Castillo, J.-C.; Agudelo, B.C.; Gálvez, J.; Carissan, Y.; Rodriguez, J.; Coquerel, Y. Periselectivity in the aza-Diels–Alder cycloaddition between α-oxoketenes and N-(5-pyrazolyl)imines: A combined experimental and theoretical study. J. Org. Chem. 2020, 85, 7368–7377. [Google Scholar] [CrossRef]
- Fulmer, G.R.; Miller, A.J.M.; Sherden, N.H.; Gottlieb, H.E.; Nudelman, A.; Stoltz, B.M.; Bercaw, J.E.; Goldberg, K.I. NMR chemical shifts of trace impurities: Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist. Organometallics 2010, 29, 2176–2179. [Google Scholar] [CrossRef]
- Bagley, M.; Davis, T.; Dix, M.; Widdowson, C.; Kipling, D. Microwave-assisted synthesis of N-pyrazole ureas and the p38α inhibitor BIRB 796 for study into accelerated cell ageing. Org. Biomol. Chem. 2006, 4, 4158–4164. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Becerra, D.; Rojas, H.; Castillo, J.-C. 3-(tert-Butyl)-N-(4-methoxybenzyl)-1-methyl-1H-pyrazol-5-amine. Molbank 2021, 2021, M1196. https://doi.org/10.3390/M1196
Becerra D, Rojas H, Castillo J-C. 3-(tert-Butyl)-N-(4-methoxybenzyl)-1-methyl-1H-pyrazol-5-amine. Molbank. 2021; 2021(1):M1196. https://doi.org/10.3390/M1196
Chicago/Turabian StyleBecerra, Diana, Hugo Rojas, and Juan-Carlos Castillo. 2021. "3-(tert-Butyl)-N-(4-methoxybenzyl)-1-methyl-1H-pyrazol-5-amine" Molbank 2021, no. 1: M1196. https://doi.org/10.3390/M1196
APA StyleBecerra, D., Rojas, H., & Castillo, J.-C. (2021). 3-(tert-Butyl)-N-(4-methoxybenzyl)-1-methyl-1H-pyrazol-5-amine. Molbank, 2021(1), M1196. https://doi.org/10.3390/M1196