1. Introduction
The miniaturization of electronic devices encourages the creation of multi-level integrated circuits with high functional density. It is possible due to the use of low-dielectric-constant (low-k) materials incorporated into interconnected media. Traditional dielectric materials based on inorganic silica are inferior to organic polymer materials due to their significant advantages in solution processability and mechanical flexibility [
1,
2,
3,
4].
Organosilicon resins with benzocyclobutene (
BCB) filler have been demonstrated to be promising as low-dielectric-constant (low-k) materials for the electronics industry and related applications. These materials are currently being actively studied [
1,
2,
3,
4,
5,
6].
In particular, Dow Chemicals (
www.dow.com) presents a line of composite commercial materials (CYCLOTENE
TM) based on divinyltetramethyldisiloxane-benzocyclobutene (
DVS-BCB) as photoresists and thermosets.
Recently, a direction has been developed associated with the use of multi-benzocyclobutene functionalized silane and siloxane monomers, which provide more dense crosslinking, high thermal stability, and low coefficient of thermal expansion. In particular, polymers are known based on tri-(benzocyclobuten-4-yl) phenylsilane and tetra-(benzocyclobuten-4-yl)silane [
7] or their siloxane analogs [
6]. However, it is a surprise that there are no data about the synthesis or physical and chemical properties of a simpler analogue, namely di(bicyclo[4.2.0]octa-1(6),2,4-trien-3-yl)dimethylsilane.
2. Results
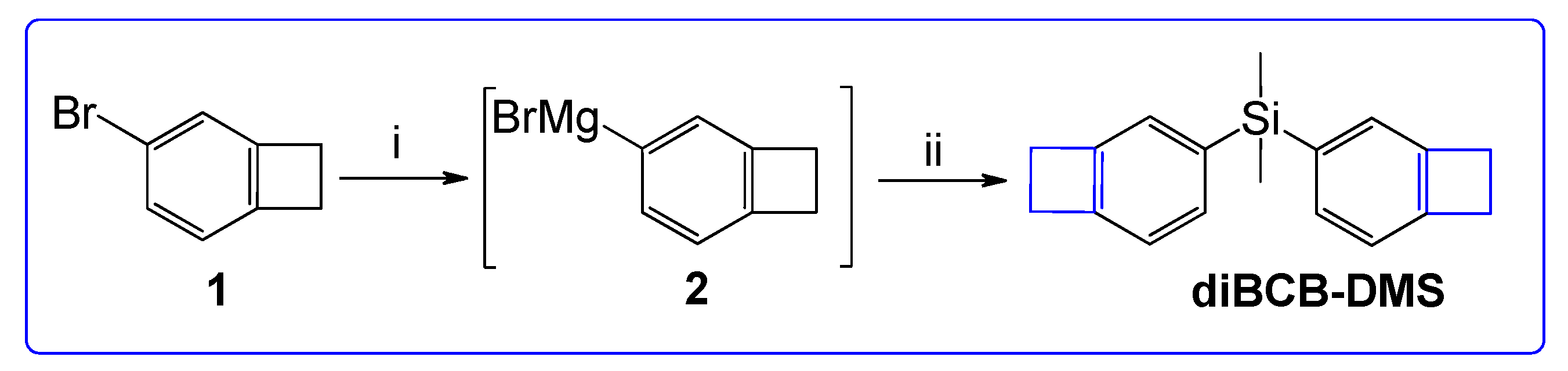
In this paper, we present a simple preparation procedure and the physical properties of a thermopolymerizable monomer containing two benzocyclobutene and dimethylsilane fragments (di(bicyclo[4.2.0]octa-1(6),2,4-trien-3-yl)dimethylsilane). The synthesis involves a two-stage process starting from 4-bromobenzocyclobutene obtained by a previously described method [
8,
9]. In the first stage, 4-bromobenzocyclobutene (
1) reacts with magnesium in the presence of iodine in anhydrous tetrahydrofuran to form a Grignard reagent (2) (
Figure 1). Further addition of dichlorodimethylsilane under ice-cooling (0–10 °C) leads to the formation of the target product
diBCB-DMS in 70% yield. The product was purified by distillation under reduced pressure (150–160 °C at 0.5–0.7 mbar) and obtained as a transparent colorless liquid crystallizing in the refrigerator.
DiBCB-DMS was characterized by
1H,
13C NMR, IR, UV/VIS, and HRMS analysis. These data can be found in the
Supplementary Materials.
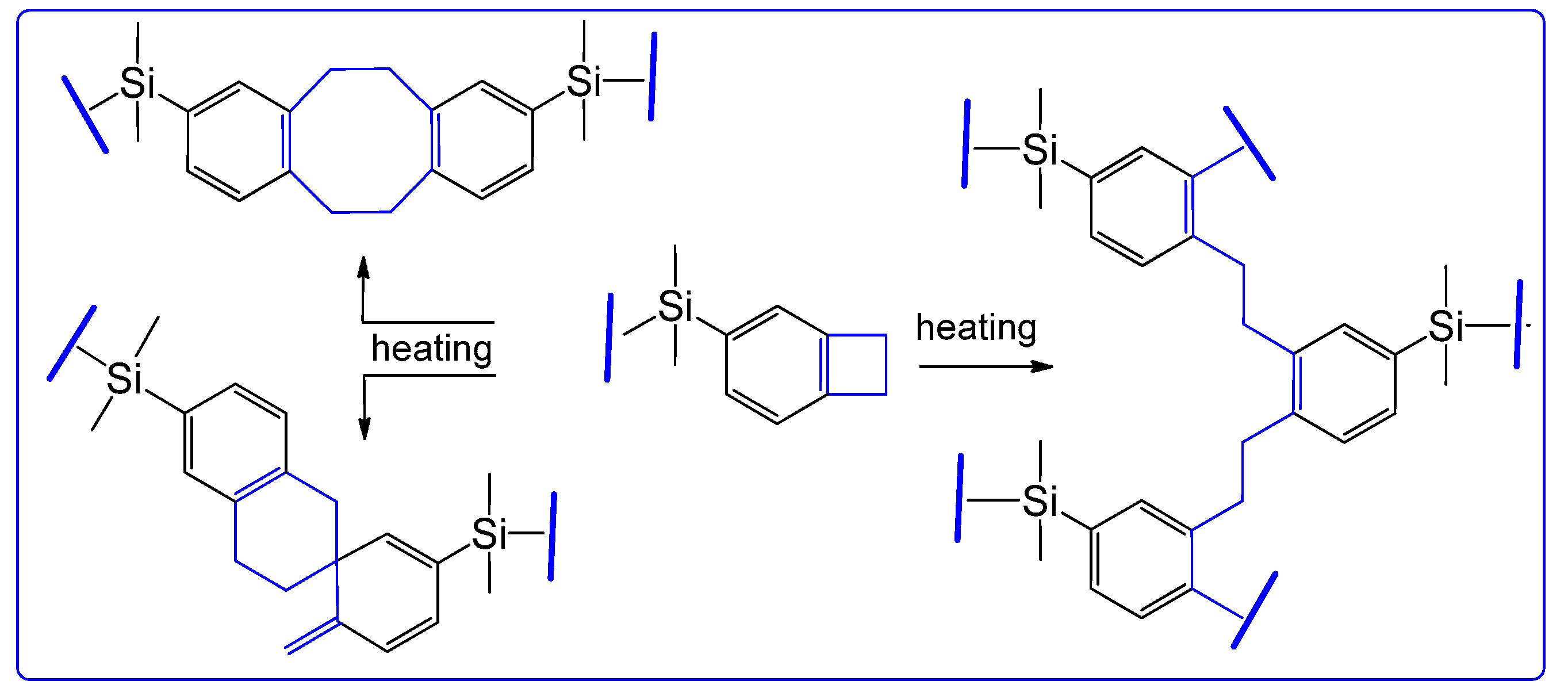
The resulting monomer
diBCB-DMS is polymerized when heated above 200 °C. It is known that the mechanism of polymerization of benzocyclobutene derivatives is multivariable [
2,
4,
6,
7,
10]. Benzocyclobutene fragments in thermo-induced o-xylylene form can react with active double bonds. Thermopolymerization in this case proceeds according to the [4+2] cycloaddition mechanism. In the absence of compounds with active double bonds in the reaction mass, the benzocyclobutene ring opens and reacts with another similar fragment in different ways. The scheme of the estimated polymerization processes is shown in
Figure 2.
The colorless liquid monomer diBCB-DMS gave a brown solid polymer upon heating at 250 °C for 30 min in air. Color change and characteristic adsorption bands of the polymer indicated that remarkable oxidative processes occurred. A decrease in mass was also observed. Therefore, a step-by-step polymerization process (160 °C—1 h, 180 °C—2 h, 200 °C—2 h, 220 °C—2 h, and 240 °C—30 min) under Ar was applied, giving a white to light yellow polymer without decrease in mass.
The resulting polymers were studied by IR spectroscopy. IR spectra of the monomer and polymers are presented in the
Supplementary Materials. According to the literature [
11,
12], the band at 1466 cm
−1 characterizes the vibrations of the CH
2 groups of the cyclobutene ring in monomer molecules and the band at 1493 cm
−1 characterizes the vibrations of dibenzocyclooctadiene and tetrahydronaphthalene structures in the crosslinked polymer. Moreover, characteristic adsorption bands of carbonyl at about 1600–1776 cm
−1 and OH groups at >3500 cm
−1 occurred, indicating the oxidation of cyclobutene moieties. The oxidation may occur primarily at the tetrahydronaphthalene group formed during the Diels–Alder reaction of BCB [
13].
We assume that the polymerization of diBCB-DMS proceeds, giving a crosslinked polymer. This fact is indirectly confirmed by high resistance of the obtained polymer to the action of organic solvents and mechanical strength at high temperatures.
3. Materials and Methods
All reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA) (Lancaster, UK) and used without further purification unless otherwise stated. Dimethyldichlorosilane was purchased from abcr GmbH (Karlsruhe, Germany).
NMR spectra were registered using a Bruker AM-300 spectrometer (Bruker Corporation, Billerica, MA, USA) in CDCl3. Mass spectra were obtained on a Varian MAT CH-6 instrument (Varian, Inc., Palo Alto, CA, USA) using a direct inlet system; the ionization energy was 70 eV and the acceleration voltage was 1.75 kV. The reaction mixtures were analyzed and the purity of all products was checked by TLC on Merck Silica gel 60 F254 UV-254 plates (Darmstadt, Germany). The optical properties of diBCB-DMS were studied in acetonitrile solution (Sigma-Aldrich, St. Louis, MO, USA) using a Lambda 35 UV/VIS spectrometer (PerkinElmer, Singapore). Absorption spectra were measured at a concentration of 0.015–0.003 mg/mL and an optical path length of 0.2 cm.
4. Synthesis of diBCB-DMS
4-Bromobenzocyclobutene (18.3 g, 0.1 mol) was added dropwise with vigorous stirring to magnesium powder (2.4 g, 0.1 mol), activated by the addition of an iodine crystal in 100 mL of freshly distilled dry tetrahydrofuran. After the start of the reaction, 4-bromobenzocyclobutene was added at such a rate that the temperature of the reaction mixture was 40–45 °C. When the addition was complete, the reaction mass was stirred for 2 h at this temperature and then was cooled to 0 °C. Dichlorodimethylsilane (6.4 g, 0.1 mol) was added dropwise at 0–10 °C and the mixture was allowed to warm to room temperature. The next day, the mixture was diluted with hexane (100 mL). The precipitate was filtered off and, washed with several portions of hexane. The filtrate was concentrated on a rotary evaporator. The remaining crude product was purified by vacuum distillation at a distillation temperature of 150–160 °C (0.5–0.7 mbar). The yield was 9.2 g (70%). diBCB-DMS is a colorless liquid solidifying in the refrigerator. 1H NMR (300 MHz, CDCl3) δ 7.62 (d, J = 7.3 Hz, 2H), 7.46 (s, 2H), 7.28 (d, J = 7.3 Hz, 2H), 3.40 (s, J = 3.4 Hz, 8H), 0.76 (s, 6H). 13C NMR (76 MHz, CDCl3) δ 147.30, 145.75, 136.85, 132.73, 128.16, 122.13, 30.14, 30.01, −1.67. HRMS (+MS) found: 265.1414; calculated: 265.1407.
5. Conclusions
New di(bicyclo[4.2.0]octa-1(6),2,4-trien-3-yl)dimethylsilane (DiBCB-DMS) was obtained via a two-stage synthesis starting from 4-bromobenzocyclobutene with good yields. DiBCB-DMS was characterized by 1H, 13C NMR, IR, UV/VIS, and HRMS analysis. The obtained product can be useful for the development of new polymerizable composite materials with good thermal stability and dielectric properties.
Supplementary Materials
Supplementary data to this article can be found online. These data include MOL file, 1H, 13C NMR, HRMS, UV/VIS, and IR of the title compound.
Author Contributions
Synthesis, K.S.L.; HRMS analysis, K.A.C.; IR data analysis, D.Y.D.; UV/VIS analysis, P.A.C.; NMR analysis, P.S.S.; polymerization, G.E.A. Writing—original draft preparation, K.S.L.; Writing—review and editing, K.S.L. and P.S.S.; Supervision and project administration, E.P.G. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation. Agreement on the provision of grants from the federal budget in the form of subsidies dated 22 November 2019 No. 075-15-2019-1694 (internal number of the Agreement 05.604.21.0227), unique identifier RFMEFI60419X0227.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Huang, Y.; Zhang, Q.; Peng, Q.; Hu, H.; Yu, H.; Yang, J.; Liu, L. Low-dielectric-constant benzocyclobutene–Organosilicon resins constructed from cyclotetrasiloxane. J. Appl. Polym. Sci. 2019, 136, 47465. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, S.; Hu, H.; Wei, X.; Yu, H.; Yang, J. Synthesis of poly(silmethylene)s via ring-opening polymerization of benzocyclobutene functionalized disilacyclobutene and their low-dielectric and thermal properties. Polym. Adv. Technol. 2017, 28, 1480–1488. [Google Scholar] [CrossRef]
- Wu, K.; Wang, Z. High-Frequency Characterization of Through-Silicon-Vias with Benzocyclobutene Liners. IEEE Trans. Compon. Packag. Manuf. Technol. 2017, 7, 1859–1868. [Google Scholar] [CrossRef]
- Yang, J.; Huang, Y.; Cao, K. Recent Progress in Benzocyclobutene Related Polymers; De Ailton, S.G., Ed.; Books on Demand: Norderstedt, Germany, 2012; pp. 201–222. [Google Scholar]
- Levchenko, K.S.; Chudov, K.A.; Demin, D.; Yu Adamov, G.E.; Poroshin, N.O.; Shmelin, P.S.; Grebennikov, E.P.; Chvalun, S.N.; Zubov, V.P. Synthesis of photo and thermosetting monomers and polymers based on benzocyclobutene. Russ. Chem. Bull. 2019, 68, 1321–1342. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Z.; Zhu, T.; Li, Z.; Wang, J.; Cheng, Y. Multi-benzocyclobutene functionalized siloxane monomers prepared by Piers-Rubinsztajn reaction for low-k materials. Eur. Polym. J. 2020, 126, 109562. [Google Scholar] [CrossRef]
- Cheng, Y.; Cai, J.; Li, J.; Wu, X.; Shi, Y.; Wang, J. Multibenzocyclobutene Functionalized Silane for Low-k Polyarylsilane Thermosets with Low Coefficient of Thermal Expansion and High Thermostability. ACS Appl. Polym. Mater. 2019, 1, 2622–2626. [Google Scholar] [CrossRef]
- Levchenko, K.S.; Chudov, K.A.; Demin, D.Y.; Shmelin, P.S.; Grebennikov, E.P. Bicyclo[4.2.0]octa-1,3,5-trien-3-yl-dimethyl((E)-styryl)-silane. Molbank 2020, 2020, M1102. [Google Scholar] [CrossRef]
- Levchenko, K.S.; Chudov, K.A.; Demin, D.Y.; Lyssenko, K.A.; Shmelin, P.S. 2-(Bicyclo[4.2.0]octa-1,3,5-trien-3-yl)-adamantan-2-ol. Molbank 2020, 2020, M1106. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, S.; Hu, H.; Wei, X.; Yu, H.; Yang, J. Photoactive Polymers with Benzocyclobutene/Silacyclobutane Dual Crosslinked Structure and Low Dielectric Constant. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 1920–1928. [Google Scholar] [CrossRef]
- Stokich, T.M.; Lee, W.M.; Peters, R.A. Real-time FT-IR studies of the reaction kinetics for the polymerization of divinyl siloxane bis-benzocyclobutene monomers. Mater. Sci. High Temp. Polym. Microelectron. 1991, 227, 103–114. [Google Scholar] [CrossRef]
- Yang, J.; Xie, L.; Zhu, F.; Sui, H.; Li, H.; Huang, Y. Incorporation of Benzocyclobutene Cross-Linkable Moieties in Poly(Methyl Acrylate): A Novel Approach to Shape-Memory Polymers Accompanied with Microphase Separation. J. Macromol. Sci. B 2011, 50, 2129–2139. [Google Scholar] [CrossRef]
- Yang, J.; Liu, S.; Zhu, F.; Huang, Y.; Li, B.; Zhang, L. New polymers derived from 4-vinylsilylbenzocyclobutene monomer with good thermal stability, excellent film-forming property, and low-dielectric constant. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 381–391. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).