3-Hydroxy-8,14-secogammacera-7,14-dien-21-one: A New Onoceranoid Triterpenes from Lansium domesticum Corr. cv kokossan
Abstract
1. Introduction
2. Results
Extraction and Isolation
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
4.3. Cytotoxic Bioassay
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Techavuthiporn, C. Langsat–Lansium Domesticum. Exotic Fruits Reference Guides; Rodrigues, S., de Brito, E.S., Silva, E.d.O., Eds.; Academic Press: London, UK, 2018; pp. 279–283. [Google Scholar]
- Mayanti, T.; Tjokronegoro, R.; Supratman, U.; Mukhtar, M.R.; Awang, K.; Hadi, A.H.A. Antifeedant triterpenoids from the seeds and bark of Lansium domesticum cv kokossan (Meliaceae). Molecules 2011, 16, 2785–2795. [Google Scholar] [CrossRef]
- Dong, S.H.; Zhang, C.R.; Dong, L.; Wu, Y.; Yue, J.M. Onoceranoid-type triterpenoids from Lansium domesticum. J. Nat. Prod. 2011, 74, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, M.; Nishide, H.; Hayashi, Y.; Kosela, S. The structure of lansioside A: A novel triterpene glycoside with amino-sugar from Lansium domesticum. Tetrahedron Lett. 1982, 23, 1349–1350. [Google Scholar] [CrossRef]
- Nishizawa, M.; Nishide, H.; Kosela, S.; Hayashi, Y. Structure of lansiosides: Biologically active new triterpene glycosides from Lansium domesticum. J. Org. Chem. 1983, 48, 4462–4466. [Google Scholar] [CrossRef]
- Tanaka, T.; Ishibashi, M.; Fujimoto, H.; Okuyama, E.; Koyano, T.; Kowiyhayakorn, T.; Hayashi, M.; Komiyama, K. New onoceranoid constituents from Lansium domesticum. J. Nat. Prod. 2002, 65, 1709–1711. [Google Scholar] [CrossRef] [PubMed]
- Habaguchi, K.; Watanabe, M.; Nakadaira, Y.; Nakanishi, K.; Kaing, A.K.; Lim, F.L. The full structures of lansic acid and its minor congener, an unsymmetric onoceradienedione. Tetrahedron Lett. 1986, 34, 3731–3734. [Google Scholar] [CrossRef]
- Mayanti, T.; Supratman, U.; Mukhtar, M.R.; Awang, K.; Ng, S.W. Kokosanolide from the seed of Lansium domesticum Corr. Acta Crystallogr. 2009, E65, o750. [Google Scholar]
- Supratman, U.; Mayanti, T.; Awang, K.; Mukhtar, M.R.; Ng, S.W. 14-Hydroxy-8,14-secogammacera-7-ene-3,21-dione from the bark of Lansium domesticum Corr. Acta Crystallogr. 2010, E66, o1621. [Google Scholar]
- Manosroi, A.; Jantrawut, P.; Sainakham, M.; Manosroi, W.; Manosroi, J. Anticaner activities of the extract from longkong (Lansium domesticum) young fruits. Pharm. Biol. 2012, 50, 1397–1407. [Google Scholar] [CrossRef] [PubMed]
- Ragasa, C.Y.; Labrador, P.; Rideout, J.A. Antimicrobial terpenoid from Lansium domesticum. Philipp. Agric. Sci. 2006, 89, 101–105. [Google Scholar]
- Leatemia, J.A.; Isman, M.B. Insecticidal activity of crude seed extracts of Annona. spp., Lansium domesticum and Sandoricum koetjape against lepidopteran larvae. Phytopatasitica 2004, 32, 30–37. [Google Scholar] [CrossRef]
- Saewan, N.; Sutherland, J.D.; Chantrapromma, K. Antimalarial tetranortriterpenoids from the seed of Lansium domesticum Corr. Phytrochemistry 2006, 67, 2288–2293. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Kitagawa, T.; Teo, S.; Anai, Y.; Ikeda, R.; Imahori, D.; Ahmad, H.S.; Watanabe, T. Structures and antimutagenic effects of onoceranoid-type triterpenoids from the leaves of Lansium domesticum. J. Nat. Prod. 2018, 81, 2187–2194. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Kitagawa, T.; Ohta, T.; Yoshida, T.; Imahori, D.; Teo, S.; Ahmad, H.S.; Watanabe, T. Structures of triterpenoids from the leaves of Lansium domesticum. J. Nat. Med. 2019, 73, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Mayanti, T.; Sianturi, J.; Harneti, D.; Darwati; Supratman, U.; Rosli, M.M.; Fun, H.K. 9,19-Cyclolanost-24-en-3-one,21,23-epoxy-21,22-dihydroxy (21R, 22S, 23S) from the leaves of Lansium domesticum Corr cv kokossan. Molbank 2015, 2015, M880. [Google Scholar] [CrossRef]
- Hou, Y.; Cao, S.; Brodie, P.J.; Miller, J.S.; Birkinshaw, C.; Andrianjafy, M.N.; Andriantsiferana, R.; Rasamison, V.E.; Tendyke, K.; Shen, Y.; et al. Euphane triterpenoids of Cassipourea lanceolata from the Madagascar rainforest. Phytochemistry 2010, 71, 669–674. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1 are available from the authors. |
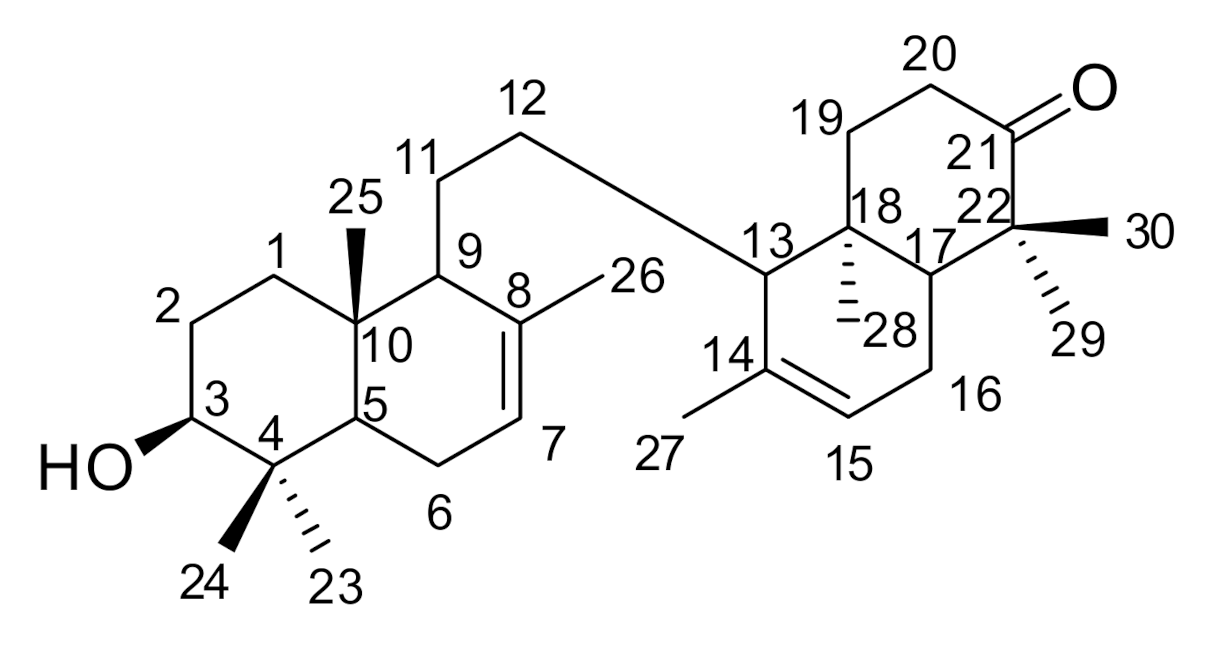

| Position | δC ppm | δH ppm (Int, mult., J = Hz) |
|---|---|---|
| 1 | 37.5 | 1.14; 1.86 (2H, m) |
| 2 | 27.4 | 1.65 (2H, m) |
| 3 | 79.1 | 3.29 (1H, dd, 11.3; 4.3) |
| 4 | 38.7 | - |
| 5 | 51.5 | 1.62 (1H, m) |
| 6 | 29.8 | 1.35; 1.50 (2H, m) |
| 7 | 121.7 | 5.40 (1H, brs) |
| 8 | 135.4 | - |
| 9 | 55.3 | 1.66 (1H, m) |
| 10 | 36.5 | - |
| 11 | 24.1 | 1.99; 2.10 (2H, m) |
| 12 | 23.5 | 1.98; 2.10 (2H, m) |
| 13 | 56.0 | 1.59 (1H, m) |
| 14 | 134.9 | - |
| 15 | 122.3 | 5.40 (1H, brs) |
| 16 | 29.9 | 1.35; 1.50 (2H, m) |
| 17 | 49.6 | 1.20 (1H, m) |
| 18 | 36.5 | - |
| 19 | 38.4 | 1.51; 2.15 (2H, m) |
| 20 | 34.7 | 2.29; 2.77 (2H, m) |
| 21 | 217.0 | - |
| 22 | 47.5 | - |
| 23 | 27.9 | 1.00 (3H, s) |
| 24 | 15.1 | 0.87 (3H, s) |
| 25 | 13.3 | 0.99 (3H, s) |
| 26 | 22.3 | 1.75 (3H, s) |
| 27 | 22.4 | 1.72 (3H, s) |
| 28 | 13.6 | 0.76 (3H, s) |
| 29 | 22.1 | 1.12 (3H, s) |
| 30 | 25.0 | 1.08 (3H, s) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zulfikar; Putri, N.K.; Fajriah, S.; Yusuf, M.; Maharani, R.; Anshori, J.A.; Supratman, U.; Mayanti, T. 3-Hydroxy-8,14-secogammacera-7,14-dien-21-one: A New Onoceranoid Triterpenes from Lansium domesticum Corr. cv kokossan. Molbank 2020, 2020, M1157. https://doi.org/10.3390/M1157
Zulfikar, Putri NK, Fajriah S, Yusuf M, Maharani R, Anshori JA, Supratman U, Mayanti T. 3-Hydroxy-8,14-secogammacera-7,14-dien-21-one: A New Onoceranoid Triterpenes from Lansium domesticum Corr. cv kokossan. Molbank. 2020; 2020(4):M1157. https://doi.org/10.3390/M1157
Chicago/Turabian StyleZulfikar, Nurul Kamila Putri, Sofa Fajriah, Muhammad Yusuf, Rani Maharani, Jamaludin Al Anshori, Unang Supratman, and Tri Mayanti. 2020. "3-Hydroxy-8,14-secogammacera-7,14-dien-21-one: A New Onoceranoid Triterpenes from Lansium domesticum Corr. cv kokossan" Molbank 2020, no. 4: M1157. https://doi.org/10.3390/M1157
APA StyleZulfikar, Putri, N. K., Fajriah, S., Yusuf, M., Maharani, R., Anshori, J. A., Supratman, U., & Mayanti, T. (2020). 3-Hydroxy-8,14-secogammacera-7,14-dien-21-one: A New Onoceranoid Triterpenes from Lansium domesticum Corr. cv kokossan. Molbank, 2020(4), M1157. https://doi.org/10.3390/M1157







