Microwave-Assisted Synthesis and Fluorescent Properties of 4-Phenyl-1,8-naphthalimide
Abstract
1. Introduction
2. Results and Discussion
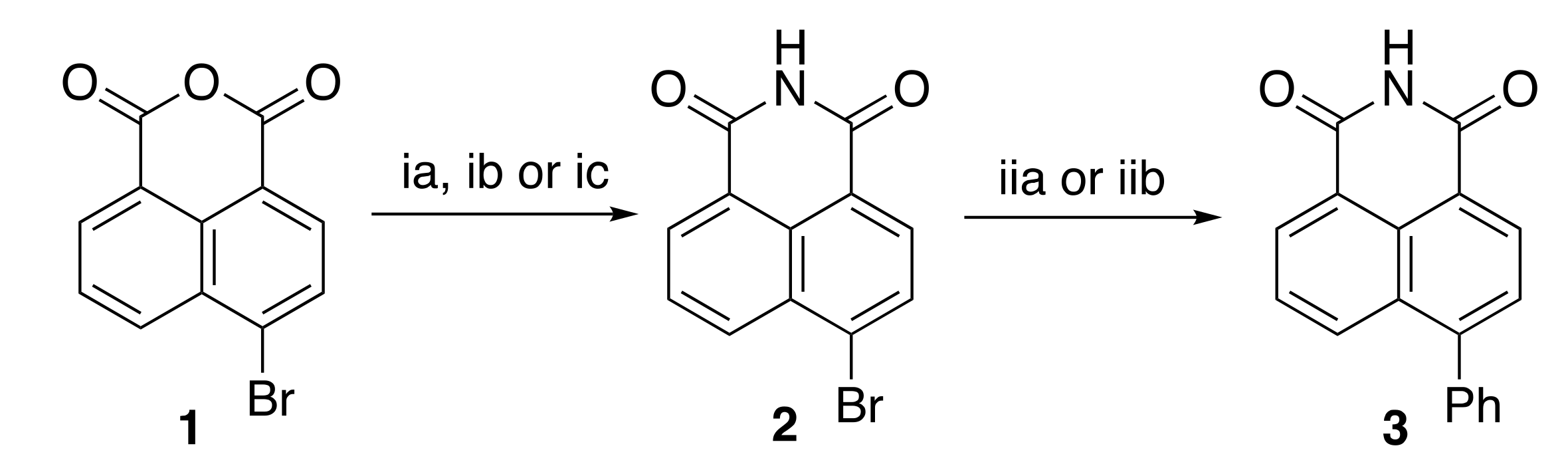
2.1. Synthesis of Compounds
2.2. Solvent-Dependent Absorption and Emission Spectroscopic Properties of 4-Phenyl-1,8-naphthalimide
2.3. Fluorescent Emission of 4-Phenyl-1,8-naphthalimide in Aqueous-Organic Solvent Mixtures
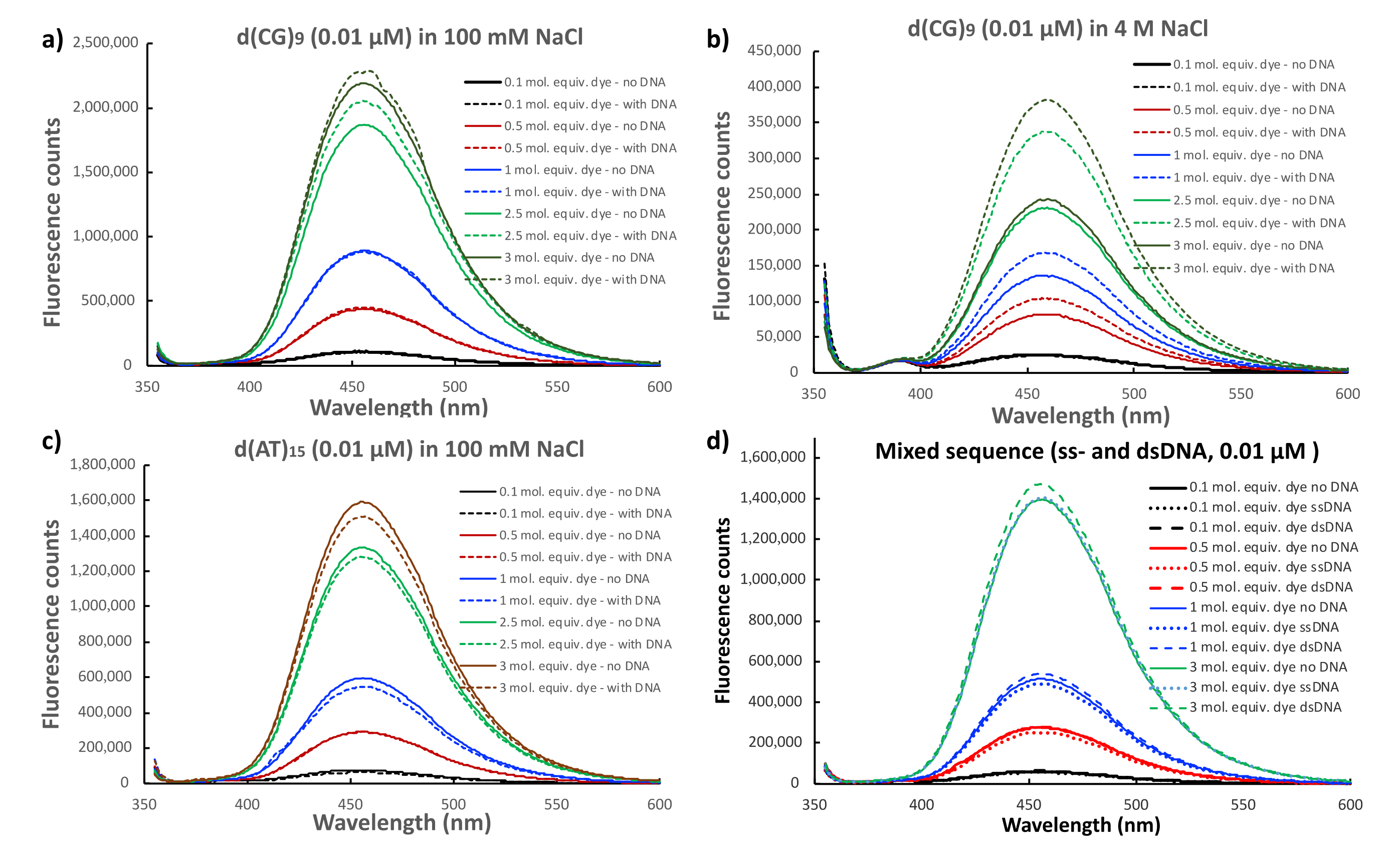
2.4. Fluorescent Properties of 4-Phenyl-1,8-naphthalimide in the Presence of DNA
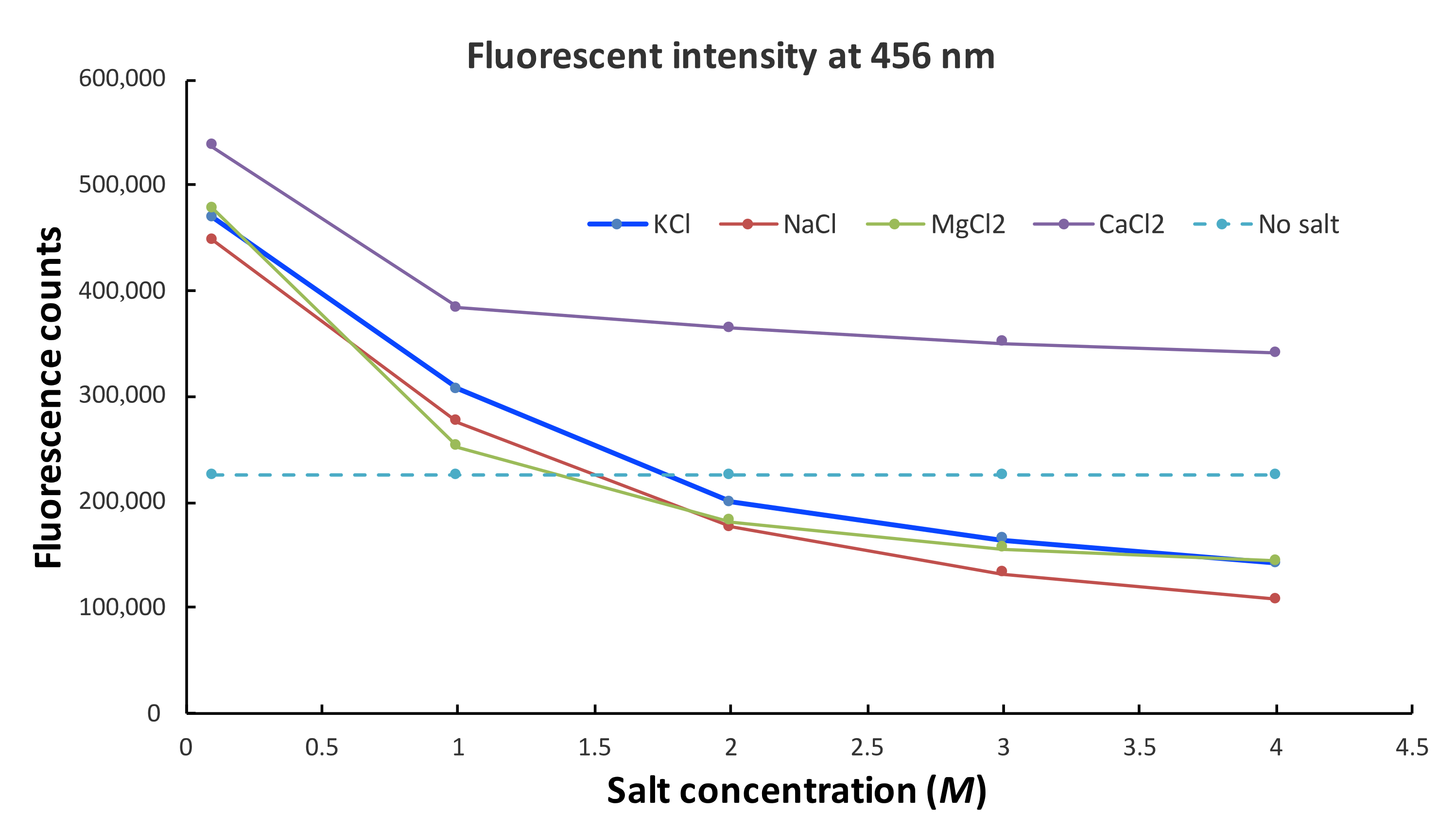
2.5. Salt Concentration-Dependent Fluorescence
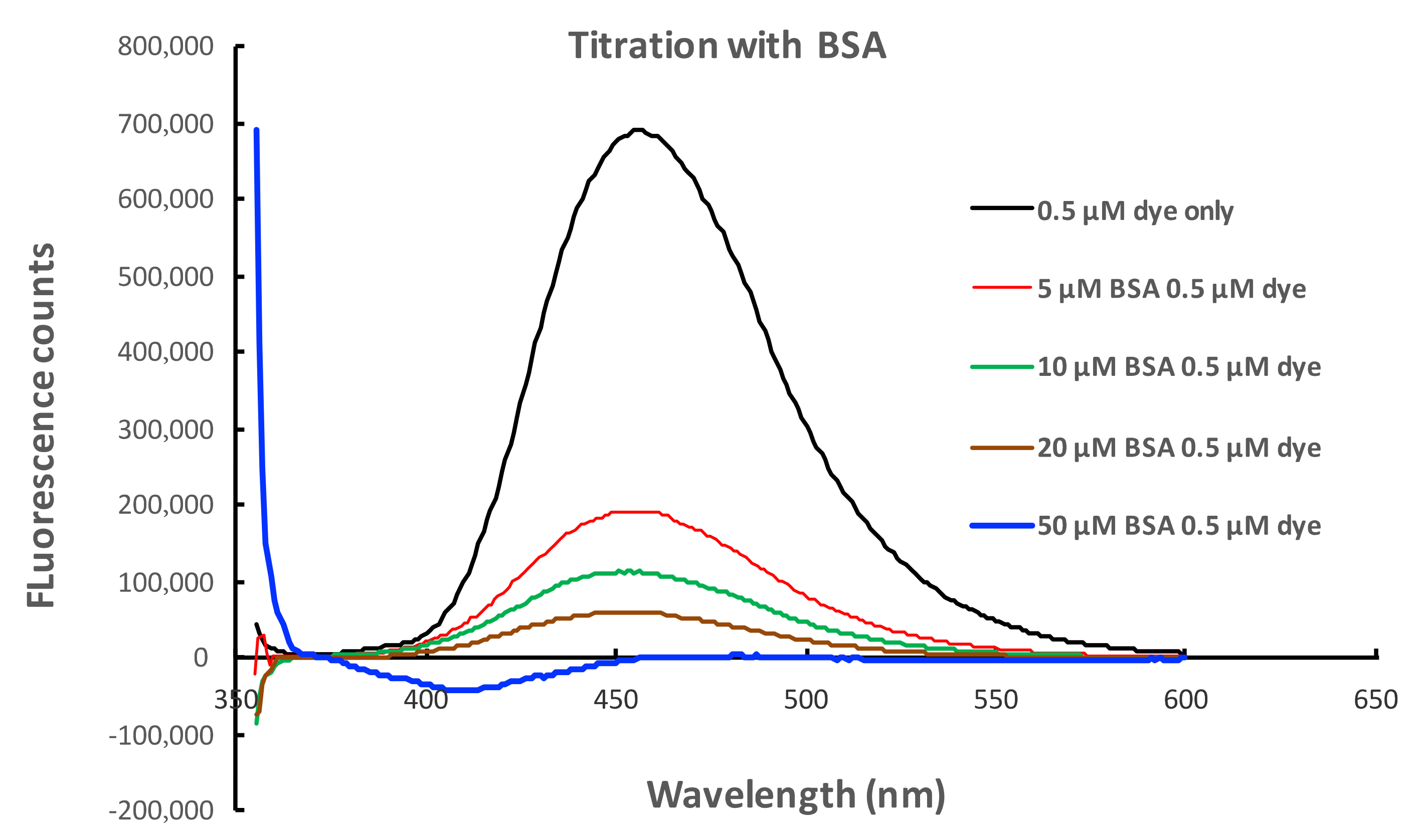
2.6. Fluorescent Properties of 4-Phenyl-1,8-naphthalimide in the Presence of Bovine Serum Albumin
3. Materials and Methods
3.1. Instrumentation
3.2. Chromatography
3.3. Solvents and Chemicals
3.4. UV/vis and Fluorescent Spectroscopy
3.5. Synthesis of 4-Bromo-1,8-naphthalimide
3.5.1. Method A (Conventional Heating with Ammonium Acetate)
3.5.2. Method B (Conventional Heating with Ammonia in Dioxane)
3.5.3. Method C (Microwave Heating with Ammonium Acetate)
3.6. Synthesis of 4-Phenyl-1,8-naphthalimide
3.6.1. Method A (Conventional Heating)
3.6.2. Method B (Microwave Heating)
3.7. Fluorescent Quantum Yield Measurement
3.8. DNA Binding
3.9. Fluorescence Measurement of 4-Phenyl-1,8-naphthalimide in Inorganic Salts of Different Concentration
3.10. BSA Binding
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Aderinto, S.O.; Imhanria, S. Fluorescent and colourimetric 1,8-naphthalimide-appended chemosensors for the tracking of metal ions: Selected examples from the year 2010 to 2017. Chem. Pap. 2018, 72, 1823–1851. [Google Scholar] [CrossRef]
- Panchenko, P.A.; Fedorova, O.A.; Fedorov, Y.V. Fluorescent and colorimetric chemosensors for cations based on 1,8-naphthalimide derivatives: Design principles and optical signalling mechanisms. Russ. Chem. Rev. 2014, 83, 155–182. [Google Scholar] [CrossRef]
- Duke, R.M.; Veale, E.B.; Pfeffer, F.M.; Kruger, P.E.; Gunnlaugsson, T. Colorimetric and fluorescent anion sensors: An overview of recent developments in the use of 1,8-naphthalimide-based chemosensors. Chem. Soc. Rev. 2010, 39, 3936–3953. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Chen, M.; Shi, G.; Wang, L.; Yin, H.; Mei, C. A novel fluorescent molecule based on 1,8-naphthalimide: Synthesis, spectral properties, and application in cell imaging. Res. Chem. Intermed. 2010, 36, 1021–1026. [Google Scholar] [CrossRef]
- Banerjee, S.; Veale, E.B.; Phelan, C.M.; Murphy, S.A.; Tocci, G.M.; Gillespie, L.J.; Frimannsson, D.O.; Kelly, J.M.; Gunnlaugsson, T. Recent advances in the development of 1,8-naphthalimide based DNA targeting binders, anticancer and fluorescent cellular imaging agents. Chem. Soc. Rev. 2013, 42, 1601–1618. [Google Scholar] [CrossRef]
- Tandon, R.; Luxami, V.; Kaur, H.; Tandon, N.; Paul, K. 1,8-Naphthalimide: A potent DNA intercalator and target for cancer therapy. Chem. Record 2017, 17, 956–993. [Google Scholar] [CrossRef]
- Tomczyk, M.D.; Walczak, K.Z. 1,8-Naphthalimide based DNA intercalators and anticancer agents. A systematic review from 2007 to 2017. Eur. J. Med. Chem. 2018, 159, 393–422. [Google Scholar] [CrossRef] [PubMed]
- Verma, M.; Luxami, V.; Paul, K. Synthesis, in vitro evaluation and DNA interaction studies of N-allyl naphthalimide analogues as anticancer agents. RSC Adv. 2015, 5, 41803–41813. [Google Scholar] [CrossRef]
- Zhang, Y.-Y.; Zhou, C.-H. Synthesis and activities of naphthalimide azoles as a new type of antibacterial and antifungal agents. Bioorg. Med. Chem. Lett. 2011, 21, 4349–4352. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Song, G.; Qian, X. Imidation of cyclic carboxylic anhydrides under microwave irradiation. Synth. Commun. 2001, 31, 1927–1931. [Google Scholar] [CrossRef]
- Kappe, C.O. Controlled microwave heating in modern organic synthesis. Angew. Chem. Int. Ed. 2004, 43, 6250–6284. [Google Scholar] [CrossRef]
- Luo, J.; Xie, Z.; Lam, J.W.Y.; Cheng, L.; Chen, H.; Qiu, C.; Kwok, H.S.; Zhan, X.; Liu, Y.; Zhu, D.; et al. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole. Chem. Commun. 2001, 18, 1740–1741. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission. Chem. Soc. Rev. 2011, 40, 5361–5388. [Google Scholar] [CrossRef] [PubMed]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: Together we shine, united we soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Powers, R.; Gibbons, A.; Joshi, D. Z-DNA: Chemistry and biological relevance. In Elsevier Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Reedijk, J., Ed.; Elsevier: Waltham, MA, USA, 2017. [Google Scholar]
- Nakai, S.; Li-Chan, E.; Arteaga, G.E. Measurement of surface hydrophobicity. In Methods of Testing Protein Functionality; Hall, G.M., Ed.; Blackie Academic & Professional, An Imprint of Chapman & Hall: London, UK, 1996; pp. 226–259. [Google Scholar]
- Xi, E.; Venkateshwaran, V.; Li, L.; Rego, N.; Patel, A.J.; Garde, S. Hydrophobicity of proteins and nanostructured solutes is governed by topographical and chemical context. Proc. Natl. Acad. Sci. USA 2017, 114, 13345–13350. [Google Scholar] [CrossRef] [PubMed]
- Lacowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: Berlin, Germany, 2006; pp. 54–55. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Betancourt, F.; Helmkay, J.; Yan, H. Microwave-Assisted Synthesis and Fluorescent Properties of 4-Phenyl-1,8-naphthalimide. Molbank 2020, 2020, M1127. https://doi.org/10.3390/M1127
Betancourt F, Helmkay J, Yan H. Microwave-Assisted Synthesis and Fluorescent Properties of 4-Phenyl-1,8-naphthalimide. Molbank. 2020; 2020(2):M1127. https://doi.org/10.3390/M1127
Chicago/Turabian StyleBetancourt, Frank, James Helmkay, and Hongbin Yan. 2020. "Microwave-Assisted Synthesis and Fluorescent Properties of 4-Phenyl-1,8-naphthalimide" Molbank 2020, no. 2: M1127. https://doi.org/10.3390/M1127
APA StyleBetancourt, F., Helmkay, J., & Yan, H. (2020). Microwave-Assisted Synthesis and Fluorescent Properties of 4-Phenyl-1,8-naphthalimide. Molbank, 2020(2), M1127. https://doi.org/10.3390/M1127



