4-[(3,5-Dimethyl-1H-pyrazol-1-yl)methyl]-4-methyl-2-phenyl-4,5-dihydrooxazole
Abstract
:1. Introduction
2. Results
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Takate, S.J.; Shinde, A.D.; Karale, B.K.; Akolkar, H.; Nawale, L.; Sarkar, D.; Mhaske, P.C. Thiazolyl-pyrazole derivatives as potential antimycobacterial agents. Bioorg. Med. Chem. Lett. 2019, 29, 1199–1202. [Google Scholar] [CrossRef] [PubMed]
- Hassan, G.S.; Abdel Rahman, D.E.; Abdelmajeed, E.A.; Refaey, R.H.; Alaraby Salem, M.; Nissan, Y.M. New pyrazole derivatives: Synthesis, anti-inflammatory activity, cycloxygenase inhibition assay and evaluation of mPGES. Eur. J. Med. Chem. 2019, 171, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Ran, F.; Liu, Y.; Zhang, D.; Liu, M.; Zhao, G. Discovery of novel pyrazole derivatives as potential anticancer agents in MCL. Bioorg. Med. Chem. Lett. 2019, 29, 1060–1064. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.B.; Tang, W.J.; Qi, X.B.; Li, R.; Liu, X.H. Novel pyrazole-5-carboxamide and pyrazole–pyrimidine derivatives: Synthesis and anticancer activity. Eur. J. Med. Chem. 2015, 90, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Sayed, G.H.; Azab, M.E.; Anwer, K.E.; Raouf, M.A.; Negm, N.A. Pyrazole, pyrazolone and enaminonitrile pyrazole derivatives: Synthesis, characterization and potential in corrosion inhibition and antimicrobial applications. J. Mol. Liq. 2018, 252, 329–338. [Google Scholar] [CrossRef]
- Prasath, R.; Bhavana, P.; Sarveswari, S.; Ng, S.W.; Tiekink, E.R.T. Efficient ultrasound-assisted synthesis, spectroscopic, crystallographic and biological investigations of pyrazole-appended quinolinyl chalcones. J. Mol. Struct. 2015, 1081, 201–210. [Google Scholar] [CrossRef]
- Xu, Z.; Gao, C.; Ren, Q.-C.; Song, X.-F.; Feng, L.-S.; Lv, Z.-S. Recent advances of pyrazole-containing derivatives as anti-tubercular agents. Eur. J. Med. Chem. 2017, 139, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Lamberth, C. Pyrazole Chemistry in Crop Protection. Heterocycles 2007, 71, 1467–1502. [Google Scholar] [CrossRef]
- Lahm, G.P.; Cordova, D.; Barry, J.D. New and selective ryanodine receptor activators for insect control. Bioorg. Med. Chem. 2009, 17, 4127–4133. [Google Scholar] [CrossRef] [PubMed]
- Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Elachqar, A.; El Hajji, S.; Kerbal, A.; Labriti, B.; Martinez, J.; Rolland, V. Synthesis of new triheterocyclic compounds, precursors of biheterocyclic amino acids. J. Mar. Chim. Heterocycl. 2008, 7, 44–49. [Google Scholar]
- Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Elachqar, A.; Kerbal, A. Simple and efficient synthesis of racemic 2-(tert-Butoxycarbonylamino)-2- methyl-3-(1H-1,2,4-triazol-1-yl)propanoic acid, a new derivative of β-(1,2,4-Triazol-1-yl)alanine. Molecules 2011, 16, 3380–3390. [Google Scholar] [CrossRef] [PubMed]
- Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Elachqar, A.; El Hajji, S.; Labriti, B.; Kerbal, A. Triheterocyclic compounds, oxazolinic precursors of biheterocyclic amino acids, Part II: Phenothiazine derivatives and structural study of regioisomers through 1H-15N 2DNMRHMBC. J. Mar. Chim. Heterocycl. 2014, 13, 39–47. [Google Scholar]
- Aouine, Y.; Alami, A.; El Hallaoui, A. N,N-dibenzyl-1-(1-[(4-methyl-2-phenyl-4,5-dihydrooxazol- 4-yl)methyl)] -1H-1,2,3-triazol-4-yl)methanamine. Molbank 2014, 2014, M819. [Google Scholar] [CrossRef]
- Boukhssas, S.; Aouine, Y.; Faraj, H.; Alami, A.; El Hallaoui, A.; Bekkari, H. Synthesis, Characterization, and Antibacterial Activity of Diethyl 1-((4-Methyl-2-phenyl-4,5-dihydrooxazol-4-yl)methyl)-1H-1,2,3-triazole-4,5-dicarboxylate. J. Chem. 2017, 2017, 4238360. [Google Scholar] [CrossRef]
- Atmani, A.; El Hallaoui, A.; El Hajji, S.; Roumestant, M.L.; Viallefont, P. From Oxazolines to Precursors of Aminoacids. Synth. Commun. 1991, 21, 2383–2390. [Google Scholar] [CrossRef]
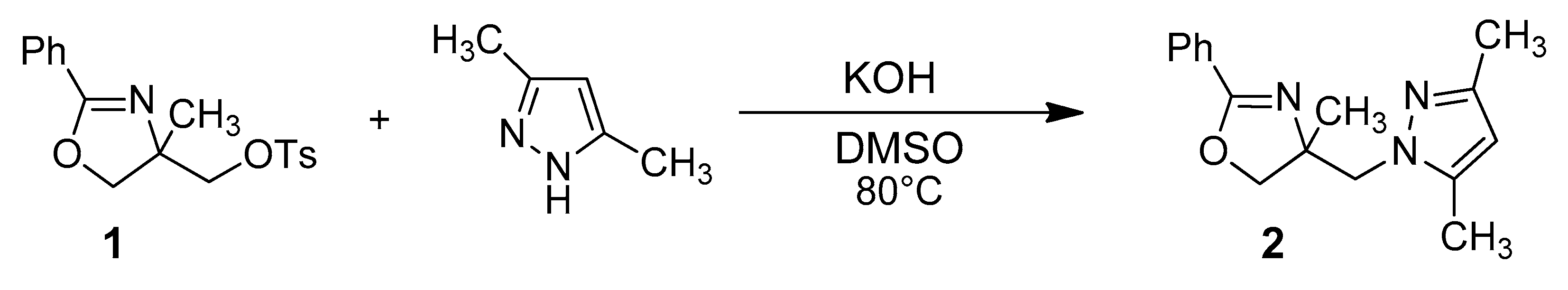
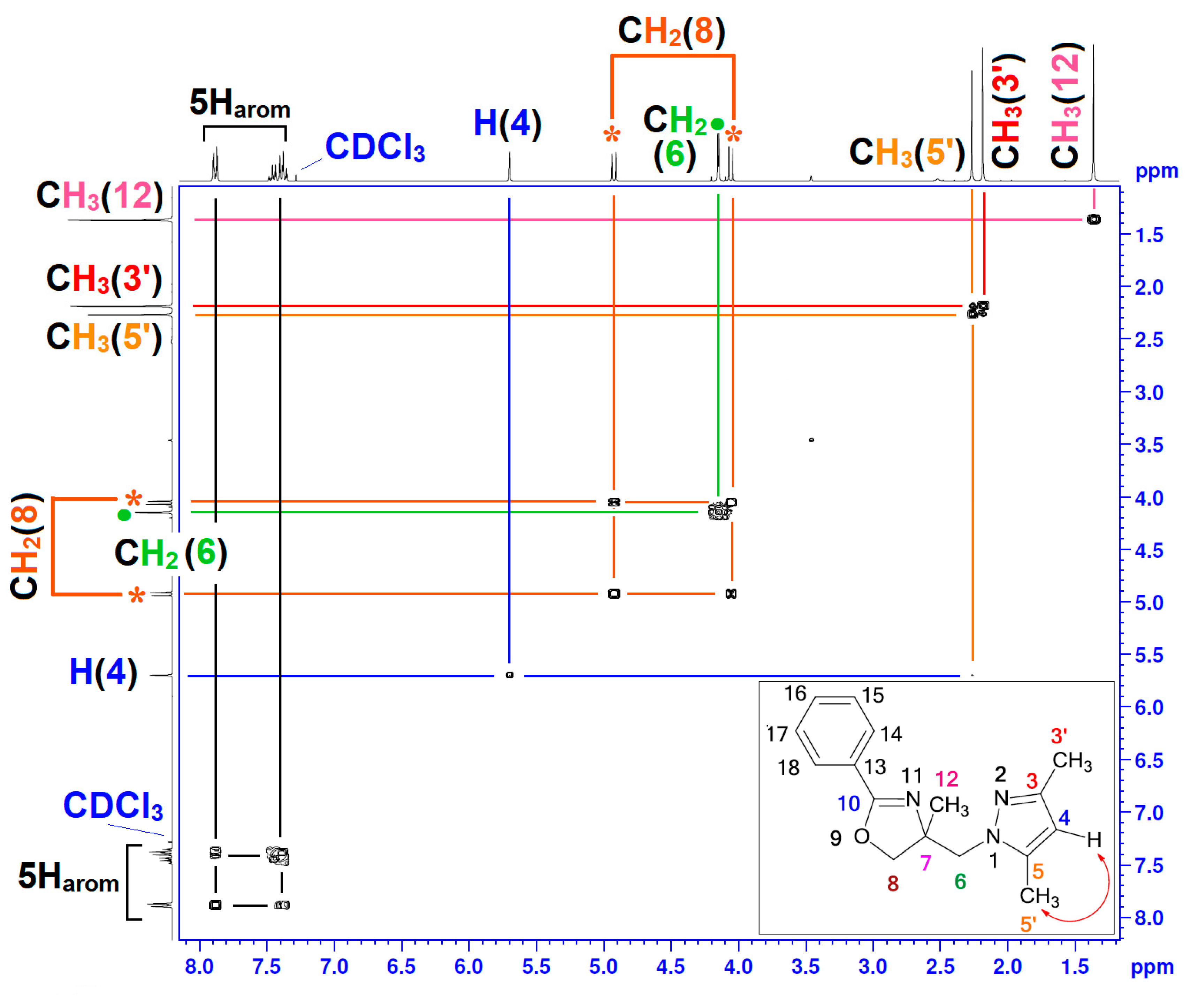
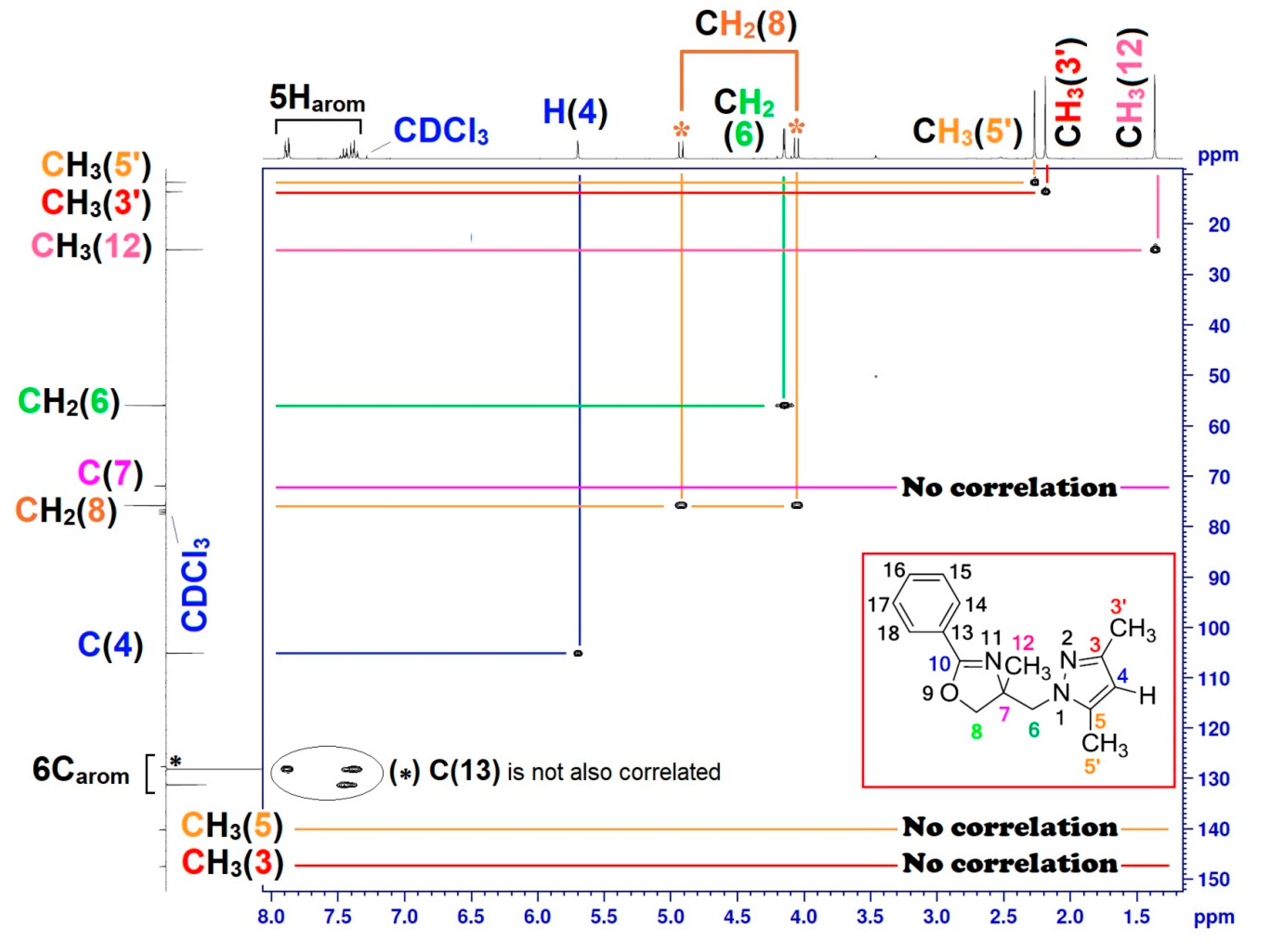
| Position | δH | δC | Correlation H-H | Correlation C-H |
|---|---|---|---|---|
| 3 | - | 147.55 | - | - |
| 3′ | 2.18 (s) | 13.46 | 3H3′–3H3′ | C3′–3H3′ |
| 4 | 5.69 (s) | 105.06 | 1H4–1H4 | C4–1H4 |
| 5 | - | 140.25 | - | - |
| 5′ | 2.266–2.268 (d, J = 0.60) | 11.60 | 3H5′–3H5′; 3H5′–1H4 | C5′–3H5′ |
| 6 | 4.096–4.2 (q, J = 14.40) | 55.95 | 2H6–2H6 | C6–2H6 |
| 7 | - | 71.90 | - | - |
| 8 | 4.042–4.939 (AB, J = 8.70) | 75.79 | 2H8–2H8 | C8–2H8 |
| 10 | - | 163.65 | - | - |
| 12 | 1.36 (s) | 25.10 | 3H12–3H12 | C12–3H12 |
| 13–18 | 7.35–7.90 (m) | 127.70–131.11 | 5Harom–5Harom | 5Carom–5Harom |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hajib, S.; Alami, A.; Faraj, H.; Aouine, Y. 4-[(3,5-Dimethyl-1H-pyrazol-1-yl)methyl]-4-methyl-2-phenyl-4,5-dihydrooxazole. Molbank 2019, 2019, M1074. https://doi.org/10.3390/M1074
Hajib S, Alami A, Faraj H, Aouine Y. 4-[(3,5-Dimethyl-1H-pyrazol-1-yl)methyl]-4-methyl-2-phenyl-4,5-dihydrooxazole. Molbank. 2019; 2019(3):M1074. https://doi.org/10.3390/M1074
Chicago/Turabian StyleHajib, Sara, Anouar Alami, Hassane Faraj, and Younas Aouine. 2019. "4-[(3,5-Dimethyl-1H-pyrazol-1-yl)methyl]-4-methyl-2-phenyl-4,5-dihydrooxazole" Molbank 2019, no. 3: M1074. https://doi.org/10.3390/M1074
APA StyleHajib, S., Alami, A., Faraj, H., & Aouine, Y. (2019). 4-[(3,5-Dimethyl-1H-pyrazol-1-yl)methyl]-4-methyl-2-phenyl-4,5-dihydrooxazole. Molbank, 2019(3), M1074. https://doi.org/10.3390/M1074






